Abstract
The largest recorded outbreak of highly pathogenic avian influenza virus of the subtype H7N7 occurred in the Netherlands in 2003. We describe the immunohistochemical and histopathologic findings of 3 chickens naturally infected during this outbreak. Influenza virus antigen occurred in endothelial cells and mononuclear cells of all tissues examined and occurred in parenchymal cells of heart, lung, kidney, pancreas, and trachea, often associated with multifocal inflammation and necrosis. These findings are consistent with the acute stage of highly pathogenic avian influenza from other subtypes. In the severely edematous wattle skin, most endothelial cells contained virus antigen, while in all other tissues virus antigen was only detected in a few endothelial cells. Virus histochemistry showed that this H7N7 virus attached to more endothelial cells in wattle skin than in other vascular beds. This might explain, at least partly, the tropism of the virus and the associated severity of lesions in this tissue.
Keywords
An outbreak of highly pathogenic avian influenza A virus (HPAIV) of the subtype H7N7 occurred in one of the most poultry-dense areas of the Netherlands in 2003. Although this outbreak lasted fewer than 3 months, it was the largest outbreak of a HPAIV of the H7 subtype ever recorded. In total, 255 flocks became infected, more than 30 million birds were culled, and the virus was transmitted to 89 people, resulting in 1 death. 3,9 Molecular analysis of virus isolated from the fatal case revealed a lysine instead of a glutamic acid at position 627 of basic polymerase 2. All other viruses isolated from poultry and people during this outbreak did not reveal this E627K mutation in the PB2 gene. 3 Further analysis revealed this mutation as the major determinant of high pathogenicity in a mammalian model. 5
Published reports on the disease in chickens during this H7N7 virus outbreak are confined to the description of clinical signs and gross pathology. The most frequent clinical signs were increased mortality, apathy, coughing, decreased water and food consumption, reduction in vocalization, and the production of pale eggs. 2 The most frequent gross lesions were peritonitis (62%), tracheitis (43%), edema of the neck and wattles (12%), and hemorrhages on the proventriculus (4%). 1 These data on gross pathology and clinical signs are similar to those caused by infection with other HPAIV. However, the virus distribution and associated histologic lesions in chickens that died during this H7N7 virus outbreak have not been described.
Although low pathogenic avian influenza virus (LPAIV) infections are normally restricted to the respiratory and intestinal system in birds, HPAIV are known to cause systemic disease in poultry. Endothelial cells become infected, resulting in altered permeability of the blood vessels and viremia within the first 2 days after infection. The majority of birds die during this acute stage. In those that survive, HPAIV continues to disseminate systemically, infecting many important organs. 10 Previous studies have shown that there might be an association between the number of endothelial cells that are infected and the progression of clinical disease that leads to death. 7 The mechanism underlying the endothelial cell tropism of HPAIV in chickens has not been studied. Interestingly, during the ongoing outbreak of HPAIV H5N1, systemic virus dissemination was also observed in ducks, although experimental studies showed that endothelial cells of the cardiovascular system were not infected. 4 These findings illustrate the lack of knowledge on the pathogenesis of HPAIV in poultry and other avian species.
Our objective was to perform a detailed histopathologic and immunohistochemical analysis on chickens naturally infected during the H7N7 virus outbreak in the Netherlands. Subsequently we wanted to investigate the endothelial cell tropism in different tissues using virus histochemistry.
Three 70-week-old layer hens (hen Nos. 1–3) were submitted for necropsy on March 1, 2003, from a poultry farm, suspected of HPAIV, in Scherpenzeel, the Netherlands. All birds from the flock had similar clinical signs: diarrhea; swollen head, comb, and wattles; respiratory signs; and yawning. During necropsy, brain, cecum, duodenum, heart (hen No. 1 and No. 2), ileum, jejunum, kidney, lung, liver, pancreas, proventriculus, skin of the wattle, spleen, and trachea were collected. Tracheal and cloacal swabs were taken for virologic analysis to confirm infection with HPAIV H7N7 virus (A/chicken/Netherlands/1/03) as described earlier by Fouchier et al. 3 Tissues for histopathologic and immunohistochemical analysis were immediately fixed in 10% neutral-buffered formalin. After 24 hours, tissues were embedded in paraffin and stained with HE. Virus antigen was detected by immunohistochemistry using a monoclonal antibody against influenza A virus nucleoprotein (clone Hb65, ATCC, Middlesex, UK) as primary antibody as described earlier. 8 An IgG2a isotype control for each tissue and a positive control tissue were included in each staining procedure. The attachment pattern of a HPAIV and LPAIV of the H7N7 subtype on endothelial cells of the skin (wattle vs. breast) and the lungs of 3 uninfected 1-year-old layer hens was determined by virus histochemistry as described earlier. 11,12 The viruses used for virus histochemistry were HPAIV H7N7 (A/Netherlands/33/03) (same HA as the virus isolated from the infected chickens 3 ) and a LPAIV H7N7 (A/Mallard/Sweden/100/02 H7N7), which was isolated during bird surveillance and closely related to the HPAIV H7N7. 6 In each staining procedure, a duck colon and the trachea of the chicken being tested were included as positive control tissues.
