Abstract
A 10-year-old Knabstrupper stallion was euthanatized because of severe dyspnea and exercise intolerance. Postmortem examination revealed diffuse severe alveolar emphysema and chronic fibrosing pleuritis of the caudal lung. Parts of both caudal lung lobes were covered with multiple raised firm gray to yellow plaques. Histologically, these areas consisted of circumscribed pleural fibroses and cysts of metaplastic keratinizing squamous epithelium. Immunohistochemistry revealed intense labeling for cytokeratins 5/6 and 10. In addition, caudal lung lobes were severely affected by a chronic partially obliterative bronchiolitis and peribronchiolitis with multifocal pleural involvement.
The mesothelium is embryologically derived from the mesoderm. This tissue, however, possesses mesodermal as well as epithelioid properties. This ambiguity is most obvious in neoplastic disorders of serosal surfaces, where often a biphasic growth in an epithelioid as well as a sarcomatoid pattern can be observed in men 5 and in animals 4 or the expression of cytokeratin can be detected. 5 Metaplasia of the mesothelium is less frequently reported. In women, metaplasia of the peritoneum toward Müllerian-type epithelium is observed with some frequency. 7 In contrast, squamous metaplasia of the peritoneal or pleural surface is uncommon in humans and limited to single case reports, 3,6,7 most of them being incidental findings after laparotomy for other reasons, e.g., acute appendicitis, or to areas within cystic mesotheliomas. 8 In this report, squamous metaplasia of the pulmonary pleura in a horse is described in a case with chronic pulmonary disease.
A 10-year-old Knabstrupper stallion was presented with labored breathing and severe exercise intolerance. A clinical diagnosis of chronic obstructive pulmonary disease was proposed. The horse was killed for humane reasons, and a complete necropsy was performed. The main findings consisted of a poorly collapsed lung with marked, caudally pronounced alveolar emphysema and a multifocal chronic pleuritis. The caudal and middle parts of both caudal lung lobes were covered with numerous raised, firm, and smooth gray to yellow plaques, up to 2 mm in diameter (Fig. 1). Airways were filled with abundant viscous white to yellow fluid. Extrathoracic lesions comprised focally mild myocardial fibrosis, moderate multifocal chronic perihepatitis, moderate chronic endarteritis of the cranial mesenteric artery, and mild splenic follicular hyperplasia.
