Abstract
A chronic ulcerative and eosinophilic dermatitis occurred in 20 captive cheetahs (Acinonyx jubatus) with persistent feline herpes virus 1 (FHV1) infection. Affected animals had erythematous, ulcerated plaques primarily on the face and forelegs in sites of contact with lachrymal and salivary secretions. The dermatitis was characterized by dense infiltrates of eosinophils and plasma cells and pseudoepitheliomatous hyperplasia. Rare keratinocytes within the lesions had nuclei with marginated chromatin and small eosinophilic inclusions composed of herpes virus nucleocapsids. Virus isolated from lesions was confirmed to be FHV1. Lesions persisted and progressed unless removed by cryoexcision. The occurrence of this unusual reaction to FHV1 in approximately 5% of captive North American cheetahs suggests a species propensity for a Th2-dominant response to herpes virus infection. This atypical immune reaction may indicate a heritable trait or modulation of the immune response by other factors such as chronic stress.
In 1991, a chronic ulcerative dermatitis developed in two cheetah cubs (Acinonyx jubatus) with feline herpes virus 1 (FHV1) infection. 10 Although at that time this cutaneous manifestation of FHV1 infection was considered unusual, 20 cheetahs in nine zoos (approximately 5% of the captive North American [NA] population) have now been confirmed with this condition, and additional cheetahs with similar lesions have been reported but not confirmed. In most cases, the cutaneous lesions first occurred as plaquelike edematous swellings at the medial canthus of the eye and