Gross necropsy showed similar lesions in all 3 hens. The trachea contained red fluid, and the lungs were diffusely heavy and wet. The subcutis of the head, comb, and wattles was thickened by clear gelatinous material. There were multiple hemorrhages on the surface of the cloaca, and the spleen appeared to be swollen.
Histologic lesions were comparable in all 3 hens (Table 1, Figs. 1a–5a). In the respiratory system there was a multifocal moderate heterophilic tracheitis, characterized by infiltration of many heterophils and fewer macrophages, plasma cells, and lymphocytes, with multifocal necrosis, characterized by karyorrhexis, karyopyknosis, and loss of cellular detail. In the lungs, there was a mild multifocal heterophilic interstitial pneumonia, with few infiltrating heterophils and macrophages in the interstitium, and air capillaries with multifocal moderate necrosis and mild interstitial edema. Perivascularly, there were few infiltrating plasma cells, lymphocytes, macrophages, and heterophils. In the lamina propria of the proventriculus, duodenum, jejunum, ileum, and cecum, there were moderate numbers of infiltrating lymphocytes, plasma cells, macrophages, and heterophils, with multifocal small foci of necrosis. In the gut-associated lymphoid tissue (GALT), there was moderate lymphocytic depletion and necrosis. In the liver, there was multifocal acute hepatocellular necrosis and infiltration of few heterophils and macrophages, and multifocal periportal moderate lympho-histiocytic hepatitis. In the kidney, there was multifocal moderate acute tubular necrosis. Hen Nos. 1 and 2 had mild multifocal cardiomyocyte degeneration of the heart. In the cerebrum and less in the cerebellum, there was mild neuronal degeneration, characterized by neuronal chromatolysis and mild focal gliosis. In the wattle, there was severe edema in the dermis with moderate hemorrhage and multifocal moderate heterophilic dermatitis, characterized by infiltration of moderate numbers of heterophils and fewer macrophages; multifocally, endothelial cells were necrotic or hypertrophic. In the spleen, there was a severe depletion of the white pulp with multifocal lymphocytic necrosis and diffuse moderate histiocytic splenitis. In the pancreas, there was multifocal moderate acute necrosis, mild hemorrhage, and infiltration of few heterophils, and multifocal moderate lympho-histiocytic pancreatitis. The peritoneum had a multifocal moderate lympho-histiocytic peritonitis, characterized by multifocal thickening with edema, mild hemorrhage, and infiltration of moderate numbers of lymphocytes, macrophages, plasma cells, and heterophils. Multifocally, there were vitelline depositions.
Summary of virus distribution and pathology in chickens naturally infected with HPAIV H7N7. The scores were similar in all 3 hens. Scores represented in the table are the mean score. Immunohistochemistry was scored based on the number of cells that contained viral antigen (+ few; ++ occasional; +++ many). Histologic lesions were scored for severity (+ mild; ++ moderate; +++ severe).

