Abstract
A newly observed syndrome characterized by progressive weight loss, pallor, and high mortality was reported in many swine herds across Thailand from February through May 2007. To determine the cause of the new syndrome, 5 pigs, 4–8 weeks of age, were submitted to the diagnostic laboratory for necropsy examination. All 5 pigs were underweight, with pallor and rough coats. The kidneys of all 5 pigs were yellowish and slightly swollen, with precipitation of crystalline material on the cut surface. Histologically, epithelial degeneration and necrosis were evident in proximal and distal tubules and collecting ducts. Round, yellow-brown crystals with radiating striations were diffusely distributed through the lumen of proximal and distal tubules and collecting ducts. Blood urea nitrogen and creatinine levels were elevated. Melamine and analogs, including cyanuric acid, were detected by gas chromatography mass spectrometry. The evidence reported here suggests that melamine- and cyanuric acid-adulterated feed caused renal failure in these pigs.
Introduction
Melamine, or 1,3,5-triazine-2,4,6-triamine, is used in the production of plastic kitchen items and as a fertilizer in some countries. Melamine contains a high nitrogen content that, when added to foods, falsely elevates the protein concentration because the added nitrogen is nonprotein nitrogen. Ruminant animals can synthesize protein from nonprotein nitrogen; however, monogastric animals cannot efficiently use nonprotein nitrogen to synthesize protein.12 Pet foods that were adulterated with melamine and its analogs have been reported to cause serious illness and death due to renal failure in dogs and cats in North America1, 4–8, 13 and dogs in Korea.14
A syndrome characterized by failure to gain weight normally, slight pallor, and increased mortality was reported from February through May 2007 in many swine herds across Thailand. Mortality began as early as 2 weeks after weaning (typically performed at approximately 24 days of age) and approached 100% within 2 months. Some pigs had no clinical signs of illness. The objective of this investigation was to determine the cause of this newly observed syndrome.
Materials and Methods
Farm and pigs
The affected swine farm had a 2-site operation with an inventory of 700 sows. The sow herd produced 250–300 weaned pigs on a weekly basis that were moved to nearby wean-to-finish facilities on contract farms. Starter feed was mixed on the farm and provided to pigs during the nursery period. Rice protein concentrate (RPC) had been included in the starter diet since January, 2007.
Pathologic examination
Necropsy was performed on 10 dead pigs on the affected farm; 5 live pigs at 4, 6, or 8 weeks of age from the affected herd were submitted to the diagnostic laboratory for necropsy examination. Tissues from the liver, urinary bladder, and kidneys were collected and fixed in 10% buffered formalin. Urinary bladder was sampled because of gross findings in the kidney. Liver was sampled because toxicosis was suspected. Sections from fixed, paraffin-embedded tissues were stained with hematoxylin and eosin for histopathologic examination.
Blood analysis
Blood samples were collected from pigs into ethylenediaminetetraacetic acid–coated tubes and analyzed for blood urea nitrogen and creatinine levels using an autoanalyzer (BT2000, Infocus, USA).
Analyses of melamine and analogs
Renal tissue from one pig was examined for the presence of melamine. The starter feed and RPC were examined for the presence of melamine and related chemicals, including cyanuric acid, ammeline, and ammelide, by gas chromatography mass spectrometry as described by the U.S. Food and Drug Administration Center for Veterinary Medicine (http://www.fda.gov/cvm/GCMSMelamine.htm). Briefly, 0.5 g of sample was extracted using a mixture of acetonitrile/water/diethylamine, and the supernatant was filtered through a 0.45-μm nylon filter. The filtrate was dried under a gentle stream of nitrogen at 70°C. The dry sample was derivatized to form trimethylsilyl derivatives and subsequently analyzed by gas chromatography mass spectrometry.