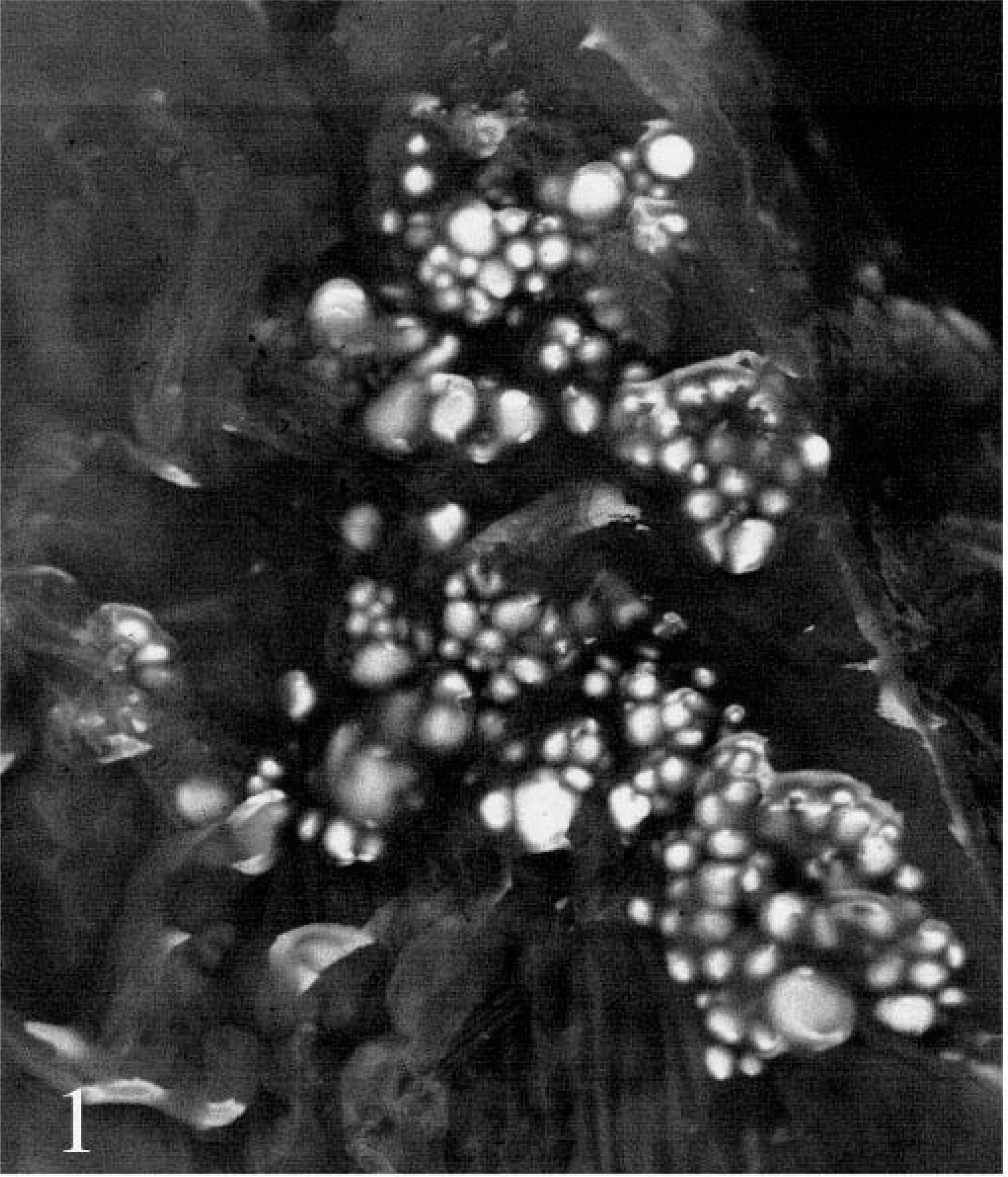
Lung; horse. Superficially protruding foci of squamous epithelium on pleural surface.
Among others, six tissue samples from different locations of the lung were collected, fixed in 10% buffered formalin, trimmed, and embedded in paraffin wax according to a standard laboratory procedure. Tissue sections stained with hematoxylin and eosin were examined by light microscopy. Additional tissue sections from the lung were dewaxed for immunohistochemistry. Antigen was demasked by heating in a microwave oven (cytokeratins 5/6 and 10; 750 W, 2 × 5 minutes) or by pronase E treatment (LP-34; 20 minutes, 0.05% in phosphate-buffered saline, 37 C). The primary antibodies anti–cytokeratin 5/6 (D5/16B4), anti–cytokeratin 10 (DE-K-10), and Clone LP34 (reacting with cytokeratins 5/6 and 18) were used (all DAKOcytomation, Glastrop, Denmark). The secondary antibody was a biotinylated goat anti-mouse antibody (Ba9200, Vector laboratories, Burlingame, California). For detection, an ABC-Kit was applied with diaminobenzidine as a chromogene according to the instructions of the manufacturer. The same procedure omitting the first antibody was used as the negative control. Sections of formalin-fixed, paraffin-embedded equine skin served as positive control.
Histologically, the lung showed severe multifocal partially obliterative bronchiolitis and peribronchiolitis with epithelial hyperplasia, hyperplasia of goblet cells, peribronchial fibrosis, and hyperplasia of bronchus-associated lymphoid tissue. Numerous bronchioli were filled with cellular debris and neutrophilic granulocytes. These lesions were most pronounced in the caudal parts of the lung. The pleura displayed multifocal chronic pleuritis with the formation of filamentous fibrotic strands, fibrosis, and multifocal infiltration with lymphocytes and plasma cells invading the underlying pulmonary tissue. Within pleural protrusions and on the plain surface of the pulmonary pleura, numerous round cystlike foci of squamous epithelium with concentric keratinization were detected (Fig. 2). Centrally, accumulations of keratin squames were discernible. In one area, rupture of a subme-sothelial cyst had induced a perilesional granulomatous response with infiltration of multinucleated giant cells and macrophages. Histologic lesions of extrapulmonary tissues were confined to mild lymphoplasmacytic rhinitis, few meningeal psammoma bodies, and mild lymphoplasmacytic periportal hepatitis.
