Abstract
Transmissible spongiform encephalopathies (TSEs) are a group of diseases that result in progressive and invariably fatal neurologic disease in both animals and humans. TSEs are characterized by the accumulation of an abnormal protease-resistant form of the prion protein in the central nervous system. Transmission of infectious TSEs is believed to occur via ingestion of prion protein–contaminated material. This material is also involved in the transmission of bovine spongiform encephalopathy (“mad cow disease”) to humans, which resulted in the variant form of Creutzfeldt-Jakob disease. Abnormal prion protein has been reported in the retina of TSE-affected cattle, but despite these observations, the specific effect of abnormal prion protein on retinal morphology and function has not been assessed. The objective of this study was to identify and characterize potential functional and morphologic abnormalities in the retinas of cattle infected with a bovine-adapted isolate of transmissible mink encephalopathy. We used electroretinography and immunohistochemistry to examine retinas from 10 noninoculated and 5 transmissible mink encephalopathy–inoculated adult Holstein steers. Here we show altered retinal function, as evidenced by prolonged implicit time of the electroretinogram b-wave, in transmissible mink encephalopathy–infected cattle before the onset of clinical illness. We also demonstrate disruption of rod bipolar cell synaptic terminals, indicated by decreased immunoreactivity for the alpha isoform of protein kinase C and vesicular glutamate transporter 1, and activation of Müller glia, as evidenced by increased glial fibrillary acidic protein and glutamine synthetase expression, in the retinas of these cattle at the time of euthanasia due to clinical deterioration. This is the first study to identify both functional and morphologic alterations in the retinas of TSE-infected cattle. Our results support future efforts to focus on the retina for the development of new strategies for the diagnosis of TSEs.
Transmissible spongiform encephalopathies (TSEs) are invariably fatal neurologic diseases and affect a number of mammalian species. Accumulation of an abnormal form of the prion protein (PrPSc) and spongiform change in the central nervous system are characteristic of these diseases. Transmission of most infectious forms of TSEs is believed to occur via ingestion of PrPSc-contaminated material. Natural transmission of bovine spongiform encephalopathy (BSE or “mad cow disease”) to humans is believed to have occurred through the ingestion of contaminated beef products, resulting in the variant form of Creutzfeldt-Jakob disease. 3,5 Interspecies transmission of other TSEs has been accomplished experimentally. Transmissible mink encephalopathy (TME), a TSE that naturally occurs in mink, has been experimentally transmitted to cattle via the intracerebral route, 11,20 with resulting histologic lesions reportedly indistinguishable from those of BSE. 11 Unfortunately, current methods for detecting TSEs in cattle are limited to postmortem analyses, creating a need for antemortem screening and diagnostic tools.
The retina is a thin, highly organized neural tissue lining the posterior aspect of the eye and is responsible for initiating vision by transducing light into neural signals. PrPSc has been demonstrated in the retinas of a number of TSE-affected species, 2,8,10,12,14–16,23,24 including cattle, 11 and accumulates primarily within the plexiform (synaptic) layers of the retina. Currently, the effect of PrPSc accumulation on specific retinal cell types in cattle is unknown. Accumulation of PrPSc in the retina of sheep with scrapie results in disorganization of rod bipolar cell axons and increased glutamine synthetase and glial fibrillary acidic protein expression by Müller glia, 10,21 suggesting that retinal function could be altered in prion-infected livestock. Electroretinography is a noninvasive tool used to evaluate retinal function by measuring the summed response of all retinal cells to a flash of light. This response is typically measured at the cornea and produces the electroretinogram (ERG), which in its most typical form consists of a small initial negative deflection (a-wave) followed by a large positive deflection (b-wave). Classic ERG analysis includes measurements of amplitude and implicit time. The amplitude, measured in microvolts, is the magnitude of each wave, and implicit time, measured in milliseconds, is the time to maximum amplitude of each wave. Reports on the use of electroretinography to evaluate retinal function in TSE-infected animals and humans are limited, 6,7,18 and to date, studies applying this methodology to TSE-infected cattle have not been reported.
This study investigated the effects of TSE infection on bovine retina using electroretinography and immunohistochemistry. Here we report that the electroretinogram b-wave is altered in TME-infected cattle before the appearance of clinical signs of disease and that there are subtle histologic and immunohistochemical differences between the retinas of TME-affected and noninoculated cattle.
Materials and Methods
Animals
Five Holstein steers were inoculated intracerebrally at 9 months of age with brain homogenate prepared from a second-passage TME-affected steer from a prior experiment. 11 TME was used in this study because of its neuropathologic similarities to BSE in cattle and because in vivo cattle experiments with BSE have not yet been approved in the USA. Antemortem testing (electroretinography, see below) was performed at monthly intervals before the onset of clinical signs of disease for all cattle and 1 or 2.5 weeks before euthanasia for 2 of 5 cattle. All cattle developed clinical disease characterized by loss of body condition, locomotor abnormalities, frequent falls, and difficulty rising and were euthanatized when deemed humanely necessary. The 5 steers were euthanatized at 16.8 (2 steers), 16.9, 19.3, and 19.6 months post inoculation. Eyes from 3 noninoculated Holstein cattle (ages 2–24 months) were used as controls for histology and immunohistochemistry. Ten approximately age-matched (16–24 months of age), noninoculated, Holstein steers housed in the same building as the TME-inoculated cattle served as the control cohort for ERG analysis. All animal procedures had the approval of the National Animal Disease Center's Animal Care and Use Committee.
Electroretinography
Holstein steers were used to gather normative (n = 10) and TSE-associated (n = 5) ERG data. ERG data were collected from TME-inoculated steers before the onset of clinical signs of disease at 12.5, 13.5, and 14.5 months post inoculation (5/5) and at 18.5 months post inoculation (1–3 weeks before euthanasia due to clinical deterioration; 2/5). Animals were lightly sedated with 0.02 mg/kg xylazine before auriculopalpebral nerve block with 2% lidocaine and electrode placement. Pupillary dilation was induced with topical administration of 1% tropicamide ophthalmic solution to the cornea. One eye was tested in each animal.
A DTL Plus microfiber electrode (LKC Technologies, Gaithersburg, MD) was placed on the unanesthetized cornea, and subdermal 12-mm, 29-gauge needle electrodes (LKC Technologies) were used as reference and ground. The reference electrode was placed subcutaneously approximately 2 cm caudal to the lateral canthus, and the ground electrode was placed subcutaneously in the region overlying the occipital bone. An EPIC 4000 visual electrodiagnostic testing system (LKC Technologies) with a CMGS-1 Color Mini-Ganzfeld Stimulator (LKC Technologies) was used to conduct the ERG experiments. Cattle were dark adapted for 20 minutes and then underwent a series of 2 scotopic recordings, 10 minutes of light adaptation, and 1 photopic recording.
