Abstract
Thymic cysts with pseudoepitheliomatous hyperplasia are described in a 7-month-old female American Eskimo Dog that died of complications from brodifacoum poisoning. Grossly, there was hemothorax with marked cranial mediastinal hemorrhage. Histologically, thymic lobules were expanded and distorted by irregular cysts, lined by single to multiple layers of plump to slightly attenuated polygonal squamous epithelial cells supported by a basement membrane (pseudoepitheliomatous hyperplasia). The thymus had a paucity of lymphocytes and lacked corticomedullary differentiation. Extensive hemorrhage within the cysts and thymic parenchyma extended into the adjacent adipose tissue. To the authors' knowledge, this is the first report of cystic thymic degeneration with pseudoepitheliomatous hyperplasia in a nonhuman species.
The third pharyngeal pouch of the foregut endoderm gives rise to the thymus and becomes colonized by lymphocytes from the primordial yolk sac. Normal involution of the thymus occurs after sexual maturity. Accelerated thymic involution, hypoplasia, and degeneration can be triggered by many causes, including infections, toxins, ionizing radiation, chemotherapeutic drugs, corticosteroids, and nutrient deficiencies. 5 In some cases, it can also lead to thymic cysts. 14, 18
Hitherto, thymic cystic degeneration with pseudoepitheliomatous hyperplasia has been described only in the human biomedical literature. 14, 15 Pseudoepitheliomatous hyperplasia was defined by Suster et al. as “epithelial proliferations that may resemble a malignant neoplastic process morphologically described under a variety of circumstances in several organs, most notably in the skin and mucous membranes.” 14 It has also been referred to as pseudocarcinomatous hyperplasia. The current article describes cystic degeneration of the thymus with pseudoepitheliomatous hyperplasia and hemorrhage in a 7-month-old, intact female American Eskimo Dog.
A 7-month-old, white, intact female American Eskimo Dog presented to the San Diego County Animal Disease Diagnostic Laboratory in December 2006 with a 24-hour history of listlessness and walking slowly before death. On necropsy, the oral mucosa was found to be white. In addition, 200 ml of uncoagulated blood filled the thorax (hemothorax), and the cranial mediastinum was markedly distended with hemorrhage. The thoracic aorta and vena cava, as well a their main branches and tributaries, were intact. Multifocal ecchymoses were found in the endocardium of the left ventricle. The tracheal ligament was elongated to 0.7 cm; the tracheal rings were flaccid and flattened, forming a D-shaped profile on transverse section, indicative of a collapsing trachea. A 2.0 × 1.0 cm area in the wall of the gastric cardia was hemorrhagic. Within the panniculus over the left dorsal thorax was a patch of hemorrhage 1.0 cm in diameter. The right kidney was absent (renal agenesis), and the left kidney was slightly enlarged (compensatory hyperplasia). There was no evidence of trauma.
Histologically, thymic lobules had a paucity of lymphocytes and lacked corticomedullary differentiation, but they were expanded and distorted by multiple irregular cysts ranging from 0.2 to 4.0 mm in diameter, separated by wide trabecular bands of thymic parenchyma containing stromal epithelial cells, lymphocytes, blood vessels, and erythrocytes (Fig. 1). The cysts were lined by 1–8 layers of plump to slightly attenuated polygonal squamous epithelial cells supported by a basement membrane. Scattered lymphocytes and Hassall’s corpuscles were admixed within the squamous epithelial lining (Fig. 2). Squamous epithelial cells had indistinct cell borders, a variable amount of wispy, pale eosinophilic cytoplasm, and polygonal and vesicular nuclei with peripheralized chromatin. Mitotic figures were not observed. In some areas, the lining of the cysts was obscured by remaining resident thymic lymphocytes (Fig. 3). Cysts contained variable numbers of lymphocytes, erythrocytes, and sloughed epithelial cells as well as variable amounts of fibrin and fluid. Extensive areas of hemorrhage were found within the capsule that extended into the adjacent adipose tissue (Fig. 1). Lobules were separated by variable amounts of edema, fibrin, hemorrhage, and lymphocytes with multifocal ectatic lymphatic vessels (Fig. 1). Cystic degeneration of the thymus with pseudoepitheliomatous hyperplasia was diagnosed.