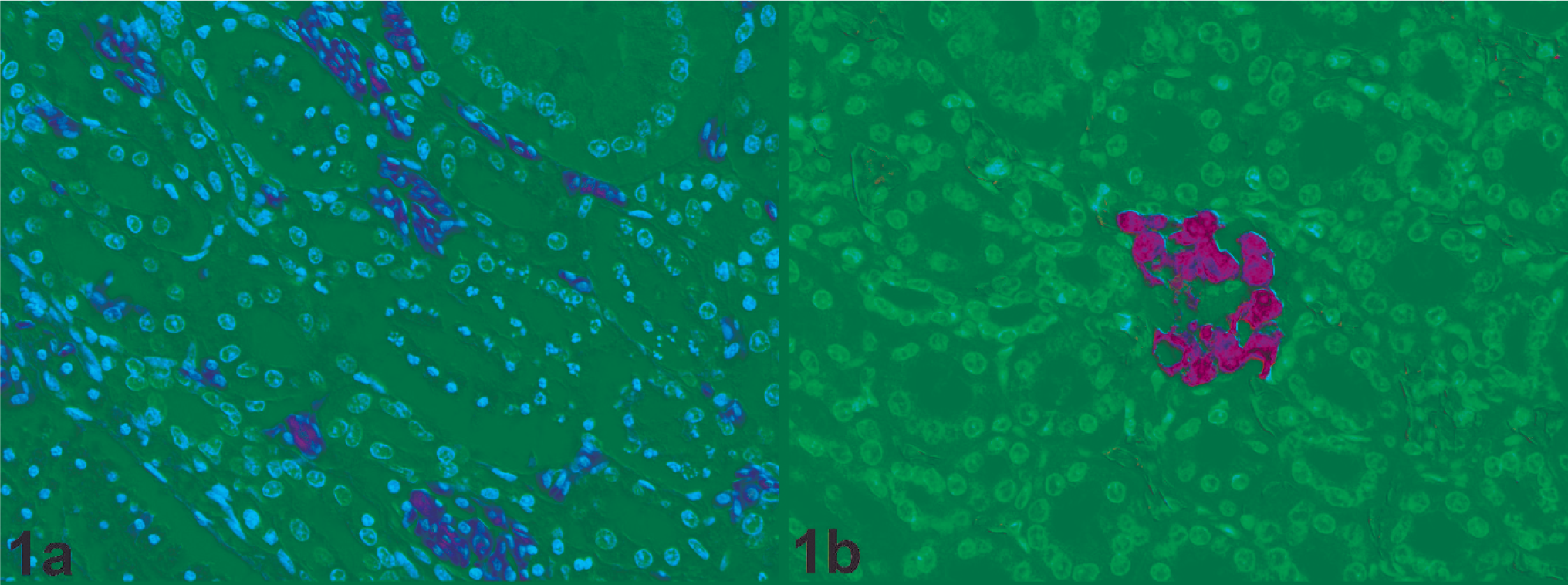

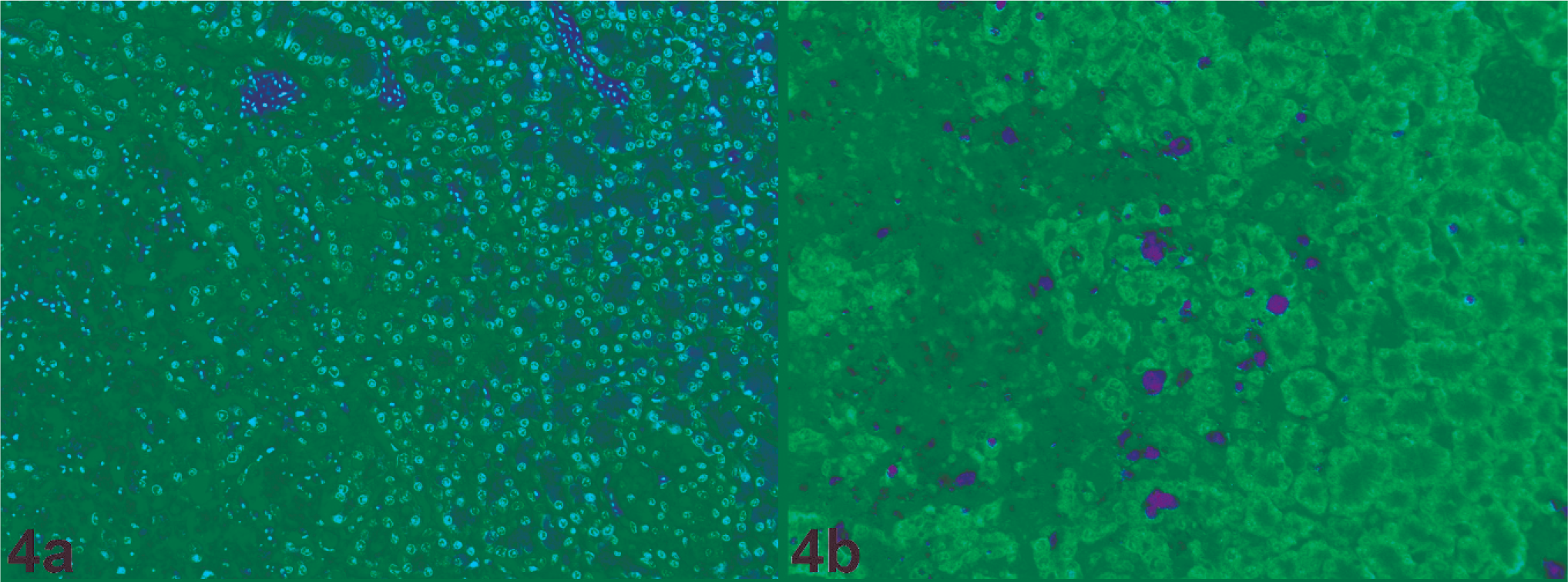
Histopathology (Fig. 2a) and virus distribution (Fig. 2b) in the liver of a chicken naturally infected with HPAIV H7N7 (original magnification 100×). A large necrotic lesion is characterized by loss of nuclei and cellular detail. Around this necrotic lesion, a few scattered individual mononuclear cells that contained virus antigen were found. Inset: Mononuclear cells that contain virus antigen (original magnification 400×).
