Abstract
Porcine circovirus type 2 (PCV2) is associated with reproductive failure in female pigs. However, the association of PCV2-positive semen in the pathogenesis has not been elucidated. The objectives of this study were to determine whether semen spiked with PCV2 causes infection in PCV2-naïve, mature female pigs and whether delivery of PCV2 via artificial insemination causes reproductive failure or fetal infection. Nine sows were randomly allocated into 3 groups of 3 sows each and artificially inseminated with PCV2 DNA-negative semen (group 1), PCV2 DNA-negative semen spiked with PCV2a (group 2), or PCV2b (group 3). All sows in groups 2 and 3 developed PCV2 viremia 7 to 14 days after insemination. None of the group 2 sows became pregnant, whereas all group 3 sows (3/3) farrowed at the expected date. At parturition, presuckle serum samples were collected, and live-born piglets, stillborn fetuses, and mummified fetuses were necropsied. All live-born piglets (n = 8) in group 3 were PCV2 viremic at birth. Stillborn fetuses (n = 2) had gross lesions of congestive heart failure. Mummified fetuses (n = 25) varied in crown-rump length from 7 to 27 cm, indicating fetal death between 42 and 105 days of gestation. PCV2 antigen was detected in the myocardium by immunohistochemistry of 7/8 (88%) live-born piglets, 2/2 (100%) of the stillborn fetuses, and 25/25 (100%) of the mummified fetuses. In addition, 4/25 mummified fetuses had PCV2 antigen associated with smooth muscle cells and fibroblasts. The results of this study indicate that intrauterine administration of PCV2 causes reproductive failure in naïve sows.
Introduction
Porcine circovirus type 2 (PCV2) is a small, nonenveloped, single-stranded, circular DNA virus of approximately 1.7 kb with 3 known genotypes capable of causing porcine circovirus-associated disease (PCVAD) in swine: PCV2a, PCV2b, and the more recently described PCV2c. 25,38 PCV2 was first described in association with wasting disease in nursery pigs with otherwise high health status. 10 Since its initial description, PCV2 has been implicated and associated with multiple disease manifestations (pneumonia, diarrhea, and reproductive failure) in growing and mature pigs. 5 Clinical disease related to PCV2 is now collectively described as PCVAD. Currently, most swine herds worldwide are seropositive for PCV2. 1 Subclinical PCV2 infections are common, and only a small percentage of infected animals develop clinical PCVAD. 25
Reproductive failure is a broad term in swine and refers to failure of implantation resulting in irregular return to estrus, failure of pregnancy progressing to term due to expulsion of all embryos or fetuses (abortion), or reduced litter size due to increased numbers of nonviable piglets at parturition. 2 Indications are that PCV2-associated reproductive failure can, depending on the timing of infection, manifest at all stages of pregnancy (early embryonic death; abortion; reduced litter size) and are more frequently observed in newly populated swine farms 4,22,40 or associated with gilts entering an existing operation. 13
PCV2-associated systemic illness of the pregnant dam can be observed after infection and can be vertically transmitted during dam viremia. 22,30,40 PCV2 has a tropism for myocardial tissues in fetuses when inoculated in utero at 57, 75, or 92 days of gestation, and infection generally results in myocardial necrosis, inflammation, and fibrosis. 22,35,40 However, if infection occurs in late gestation, the cellular tropism of PCV2 is shifted from myocardiocytes to antigen-presenting cells and lymphoid tissues. 34
In recent years, PCVAD rapidly spread across major swine-producing regions of North American, causing devastating morbidity and mortality, especially in postweaned pigs, which subsequently was thought to be associated with introduction of PCV2b in affected areas. 6 With the overwhelming incidence of PCVAD, potential transmission routes of PCV2 across pig populations were reevaluated. Currently, the main route of PCV2 transmission is thought to be fecal-oral. 36 However, PCV2 can been detected in semen of infected boars. 12,14,15,17 Infected boars shed PCV2 continuously for extended periods without showing clinical signs or changes in semen quality parameters. 15,17 Furthermore, artificial insemination techniques are commonly used for dam matings, and semen may be transported over great distances. Important swine viral diseases, such as porcine reproductive and respiratory syndrome virus infection, have been shown to be transmitted with boar semen. 9 Thus, the objectives of this study were to determine if semen spiked with PCV2 has the ability to cause infection in PCV2-naïve, mature female swine and if delivery of PCV2 via artificial insemination causes reproductive failure or fetal infection.