Results
A total of 4,000 weaned pigs died from February through May 2007. Clinical abnormalities and mortality varied among groups of pigs. The mortality started as early as 2 weeks after weaning and approached 100% within 1–2 months, depending on the group. Some affected pigs had clinical signs of pale skin and decreased weight gain, but death with no history of clinical signs was reported more often. The differential diagnosis included porcine respiratory disease complex, postweaning multisystemic wasting syndrome (porcine circovirus–associated disease), and toxicosis.
Pathologic examination
The body weights of all 5 pigs presented for necropsy at the diagnostic laboratory were lower than normal for their age. The skin was slightly pale with rough hair. The kidneys of all 5 pigs were yellowish, slightly swollen, and glistening, with perirenal edema (Fig. 1). The renal pelvis was dilated; the renal cortex and medulla were atrophied; crystalline precipitates were evident on the cut surface (Fig. 2).
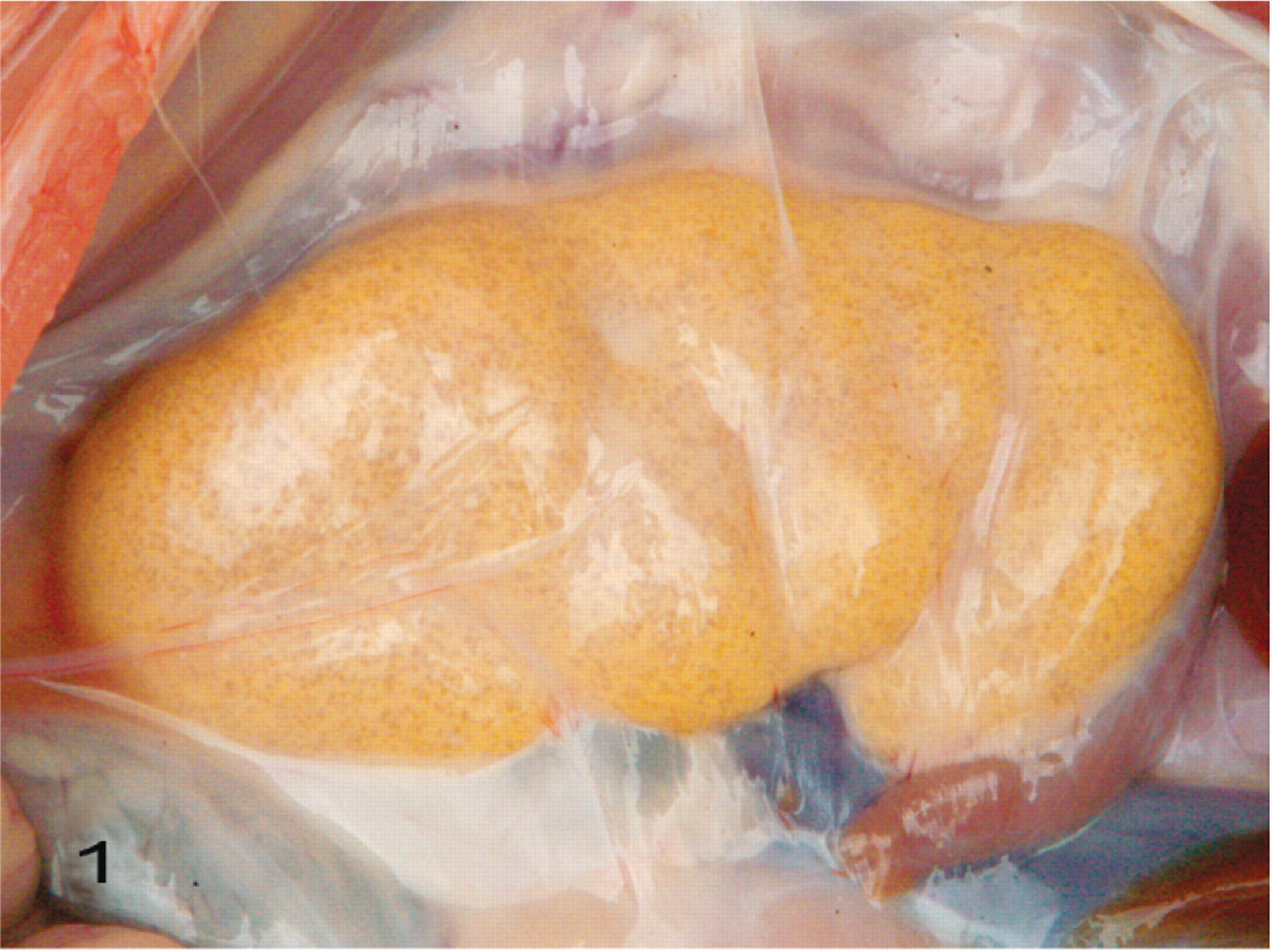
Kidney; pig No. 1. The kidney is yellowish and slightly swollen. Perirenal edema and paucity of perirenal fat are evident.