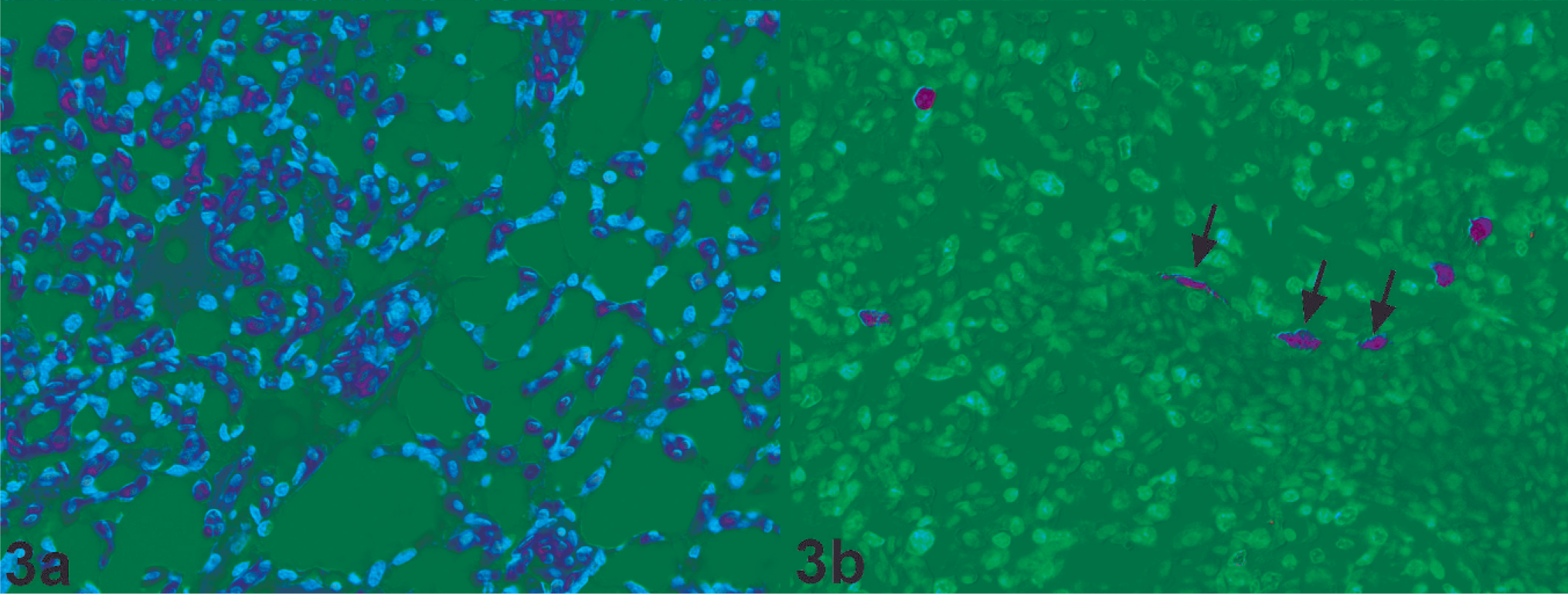
Histopathology (Fig. 3a) and virus distribution (Fig. 3b) in the lung of a chicken naturally infected with HPAIV H7N7 (original magnification 400×). A few air capillaries contain edema fluid (lumen filled with fluid) and scattered individual cells that express virus antigen. By immunohistochemistry, individual endothelial cells contain virus antigen (arrow).