Materials and Methods
Animals and housing
Nine primiparous and multiparous 7- to 30-month-old crossbred conventional specific pathogen-free sows were used in the study. The animals were purchased from a source farm known to be serologically negative for PCV2, porcine reproductive and respiratory syndrome virus, swine influenza virus, procine parvovirus (PPV), swine hepatitis E virus, and encephalomyocarditis virus. At the Iowa State University research facility, the sows were individually housed in 9 rooms in a Biosafety Level 2 facility. Each room was approximately 2.5 × 3 m in size, had separate ventilation, contained an individual water nipple, and had an anteroom where personal protective gear was applied to personnel entering the room. The sows were fed approximately 2 kg of a balanced corn-soybean–based ration formulated for gestation daily until day 90 of pregnancy, when the amount of feed was increased to approximately 3 kg.
Experimental design
The experimental protocol for the study design was approved by the Iowa State University Institutional Animal Care and Use Committee. At the research facility, the sows were randomly allocated into 1 of 3 groups. Group 1 (n = 3) sows served as negative controls and were artificially inseminated with extended PCV2 DNA–negative semen upon estrus detection. Groups 2 (n = 3) and 3 (n = 3) sows were artificially inseminated with extended semen spiked with PCV2a or PCV2b, respectively, as described below. The sows were allowed to gestate, carry pregnancy to term, and farrow naturally. At parturition, presuckle serum samples were obtained from all piglets. Immediately after collection of blood, all live-born piglets were humanely euthanatized by intravenous overdose of pentobarbital for tissue evaluation.
Estrus synchronization
All sows were synchronized for estrus detection prior to artificial insemination. A commercially available product (Matrix, Intervet Inc., Millsboro, DE) was administered to each sow at the recommended dosage for 15 connective days. Twenty-four hours after the last administration, each sow received 5 ml of gonadotropin (P.G. 600, Intervet Inc.) intramuscularly in the neck.
Semen processing
Semen, which was determined to be free of PCV2 DNA by quantitative real-time polymerase chain reaction (PCR), 29 was extended using a commercially available semen extender (TriXcell, IMV International, Minneapolis, MN) that had been reconstituted with sterile water and warmed to match semen temperature. Each 80-ml insemination dose was adjusted to contain approximately 5 billion sperm per milliliter. Extended semen was stored at 17°C, rotated daily, and used within 4 days of collection.
Inoculation
One day prior to expected estrus, all sows were crated for breeding in individual 0.7 × 2 m crates that allowed the sows to lie down but prevented full turns and access to the floor drain. During estrus, all groups were artificially inseminated with extended semen once a day for 3 consecutive days. Immediately prior to artificial insemination of group 2 and 3 sows, 5 ml of PCV2a or PCV2b was added to the 80-ml dose of extended semen, respectively.
The PCV2 isolates used in this study originated from finishing pigs clinically affected by PCVAD. 7,26 The PCV2a isolate ISU-40895 and the PCV2b isolate NC-16485 had an infectious titer with respective infectious DNA clones of 104.4 and 104.2 TCID50 per milliliter, respectively. Both PCV2 isolates were produced in a similar manner by transfecting PK-15 cells as described previously. 8 The PCV2a and PCV2b isolates shared 95.7% nucleotide sequence identity. Inocula titers were determined by using an immunofluorescence assay with a PCV2 specific antibody, 8 and the PCV2 inocula were stored at −80°C until use.
Sample collection
Serum samples were collected via venapuncture of the jugular vein using a Vacutainer system from all sows prior to PCV2 inoculation on days post inoculation (DPI) −1 and then weekly thereafter until parturition. At parturition, colostral samples from each sow were obtained in a sterile centrifuge tube, and presuckle serum samples were collected from all viable piglets by venapuncture using a vacutainer system. All samples were stored at −80°C until tested.
Serology
Presuckle piglet sera, sow sera, and colostral samples were tested for anti-PCV2 IgG antibodies using a PCV2 recombinant capsid protein (ORF2)-based ELISA. 21 Samples with a sample-to-positive ratio equal or greater than 0.2 were considered positive.
Sow serum samples collected on DPI −1 and DPI 112 were tested for the presence of anti-porcine reproductive and respiratory syndrome virus, anti-swine influenza virus H1N1, anti-swine influenza virus H3N2 antibodies by ELISA (IDEXX Laboratories, Inc., Westbrook, ME), anti-PPV antibodies by hemagglutination inhibition, 19 and anti-encephalomyocarditis virus antibodies by virus neutralization (National Veterinary Service Laboratory, Ames, IA; protocol number BPPRL2109).