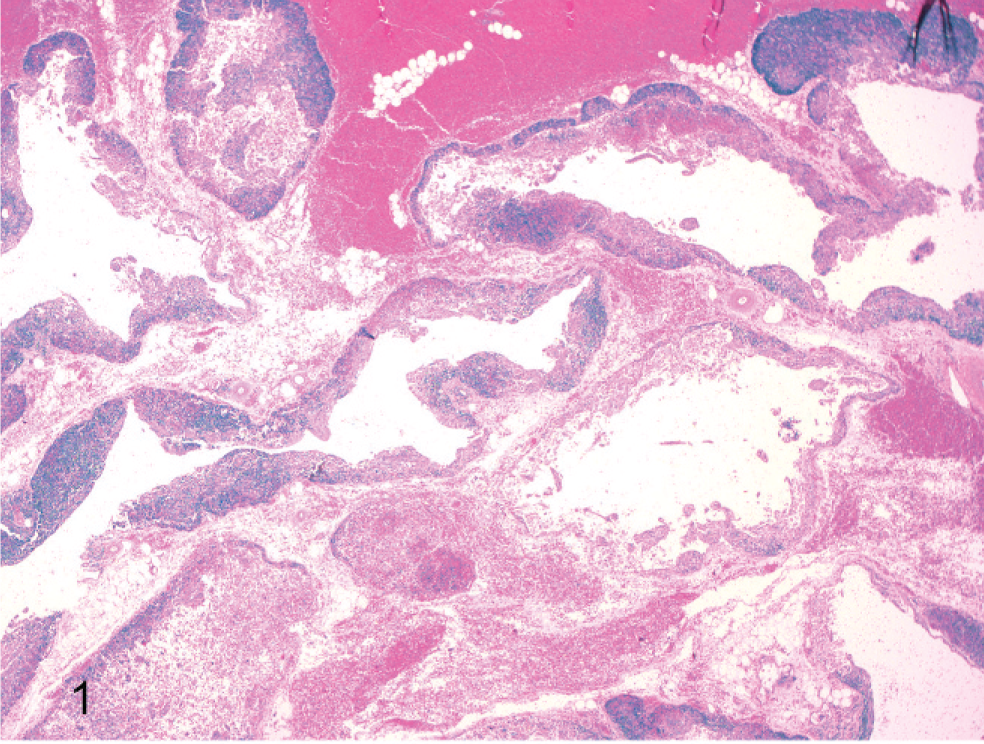
Thymus; dog. Several irregular cystic spaces expand and distort the thymic parenchyma. Extensive hemorrhage separates cysts and variable amounts of hemorrhage are within cystic cavities. HE.