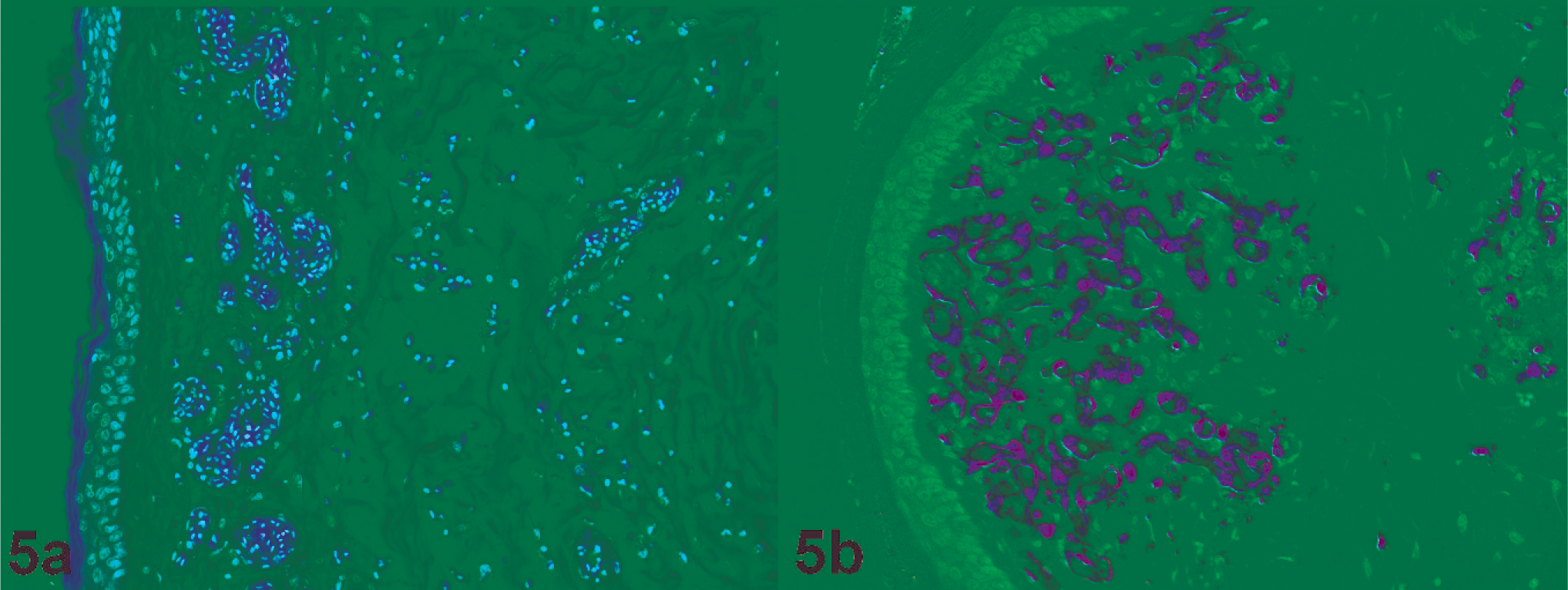
Histopathology (Fig. 5a) and virus distribution (Fig. 5b) in the skin (wattle) of a chicken naturally infected with HPAIV H7N7 (original magnification 200×). Edematous wattle characterized by large amounts of intercellular fluid with moderate hemorrhage is shown. Virus antigen is present in many endothelial cells.