Polymerase chain reaction
Colostrum samples, presuckle piglet serum samples, and sow serum samples were tested for the presence and amount of PCV2 DNA by quantitative real-time PCR as previously described. 28 PCV2 DNA was extracted using a commercially available isolation kit (QIAamp DNeasy blood & tissue kit, Qiagen, Valencia, CA) following the manufacturer's protocol and recommendations. Quantitative real-time PCR parameters were the same as previously described. 28
Virus isolation and sequencing
A tissue pool containing myocardium, spleen, and tonsil from group 3 stillborn piglets was homogenized in phosphate-buffed saline and used for PCV2 isolation on a continuous porcine kidney cell line as previously described. 31 Immunofluorescence microscopy using polyclonal anti-PCV2 was used to demonstrate PCV2 within transfected cells. 31 The isolated PCV2 was partially sequenced after amplification of the ORF2 by nested PCR as previously described. 24 The obtained sequence was compared to that of the original inoculum.
Necropsy
All expelled live-born piglets were humanely euthanatized, and all piglets including stillborns and mummified fetuses were necropsied. Gross lesions were evaluated, and crown-to-rump length was used for determination of the approximate time of fetal death during gestation. 39 Tissues collected from the piglets included heart, lungs, liver, kidney, brain, spleen, tonsil, thymus, and pinna (ear notch). In addition, placental sections were collected. A similar set of tissues was collected for mummified fetuses, depending on the degree of decomposition.
Microscopic evaluation and immunohistochemistry
Tissue sections were collected in 10% neutral-buffered formalin, routinely processed, stained with hematoxylin and eosin, and evaluated by a veterinary pathologist who was blinded to the treatment status. Immunohistochemistry (IHC) to detect PCV2 antigen was performed as previously described using rabbit polyclonal antisera. 37 The presence and amount of PCV2 antigen in selected tissue sections (myocardium, lungs, pinnae, tonsil, and placenta) was blindly scored as previously described. 27
Statistical analysis
Summary statistics (mean and SE) for log-transformed genomic copies of PCV2 DNA in collected samples (sow colostrum and presuckle piglet serum) were performed using JMP 7.0.2 (SAS Institute, Cary, NC).
Results
Estrus and pregnancy
Four to 5 days after gonadotropin administration, all synchronized females exhibited signs of estrus as expected and were artificially inseminated. Visible signs of estrus detection were not observed in group 1, 2, or 3 animals during the subsequent 2 estrus cycles (∼21 and 42 days). Starting at approximately 12 weeks of gestation, all group 1 and 3 sows showed visible abdominal distension and mammary development. None of the group 2 sows had visible signs of pregnancy, and these sows were later confirmed to be not pregnant.
Group 1 and 3 animals carried pregnancy to term and farrowed between 114 and 117 DPI. The numbers of live-born piglets, stillborns, and mummified fetuses are summarized in Table 1. Group 3 mummified fetuses varied in crown-to-rump length from 7 to 27 cm (Fig. 1), indicating fetal death between 42 and 105 days of gestation.
Total number of expelled piglets and fetuses by litter at parturition after artificial insemination of porcine circovirus type 2 (PCV2)-negative semen or extended semen spiked with PCV2a or PCV2b.


Stillborn and mummified fetuses expelled at parturition from a group 3 (intrauterine porcine circovirus type 2 b inoculation) sow with varying crown-to-rump lengths.
Serology
All serum samples collected on DPI −1 were negative for anti-PCV2 antibodies. PCV2-specific antibodies were not detected in presuckle serum samples from group 1 piglets or group 1 sows at parturition, and colostral samples were negative for anti-PCV2 antibodies in this group.
All group 2 and 3 sow serum samples were positive for anti-PCV2 IgG antibodies on DPI 112, and ELISA sample-to-positive ratios ranged from 0.4 to 1.0. Anti-PCV2 antibodies were detected in 1/1, 2/2, and 1/5 presuckle live-born piglet serum samples of individual group 3 litters. All group 3 colostral samples were positive for PCV2 antibodies at parturition, with sample-to-positive ratios ranging from 0.4 to 0.8.