The b-wave amplitude and implicit time was measured for each ERG. The noninoculated group was compared with the preclinical TME-inoculated group using the Mann-Whitney U-test with a 95% confidence interval. P values < 0.05 were regarded as statistically significant.
Histopathology
Entire globes with a segment of optic nerve were extracted at necropsy, immersed in Bouin's fixative for at least 24 hours, rinsed in tap water, and then immersed in alkaline alcohol. A 5-mm-thick vertical section from the caudal aspect of the globe containing retina and optic nerve were processed by routine histologic methods and embedded into paraffin blocks. Serial 4-µm sections were cut from the retina and stained with HE. Cell density in the ganglion cell layer was determined in 2 control animals and 2 TME-affected animals by identifying sections with optic nerve head moving 1 40× field dorsally and manually counting nuclei within the ganglion cell layer of the subsequent 5 40× fields.
Immunohistochemistry
The distribution of PrPSc and various retinal cell type-specific markers in the retinas of 3 control and 5 TME-affected cattle were examined. Slides were immunolabeled to detect PrPSc as previously described 12 using primary antisera containing monoclonal antibodies F89/160.5 19 and F99/97.6.1 22 each at a concentration of 5 µM/ml. Sections immunolabeled to detect retinal cell type-specific antigens were processed as previously described. 21 Primary antibodies used in this study included the following: rabbit anti-protein kinase C-alpha isoform (Sigma, St. Louis, MO), which in retina specifically labels rod bipolar cells; guinea pig anti-vesicular glutamate transporter 1 (Chemicon International, Inc., Temecula, CA), which labels glutamatergic terminals of photoreceptors and rod bipolar cells; rabbit anti-glutamine synthetase (Sigma), which specifically labels Müller glia in the retina; and rabbit anti-glial fibrillary acidic protein (DakoCytomation, Carpinteria, CA), which labels astrocytes and reactive Müller glia. Secondary antibodies included fluorescein isothiocyanate–conjugated donkey anti-guinea pig IgG (Jackson ImmunoResearch, West Grove, PA) or fluorescein isothiocyanate–conjugated donkey anti-rabbit IgG (Jackson ImmunoResearch). Labeling patterns were imaged with a fluorescence-capable microscope (Nikon Eclipse E800) equipped with a digital camera and prepared using Adobe Photoshop CS Version 8.0 and Macromedia Freehand MX Version 11.0 for the Macintosh.
Results
To evaluate retinal function in TME-infected cattle, we compared ERGs between noninoculated control and TME-inoculated cattle. All TME-infected cattle were displaying some degree of neurologic dysfunction, identified via extensive neurologic examination, at approximately 15 months post inoculation (F. Vargas, personal communication). Representative ERGs from a noninoculated control and a preclinical TME-infected animal are shown in Figures 1 through 3. A statistically significant increase in b-wave implicit time in TME-infected cattle was shown at all preclinical time points under all testing conditions, with the exception of test 2 at the earliest time point (Fig. 4, Table 1). A significant difference in b-wave amplitude was, however, not observed at any preclinical time point. Analysis of b-wave values from 2 of the 5 TME-infected cattle during the clinical stage of disease demonstrated decreased average b-wave amplitude and prolonged implicit time under scotopic conditions relative to controls (Table 1). B-wave amplitude was decreased, but implicit time was not prolonged under photopic conditions.
Summary of mean electroretinogram b-wave amplitude and implicit time data from control (noninoculated) and transmissible mink encephalopathy-inoculated cattle.∗
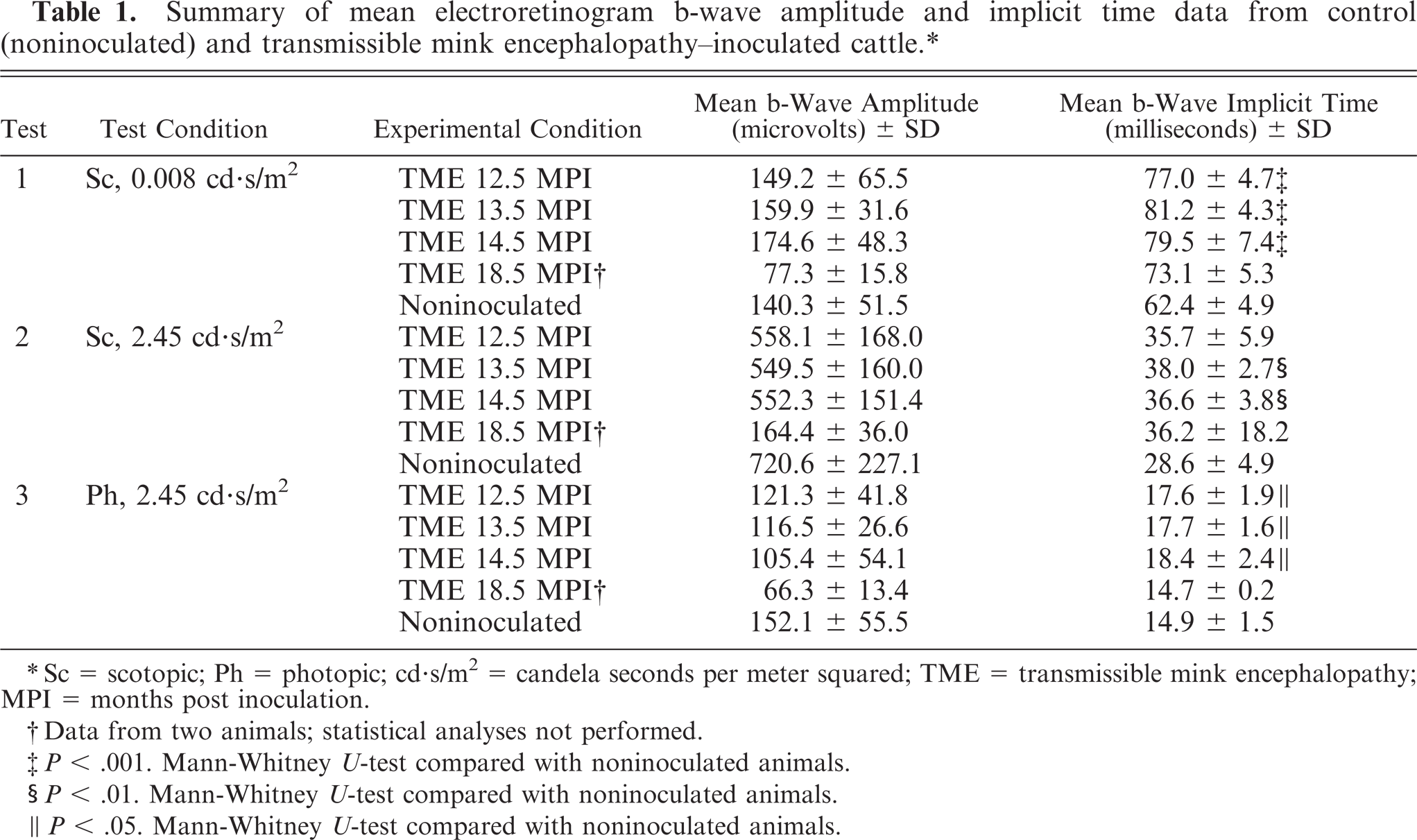
∗ Sc = scotopic; Ph = photopic; cd.s/m2 = candela seconds per meter squared; TME = transmissible mink encephalopathy; MPI = months post inoculation.
