Abstract
Between November 2003 and September 2006, 300 to 400 45–60-day-old Iberian piglets developed anorexia, polydipsia, and lethargy. Piglets were from 5 different farms in the western part of Spain. Morbidity was between 40% and 60%, and mortality ranged from 20% to 40% of the total population of postweaning piglets. In the 9 piglets in which postmortem examinations were conducted, kidneys were enlarged with yellow foci in the cortex and medulla. Microscopically, these foci were accumulations of crystals within the lumina of dilated distal tubules and collecting ducts, causing flattening of the renal tubular epithelial cells. The crystals displayed a multicolored birefringence under cross-polarized light. The multinucleated giant cells surrounding the crystals, interstitial fibrosis, and nonsuppurative infiltrates indicated a chronic inflammatory response. Toxicologic analysis of fixed kidney tissues from 4 piglets demonstrated the presence of melamine, ammeline, ammelide, and cyanuric acid. Ammelide concentrations were highest, ranging from 39,000 to 92,000 mg/kg, followed by ammeline (20,000–34,000 mg/kg), melamine (9,200–29,000 mg/kg), and cyanuric acid (2,200–9,100 mg/kg). The clinical, histologic, and toxicologic findings in affected piglets were similar to those reported in dogs and cats that died of melamine and melamine analogue-associated renal failure in 2004 and 2007. To the authors' knowledge, this is the first documented report of poisoning due to melamine and its analogues in pigs and demonstrates that contamination of pig feed occurred as early as 2003.
In 2004 and 2007, major outbreaks of renal failure due to melamine and its analogues occurred in thousands of dogs and cats in Asia and North America. 3,4 Pet foods contaminated with melamine and related compounds were confirmed as the cause of this acute renal failure. 3,17 In addition, the 2008 illness and death of children in China has been linked to melamine-contaminated infant formula (Wong E: 2008, Milk scandal pushes China to set limits on melamine. New York Times. Available at http://www.nytimes.com/2008/10/09/world/asia/09milk.html).
Melamine (2,4,6-triamino-1,3,5-triazine) is a chemical used primarily for the production of melamine resin, plastics, glues, and inks. 20 It has also been marketed as a fertilizer because of its high nitrogen content. The use of melamine as a nonprotein nitrogen (NPN) source for cattle is considered unacceptable because its hydrolysis is slower and less complete than that of other nitrogen sources, such as cottonseed meal and urea. 15 Alkaline hydrolysis of melamine can produce 3 melamine derivatives: ammeline, ammelide, and cyanuric acid. 14 Ammeline and ammelide have been evaluated as NPN supplements in ruminants but were found to be inadequate. 11 However, cyanuric acid was considered an adequate NPN source for sheep. 1 In the case of the contaminated pet food, it has been speculated that melamine was added intentionally to raise the apparent protein content of the food as the protein concentration is measured by analyzing total nitrogen content (Barboza D, Barrionuevo A: April 30, 2007, Filler in animal feed is open secret in China. New York Times. Available at: http://www.nytimes.com/2007/04/30/business/worldbusiness/30food.html?ex=1189742400&en=9565d7aa6a8ed9bb&ei=5070).
