Abstract
A 6-mo-old female Beagle dog was inappetent and depressed. The radiographic, ultrasonographic, and computed tomographic examination of the chest revealed a 10 × 7 cm multicystic mediastinal structure interpreted as altered thymus, in association with moderate pleural effusion that laboratory tests confirmed as hemothorax. No history of trauma or anticoagulant drug intoxication was reported, and no coagulation disorders were detected. Afterward, medial cranial sternotomy was performed to remove the altered tissue. Histologically, this tissue was compatible with a thymic remnant, characterized by numerous cystic lesions, mostly blood filled and lined by flattened-to-cuboidal epithelial cells, occasionally projecting into the surrounding stroma, and forming cytokeratin-positive ribbons, trabeculae, and papillae. Lymphocytes were scant, and numerous areas of congestion and hemorrhage were present throughout the samples. This case of idiopathic thymic hemorrhage with cystic degeneration of the thymus and pseudoepitheliomatous hyperplasia was an incidental finding; the dog recovered from surgery uneventfully.
Thymic hemorrhage or thymic hematoma is a life-threatening condition that occurs most frequently in young dogs. It has been associated with different causes, from anticoagulant intoxication, trauma, or neoplasia to idiopathic pathogenesis, particularly during the involution phase of the organ. 18
A 6-mo-old female Beagle was referred to the Veterinary Teaching Hospital of the Department of Veterinary Medicine (University of Perugia, Italy) because of reluctance to move, decreased appetite, and depression. Clinical examination revealed pale mucosae, tachycardia (140 bpm), mild dyspnea, and hypothermia (37°C). A complete blood cell count showed mild regenerative anemia, mild thrombocytopenia, and leukocytosis. The biochemical profile and coagulation test were unremarkable. Radiographic and ultrasonographic examination of the thorax revealed moderate pleural effusion and a mildly heterogeneous, vascularized, cranial mediastinal structure of ~ 10 × 7 cm. Pleural fluid was sampled by ultrasound-guided fine-needle aspiration. The patient was promptly stabilized with oxygen and intravenous fluids at 5 mL/kg/h. Computed tomography of the thoracic cavity showed a heterogeneous density and mild post-contrast enhancement of the thymic tissue (Fig. 1).
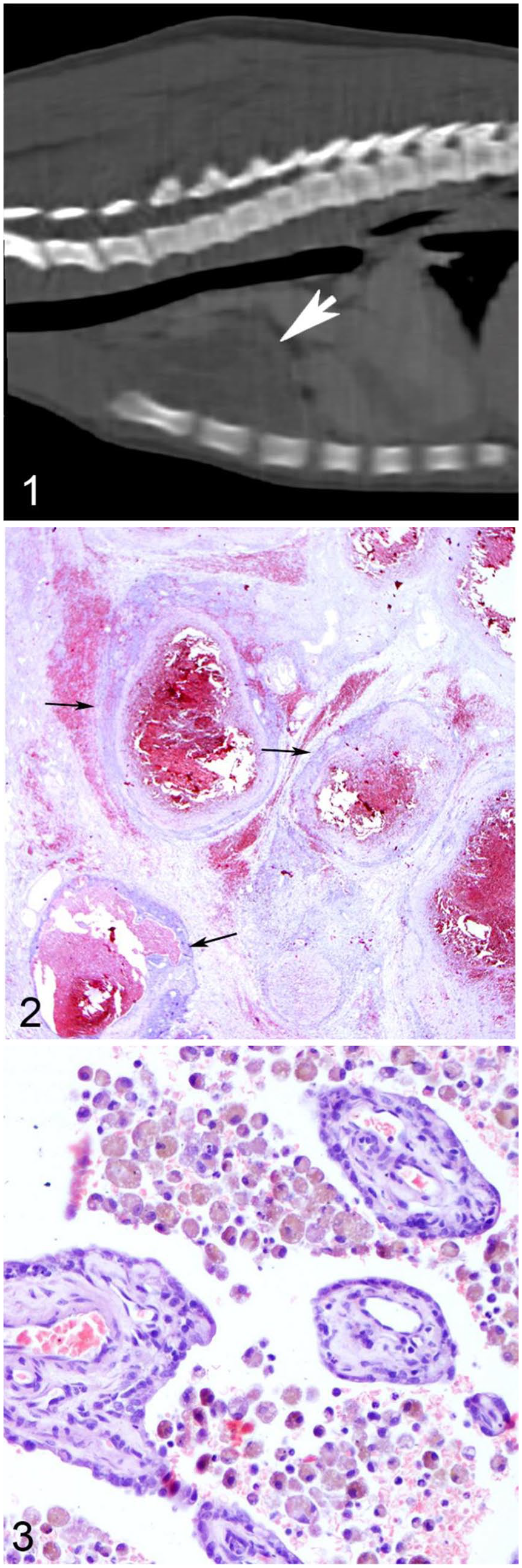
Cystic thymic degeneration with pseudo- epitheliomatous hyperplasia in a Beagle puppy.