† Data from two animals; statistical analyses not performed.
‡ P < .001. Mann-Whitney U-test compared with noninoculated animals.
§ P < .01. Mann-Whitney U-test compared with noninoculated animals.
‖ P < .05. Mann-Whitney U-test compared with noninoculated animals.
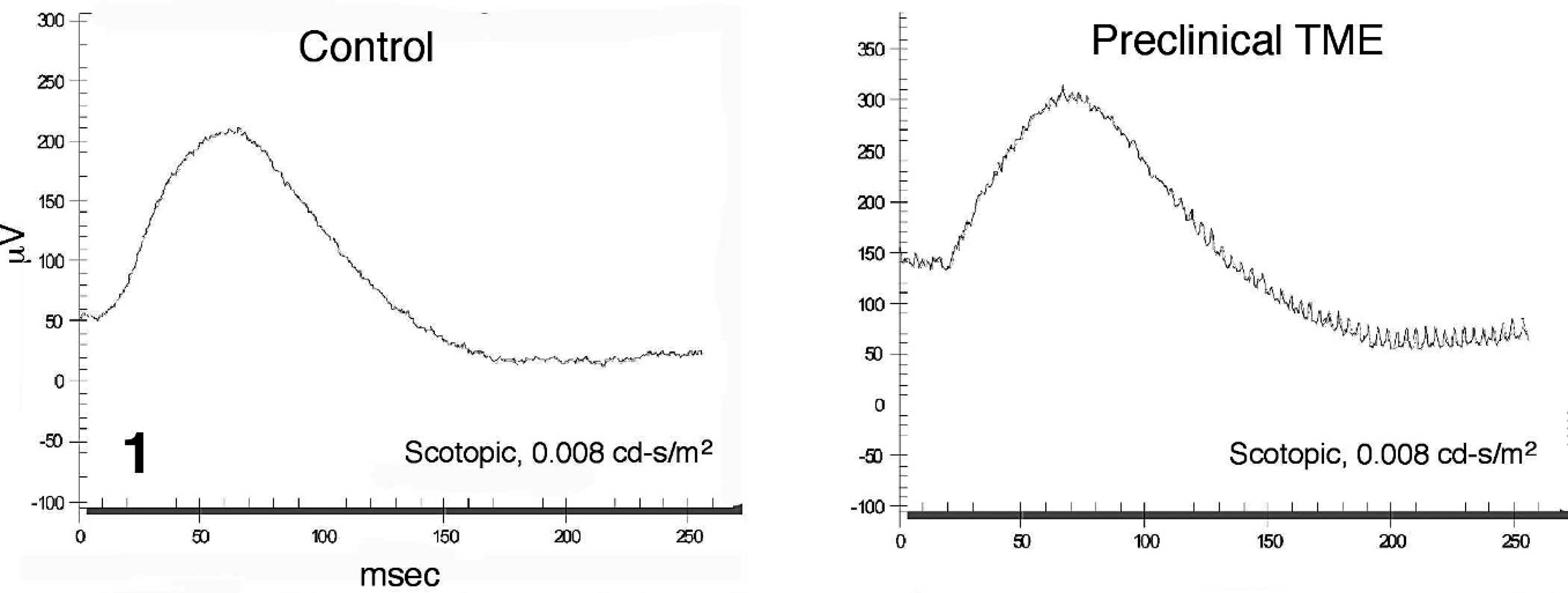
Representative electroretinogram from a noninoculated control (left) and a preclinical transmissible mink encephalopathy–inoculated Holstein steer (right) recorded under dark-adapted testing conditions using a light stimulus intensity of 0.008 cd·s/m2. The b-wave is the large positive deflection.
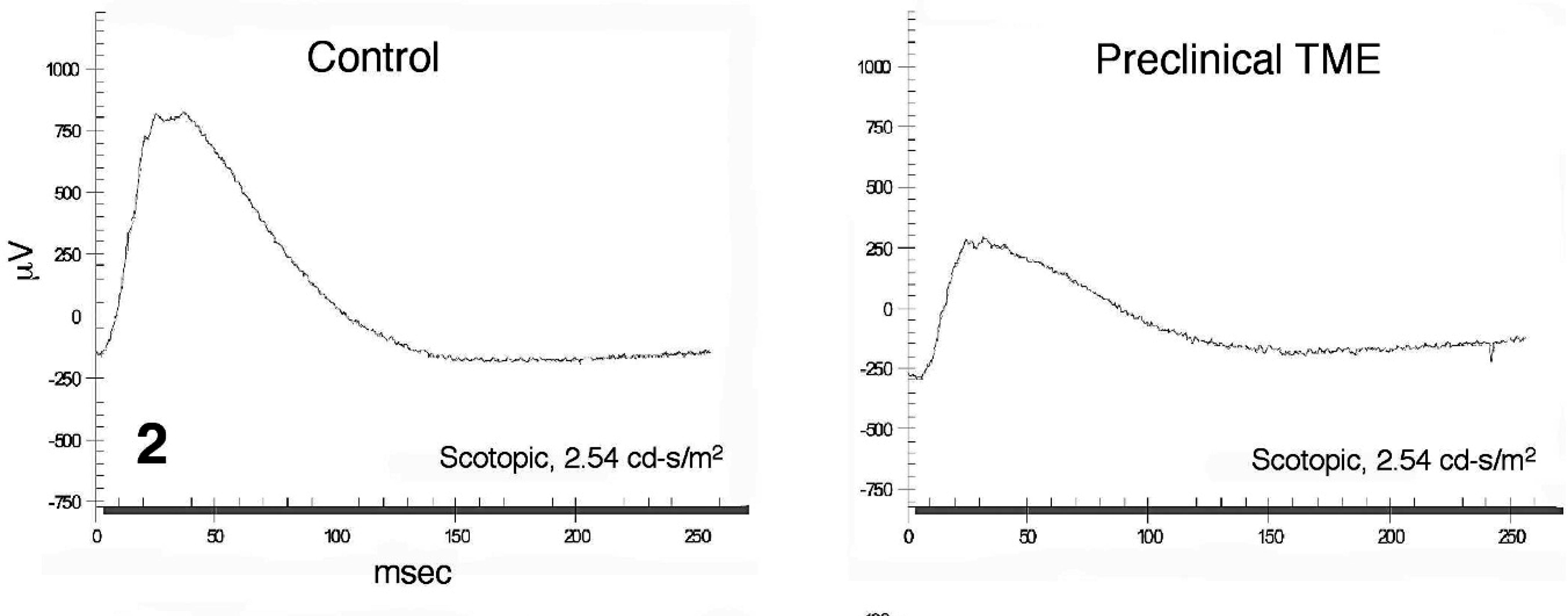
Representative electroretinogram from a control (left) and a preclinical transmissible mink encephalopathy–inoculated Holstein steer (right) recorded under dark-adapted testing conditions using a light stimulus intensity of 2.45 cd·s/m2.