By immunohistochemistry, viral antigen was detected in all tissues with comparable patterns in all 3 hens (Table 1, Figs. 1b–5b). Virus antigen was often associated with histologic lesions, although virus antigen was also present in areas without detectable lesions. Viral antigen was most commonly observed in endothelial and mononuclear cells in all tissues. A remarkable finding was that viral antigen was detected in many endothelial cells in the wattle, while in all other tissues viral antigen was only detected in a few, individual endothelial cells. Parenchymal cells of the heart (cardiomyocytes), kidney (tubular epithelial and glomerular cells), lung (epithelial cells), pancreas (acinar cells), and trachea (epithelial cells) also contained viral antigen. Although there was hepatocellular necrosis, virus antigen was not detected in hepatocytes. In the wattle, keratinocytes of the skin contained viral antigen in 1 focus, and a few cells in the feather pulp of 1 feather follicle contained viral antigen.
By virus histochemistry, attachment of virus was visualized as a red precipitate (Fig. 6). In all 3 hens, there was abundant attachment of the HPAIV H7N7 and LPAIV H7N7 to epithelial cells in the trachea. Both H7N7 viruses attached rarely to endothelial cells in the skin of the wattle, but not to endothelial cells in breast skin, trachea, or lung although capillaries of the same diameter were present. No differences were observed between the attachment pattern of HPAIV H7N7 and that of LPAIV H7N7.
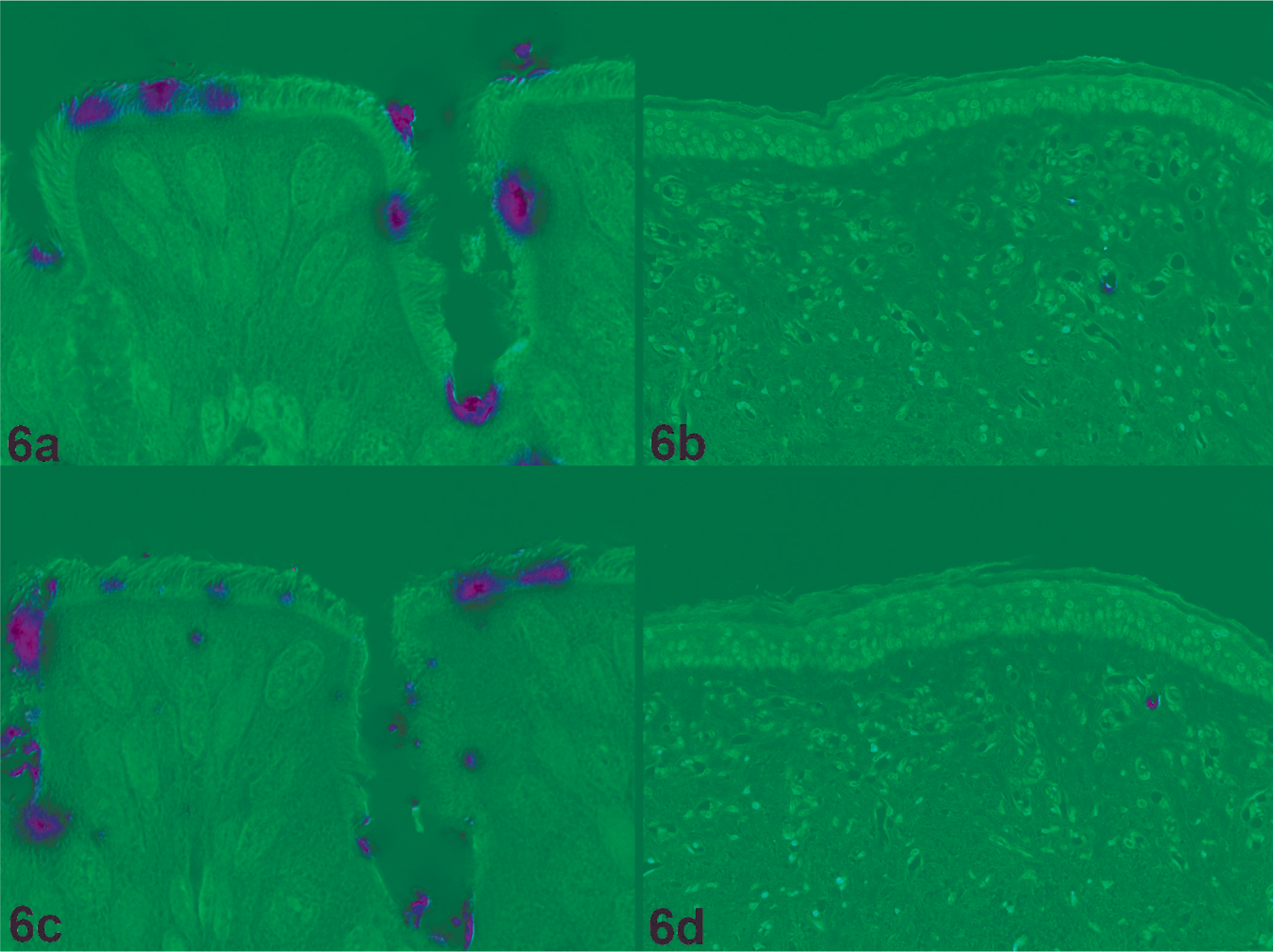
Virus attachment of HPAIV H7N7 (Fig. 6a and Fig. 6b) and a LPAIV H7N7 (Fig. 6c and Fig. 6d). Both viruses attach to the ciliated epithelial cells in the trachea (Fig. 6a and Fig. 6c, original magnification 1,000×). Rarely, viruses attach to vascular endothelial cells in the skin of the wattle (Fig. 6b and Fig. 6d, original magnification 200×).