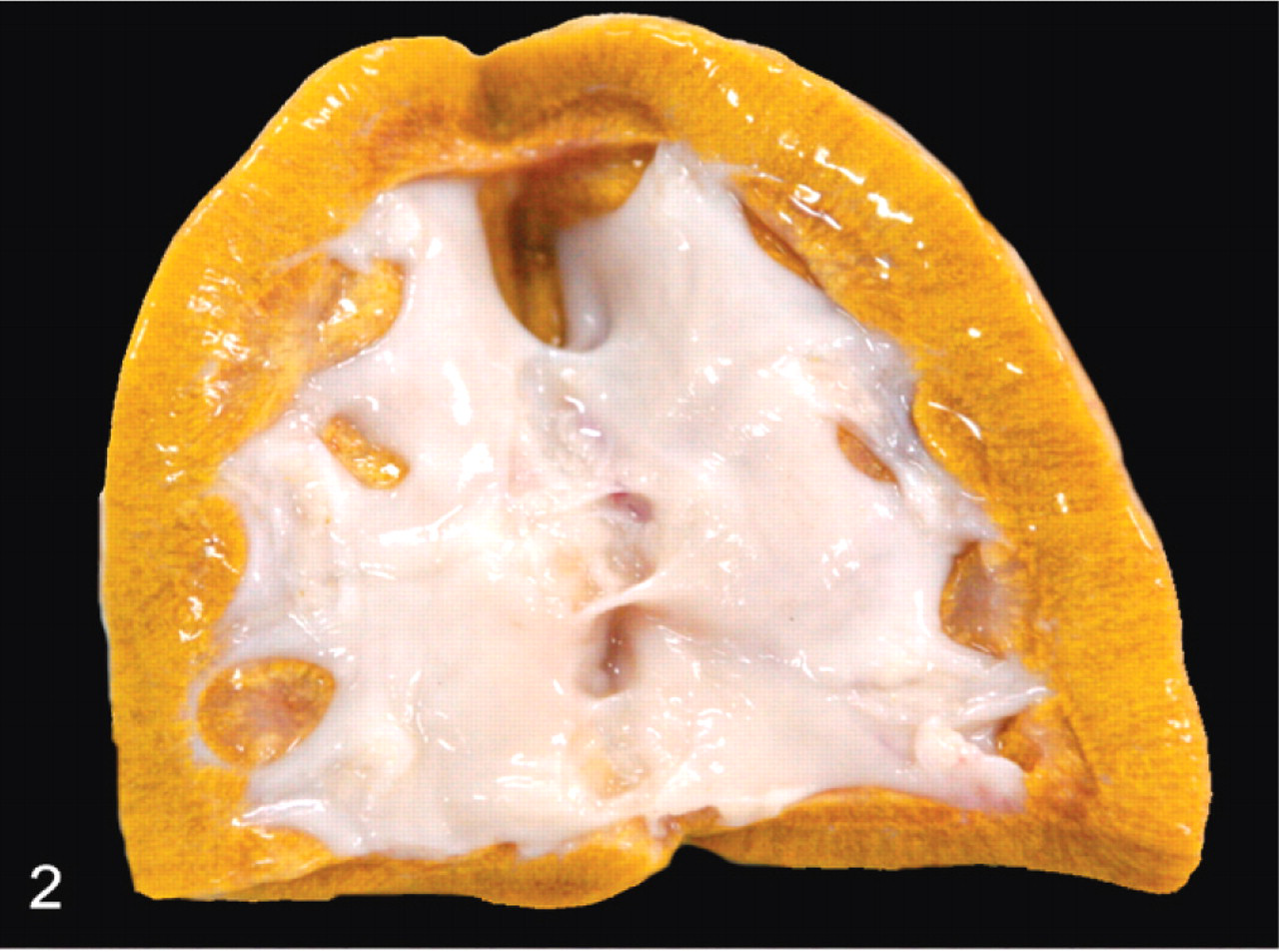
Kidney; pig No. 1. The renal pelvis is dilated. The cortex and medulla are thin with indistinct corticomedullary junction. Renal parenchyma is wet and swollen with yellow mottling due to the crystalline deposits.