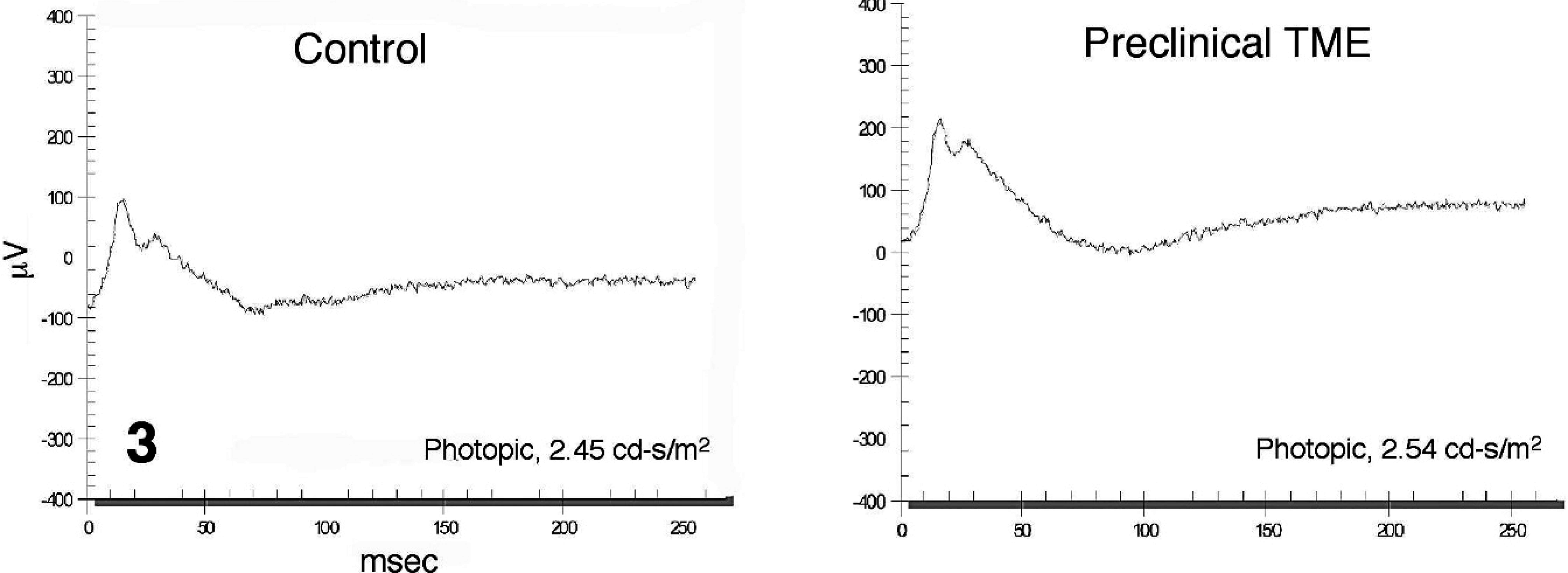
Representative electroretinogram from a control (left) and a preclinical transmissible mink encephalopathy–inoculated Holstein steer (right) recorded under light-adapted testing conditions using a light stimulus intensity of 2.45 cd·s/m2.
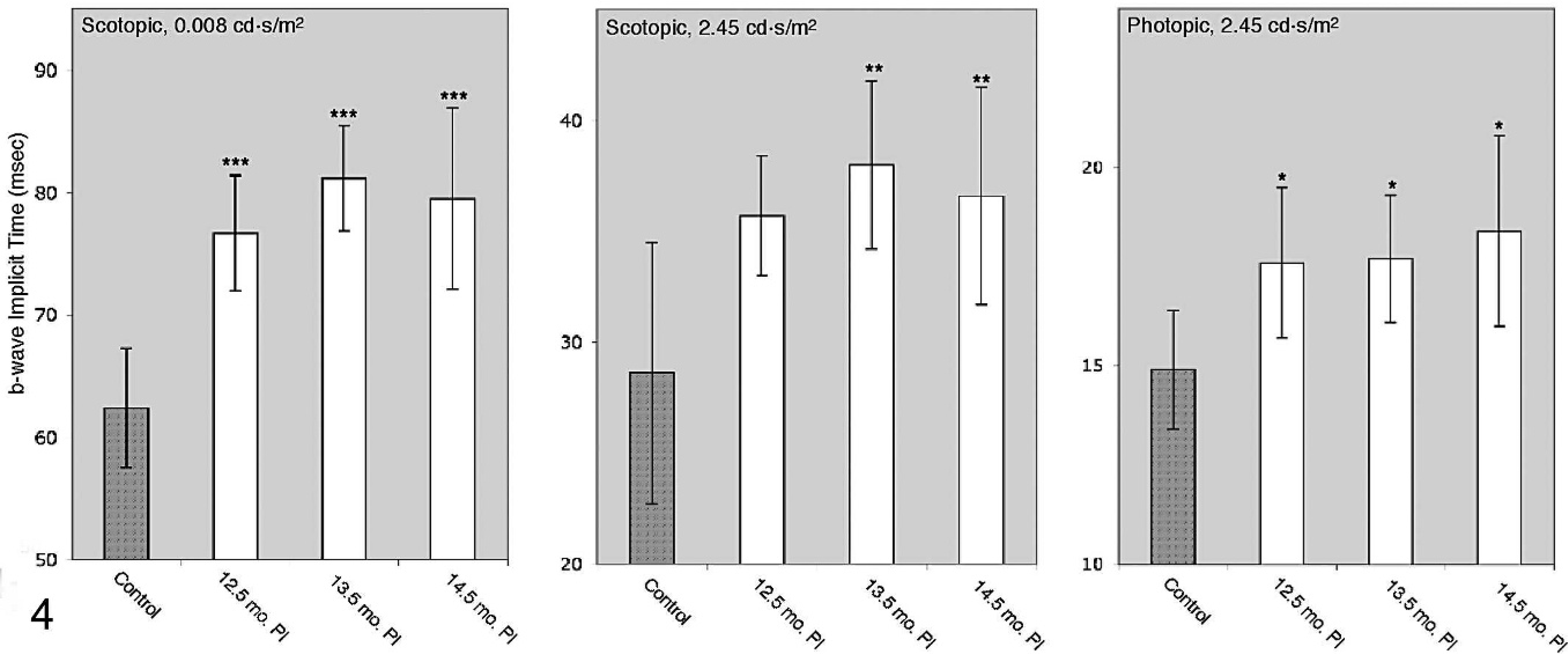
Comparison of mean electroretinogram b-wave implicit time data among control and preclinical transmissible mink encephalopathy–inoculated cattle at serial postinoculation time points under scotopic or photopic testing conditions. Implicit time was significantly prolonged in transmissible mink encephalopathy–inoculated cattle versus controls at all time points, with the exception of 12.5 months postinoculation for the second test. Implicit time data represented here are shown in Table 1.