Specific IgG antibodies against porcine reproductive and respiratory syndrome virus, swine influenza virus, PPV, and encephalomyocarditis virus were not detected in DPI −1 or DPI 112 serum samples collected from group 1, 2, or 3 sows.
Polymerase chain reaction
All sows were negative for PCV2 DNA on DPI −1. PCV2 genomic DNA was not detected in weekly serum samples or in colostral samples of group 1 sows during the study. Similarly, all presuckle serum samples collected from group 1 piglets were negative for PCV2 genomic DNA.
All group 2 females had detectable PCV2 DNA in serum samples by DPI 7. Length of viremia varied among group 2 sows; individual females had detectable PCV2 DNA in serum for 7, 11, and 14 consecutive weeks (Table 2).
Summary of porcine circovirus type 2 (PCV2) DNA detected in sow sera by polymerase chain reaction after artificial insemination of sows with PCV2-negative semen (group 1, n = 3) or extended semen spiked with PCV2a (group 2, n = 3) or PCV2b (group 3, n = 3).
In group 3, 2/3 sows were PCV2 viremic on DPI 7, and 3/3 were viremic on DPI 14. Similar to group 2, the length of PCV2 viremia varied among the group 3 sows. PCV2 genomic DNA was detected for 3 weeks and 6 weeks in 2/3 sows. The third sow in group 3, in which serum PCV2 DNA was first detected on DPI 14, remained viremic until parturition, which corresponds to 15 weeks (Table 2). All group 3 colostral samples were positive for PCV2 genomic DNA with a log10 mean genomic PCV2 copy numbers ± SE of 105.0 ± 0.5 per ml colostrum. Presuckle serum samples from all live-born piglets in this group (n = 8) were positive for PCV2 DNA with log10 mean genomic PCV2 copy numbers ± SE of 106.7 ± 0.4 per ml serum.
Virus isolation and sequencing
PCV2 was isolated from the homogenized tissue pool of myocardium, spleen, and tonsil from group 3 stillborn piglets after 2 blinded cell culture passages. Sequencing of the cultured PCV2 isolate showed 100% sequence identity to the ORF2 gene of the original PCV2b inoculum used for inoculation of the dams.
Gross lesions
Gross abnormalities were not observed in group 1 live-born piglets, group 1 stillborn fetuses, or group 3 live-born piglets.
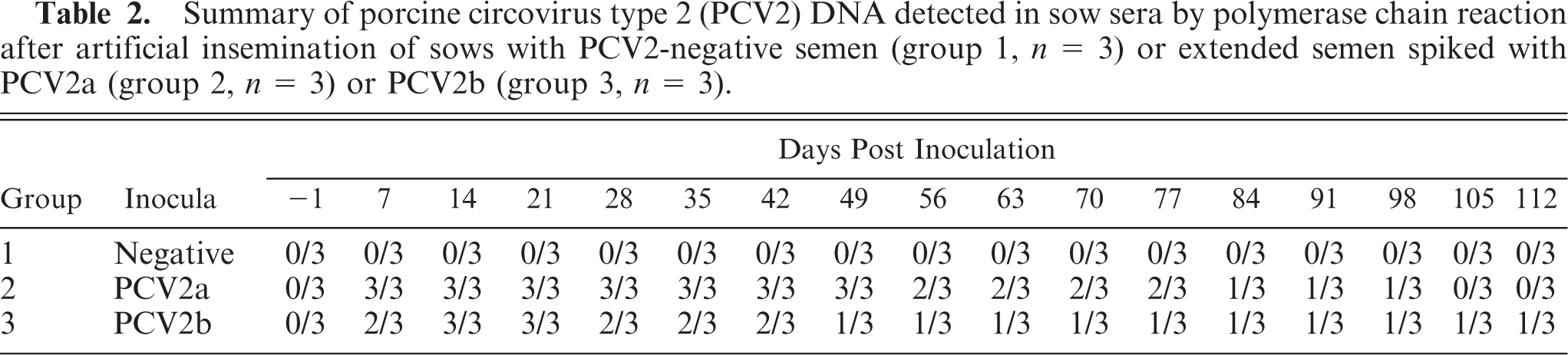
The 2 group 3 stillborn fetuses were 23 and 27 cm in crown-to-rump length. Both had mild to moderate abdominal distension with approximately 8 to 10 ml of straw-colored fluid in the abdominal cavity with occasional strands of fibrin adhered to viscera. The thoracic cavities of both pigs contained approximately 10 and 30 ml of a similar fluid. The hearts were 2 to 3 times enlarged, had a globose appearance, and had irregular pale streaks within the myocardium (Fig. 2). On cut section, pale streaks were present within the walls of both ventricles and the interventricular septum. Eccentric dilation of both the left and right ventricle was evident, and there was mild interlobular pulmonary edema. Hepatomegaly with an enhanced lobular pattern was present in both stillborn piglets along with mild to moderate perirenal and mesocolonic edema. In addition, 1/2 piglets had multifocal splenic petechial hemorrhages and a small, barely visible thymus.