Histologic lesions occurred in the kidneys and affected both cortex and medulla (Fig. 3). Crystals were not observed in glomeruli. Crystals were distributed through the rest of the nephron and were more in distal than proximal parts. Intratubular round, yellow-brown crystals with radiating striations were associated with epithelial disruption and necrosis. The proximal and distal renal tubules were dilated and distorted with attenuated epithelium; focally, there were tubular epithelial hypertrophy and proliferation. Glomerular tufts were distorted with widening of Bowman's space. Moderate numbers of intratubular and peritubular neutrophils were observed in distal tubules and collecting ducts. Multinucleated giant cells surrounded crystals in and around distal tubules and collecting ducts. Areas of interstitial fibrosis were infiltrated by lymphocytes and plasma cells (Fig. 4). The urinary bladder and liver were histologically normal.
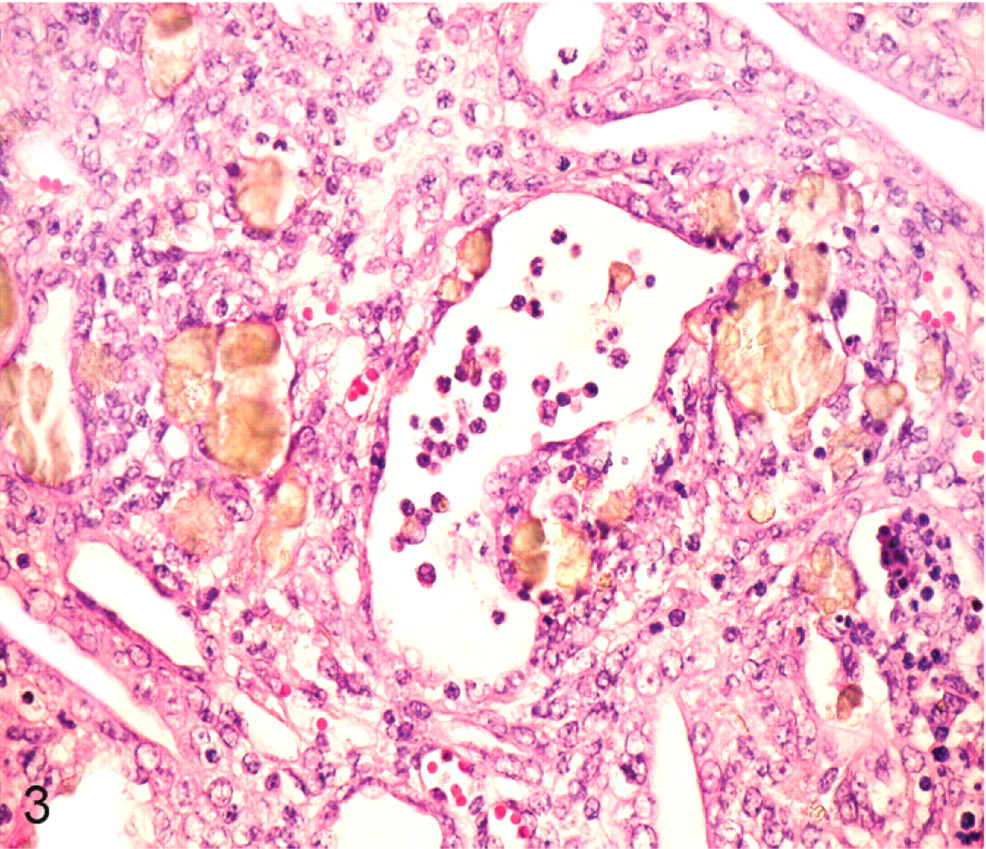
Kidney; pig No. 1. Round, yellow-brown crystals with radiating striations are in the lumen of renal tubules. Affected tubules have epithelial disruption and necrosis. Moderate numbers of peritubular and intratubular neutrophils and macrophages are evident. HE.