Examination of HE-stained retinal sections (Figs. 5,6) revealed mild pathologic change. Evidence of spongiform change was observed within the inner plexiform layer of TME-affected cattle but not in controls (Fig. 6). Cell density within the ganglion cell layer differed between the 2 groups, with controls having an average of 114 nuclei per 5 40× fields versus 44 nuclei per 5 40× fields in TME-affected cattle. Immunoreactivity for PrPSc was detected in the retinas of all TME cattle and was localized primarily to the synaptic layers and the cytoplasm of retinal ganglion cells (Fig. 8).

Retina; noninoculated control bovine. Histopathologic lesions were not detected in retinas from control cattle. OS = outer segments; ONL = outer nuclear layer; OPL = outer plexiform layer; INL = inner nuclear later; IPL = inner plexiform layer; GCL = ganglion cell layer; OFL = optic fiber layer. HE. Bar = 20 µm.

Retina; transmissible mink encephalopathy–inoculated bovine. Subtle changes detected in the retinas of transmissible mink encephalopathy–affected cattle included multifocal, distinct, clear spaces within the inner plexiform layer (arrows) and decreased numbers of nuclei in the ganglion cell layer. HE. Bar = 20 µm.
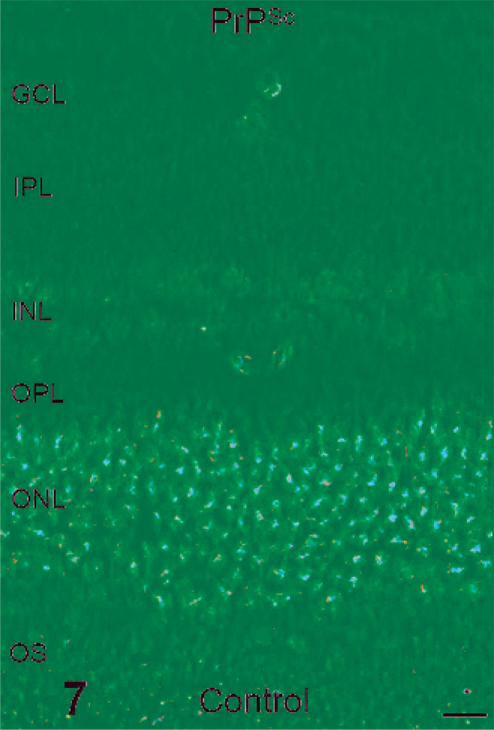
Retina; control bovine. Prion protein was not detected in control retina. Prion protein immunohistochemistry with monoclonal antibodies F89/160.5 and F99/97.6.1 and hematoxylin counterstain. Bar = 20 µm.

Retina; transmissible mink encephalopathy–inoculated bovine. Intense punctate prion protein immunoreactivity was detected throughout both plexiform layers, within retinal ganglion cells (arrows), and sporadically within the inner nuclear layer and at the outer limiting membrane (arrowheads). Prion protein immunohistochemistry with monoclonal antibodies F89/160.5 and F99/97.6.1 and hematoxylin counterstain. Bar = 20 µm.
Antibodies directed against retinal cell types contributing to the ERG b-wave (rod bipolar cells and Müller glia) were used to examine these cell populations in cattle clinically affected with TME. In control retinas, immunoreactivity for the alpha isoform of protein kinase C was present in rod bipolar cells. Relative to controls, alpha isoform of protein kinase C immunoreactivity in TME-affected cattle appeared less uniform across the outer plexiform layer, and there were fewer distinct, large, ovoid puncta at the level of rod bipolar cell terminals (Fig. 9). Glutamatergic photoreceptor and bipolar cell terminals were immunoreactive for vesicular glutamate transporter 1 in control retinas. Compared with controls, vesicular glutamate transporter 1 in retinas from TME-affected cattle was less obviously associated with larger bipolar cell terminals along the vitreal border of the inner plexiform layer (Fig. 10). Müller glia were labeled in their entirety by antibodies against glutamine synthetase in both control and TME-affected retinas (Fig. 11), but subtly higher levels of glutamine synthetase immunoreactivity were observed in the processes of Müller glia, specifically the portion coursing through the inner plexiform layer, in TME-affected cattle versus controls. Immunoreactivity for glial fibrillary acidic protein was detected in the optic fiber layer of both control and TME-affected cattle, but markedly fewer and less prominent immunoreactive radial processes were observed in control retina compared with TME-affected retina (Fig. 12).