Cytologic examination of the pleural effusion confirmed hemothorax; the mass was poorly cellular and represented by a few small and mature lymphocytes, admixed with occasional medium-to-large mononuclear cells (interpreted as immature lymphocytes) and rare neutrophils. Flow cytometric immunophenotyping indicated the presence of both CD4+ and CD8+ cells. Complete surgical removal of the thymic tissue with a cranial sternotomy was scheduled in order to prevent further intrathoracic bleeding and to characterize the lesion. Percutaneous biopsy was not performed because of the risk of intrathoracic hemorrhage. In agreement with the owner, surgery to remove the mediastinal mass was scheduled a week later. The dog was discharged from the hospital and in the interim week, the referring veterinarian prescribed an antibiotic and a corticosteroid because of progressive leukocytosis.
Macroscopically, the thymus was reduced in size (5 × 3 cm) compared to the time of presentation, with an irregular appearance; it was completely adherent to the cranial mediastinal structures and to both sides of the cranial thorax and, in order to safely remove the mass, a careful dissection of all the adhesions was necessary. A thoracic drainage tube was inserted through the seventh left intercostal space, and the sternotomic access was closed routinely. Intravenous methadone (0.2 mg/kg, IV, every 4–8 h for 48 h) and, through the thoracic drainage tube, a mixture of 0.5% bupivacaine (1.5 mg/kg, diluted into 10 mL in lactated Ringer solution, administrated every 6–8 h) was administered as post-operative analgesia. The chest tube was removed 24 h post-operative and, after 3 d of hospitalization, the dog was discharged. Ten days after surgery, when cutaneous sutures were removed, clinical examination showed complete resolution of the clinical signs. In addition, no pleural effusion and no signs of residual thymic tissue in the cranial mediastinum area were observed by radiographic and ultrasonographic examination of the thorax.
On gross examination, the mass was dark red, soft, with a multicystic appearance, supported by white, fibrous septa. On cut section, the tissue oozed blood. The mass was formalin-fixed and paraffin-embedded, processed routinely, and stained with hematoxylin and eosin for light microscopy. Immunohistochemistry was performed with standard methods. 3 Briefly, antigen retrieval was performed in a microwave oven with appropriate buffers (Table 1). Primary antibodies (cytokeratin AE1-AE3, vimentin, α-SMA, von Willebrand factor, CD3, CD20, and MAC387) were incubated for 2 h at room temperature in a humidity chamber. Then, slides were rinsed and incubated with secondary antibody and streptavidin from a ready-to-use kit (Mouse and rabbit specific HRP; Abcam, Cambridge, UK) as per the manufacturer’s instructions. Positive reaction was revealed with 3-amino-9-ethylcarbazole (AEC substrate; Abcam). Appropriate positive and negative controls were included for each antibody.
Immunohistochemical markers used to characterize the lesion and their protocols.
Histologically, the thymic architecture was completely altered; medulla and cortex were not recognizable, and the lymphocytic population was severely depleted. The organ was replaced by numerous variably sized cysts, mostly filled with blood and delicate strands of eosinophilic fibrin (Fig. 2) and occasionally with numerous foamy hemosiderin-laden macrophages (Fig. 3). The cysts were lined by flattened or cuboidal epithelial cells arranged mostly in a single layer (Fig. 4). These epithelial cells occasionally grew in anastomosing trabeculae and papillae, intermingled with the surrounding stroma, but still in connection with the cyst-lining component (Fig. 5). Proliferating ribbons and trabeculae were also observed in areas not associated with the cysts. These epithelial cells were characterized by scant, finely granular, eosinophilic cytoplasm with mostly well-defined cell borders and a central round-to-oval nucleus with finely stippled chromatin; the nucleolus was often visible. Anisocytosis and anisokaryosis were moderate. Rare mitotic figures were seen within both the epithelial and the mesenchymal population. Occasionally, the cytoplasm of these cells had a glassy appearance (probably squamous differentiation). Peculiarly, Hassall corpuscles were not identified in any of the sections examined. Immunohistochemistry with anti-pancytokeratin (cytokeratin AE1-AE3) confirmed the epithelial nature of the proliferating cells described previously (Fig. 6). No invasion of the capsular space was evident in the sections examined. Multifocally, the monolayered wall of the cysts was interrupted, and the central cystic space was invaded by stroma represented by fibroblastic reaction associated with proliferation of small capillaries, interpreted as early granulation tissue. The stroma was scantily cellular, with multifocal areas characterized by hemorrhage, congestion, and sporadic, moderate edema. Occasionally, dense bundles and fascicles of spindle cells, positive for both vimentin and SMA, were interpreted as reactive myofibroblasts (Fig. 7). In stromal areas, frequent aggregates of macrophages (positive for MAC387) with cytoplasm filled with hemosiderin were identified, mostly near areas of hemorrhage. The immunohistochemical investigation also revealed a residual population of CD3+ T lymphocytes, frequently infiltrating the epithelial structures, and rare aggregates of B lymphocytes (CD20+). 12 Throughout the sections, there were also numerous venous and arterial blood vessels of different diameter, with variable congestion.