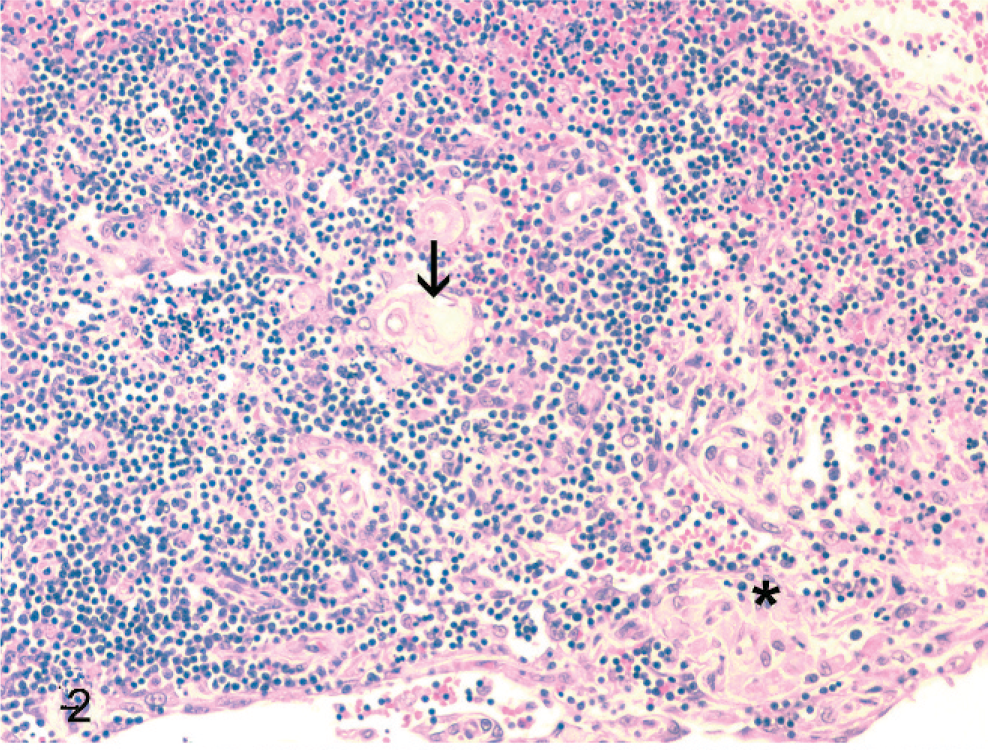
Thymus; dog. There is loss of cortical and medullary architectural distinction with scattered Hassall’s corpuscles (arrow) and a focus of disorganized epithelial cells (asterisk). Along the bottom margin of the figure are slightly attenuated epithelial cells lining the cyst. HE.
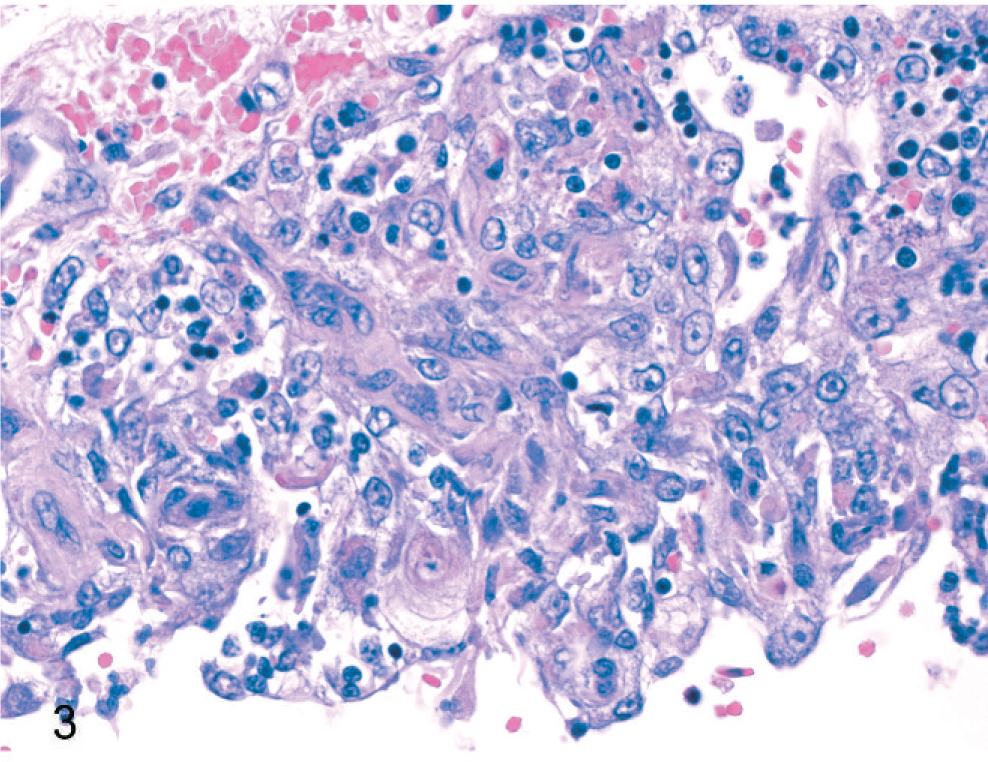
Thymus; dog. Cyst lined by multiple disorganized layers of polygonal squamous epithelial cells (hyperplasia). HE.