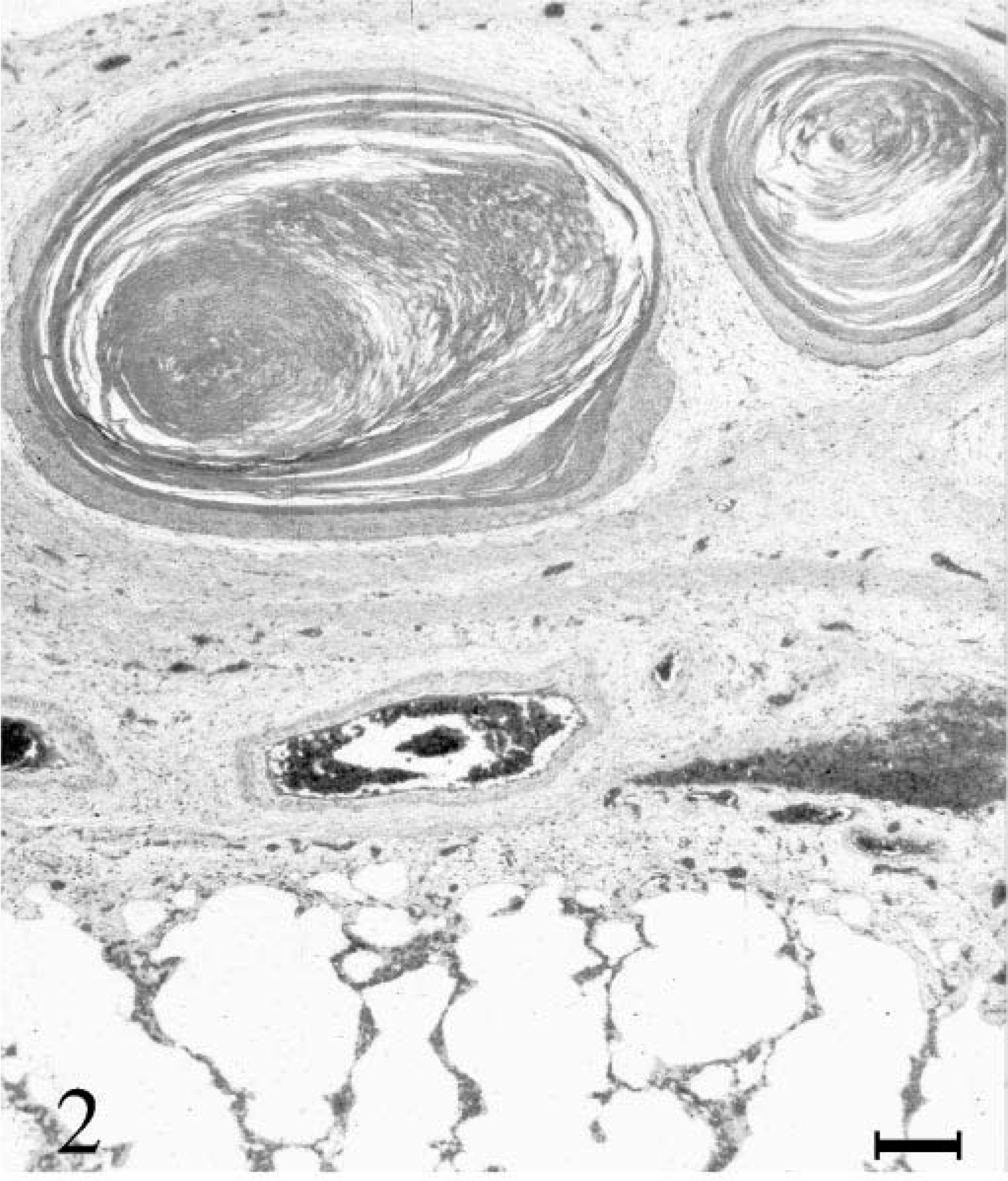
Lung; horse. Thickened pleura with cystic squamous foci. Hematoxylin and eosin. Bar = 200 µm.
Immunohistochemistry revealed intense labeling of cytokeratin 5/6 and LP34 antibodies in bronchial epithelia. Pleural squamous foci displayed strong staining in the basal layers. With progradient keratinization of the cells, the staining intensity decreased (Fig. 3). Cytokeratin 10 could not be detected in pulmonary tissue. The cytokeratin 10 labeling of the squamous areas of the pleura showed an increasing intensity starting from the spinous layer toward the fully keratinized centers (Fig. 4).