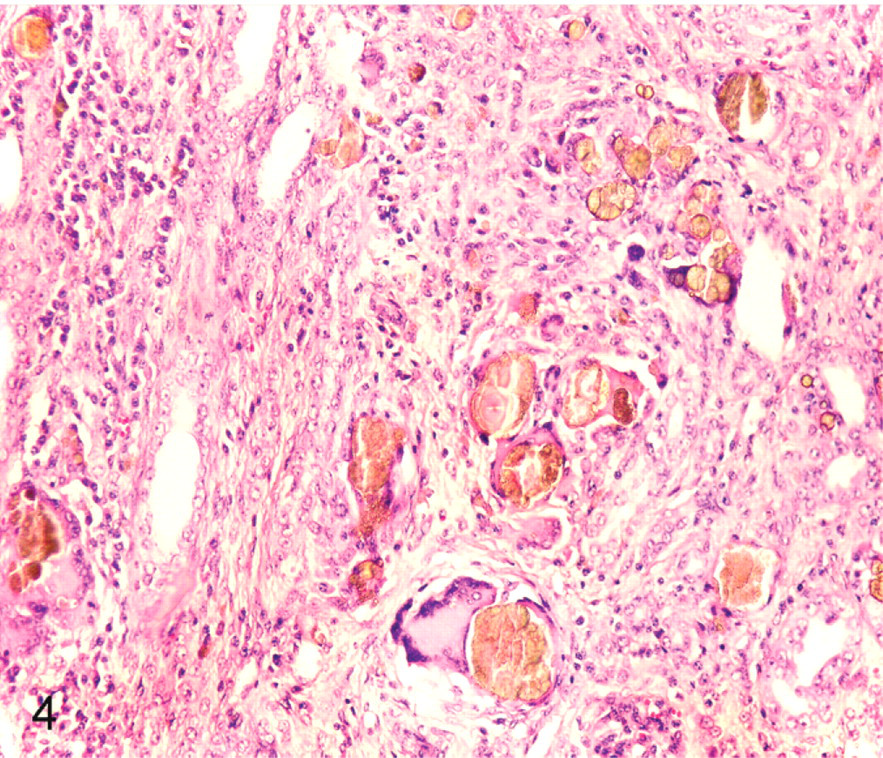
Kidney; pig No. 1. Macrophages and multinucleated giant cells surround the renal crystals. The renal interstitium is expanded by fibrous tissue with infiltration by lymphocytes and plasma cells. HE.
Blood analysis
The blood urea nitrogen concentration in 2 pigs was elevated at 121.2 and 157.9 mg/dl, respectively. Creatinine concentration of both pigs was also elevated at 11.9 and 15.0 mg/dl, respectively. The blood urea nitrogen and creatinine concentrations in the other 3 pigs were within the reference range (8–24 mg/dl and 1–2.7 mg/dl, respectively).