Separately, melamine and cyanuric acid are relatively nontoxic. The acute toxicity of melamine in rodents has been reported with oral median lethal doses (LD50) of 3,100 mg/kg (male rats) and 3,900 mg/kg (male mice). 13 Chronic oral administration of melamine to mice and rats causes urolithiasis, irritation of urothelium, and bladder neoplasia. 13,16 In sheep, a single dose of melamine at 2.17 g/kg body weight via rumen fistula resulted in increased serum urea nitrogen concentration. 5 When 10 g melamine was administered to each animal daily, 2 sheep developed anorexia and anuria and subsequently died. 5 Subchronic oral administration of sodium cyanurate to rats and mice at 700 mg/kg and 2,200 mg/kg, respectively, caused bladder calculi and associated bladder epithelial hyperplasia but no other adverse effects. 8 Of 3 dogs given 8% monosodium cyanurate in their daily diet, 2 died at 16 and 21 months old, and the third dog was euthanized at the end of the 2-year study. 9 In contrast, dietary monosodium cyanurate given to dogs for 6 months at a concentration of 0.8% in their diet resulted in no ill effects or renal lesions. Cyanuric acid fed daily to sheep for 74 days at approximately 200, 400, and 600 mg/kg body weight did not result in any adverse effects. 10 In cats, separate oral administration of high doses of melamine (181 mg/kg) or cyanuric acid (243 mg/kg) was not toxic, but a combination of both compounds, even at low doses (32 mg/kg each), resulted in crystal formation, obstructive nephropathy, and acute renal failure within 24 hr after exposure. 17 Similar findings have been observed in pigs and fish experimentally fed with these compounds. 18
Limited data are available on the toxicity of ammeline and ammelide. A single oral dose of 100 mg/kg ammeline or ammelide given to rats did not result in adverse effects. 6 Sheep given a single oral dose of ammeline (647–1,887 mg/ kg) or ammelide (696–1,823 mg/kg) had no adverse clinical signs. 11 However, sheep dosed daily with ammeline or a mixture of ammeline and ammelide at a total NPN dosage of 200–300 mg/kg body weight developed anorexia, excessive urination, and diarrhea. Clinical signs appeared approximately 4 weeks after the first dose, and 15 of 30 sheep died between days 39 and 59 of the study. 11