on the palpebra that progressed to nonhealing ulcers. Lesions then extended across the nasal planum or ventrally to the nasal philtrum and skin beneath the eyes (Table 1, Fig. 1). Lesions also occurred commonly at the commissures of the lips, top of the head, distal forelegs, flank, tail, and footpads (Fig. 2). Three cheetahs had concurrent ulcerative keratitis, and one affected cheetah had a lingual ulcer.
Signalment, treatment, and outcome in 20 captive North American cheetahs (Acinonyx jubatus) with chronic herpes virus dermatitis.
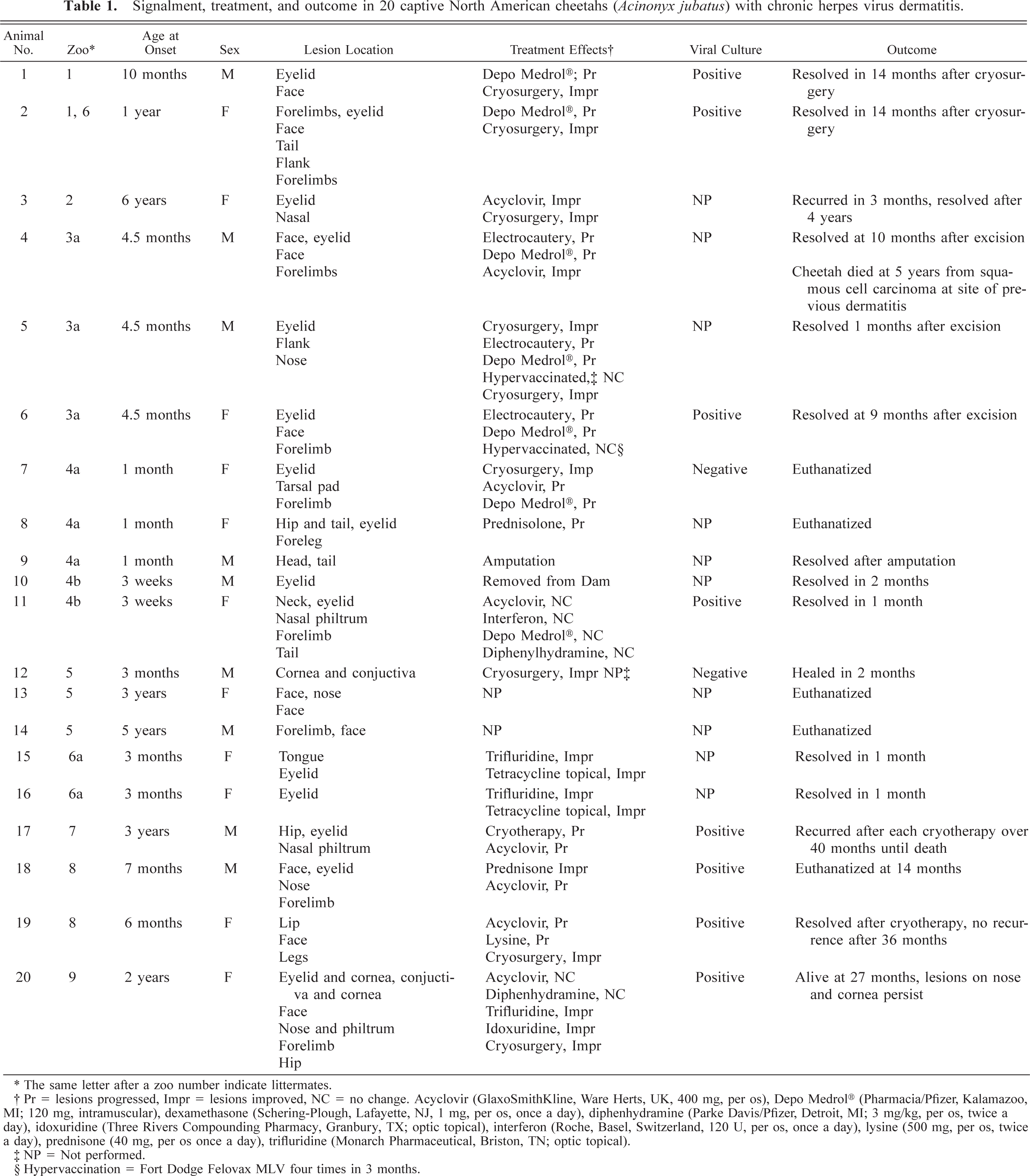
∗ The same letter after a zoo number indicate littermates.
† Pr = lesions progressed, Impr = lesions improved, NC = no change. Acyclovir (GlaxoSmithKline, Ware Herts, UK, 400 mg, per os), Depo Medrol® (Pharmacia/Pfizer, Kalamazoo. MI; 120 mg, intramuscular), dexamethasone (Schering-Plough, Lafayette, NJ, 1 mg, per os, once a day), diphenhydramine (Parke Davis/Pfizer, Detroit, MI; 3 mg/kg, per os, twice a day), idoxuridine (Three Rivers Compounding Pharmacy, Granbury, TX; optic topical), interferon (Roche, Basel, Switzerland, 120 U, per os, once a day), lysine (500 mg, per os, twice a day), prednisone (40 mg, per os once a day), trifluridine (Monarch Pharmaceutical, Briston, TN; optic topical).
‡ NP = Not performed.
§ Hypervaccination = Fort Dodge Felovax MLV four times in 3 months.

Facial skin; cheetah. Chronic herpesviral dermatitis characterized by an ulcerated plaque with peripheral epidermal hyperplasia extending from the medial canthus over the nasal planum in areas in contact with lachrymal secretions.