Analyses of melamine and analogs
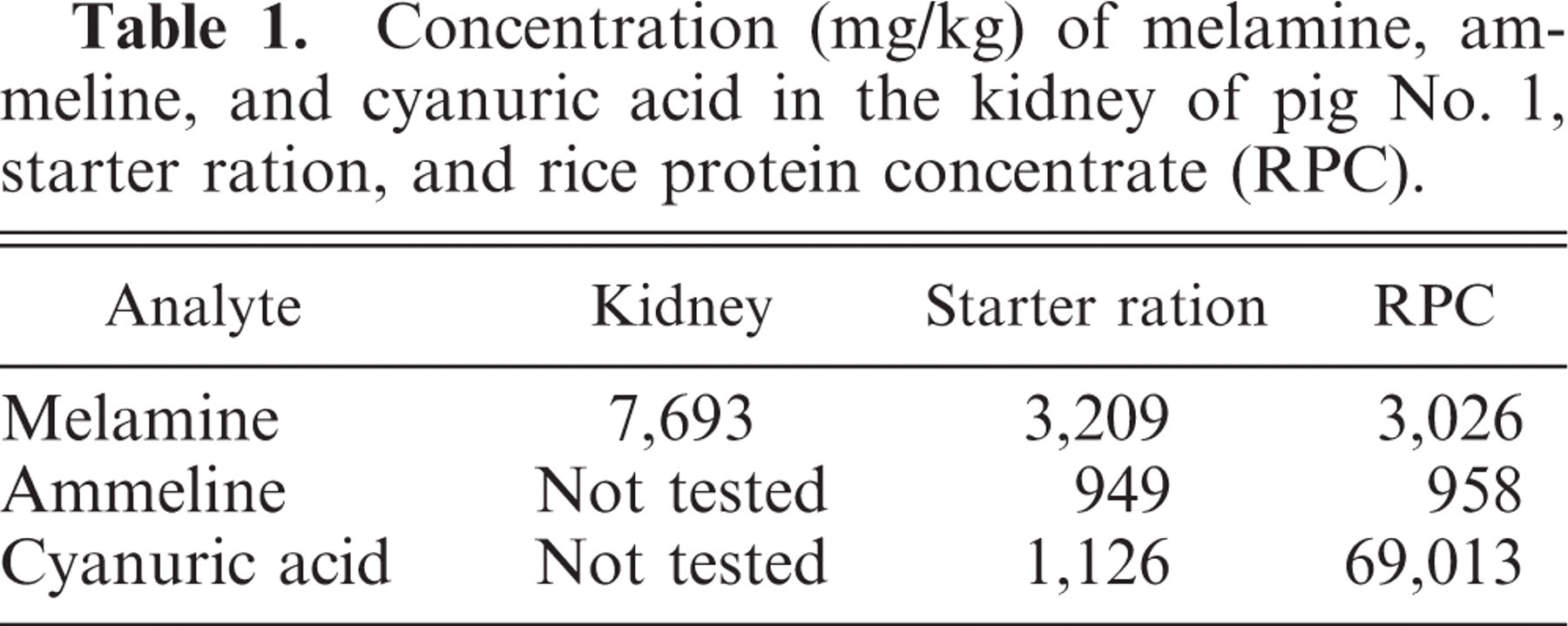
The starter diet contained melamine, ammeline, and cyanuric acid at 3,209, 949, and 1,126 mg/kg, respectively (Table 1). The RPC contained 3,026, 958, and 69,013 mg/kg of melamine, ammeline, and cyanuric acid, respectively. The concentration of melamine in the kidney was 7,692 mg/kg. The concentrations of melamine and ammeline in feed did not differ significantly from those in the RPC, whereas the concentration of cyanuric acid in the RPC was markedly higher than that in the starter diet.
Concentration (mg/kg) of melamine, ammeline, and cyanuric acid in the kidney of pig No. 1, starter ration, and rice protein concentrate (RPC).