Between November 2003 and June 2006, 300 to 400 45–60-day-old (postweaning) piglets from 5 different farms in Castilla y Leon (in the west of Spain) developed anorexia, depression, lethargy, and polydipsia. Clinical signs began several days after weaning, and morbidity was between 40% and 60%. Initial mortality was estimated between 20% and 40% of the total population of the postweaning group. Piglets that survived the initial illness were weak and had poor growth rates for several months. Because of the poor growth rates, 5–10% of the affected piglets were euthanized in the weeks after the acute toxicosis. Piglets were kept in semiextensive conditions, with approximately 20 animals/ha and ad libitum feeding. Investigation revealed that all 5 affected farms fed an identical protein concentrate. All affected piglets had access to this protein concentrate from birth to weaning. Older pigs that had no access to the protein concentrate failed to develop clinical signs. Other feed components, such as wheat and corn, varied from farm to farm. Once the protein concentrate was removed from the diet, no new cases of toxicosis were reported.
Tissue samples from 9 affected piglets were included in the current study. A complete necropsy was performed on 2 piglets by the Pathology Diagnostic Service of the Veterinary Faculty of León (PDSVFL; Spain). Necropsies of the remaining 7 piglets were performed by the practitioners, and tissue samples from different organs were submitted to the PDSVFL. All tissues were fixed in 10% neutral buffered formalin, processed routinely, embedded in paraffin, sectioned at 3 μm, stained with hematoxylin and eosin, and coverslipped. Selected kidney samples from 2 affected pigs and from a dog with oxalate nephrotoxicosis (used as negative control) were also stained with von Kossa (VK) and oil-red-O 72 hr (ORO 72 hr). 20 The use of both stains permitted the distinction between calcium oxalate and melamine derivative crystals. 20
Renal tissues from 4 piglets that were fixed in formalin and embedded in paraffin were submitted to the California Animal Health and Food Safety Laboratory (Davis, CA) for analysis of melamine, ammeline, ammelide, and cyanuric acid (Table 1). Renal tissue was retrieved from the paraffin blocks by heating and melting the paraffin. The paraffin was subsequently removed from the tissue with 3 changes of 100% xylene, rehydration through graded concentrations (100–70%) of alcohol, and a final wash with flowing tap water. Briefly, melamine cyanurate, melamine, and cyanuric acid were extracted from renal tissue by homogenization in acetonitrile/water/diethylamine. After dilution and sonification, the reconstituted extracts and urine samples were analyzed by liquid chromatography and mass spectrometry (LC/MS) using atmospheric pressure chemical ionization. Cyanuric acid and melamine were analyzed in negative and positive ion modes, respectively. Positive control tissue samples were fortified with crystalline melamine cyanurate or melamine and cyanuric acid. This technique allowed detection of melamine and cyanuric acid at a minimum concentration of 5 μg/g. 7