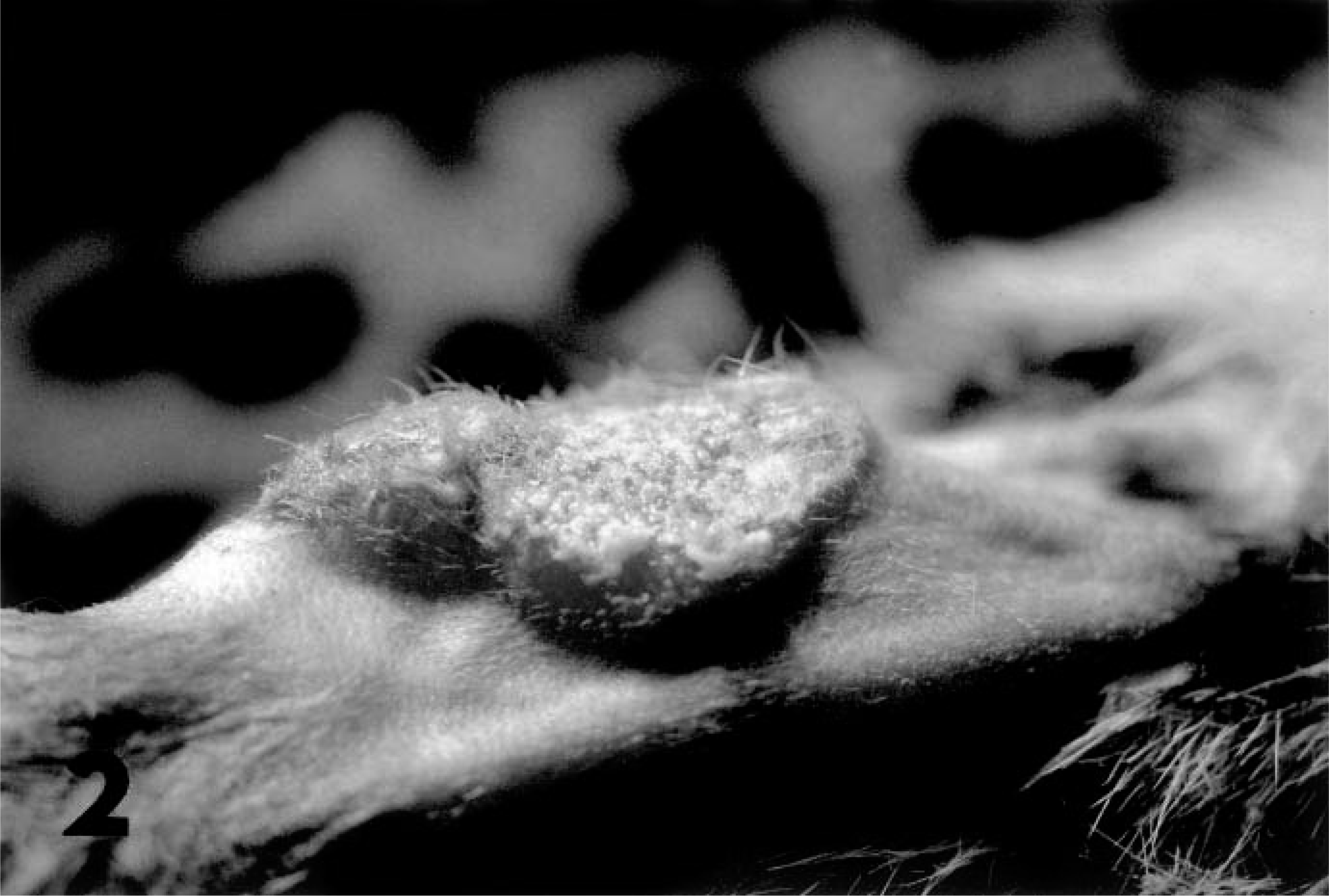
Skin on forelimb; cheetah. Discrete plaque of epidermal thickening from chronic herpesviral dermatitis at sites of grooming.
The skin lesions were typically firm irregular plaques with a raised rim and central ulceration. A white, eosinophil-rich exudate was present in many cases. Over time, these plaques expanded centrifugally, and the epidermis became thickened and verrucous, resembling squamous cell carcinomas. The histopathologic appearance was similar in all cases and was characterized by dense dermal infiltrates of eosinophils, mast cells, plasma cells, and lymphocytes and by pseudoepitheliomatous hyperplasia (Fig. 3). Approximately 40% of the inflammatory cell infiltrates were eosinophils and 30% were plasma cells. In some cases, eosinophilic pustules occurred in the epidermis, and aggregates of eosinophils were associated with degenerate collagen in the dermis. The hyperplastic epithelium formed raised plaques and had prominent rete ridges. Within the stratum spinosum, there were individual necrotic keratinocytes and swollen, degenerating cells with karyomegaly. The nuclei of karyomegalic cells had marginated chromatin, and some nuclei had discrete eosinophilic inclusions separated from the marginated chromatin by a clear rim (Cowdry's type A inclusions) (Fig. 4). Small numbers of 100-nm diameter herpesvirus nucleocapsids were detected in nuclei by electron microscopy (Figs. 5, 6).