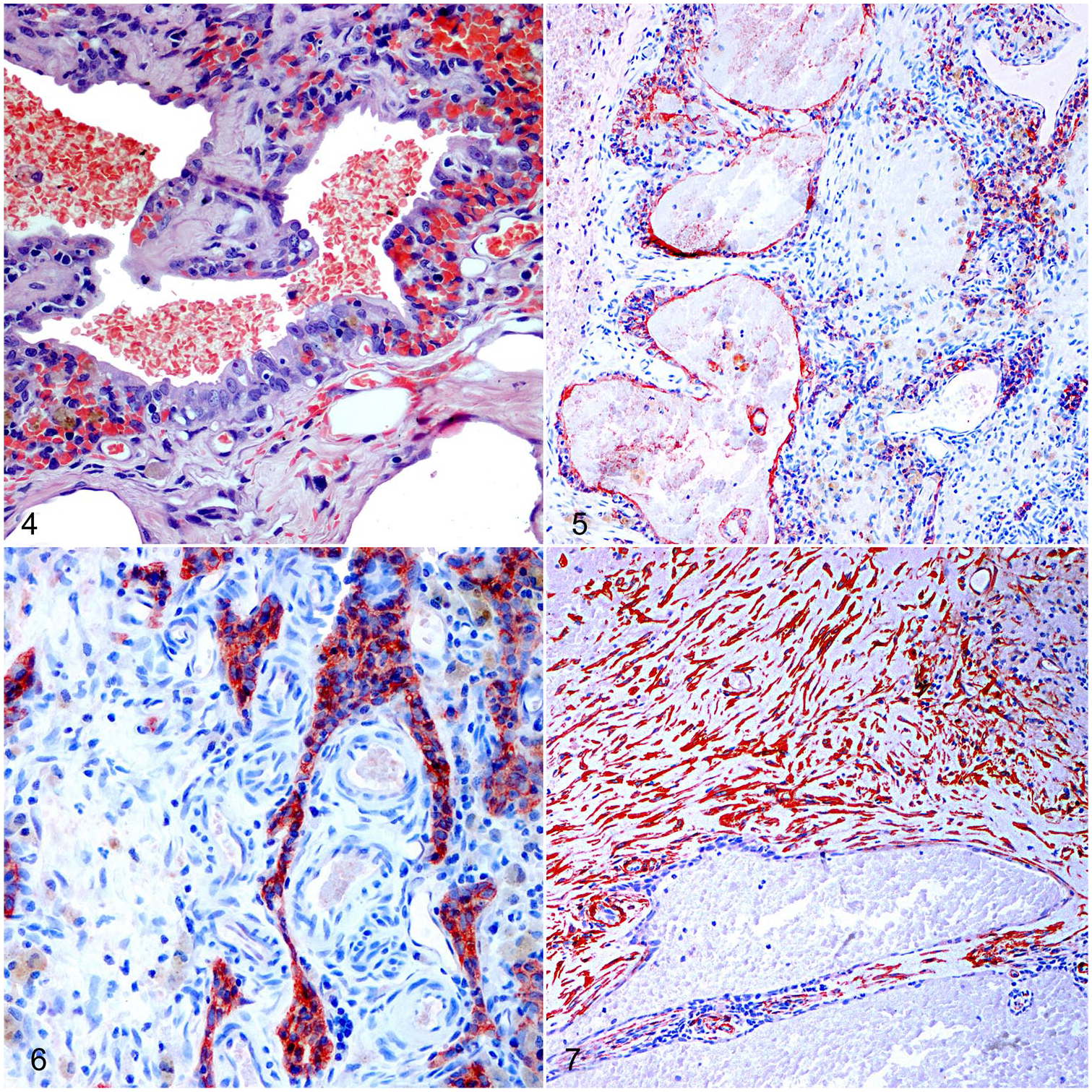
Cystic thymic degeneration with pseudoepitheliomatous hyperplasia in a Beagle puppy.