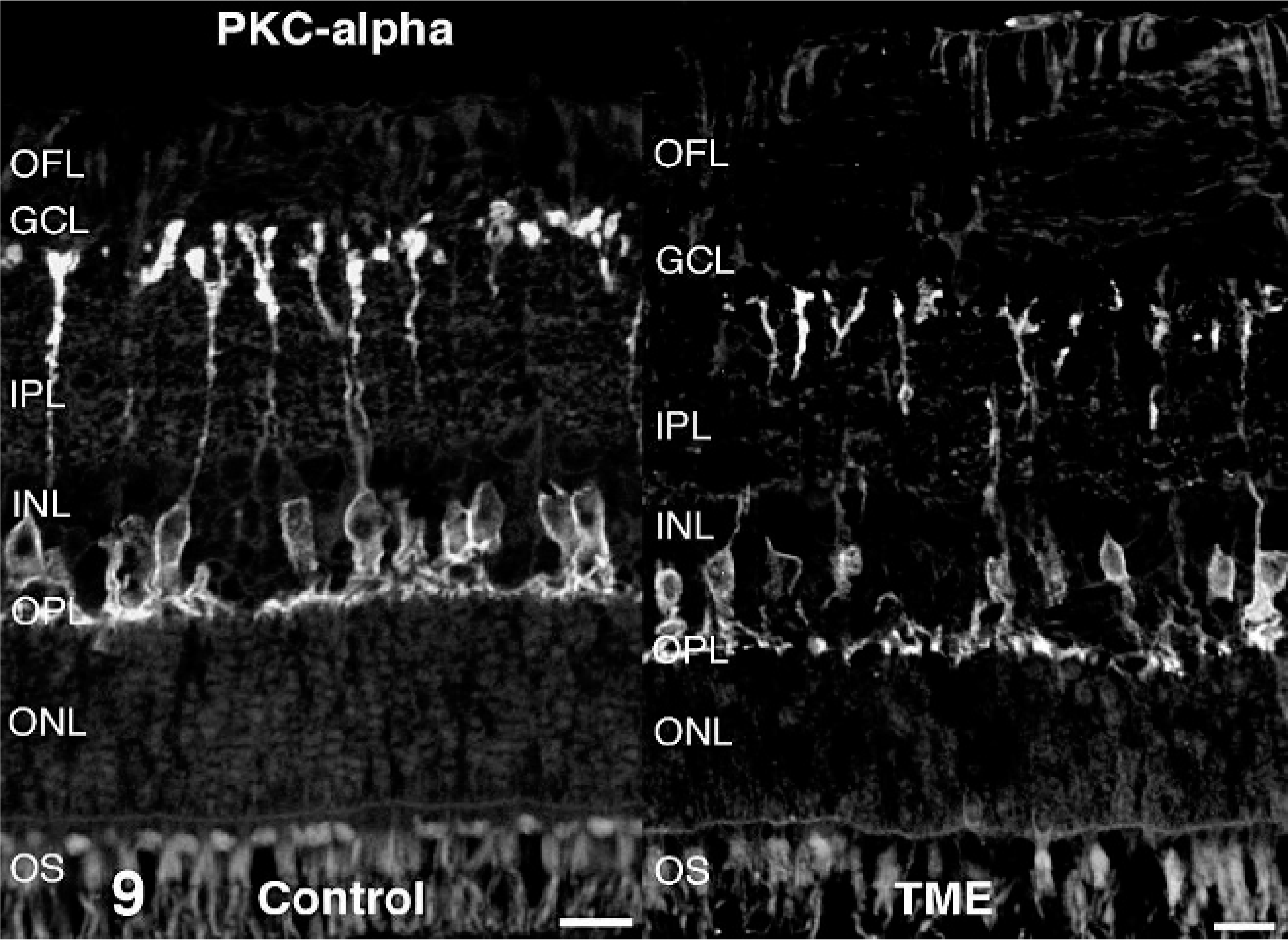
Retina; control bovine (left) and transmissible mink encephalopathy–inoculated bovine (right). Compared with control retinas, the alpha isoform of protein kinase C immunoreactivity in transmissible mink encephalopathy–affected retinas was less uniform across the outer plexiform layer and rod bipolar cell terminals were less obvious as large, ovoid puncta of immunoreactivity at the vitreal border of the inner plexiform layer. OS = outer segments; ONL = outer nuclear layer; OPL = outer plexiform layer; INL = inner nuclear later; IPL = inner plexiform layer; GCL = ganglion cell layer; OFL = optic fiber layer. Alpha isoform of protein kinase C fluorescence immunohistochemistry. Bar = 20 µm.
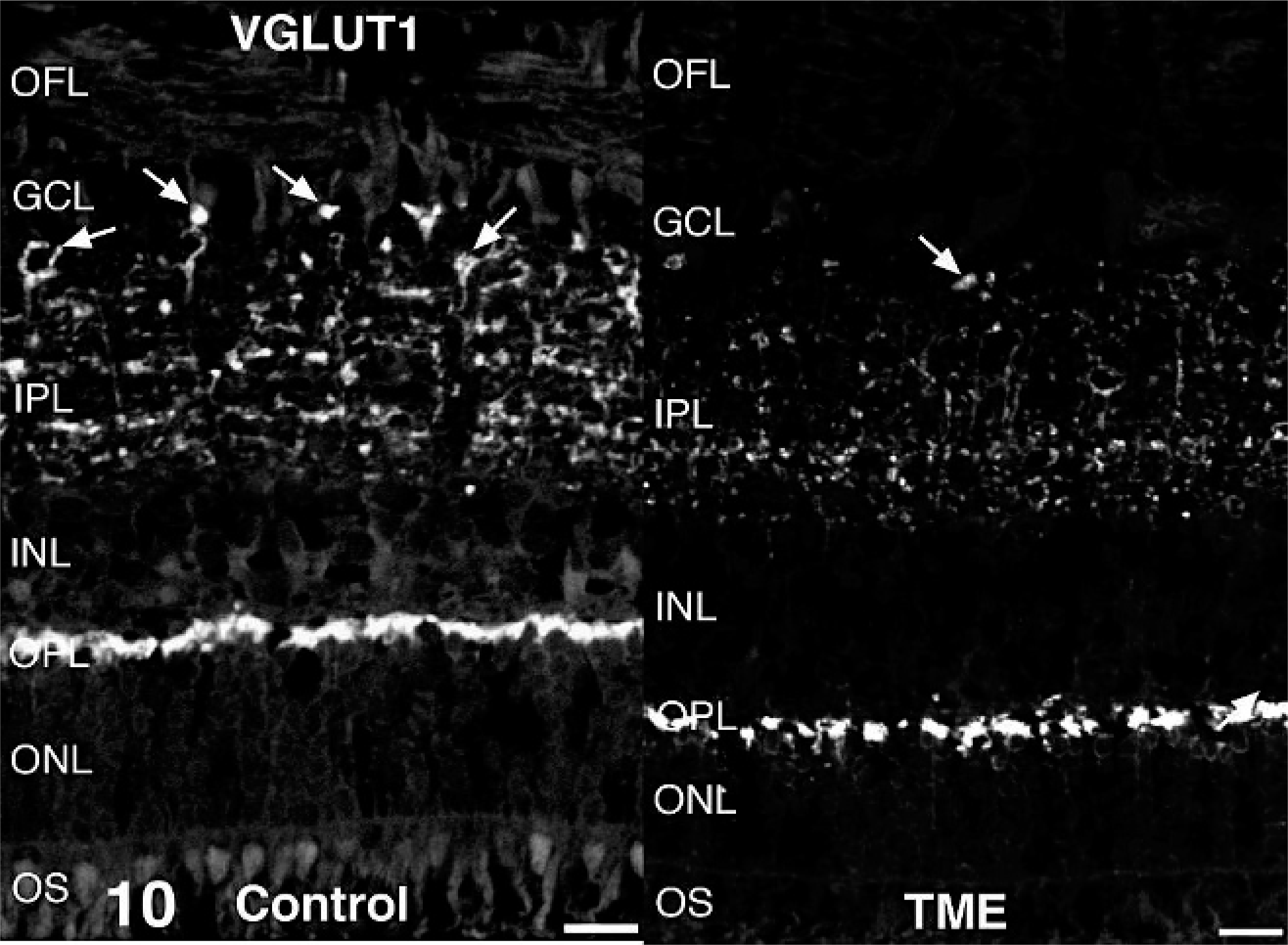
Retina; control bovine (left) and transmissible mink encephalopathy–inoculated bovine (right). Vesicular glutamate transporter 1 immunoreactive bipolar cell terminals (arrows) in transmissible mink encephalopathy–affected retinas were less prominent along the vitreal border of the inner plexiform layer compared with control retinas. Vesicular glutamate transporter 1 fluorescence immunohistochemistry. Bar = 20 µm.