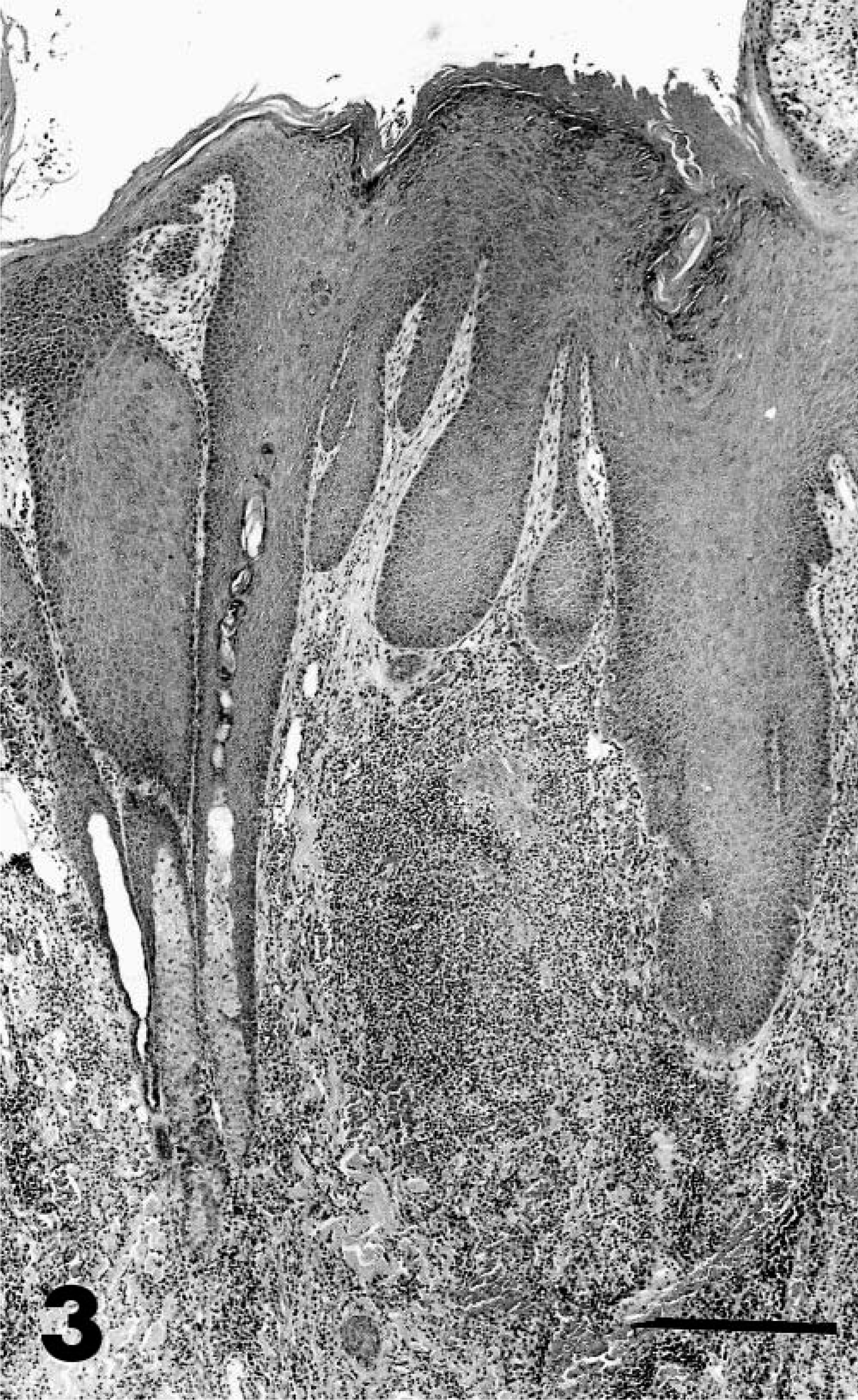
Skin; cheetah. Pseudoepitheliomatous hyperplasia and dense eosinophilic, mastocytic, and plasmacytic infiltrates in the dermis from chronic herpesviral dermatitis. HE. Bar = 250 µm.