This histopathologic study, including immunohistochemistry and virus histochemistry, describes for the first time the pathology of natural HPAIV H7N7 infection in chickens from the 2003 outbreak in the Netherlands. Furthermore, for the first time we determined the attachment pattern of HPAIV and LPAIV of the H7N7 subtype in endothelial cells. The distribution of HPAIV H7N7 and associated lesions in naturally infected chickens fits with the acute stage of HPAIV infection in poultry. 10
The attachment of HPAIV H7N7 virus to endothelial cells in the wattle compared with no attachment to endothelial cells in other tissues—although the difference was small—fits with the more abundant expression of influenza virus antigen in wattle endothelium than in endothelium elsewhere. Although virus attachment to host cells is only the first step in the replication cycle of influenza viruses, it is considered an important determinant in tissue tropism and might explain, at least in part, the abundant replication of HPAIV H7N7 in the wattle and the severity of the associated edema.
The lack of difference between the attachment between HPAIV and LPAIV H7N7 indicates that the ability to attach is not the main determinant for the systemic spread of HPAIV H7N7. The presence of a polybasic cleavage site in HPAIV, resulting in the cleavage of HA by enzymes present in all cells, probably contributes more to the systemic infection of HPAIV than the receptor binding properties. 10
In addition to the main findings above, there were 3 additional observations on the pathogenesis of this infection. First, influenza virus antigen was expressed in keratinocytes and in a feather follicle, suggesting that skin and feathers may be an additional source for virus. Influenza virus antigen has been detected before in feather follicles in gallinaceous species 7 and in feather epidermal cells of domestic ducks and geese. 13
Secondly, although foci of necrosis colocalized with the presence of virus antigen in most tissues, this was not the case in the spleen, gut-associated lymphoid tissue, and liver. Either the necrosis in these tissues had another cause; or virus-infected cells in these tissues underwent such a rapid apoptosis or necrosis that insufficient amount of nucleoprotein was produced to be detectable by immunohistochemistry.
Finally, the detection of H7N7 virus in cloacal swabs in the absence of virus antigen in intestinal epithelial cells raises the question of where this virus originated from. Because the intestinal tract was only sampled on 4 different levels, we cannot exclude focal spots of influenza virus replication that were missed during sampling in all 3 hens. Another source for the virus could be the kidney and the pancreas. They both contained virus antigen and drain into the cloaca, the pancreas via the pancreatic duct, and the kidney via the ureter.
In conclusion, distribution of virus antigen and associated lesions in chickens naturally infected during the HPAIV H7N7 outbreak in the Netherlands fits with the acute stage of disease in chickens naturally infected with HPAIV. The presence of more attachment to endothelial cells in the skin of the wattle compared with endothelial cells in other tissues might explain the tropism of this H7N7 virus for endothelial cells in the wattle.
Footnotes
Acknowledgements
We thank the poultry farmer and the Food and Consumer Product Safety Authority for providing specimens, Lonneke Vervelde for providing control tissues of uninfected layer hens, Frank van der Panne for the help with microphotographs, and Peter van Run for excellent technical assistance.