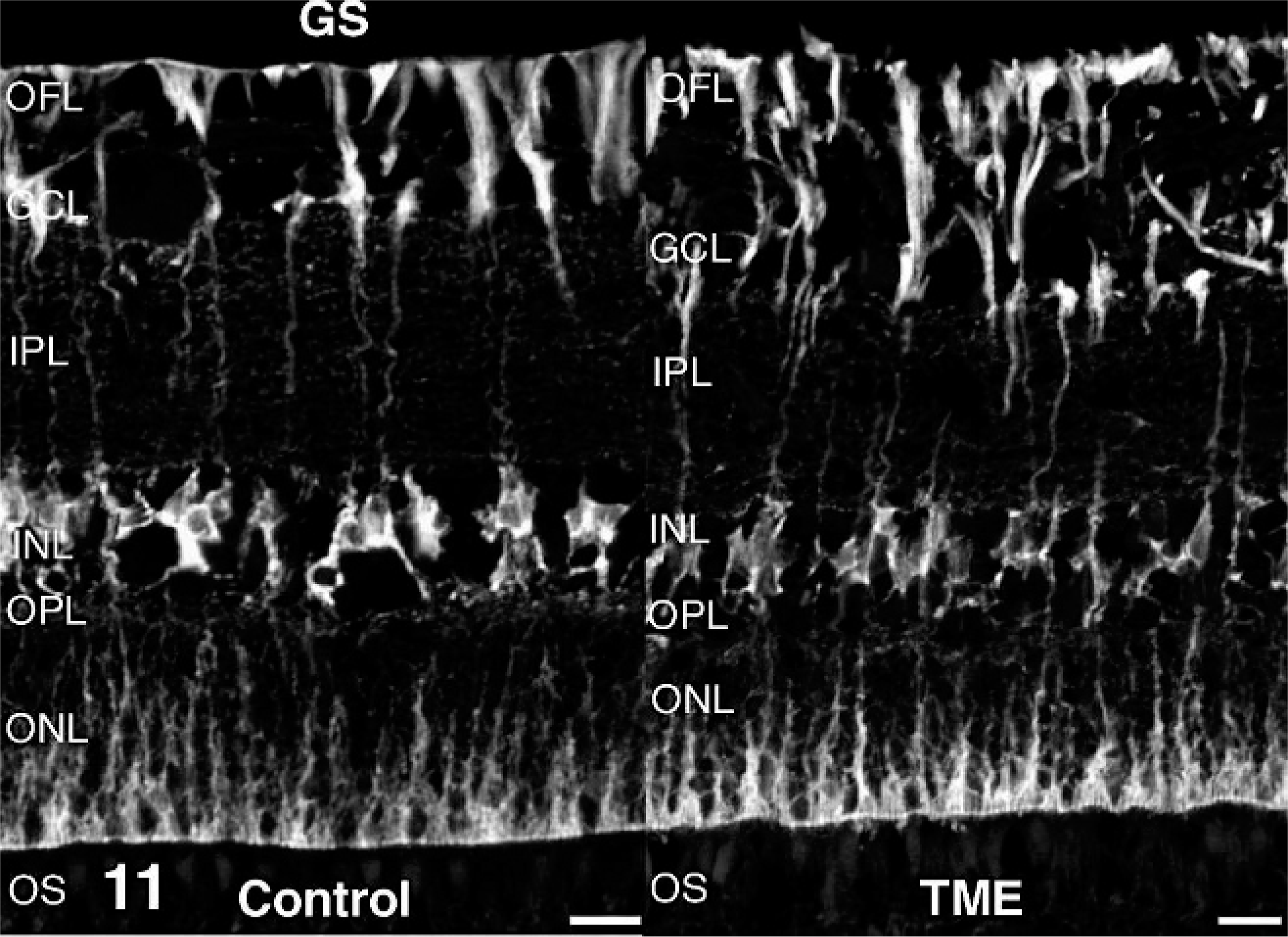
Retina; control bovine (left) and transmissible mink encephalopathy–inoculated bovine (right). Relative to control retinas, glutamine synthetase immunoreactivity in transmissible mink encephalopathy–affected retinas was slightly increased in Müller glial processes coursing through the inner plexiform layer. Glutamine synthetase fluorescence immunohistochemistry. Bar = 20 µm.