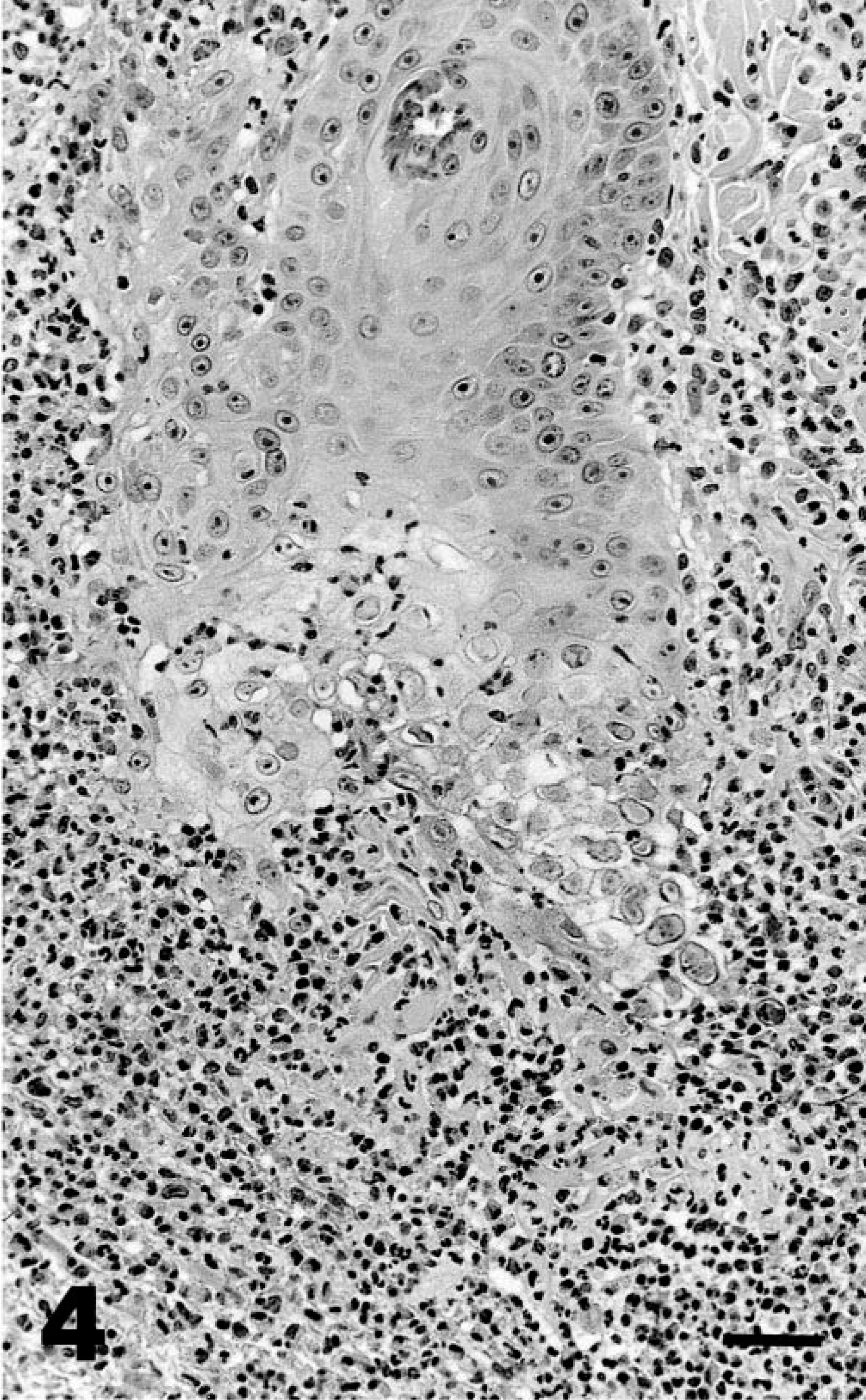
Skin; cheetah. Dense infiltrates of eosinophils, mast cells, and plasma cells associated with kerotinocyte degeneration in deep rete ridges. Some keratinocytes have karyomegaly and Cowdry's type A intranuclear inclusions. HE. Bar = 50 µm.