Additional histologic findings included the following: 1) centrilobular hepatocellular atrophy and sinusoidal dilation with diffuse microvesicular degeneration, 2) subendocardial hemorrhages, 3) multifocal adrenal cortical hemorrhages, 4) tonsillar follicular lymphocytolysis, 5) gastric submucosal hemorrhages, and 6) aortic adventitial hemorrhage and mesothelial hypertrophy.
The hemothorax and hemorrhages warranted investigation for a coagulopathy. Toxicologic analysis of the liver detected brodifacoum, a second-generation anticoagulant poison, at 1.7 ppm wet weight (performed by California Animal Health and Food Safety Laboratory, Davis, CA).
The thymus is composed of epithelial and lymphoid components. The epithelial component develops from 2 streams of epithelium arising from the foregut endoderm: thymic duct epithelium and reticular epithelium. Thymic duct epithelium forms a tubular system between thymic lobules and is supported by a basement membrane. 17 It is thought that thymic cysts result from either congenital malformations of this ductal epithelium or attempts to recapitulate its embryologic origin in disease states, leading to formation of acquired cysts. 17
Reports of thymic cysts are rare in human beings 14 and have been reported in animals. Congenital thymic cysts are usually unilocular, thin walled, and lined by thymic epithelial cells, flattened epithelial cells, cuboidal to columnar epithelial cells, or thymic tissue without an epithelial lining. 6, 14 Acquired thymic cysts are often multilocular, thick walled, 13 and partially lined by squamous, columnar, or cuboidal epithelium. 11, 15 Neoplasia, 10, 13 inflammation, 14 trauma, 13 or systemic disease 8, 13 can give rise to acquired thymic cysts. Reports of congenital and/or acquired thymic cysts have been reported in dogs, 4, 16 a cat, 4 harbor porpoises,19 bottlenose dolphins, 3 pigeons, 1 and rats. 12 Epithelial thymic cyst formation has been recorded in rats treated with exogenous estrogen. 12 The ovaries of the dog in this case report were grossly and histologically normal with the presence of primary and secondary follicles. Ovarian histologic features were not suggestive of hyperestrogenism.
Pseudoepitheliomatous hyperplasia within thymic cysts has not been previously reported in animals to the author’s knowledge and has only rarely been reported in human beings. It is believed to be caused by an exaggerated response to thymic injury or inflammation. 14 Pseudoepitheliomatous hyperplasia may develop in thymic cysts as an expression of regeneration of the lining epithelium in response to the inflammatory, hemorrhagic, and necrotizing changes that often accompany these lesions. 14 In aging rodents, thymic epithelial hyperplasia has been reported in the presence of thymic involution. 12 Hyperplastic thymic epithelial cells are cuboidal to columnar, often form tubules or cords, occasionally are ciliated, and may have an admixture of secretory goblet cells.