Dates of pathological evaluation of 9 Iberian piglets with melamine and melamine analogue-associated renal failure, and concentrations of melamine, ammeline, ammelide, and cyanuric acid in kidneys of 4 piglets.*
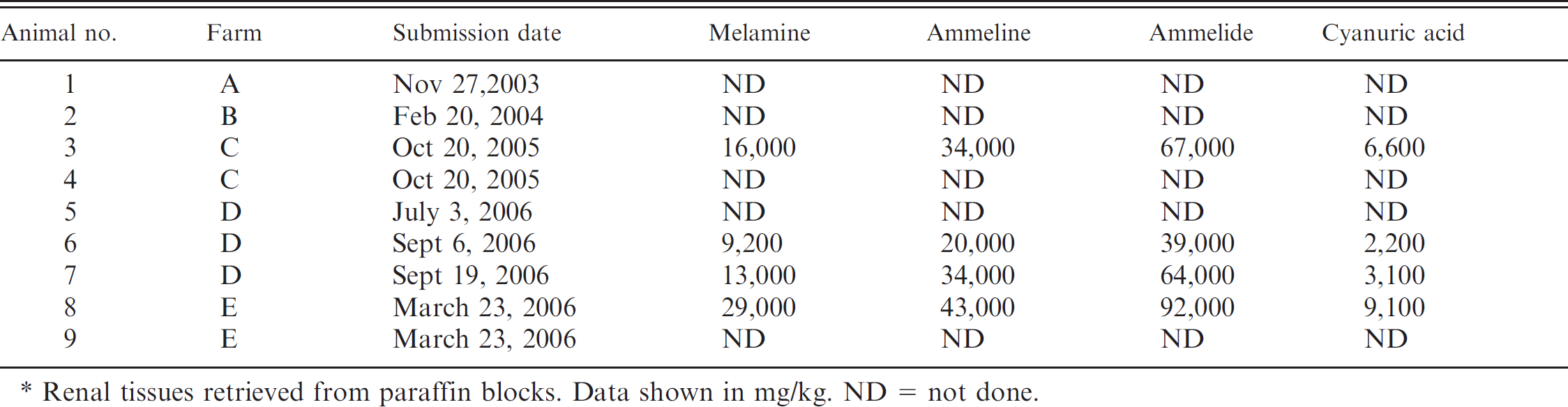
Renal tissues retrieved from paraffin blocks. Data shown in mg/kg. ND = not done.
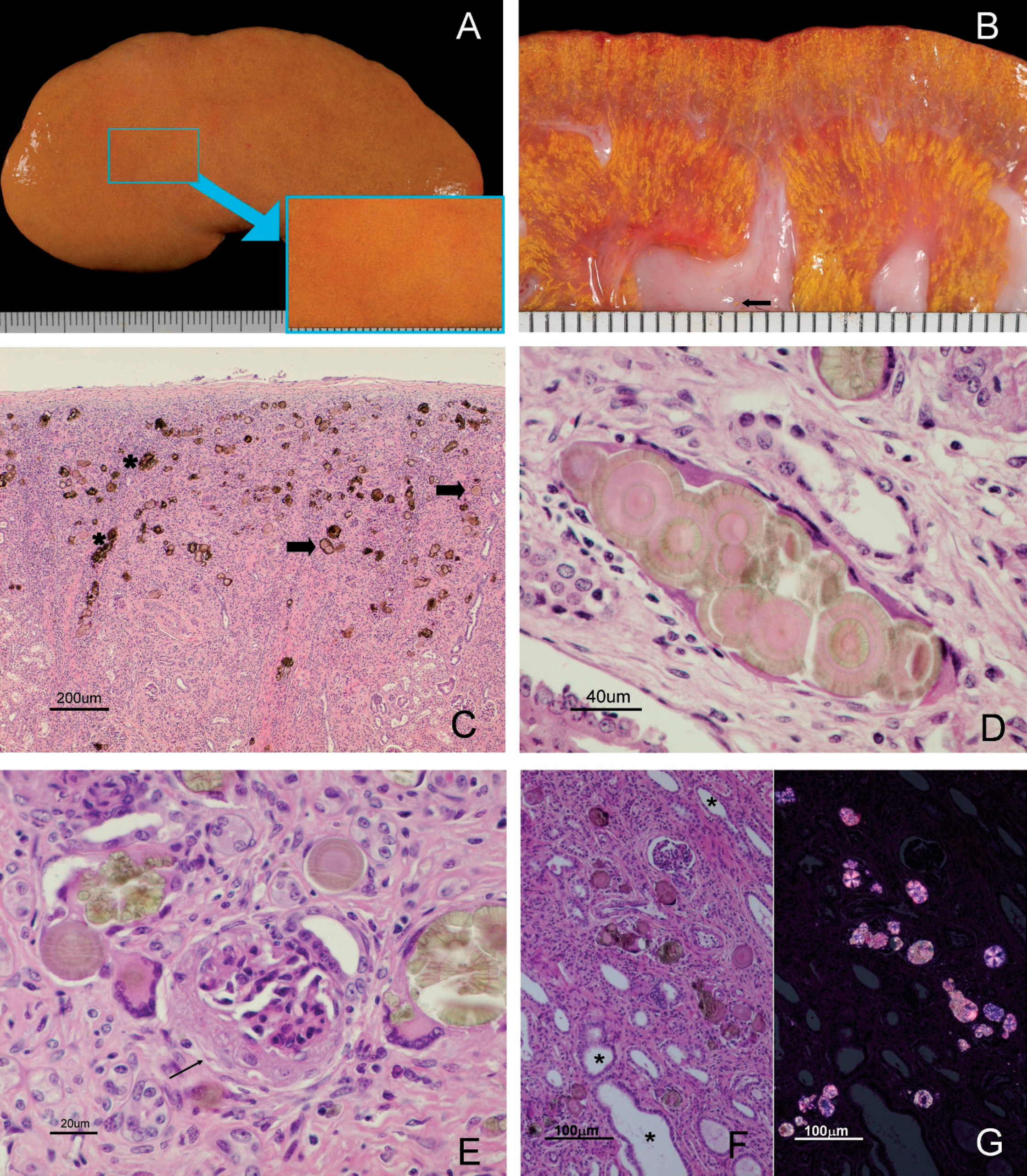
Macroscopic and histopathologic lesions of porcine kidneys as a result of intoxication with melamine analogues. A, kidney, piglet no. 7: orange discoloration of the renal cortex with irregular contours and fine yellow dots (detailed in the box). B, kidney, piglet no. 7, sagittal section: cortex and medulla with yellow material arranged in a radiating pattern. Deposit of this material in the renal pelvis (arrow). C, kidney cortex, piglet no. 3: yellow-pale brown crystals located in the distal convoluted tubules. Severe interstitial nephritis (in the left) and fibrosis (in the middle). Two types of crystals are visible: crystals with an eosinophilic center and a concentric appearance (arrows) and crystals with an irregular form (asterisks). Hematoxylin and eosin (HE). Bar = 200 μm. D, kidney cortex, piglet no. 5: cluster of crystals with eosinophilic centers, concentric laminates, and radial striation. Tubular epithelium is attenuated. HE. Bar = 40 μm. E, kidney cortex, piglet no. 2: multinucleated, foreign body-type giant cells surrounding, or adjacent to, crystals. Sclerosis of Bowman's capsule and atrophy of the glomerulus (arrow). HE. Bar = 20 μm. F, kidney cortex, piglet no. 6: multiple crystals distending distal convoluted tubules (asterisks). Inflammatory cells are present in the adjacent interstitium. HE. Bar = 100 μm. G, Kidney cortex, piglet no. 7, same field as previous image (panel F) seen with polarized light. Crystals are strongly birefringent and some show a radial striation and blue reflection. HE. Bar = 100 μm.
Among the 9 piglets examined, gross abnormalities were similar and limited to the kidneys. Kidneys were moderately enlarged and firm. The capsule could be removed smoothly, revealing a slightly irregular, orange, cortical surface with numerous, multifocal, yellow foci 0.5 mm in diameter (Fig. 1A). Upon sectioning, the fine, yellow foci extended from the cortex to the medulla in thin radial lines that paralleled the medullary rays and were concentrated at the renal crest. Deposits of this fine yellow material were also observed at the renal pelvis (Fig. 1B).