Possible differential diagnoses included: thymic hemorrhage with concurrent hematoma (either traumatic, toxic, or secondary to thymic involution), thymoma with intralesional hemorrhage, vascular tumor of the thymus (i.e., hemangioma or hemangiosarcoma), parathyroid cyst(s) or branchial cyst(s), and thymic hyperplastic lesion, such as true thymic hyperplasia or the seldom reported pseudoepitheliomatous hyperplasia.
Thymic hemorrhage was suspected given the young age of the dog. The most frequent causes of thymic hemorrhage are anticoagulant rodenticide toxicosis, trauma, and dissecting aneurysms of the aorta; when no cause of hemorrhage can be identified, the lesion is classified as idiopathic.6,19 The normal coagulation profile excluded anticoagulant rodenticide toxicosis for this dog. Traumatic events including minor ones such as excessive pulling on the dog’s collar with a leash was considered unlikely because the owners reported the use of a harness for daily walks, and no grooming or pet service procedures had been performed recently on the animal.7,8 Nevertheless, we observed histologic lesions that can be associated with thymic hematoma(s) (i.e., disseminated foci of hemorrhage, diffuse loss of lymphocytes, fibroangioblastic tissue, and rare infiltration of adipose tissue). 6
The thymic modifications and the histologic findings in our case were not compatible with the reported findings of physiologic thymic involution.2,6,11,14 We considered epithelial proliferation, namely a type B3 thymoma, which is characterized by round or polygonal epithelial cells, with absent-to-mild atypia, and a minor lymphocytic component. 18 This hypothesis was considered unlikely because of the histologic appearance of the lesion, together with the presence of hemorrhagic lacunae and of abundant, variably cellular myofibroblastic stroma, and the young age (6 mo) of the dog.5,6,20
Unlike Labrador Retriever and Labrador crosses, which are overrepresented in case series of thymomas and thymic epithelial tumors,4,5 Beagles are rarely reported to develop these types of tumors. 9 The presence of numerous, variably sized, blood-filled lacunae was also suggestive of a vascular tumor, such as cavernous hemangioma. 10 However, immunohistochemical pan-cytokeratin positivity of the cells lining the cystic spaces together with von Willebrand factor negativity excluded this diagnosis.
Solitary or multilocular thymic cysts have been also reported in association with remnants of embryonic ducts, connecting the parathyroid and the thymus. These cysts are lined by cuboidal epithelium, partially ciliated, and usually contain eosinophilic proteinaceous material, which can also be observed in parathyroid cysts. 17 This diagnostic hypothesis was excluded because of the lack of evidence of cilia and of eosinophilic proteinaceous material (except from fibrin) in the sections examined. Branchial cyst origin was also ruled out because pseudostratified ciliated epithelium was not observed.6,15 In humans, rare cases of true thymic hyperplasia are also reported, both in children and adults, caused by expansion of both epithelial and lymphoid cellular populations. Because histologic and clinical features were different for this dog, we also excluded this diagnosis. 1
We hypothesized that the 1-wk administration of corticosteroids, just before the surgical removal of the organ, could have caused rapid lymphocytic depletion, leading to the decrease in size of the thymus and increasing the dimension and the presence of cystic lesions, as reported previously. 13 In that case report, thymic degeneration with pseudoepitheliomatous hyperplasia was described in the thymus of an American Eskimo Dog with brodifacoum toxicosis. 13 Pseudoepitheliomatous hyperplasia is an uncommon finding in humans, defined as “epithelial proliferations that may resemble a malignant neoplastic process morphologically described under a variety of circumstances in different organs.” 16 We considered this diagnosis the most likely. The pathogenesis of pseudoepitheliomatous hyperplasia is still poorly understood, but a regenerative response to thymic injury or inflammation is suspected. 17 We also hypothesize that the physiologic process of thymic involution, common in dogs 6–12 mo old, could have participated in this clinical and pathologic picture. 6
We therefore diagnosed the lesion as cystic degeneration and hemorrhage of the thymus with pseudoepitheliomatous hyperplasia. The lymphocytic depletion might have been the result of the administration of corticosteroids. Some authors suggest that thymic hemorrhages in juvenile dogs could be caused by a sudden rise in pressure within the thin-walled thymic veins and arteries, not receiving counterpressure from the surrounding tissue. 6 We postulate that pseudoepitheliomatous hyperplasia was already present at the time of presentation of the dog, possibly the result of previous thymic injury or inflammation. This alteration, in association with the cystic degeneration of the organ, may have predisposed to the fragility and rupture of the organ. To our knowledge, this is the second report 12 of pseudoepitheliomatous hyperplasia in a dog.
Footnotes
Acknowledgements
We thank Sara Leto, Valeria Migni, and Luca Stefanelli for their valued technical support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