The pleomorphic presentations of thymic epithelial hyperplasia are similar to the spectrum of morphologic variants of thymoma. Thymoma is a neoplasm of the anterior mediastinum and is composed of thymic epithelium in which there are various degrees of benign lymphocytic infiltration. 7 It can be difficult to distinguish extensive thymic epithelial hyperplasia from early benign thymomas. 12 Thymoma can be divided into 3 histologic groups, depending on the relative amount of epithelial and lymphoid cells: predominantly lymphocytic, predominantly epithelial, or mixed. There are many morphologic variations within the groups, including epidermoid, keratinizing, papillary, ribbons, cords, tubules, spindeloid, endocrine, neuroendocrine, and myxoid thymoma; however, none are similar to the current case. 12 Thymoma engrafted on a thymic cyst was considered in the differential diagnosis in this case. However, the association of the cystic and hyperplastic changes with hemorrhage, degeneration, and lymphocytolysis is unlike thymoma. In addition, the median age of dogs with thymoma is 10 years (minimum age, 2.5 years). In dogs and human beings, it has been reported that cyst formation can be present within thymoma. 16 The age of the dog in this case report was 7 months, much younger than any reported case of canine thymoma.
Other midline cysts were considered, including branchial cysts. Branchial cysts are located lateral to the parathyroid-thyroid area 17 and have been described in the thymus of dogs. 4 They are lined by pseudostratified columnar or focally squamous cells that are partially to fully ciliated. 4, 17 In this case, the epithelial cells were not ciliated.
Based on the multilocular characteristics of the thymic cysts, we believe them to be an acquired process. Both the thymic cysts and the pseudoepitheliomatous hyperplasia were of unknown cause and probably reflect a nonspecific response to thymic damage and inflammation.
The thymus and anterior mediastinum are sites of predilection for spontaneous hemorrhages in dogs poisoned by anticoagulant rodenticides. 9 Described lesions associated with thymic hemorrhages include expansion of thymic lobules and interlobular septa, medullary and inner cortical hemorrhage, loss of normal lobular architecture, infarcts, leukocytoclastic vasculitis, and angiofibroplasia. Idiopathic thymic hemorrhage of 2 young littermate dogs has been described. 2 In these cases, there was no known exposure to rodenticides. One dog died, and a liver sample was negative for anticoagulant rodenticides; the other dog survived and was not tested for anticoagulants. Thymic hemorrhage has also been associated with rupture of dissecting aortic aneurysms and trauma from automobile accidents. 5 Two theories have been postulated and reviewed in the article by Liggett et al on the pathogenesis of thymic hemorrhage in young dogs with thymic involution. 9 One theory suggests that the involuting thymus is predisposed to hemorrhage because of the thin-walled veins and arteries not receiving sufficient counterpressure from adipose and loose connective tissue. A sudden rise in blood pressure could then trigger fatal hemorrhage. Another author proposed the theory that trauma could result in overstretching the neck, causing rupture of vessels in a regressing thymus. 9 However, the previously described cases of thymic hemorrhages in animals did not include thymic cyst formation or pseudoepitheliomatous hyperplasia. In the present case, thymic cysts may have predisposed the organ to hemorrhage. Unilocular thymic cysts associated with hemorrhage have been reported in 2 children—1 with aplastic anemia and a 13-year-old boy with no other symptoms. 11
The initiating cause of cystic thymic degeneration and pseudoepitheliomatous hyperplasia is unknown. The thymic cysts and pseudoepitheliomatous hyperplasia were chronic lesions in contrast to the acute thymic hemorrhage. Although brodifacoum clearly has anticoagulant effects, to the author’s knowledge there are no reported direct toxic effects of brodifacoum on the thymus.
Cystic thymic degeneration with pseudoepitheliomatous hyperplasia has been reported rarely in human beings. To our knowledge, this is the first reported case of cystic thymic degeneration with pseudoepitheliomatous hyperplasia in a dog or any animal. Hemorrhage associated with the cysts was probably due primarily to the effects of brodifacoum intoxication. The contribution of the cysts and pseudoepitheliomatous hyperplasia to the hemorrhage is unknown.
Footnotes
Acknowledgements
We thank Dr. Robert H. Poppenga (CAHFS Toxicology Laboratory, School of Veterinary Medicine, University of California) for toxicologic analysis of the liver.