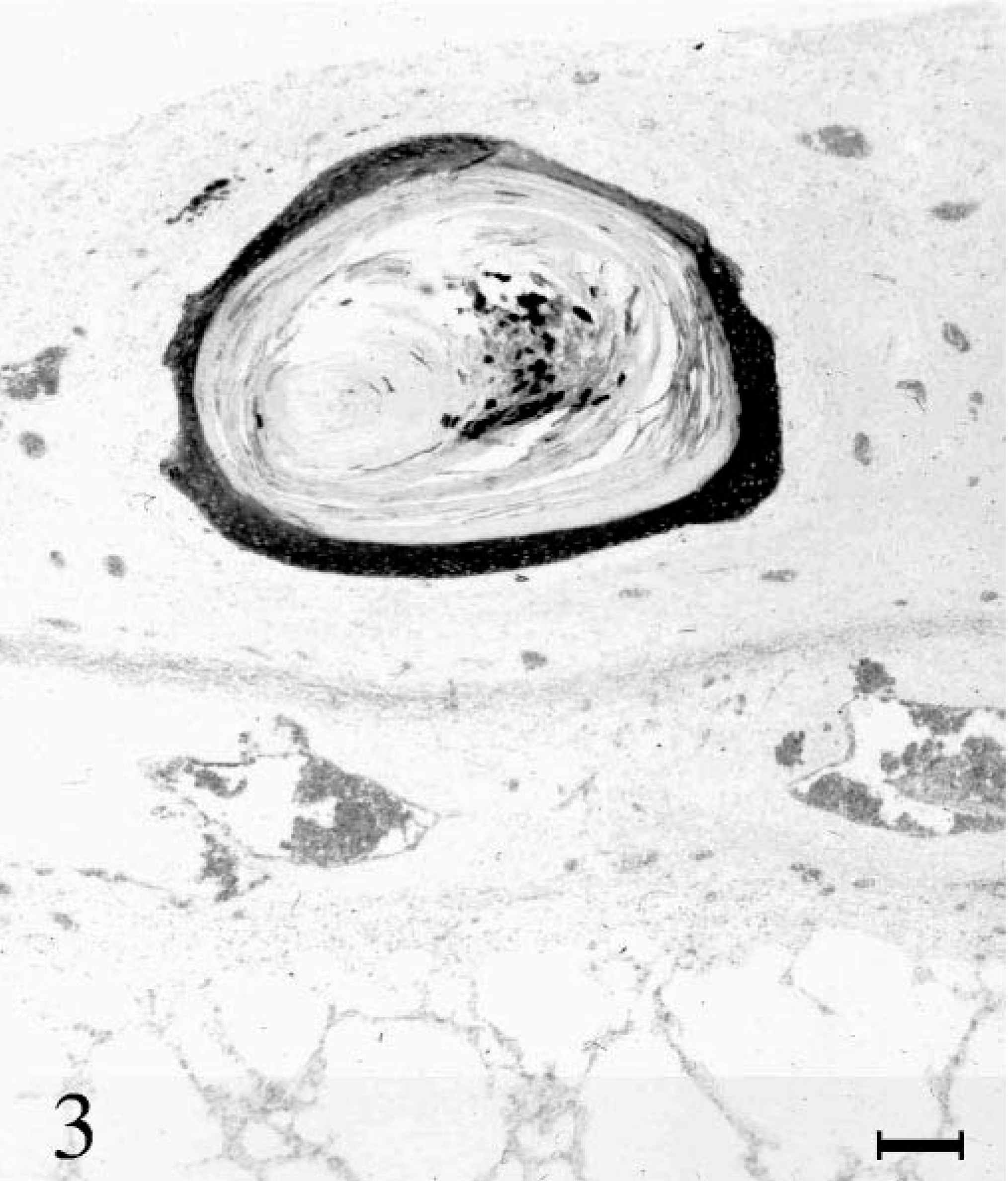
Lung; horse. Detection of cytokeratin in basal layers of a cystic squamous focus. Immunohistochemistry with LP 34, hemalaun counterstain. Bar = 200 µm.