Histologic lesions in all 9 piglets were limited to the kidneys. The most prominent findings were the presence of large (20–60 μm), round crystals in the cortex and medulla. All crystals displayed a multicolored birefringence with cross-polarized light. Crystals were located in the lumina of distal convoluted tubules and collecting ducts. The appearance of the crystals was variable. Some crystals had basophilic round centers surrounded by concentric lamellae with blue radial striations (Figs. 1C, 1D, 1F, 1G), while other crystals were pale green and irregular or striated aggregates (Figs. 1C, 1E–1G). Both types of crystals were positive by the ORO 72 hr technique, but did not stain with the VK method. In contrast, oxalates in the control dog kidney were stained intensely with the VK method but did not stain with ORO 72 hr. Affected tubules were dilated, and the lining epithelium was flattened with only a few intraluminal epithelial cells. No evidence of epithelial cells necrosis, anisokaryosis, or regeneration was detected. Chronic inflammation was associated with the crystals, and the infiltrates were composed of macrophages, lymphocytes, plasma cells, and multinucleated, foreign body-type giant cells (Fig. 1E). Inflammatory cells were inside the lumina of the affected tubules with giant cells surrounding the crystals. More frequently, the renal tubules were disrupted and the crystals were located in the adjacent interstitium and surrounded by the inflammatory infiltrate (Fig 1E). Neutrophils were rarely seen. In all cases, a moderate to severe, diffuse, chronic interstitial nephritis was present. Mononuclear infiltrates and fibrosis were always associated with the presence of crystals, but either cellular infiltration or fibrosis predominated depending on the area of the kidney that was examined (Fig. 1C). Occasionally, there was thickening of Bowman's capsule of some nephrons (Fig. 1E). Melamine, ammeline, ammelide, and cyanuric acid were detected by LC/MS in all 4 kidneys analyzed. Concentrations of ammeline and ammelide were much higher than those of melamine and cyanuric acid, but all of these compounds were present in each kidney (Table 1).
Renal failure with high rates of morbidity and mortality were present in the 5 episodes of toxicosis in Iberian piglets. The clinical, pathological, and toxicologic findings in affected piglets were similar to those reported in previous cases of melamine and cyanuric acid toxicosis in pets that had been reported in 2004 and 2007. 3,4 In addition, the pathological findings were similar to those described in a pig experimentally dosed with melamine and cyanuric acid. 18
The diagnosis of toxicosis was based on the presence of characteristic crystals in distal tubules and collecting ducts and the subsequent demonstration of ammeline, ammelide, cyanuric acid, and melamine in affected kidneys. The morphology of the crystals in porcine kidneys was identical to that observed in the kidneys of cats and dogs in the 2007 outbreaks, as well as in cats experimentally dosed with melamine and cyanuric acid. 3,4,17 In cats, the coadministration of melamine and cyanuric acid resulted in characteristic crystal formation and nephropathy. 17 The fact that melamine and cyanuric acid combined rapidly into a lattice structure via hydrogen bonds21 was considered a major driving force in crystal formation.
In the present report, the kidneys of Iberian pigs with toxicosis contained much higher concentrations of ammeline and ammelide than melamine and cyanuric acid. This is in sharp contrast to the findings in dogs and cats affected in 2007, where kidneys contained much higher concentrations of melamine and cyanuric acid and only low concentrations of ammeline and ammelide. Because of the similar chemical structure of melamine analogues, it is possible that ammeline and ammelide can substitute for melamine and cyanuric acid in the formation of crystals. 6
Crystals described in sheep dosed with ammeline and ammelide11 appeared identical to the renal crystals of the piglets in the present report. However, kidneys of affected sheep were not analyzed for concentrations of melamine and its analogues, and these crystals may have consisted of metabolites of ammeline and ammelide. There are limited data with regard to melamine or cyanuric acid kinetics or metabolism after ingestion. In rats, these compounds are not considered to be metabolized, 12,19 but no kinetic data exist for pigs. It is possible that melamine and cyanuric acid were the primary compounds in the contaminated pig feed and were subsequently metabolized to ammeline and ammelide, which might have contributed to crystal formation in the kidneys. 6 Unfortunately, the protein concentrate implicated in all 5 outbreaks of toxicosis was not available for analysis, so the definitive contaminants could not be identified. From all information available, ammeline and ammelide probably were the major contaminants of the implicated protein concentrate.