Heart, stillborn fetus, group 3. Globose appearance with multifocal pale streaks within the myocardium. Bar = 1 cm.
The larger mummified fetuses (>23 cm crown-to-rump length) in group 3 litters had abdominal distension with marked amounts of serosanguineous to clear subcutaneous edema and mild to moderate amounts of serosanguineous abdominal fluid (∼8–15 ml). Similar to the stillborn piglets, the mummified fetuses had hepatomegaly and cardiomegaly.
Microscopic evaluation
Microscopic lesions were not observed in placental sections from any groups or tissues from group 1 live-born or stillborn piglets.
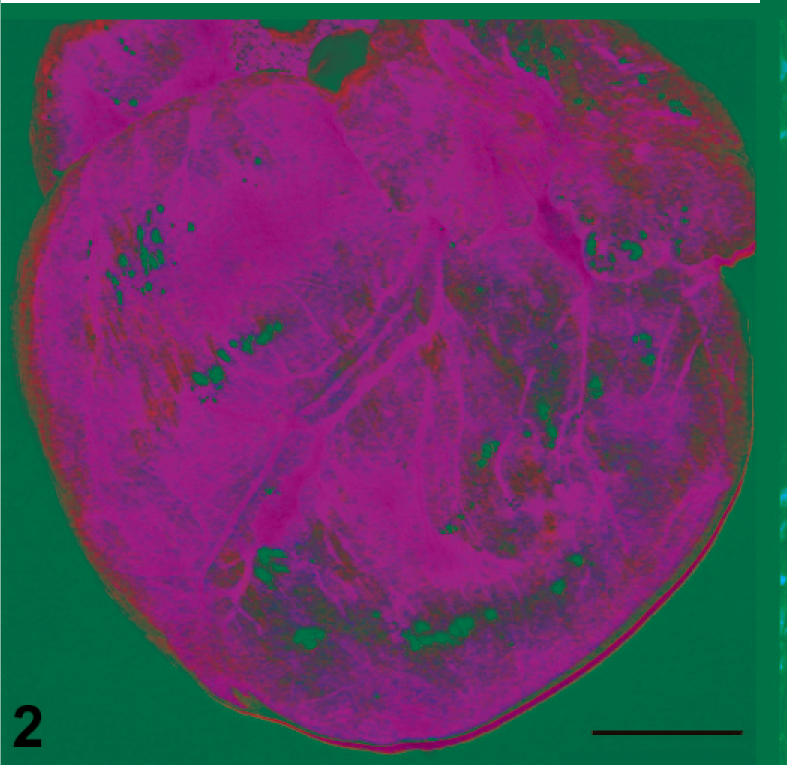
In group 3 live-born piglets, 4/8 pigs had multifocal to locally extensive regions where myocardiocytes were lost, fragmented, or occasionally vacuolated with absent or condensed nuclei. Replacing lost myocardiocytes were low amounts of loosely arranged fibrous connective tissue and edema, which also separated adjacent myocardiocytes. Low numbers of lymphocytes were occasionally present within regions of myocardial necrosis or separating myocardiocytes. Other microscopic changes in live-born group 3 piglets included splenic periarterial sheaths composed of macrophages and rare multinucleated giant cells rather than lymphocytes in 2/8 piglets (Fig. 3) and mild to moderate interlobular pulmonary edema in 2/8 piglets.
Spleen, live-born piglet, group 3. Splenic periarterial sheath composed of macrophages and rare multinucleated giant cells (arrows) rather than lymphocytes. Hematoxylin and eosin, 400×. Bar = 50 µm.