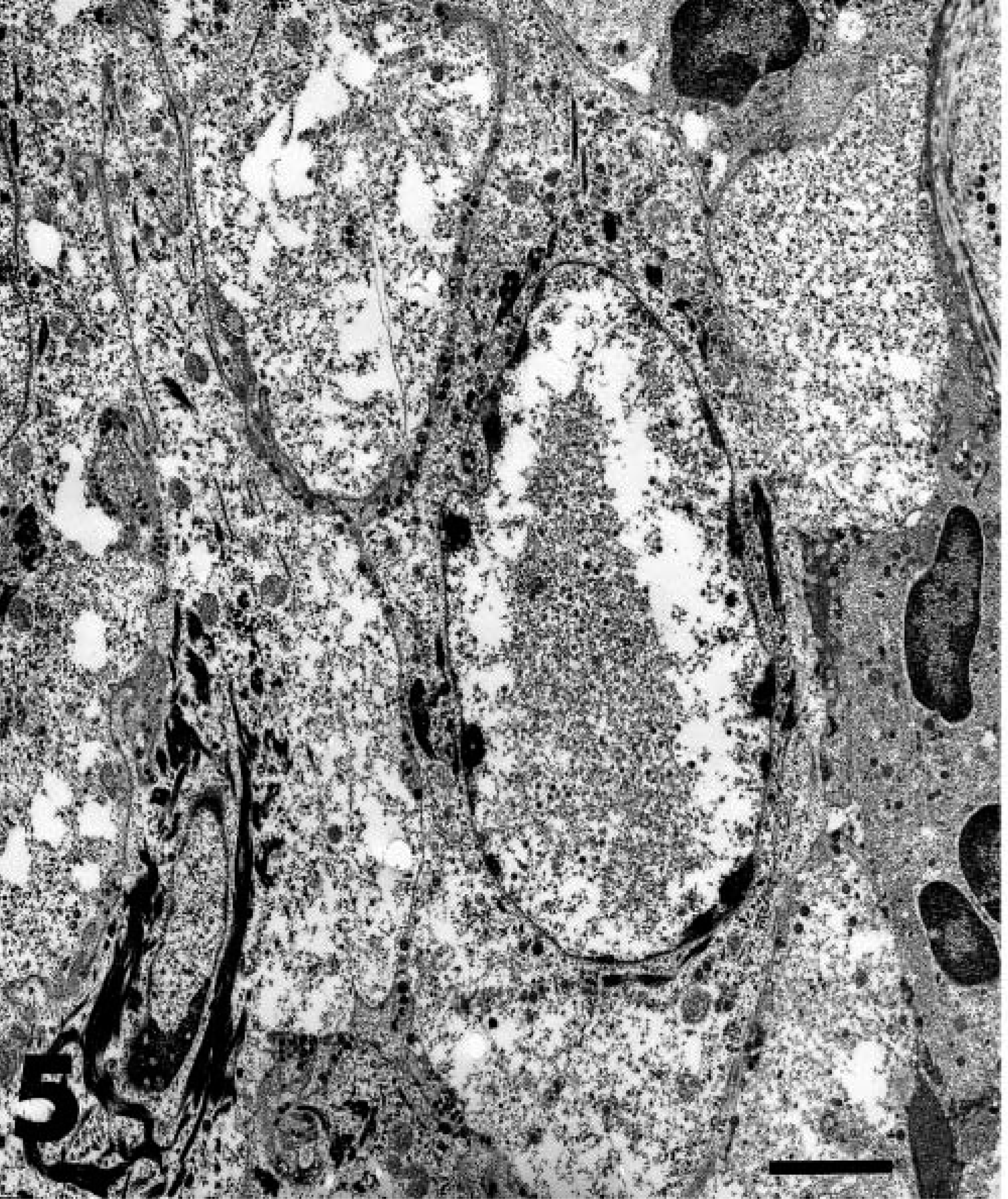
Transmission electron micrograph. Skin; cheetah. Degenerate keratinocyte with intranuclear herpes viral nucleocapsids. Bar = 4 µm.