The characteristics of the renal lesions in the affected piglets were consistent with the chronic form of melamine and melamine analogue-associated renal failure previously reported. 3,4,17,20 However, some differences exist between the lesions observed in these piglets and those reported in cats and dogs, most notably a higher number of multinucleated giant cells associated with crystals inside the tubular lumina and within the interstitium of affected piglets. The relatively mild damage to the renal tubular epithelial cells in piglets also differs from histologic lesions of dogs and cats. 3,17 These differences may be a result of variations in the concentrations of melamine and its analogues in affected feed. Alternatively, species and age differences may contribute to these differences. In the 2007 intoxications, cats were considered more susceptible to melamine and cyanuric acid than dogs, perhaps because of physiologic differences in tubular function. 3
Neither of the 2 pigs that underwent a full necropsy had lesions associated with uremia, which were common findings in pets. 3 Nevertheless, a significant number of pets from the 2007 outbreaks had laboratory evidence of uremia without extrarenal lesions. Unfortunately, clinical pathology data related to renal function were not available for the affected pigs. Thus, the presence and severity of azotemia could not be evaluated; however, azotemia was likely present based on the renal lesions. Death of the piglets was attributed to renal damage caused by renal tubular obstruction as opposed to a direct toxic effect of the ingested compounds or a morphologic alteration of the tubular epithelial cells. 18
The confirmed exposure of pigs to melamine and melamine analogues emphasizes the potential health risk to humans of consuming contaminated meat. During the 2007 outbreak in pets in the United States, it was discovered that melamine-contaminated pet food waste was incorporated at very low levels into swine and poultry rations, which were subsequently consumed by approximately 56,000 swine and 80,000 chickens. Melamine appears to be cleared rapidly by renal filtration in pigs 2 ; however, melamine was still detected in muscle tissue of hogs that ingested approximately 70 mg/kg melamine in their food but never developed clinical signs (U.S. Department of Agriculture [USDA]: 2007, Interim melamine and analogues safety/risk assessment. Available at: http://www.cfsan.fda.gov/dms/melamra.html). The safety and risk assessment of melamine and its derivatives to human health is still ongoing (USDA: 2007, Interim melamine and analogues), but results of an interim risk assessment indicate that melamine residues in edible tissues of animals are unlikely to pose a human health risk. However, there are no data on the risk to humans if food is contaminated with a combination of melamine and cyanuric acid. In addition, the findings of the current study suggest that ingestion of a combination of ammeline and ammelide also requires a careful risk evaluation. Furthermore, concentrations of melamine and melamine analogues in edible tissues of animals that develop renal disease but subsequently recover have not been evaluated. In Spain, most Iberian pigs are slaughtered at approximately 1 year of age with a body weight of 120–150 kg. However, it is also common in some areas of Spain to consume 20-day-old piglets with a body weight of 5–10 kg. Thus, evaluation of feed for all age groups is necessary to prevent exposure to melamine and melamine analogues.
The current study suggests that melamine and melamine analogue-associated renal failure occurred in pigs as early as 2003, well before the confirmed intoxications of dogs and cats in 2004 and 2007. To the authors' knowledge, this is the first documented natural occurrence of toxicosis in livestock. In Spain, no other cases of melamine-associated toxicosis have been reported in pets or livestock. It is particularly troublesome that the 5 episodes of confirmed toxicosis occurred over a period of 3 years, between November 2003 and September 2006. This indicates continuous contamination with melamine and melamine analogues in the feed supply. Although the source of melamine and cyanuric acid responsible for the 2007 outbreak has been identified as vegetable protein concentrates imported from China, the source for the 5 outbreaks in Spain between 2003 and 2006 remains undetermined. It is remarkable that all affected piglets belonged to the Iberian breed whereas no white pigs examined in the PDSVFL during these last few years has shown pathological or clinical evidence of melamine or melamine analogue-associated renal failure. A possible explanation could be that the contaminated protein concentrate was only distributed to farms with Iberian swine. Pigs of this breed are managed in a semiextensive system with slightly different feeding management compared with the intensive pig farms that raise white pigs. Furthermore, all affected piglets were less than 60 days old, suggesting that only the starter ration contained was contaminated. Adult pigs on affected farms lacked clinical signs, suggesting that their rations were not contaminated or that concentrations of melamine or its analogues were too low to produce toxicity.
These outbreaks in piglets emphasize that livestock food supplies remain vulnerable to chemical contamination. Although 5 separate episodes of toxicosis over a 3-year period were documented in the current study, it is likely that many more cases of toxicosis occurred in young pigs but were not diagnosed. Exposure to melamine, cyanuric acid, ammeline, and ammelide should be considered in pigs with renal failure, especially if there is clustering of affected animals with a similar dietary intake.
Acknowledgements. The authors thank Gloria Belver for technical support.