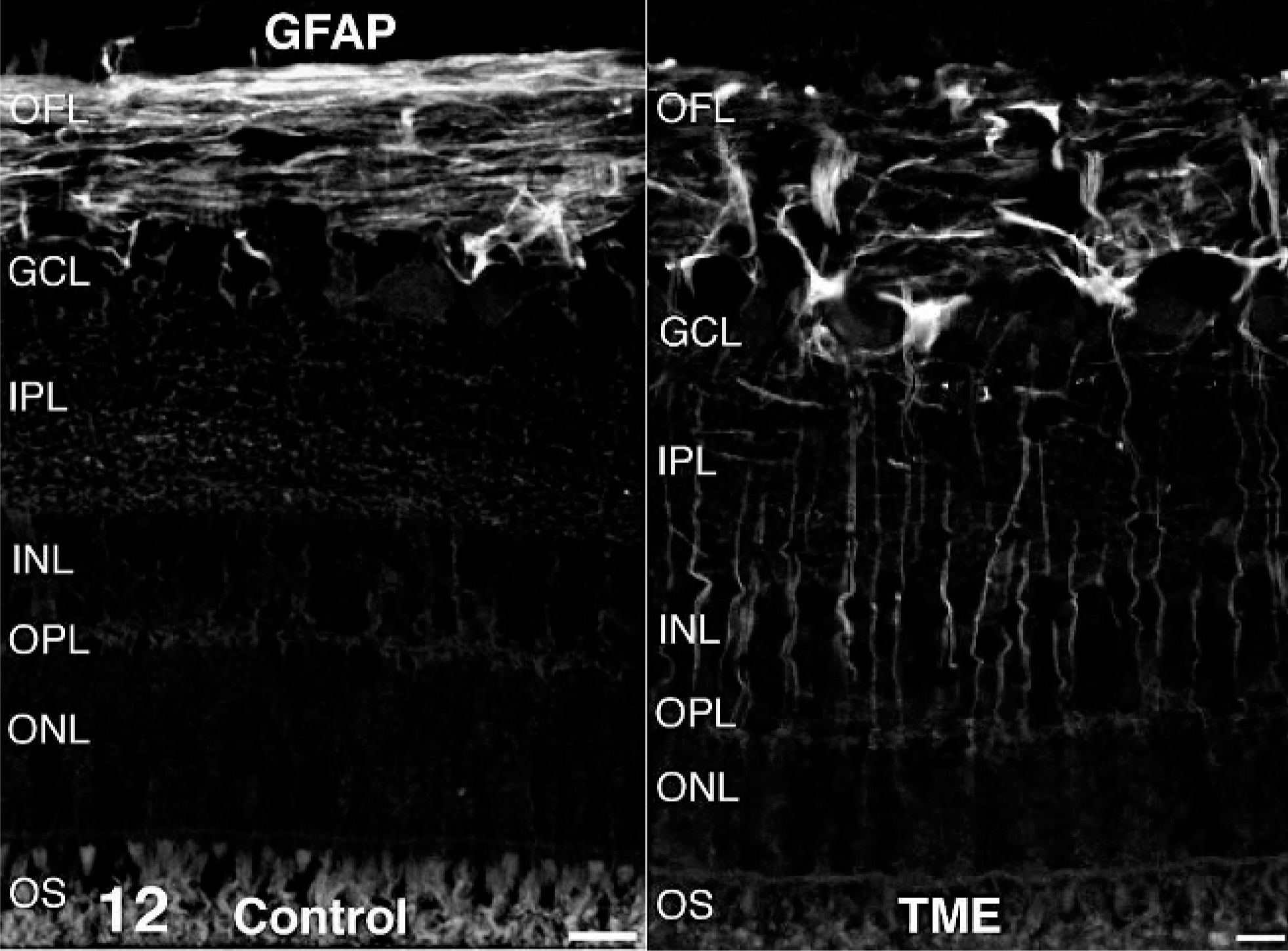
Retina; control bovine (left) and transmissible mink encephalopathy–inoculated bovine (right). Glial fibrillary acidic protein–positive radial processes were more numerous and prominent in retinas from transmissible mink encephalopathy–affected cattle versus controls. Glial fibrillary acidic protein fluorescence immunohistochemistry. Bar = 20 µm.
Discussion
In this study, we examined both functional and morphologic consequences of PrPSc accumulation in the bovine retina using electroretinography and immunohistochemistry. Our electroretinography results demonstrate altered retinal function in TME-inoculated cattle before the onset of clinical signs of disease. This is the first report of a detectable change in a neurophysiologic parameter in a TSE-infected animal before the onset of clinical signs of disease. Further, we demonstrate morphologic abnormalities in the retinas of these cattle during clinical stages of disease.
Several lines of evidence suggest that the rod bipolar cell is primarily responsible for generating the ERG b-wave; Müller glia are currently regarded as a lesser participant. 9 Our results demonstrate prolonged ERG b-wave implicit time in preclinical cattle. We also demonstrate altered immunolabeling patterns of rod bipolar cell markers (alpha isoform of protein kinase C, 13 vesicular glutamate transporter 1 17 ) and Müller glia markers (glutamine synthetase 13 , glial fibrillary acidic protein 10 ) in clinically affected cattle. Taken together, these data indicate PrPSc-induced pathology may be occurring in the retina before the onset of clinical signs of disease, possibly resulting in compromise of the morphologic integrity of rod bipolar cells, thereby disrupting their connectivity and synaptic function, and alteration of the normal homeostatic mechanisms of Müller glia, such as potassium buffering and extracellular neurotransmitter regulation. Alteration of the b-wave in TME-inoculated cattle, which have PrPSc accumulation in synaptic layers at clinical stages of disease, suggests that synaptic transmission in the retina may be altered, even at preclinical stages of disease. This could indicate early impairment or loss of functional normal cellular prion protein or disruption of synapses or synaptic proteins secondary to PrPSc accumulation. Cellular stress may also play a role, because antioxidant properties have been ascribed to cellular prion protein, 25,26 and cellular prion protein has been implicated in transducing neuroprotective signals in retinal explants. 4 Prolonged b-wave implicit time for both scotopic and photopic tests suggests that both rod and cone photoreceptor pathways are affected.
Human Creutzfeldt-Jakob disease patients have been reported to exhibit various symptoms of visual disturbance. 1 Similar to ERG reports from Creutzfeldt-Jakob disease–affected patients, 7,18 b-wave amplitude was decreased in 2 clinically affected TME cattle. This observed decrease in b-wave amplitude during the clinical, versus the preclinical, stage is likely a function of the extent of PrPSc accumulation in the retina and its associated pathology. For the photopic ERG, average b-wave implicit time for these 2 clinically affected cattle was similar to the mean control value. This observation was not consistent with our findings during the preclinical period but could be due to the small sample size and relatively small duration of the photopic response.
We have demonstrated altered retinal function in TME-infected cattle before the onset of clinical signs of disease. Further studies are needed to establish whether correlative changes in retinal cellular morphology are present during this preclinical period and whether similar functional alterations are present in cattle with their naturally occurring TSE, bovine spongiform encephalopathy. Definitive diagnosis of TSE in livestock is currently based on a combination of postmortem testing that includes histopathology, immunohistochemistry, and molecular techniques, but with sufficient technological advances in both data collection and analysis, electroretinography, or a derivative of electroretinography, may one day provide a new TSE screening strategy for live cattle. Ongoing studies in the laboratory are investigating automated high-throughput methods of ERG waveform analysis that would be necessary for the practical application of this technique.
Footnotes
Acknowledgements
We thank L. Mandell, M. Church, and J. Lesan for technical assistance. This work was supported by Specific Cooperative Agreement #58-3625-5-114 with the Agricultural Research Service, US Department of Agriculture. Mention of trade names or commercial products in this paper is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