Stillborn group 3 fetuses had similar but more extensive myocardial lesions compared with the live-born piglets. Rare basophilic intranuclear inclusion bodies were occasionally observed in myocardiocytes of stillborn fetuses. Furthermore, 1/2 stillborn fetuses had locally extensive hemorrhage that separated myocardiocytes with prominent dense linear bands of fibrous connective tissue replacing lost myocardiocytes (Fig. 4). Occasional cardiomyocytes were mineralized. In liver sections, both fetuses had apoptotic and coagulative necrosis of hepatocytes with individualization. Mild vacuolar change, congestion, and hemorrhage were also apparent. Splenic lesions were observed in 1/2 stillborn fetuses, with periarterial sheaths being composed of macrophages and rare multinucleated giant cells rather than lymphocytes.

Heart, stillborn fetus, group 3. Myocardiocytes are multifocally lost and replaced by prominent linear bands of fibrous connective tissue. The adjacent myocardium is necrotic and degenerative with separation of myocardiocytes by lymphocytes and edema. HE, 100×. Bar = 200 µm. Insert: Heart, live-born piglet, group 3. Immunohistochemistry, abundant porcine circovirus type 2 antigen with myocardiocytes. 400×. Bar = 50 µm.
All 25 group 3 mummified fetuses had myocardial necrosis with fibrous connective tissue similar to stillborn fetuses. In addition, locally extensive regions of myocardium were mineralized in 8/25 mummified fetuses.
Immunohistochemistry
Immunohistochemistry for PCV2 antigen on heart, lung, tonsil, and pinna (ear notch) tissues of group 1 live-born piglets and stillborn fetuses revealed no detectable PCV2 antigen. Placental tissue from all sows was also negative for PCV2 antigen.
Seven of 8 group 3 live-born piglets had low to abundant amounts of PCV2 antigen within the myocardium and 1/8 had moderate tonsil-associated PCV2 antigen. PCV2 antigen was not detected in sections of lung tissues or ear notches.
Both stillborn fetuses in group 3 had moderate to abundant staining of PCV2 antigen in myocardial tissue (Fig. 4). PCV2 antigen was also detected in tonsillar tissue sections with mild to moderate IHC staining. Lung and pinna were negative.
All 25 group 3 mummified fetuses had abundant PCV2 antigen in myocardial tissue and in tonsils. Furthermore, smooth muscle cells and fibroblasts stained positively for PCV2 antigen in the lung of 4/25 and the pinna of 2/25 of the mummified fetuses in group 3 (Fig. 5).
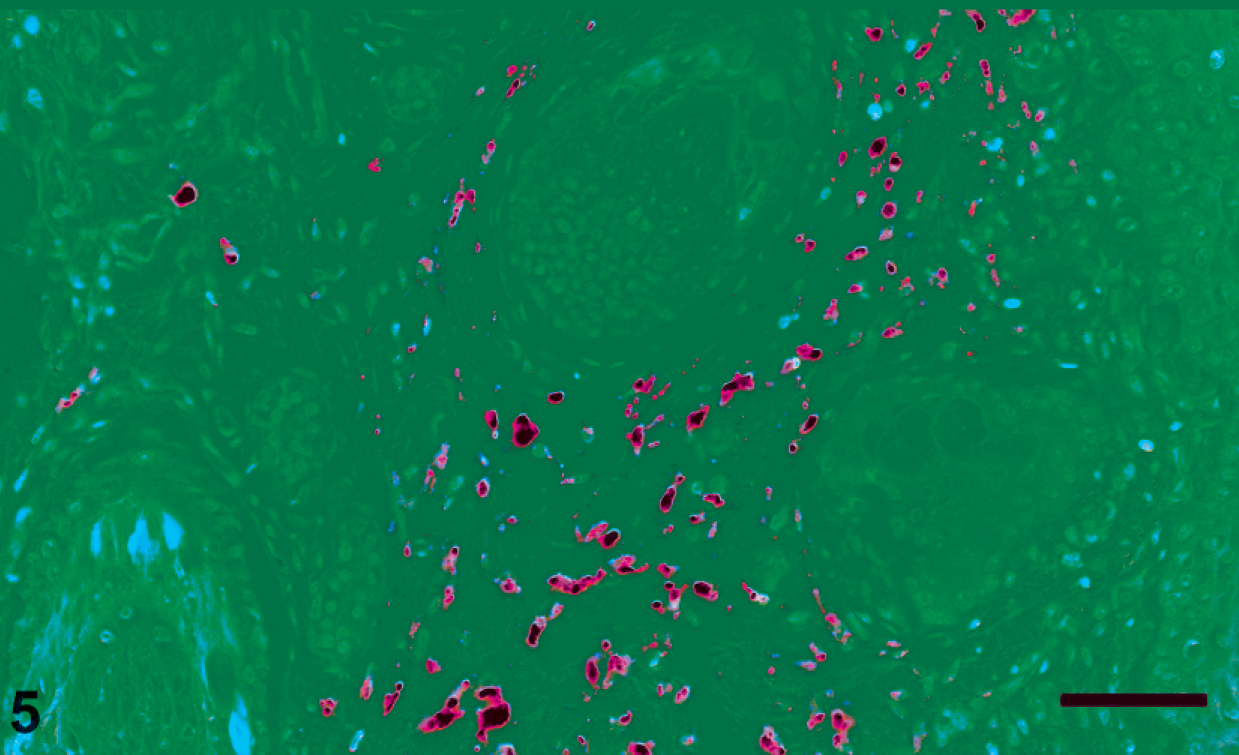
Pinna, mummified fetus, group 3. Immunohistochemistry, porcine circovirus type 2 antigen present in fibroblast-like cells within the dermis surrounding hair follicles. 400×. Bar = 50 µm.