Discussion
This may be the first report of melamine- and cyanuric acid–associated nephrotoxicosis in pigs. High concentrations of melamine and cyanuric acid were found in the RPC and starter ration for these pigs. High concentrations of melamine were also found in the kidney, but analogs were not analyzed. Pigs developed clinical abnormalities as early as 2 weeks after beginning the starter ration. Clinical signs included reduced food consumption, decreased weight gain, pale skin, and increased mortality rates. Deaths were observed in both pigs doing poorly and those appearing healthy. Pathologic examination revealed renal tubular degeneration and necrosis with intraluminal crystals that had radiating striations typical of those found in melamine- and cyanuric acid–associated nephrosis. Disease in these pigs was attributed to renal failure.
The RPC was formulated in the starter ration at 10%. Therefore, the melamine concentration in the starter ration theoretically should have been 10% of that in the RPC. Instead, the concentrations of melamine in the starter ration and in the RPC were equivalent. In contrast, the concentration of cyanuric acid in the starter ration was significantly lower than that in the RPC. These findings suggest the possibility of other sources of melamine in the starter ration. Alternatively, the RPC might not have been distributed evenly in the starter ration due to inadequate mixing, which could have biased the sampling technique.
Melamine allegedly has been used in the feed industry for several years as a nonprotein nitrogen source to cause falsely elevated protein concentration in raw feed materials. Grain proteins, including wheat gluten, corn gluten, and RPC, have reportedly been adulterated with melamine and its analogs.8 Studies in ruminants suggest that melamine can cause fatal uremia.10 In other species, the toxicity of melamine has been reported to be low, with few reports of clinically adverse effects. In 1 study, large doses of melamine given to dogs, rabbits, and rats reportedly had no significant clinical effects.9 Another study reported a toxic effect of melamine in rodents and dogs (http://www.cdpr.ca.gov/docs/risk/toxsums/pdfs/1023.pdf). The oral 50% lethal dose in rats was 3,161 mg/kg. Dogs that ingested 1,200 mg/kg/day of melamine for 1 year developed no clinical signs, except crystalluria. The difference between the finding in rats and dogs may have been due to the level and duration of melamine exposure or to presence or absence of the cyanuric acid analog. The dose administered to rats was relatively higher, which could account for the acute deaths. The exposure level in the dogs was lower, but the duration of exposure was longer. Thus, the toxic effect of melamine may depend on the level and duration of exposure.
Other conditions, including dehydration, high uric acid levels in pets with calcium oxalate crystals/uroliths, and/or the presence of other toxic substances (e.g., cyanuric acid), can exacerbate the toxic effect of melamine.3, 13, 14 These factors could enhance melamine crystal formation and precipitation. Crystal formation can be produced in the urine of cats in vitro by adding melamine and cyanuric acid (http://www.labservices.uoguelph.ca/urgent.cfm). When combined, melamine and cyanuric acid precipitate in the urine as crystals that obstruct distal tubules and collecting ducts and cause extensive tubular epithelial damage. Melamine combines with cyanuric acid to form melamine cyanurate, which is more toxic than either melamine or cyanuric acid alone.2, 3, 11, 13, 14 Animals with few crystals can compensate for minor loss of renal function; however, those with numerous crystals experience acute renal failure and (usually) death.
In this study, renal failure was attributed mainly to the physical presence of crystals formed from melamine and cyanuric acid, both of which were found in the ration fed to these pigs. The pigs remaining on the farm that had consumed feed adulterated with melamine and its analogs failed to gain weight when subsequently switched to other commercial feeds. However, groups of weaned pigs that were sent to other facilities and fed a different commercial feed grew normally and did not have clinical signs of renal disease.
Footnotes
Acknowledgements
We thank the owners of the farm and friends who contributed cases. We also thank Drs. Steve Ensley and Erin Strait from Iowa State University, USA, for critical review of the manuscript.