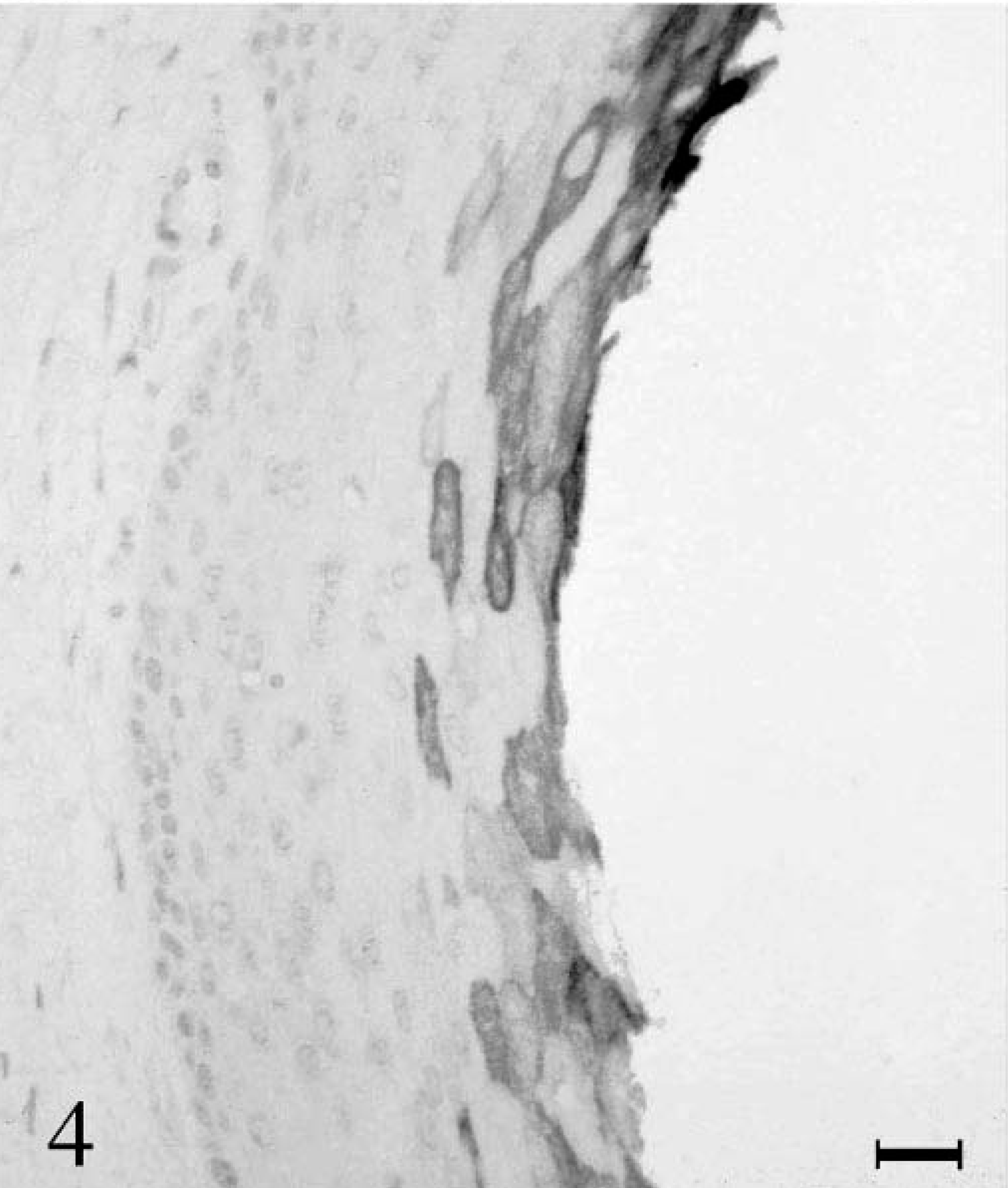
Lung; horse. Expression of cytokeratin 10 in squamous epithelium increases with proceeding keratinization. Immunohistochemistry for cytokeratin 10. Bar = 50 µm.
No case of spontaneous keratinizing squamous epithelium in and on the serosal surface is described in veterinary literature accessible from databases (PubMed, 12 million citations). Young (1928) (cited in Crome 3 ) observed the development of transitional and stratified epithelia after the experimental injection of Sudan III, sodium cholate, and olive oil into the pleural cavity of rabbits.
In rats, a cystic keratinizing lesion can be induced in the pulmonary tissue. Their origin is suspected to take place from metaplastic alveolar epithelia or clara cells. 1,2 Pleural involvement is uncommon, however, whereas in the presented equine case, no affected pulmonary parenchyma was detected. The strong staining of affected foci for cytokeratin 10 further supports an extrabronchial origin.
Because of the cystic shape of most foci, a differential diagnosis of hamartomatous epithelial cysts has to be considered. The occurrence of keratinizing cysts within post-pleuritic adhesions strongly suggests a postembryologic development of the lesion. Epidermal epithelial cysts, especially dermoid cysts, are believed to be caused by traumatic implantation of surface epithelia. This pathogenesis appears improbable for intrathoracic lesions.
The pathogenesis of pleural metaplasia in this case most probably comprises persistent noxious stimuli by chronic inflammation on one hand and mechanical alterations by extremely forced muscle sustained expiration on the other hand. A proof for the latter can be found in the rupture of some cysts despite the mechanical coverage inside the thoracic cavity. Based on a single case, however, it cannot be decided whether the simultaneous occurrence of inflammation and metaplasia is pathogenically related. In the history of this case, there was no known exposure toward fibers, e.g., asbestos. Crome 3 describes human cases of peritoneal squamous metaplasia as incidental findings without inciting inflammation. The reported cases differed from the presented equine, however, in that they did not show keratinization. In a case of peritoneal squamous metaplasia after long-term peritoneal dialysis, in contrast, keratinization could be demonstrated. 7