Discussion
Reproductive failure, manifesting as either irregular return to estrus, abortion, or increased numbers of nonviable pigs in term litters, has been previously attributed to PCV2 infection in both field and experimental scenarios. 22,30,40 However, the role of semen transmission has not been appropriately evaluated as a potential source of dam or fetal infection. In boars, PCV2 infection is not typically associated with clinical disease or abnormalities in sperm morphology; however, low amounts of PCV2 DNA are frequently detected in semen. 14,15,17 The potential for semen transmission is apparent; however, previous epidemiologic reports have not found a direct correlation between semen and PCV2 transmission. 11,33
Previously, PCV2-positive semen collected from experimentally inoculated Landrace boars was found to be infectious in a swine bioassay model; however, when the same semen was extended and used to artificially inseminate PCV2-naïve gilts, PCV2 transmission was not observed. 16 Thus, to further investigate the role of semen in the transmission of PCV2, the virus inoculum was added to each extended semen dose to determine if PCV2 is capable of infecting the dam via this route. To ensure that PCV2 infection did not occur by other routes (orally), the inseminated mature females were crated and not allowed to turn around, from the day of estrus detection until 3 days after the last inoculation. The results showed that both PCV2a and PCV2b were capable of inducing infection via the intrauterine route as evidenced by PCV2 viremia and seroconversion. This indicates that the intrauterine route is a potential route of PCV2 infection.
In group 1, pregnancy was achieved in all females after insemination, and pregnancy was maintained to term. Similarly, all group 3 sows artificially inseminated with extended semen spiked with PCV2b became pregnant and maintained pregnancy to term. Alternatively, all group 2 females failed to become pregnant or maintain pregnancy after artificial insemination. This may in part be due to the low numbers of sows used in the study (because of cost prohibitions and lack of availability of PCV2-negative animals), usage of the PCV2a genotype, the slight difference in inocula titer, or combinations of these events. However, estrus was not observed in any group 2 sow around 21 or 42 DPI, which corresponded to the regular return to estrus intervals. Thus, it can be speculated that conception may have occurred but was terminated with subsequent resorption similar to what is reported with PPV infection. 18
In previous field and experimental cases of PCV2-associated reproductive failure, abortion was observed 30,40 but was not a feature in the current study. The occurrence of early termination of a pregnancy may be related to differences in virulence between viruses, acute severe systemic illness of the pregnant female, or acute fetal death of the majority of the fetuses. Based on the observations of this study, previous findings, and results reported by other groups, 4,20 we believe that PCV2-associated reproductive failure is more insidious and causes gradual embryonic loss or fetal death. Thus, PCV2-associated reproductive failure should be considered in situations in which increased numbers of stillborn and/or mummified fetuses in term litters are observed.
When fetuses were inoculated intrauterinally with PCV2 at different stages of gestation, it was found that transmission between fetuses did not occur but that the virus was able to replicate in fetal tissues, especially in the myocardium. 35 In contrast, the results of this study indicated that in utero transmission between fetuses occurred. This is supported by the variable crown-to-rump lengths of the mummified fetuses, indicating death of individual fetuses anytime between 42 and 105 days of gestation, similar to what occurs with PPV infection. 18 The long PCV2 viremia noted in some of the sows may have contributed to transplacental infection at different times during gestation; however, 2/3 sows did not have detectable PCV2 DNA in serum samples by 49 days of gestation, although live-born piglets in the corresponding litters had anti-PCV2 antibodies, indicating infection after the onset of fetal immunocompetence (∼70 days).
After artificial insemination with PCV2-spiked semen in the current study, gross lesions of chronic myocardial dysfunction and cardiac dilatation were present in the 2 stillborn fetuses in group 3. These results are similar to those of previously described field cases. 4,22 The observed pulmonary, mesocolonic, and perirenal edema can be attributed to the associated heart failure or related to PCV2-associated vasculitis. 23 Microscopic evaluation of fetal myocardial tissues showed extensive necrosis, lymphocytic inflammation, edema, mineralization, and fibrosis similar to what have been previously reported. 4,22,40 Occasionally, mummified fetuses in this study had abundant myocardial fibrosis and extensive mineralization. Other groups have reported depletion of lymphocytes from splenic periarterial sheaths in live-born and stillborn fetuses, 4 which we also observed and additionally demonstrated the presence of macrophages and multinucleated giant cells, suggestive of chronic infection.
In this study, and similar to previous studies, abundant PCV2 antigen was detected in myocardial tissue. Interestingly, in PCV2-infected live-born, stillborn, and mummified fetuses in which antigen was observed by IHC, the myocardium always contained antigen while lymphoid tissue (tonsil) contained detectable PCV2 antigen only sporadically (Table 3). This reinforces the idea that myocardial tissue is the best tissue for accurate diagnosis of PCV2-associated fetal infection.
Summary of findings in expelled piglets and fetuses at parturition from group 3 sows artificially inseminated with porcine circovirus type 2b (PCV2b)-spiked extended semen.
∗ Estimation of fetal death based on crown-to-rump length.
† Fetal crown-to-rump length.
‡ PCR = polymerase chain reaction; NA = not applicable.
At parturition, all presuckle serum samples from group 1 piglets were negative for PCV2 DNA and specific antibodies. In contrast, all presuckle serum samples collected from normal-appearing live-born piglets born in group 3 contained PCV2 DNA. In a previous report, when the timing of fetal infection was manipulated by direct fetal inoculation after 75 days of gestation, PCV2 antibodies were generally detected in presuckle serum samples and the fetuses were nonviremic. 35 Alternatively, if fetuses were infected at 57 days of gestation, anti-PCV2 antibodies were not detected 21 DPI, but fetal serum samples were positive for PCV2 DNA. 35 Interestingly, approximately 50% of the group 3 live-born piglets had detectable anti-PCV2 antibodies. This indicates that fetal infection in these piglets occurred after the fetuses reached immunocompetence (∼70 days of gestation).
The remaining live-born piglets without measurable amounts of PCV2 antibodies may have been infected just prior to parturition and therefore did not have time to develop PCV2 antibodies, or they may have been infected prior to immunocompetence. Interestingly, of the live-born piglets in group 3 that did not have anti-PCV2 antibodies, all had mild to marked amounts of PCV2 antigen in heart detected by IHC. Previous reports suggest that PCV2 replication in myocardial tissue diminishes as fetuses near full development. 34 With marked PCV2 antigen in a portion of the seronegative live-born piglets, it can be speculated that these animals may have been chronically infected. The exact mechanism of PCV2 persistent (chronic) infection is unknown, but persistent infection is supported by previous reports detecting PCV2 viremia of experimentally infected animals at 90 DPI, 15 PCV2 DNA in tissues at 125 DPI, 3 and detectable viremia in naturally infected pigs for 22 weeks. 32
In conclusion, intrauterine placement of PCV2 was capable of causing both viremia in the sow and fetal infection but did not result in abortion. The role of PCV2 in early embryonic death resulting in resorption and irregular returns to estrus is less clear based on the results. However, semen transmission should be considered as a means for transmission of PCV2, potentially resulting in intrauterine infection of piglets and perhaps leading to persistent infection. Furthermore, PCV2 infection should be considered if increased numbers of stillborn and mummified fetuses are expelled at parturition. Myocardium is preferred for identification of PCV2 antigen and DNA in fetuses.
Footnotes
Acknowledgements
This project was supported by Pork Check Off Dollars from the National Pork Board and by Boehringer Ingelheim Vetmedica, Inc. The authors thank Joseph Bender, Paul Thomas, and Troy Worth for their assistance with animal care and collection of samples throughout the study. We also thank the Iowa State University Laboratory Animal Resources staff for daily animal care.