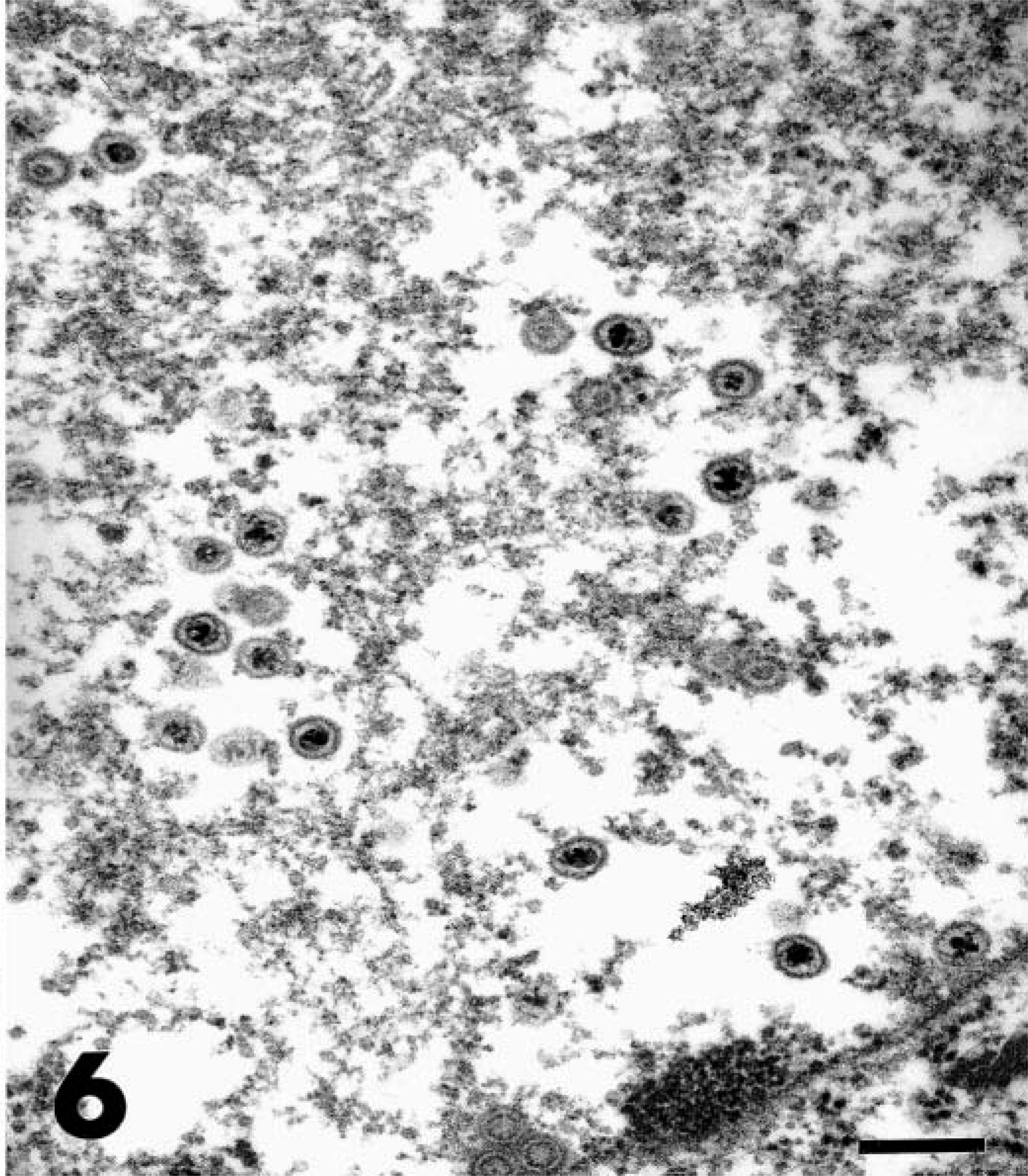
Transmission electron micrograph. Skin; cheetah. Higher magnification of intranuclear herpes viral nucleocapsids from Fig. 5. Bar = 200 µm.
Viral inclusions were evident only in keratinocytes and were more plentiful in early lesions with less intense inflammation. Inclusions in early lesions were present in the stratum spinosum in association with mild acanthosis and dermal infiltrates of eosinophils and mast cells. In more chronic and severe lesions, intranuclear inclusions and karyomegaly were found only rarely in keratinocytes of the deep rete ridges surrounded by florid eosinophilic and plasmacytic infiltrates. Extensive destruction of the epidermis by the inflammation possibly accounted for the paucity of inclusions in chronic lesions.
Many affected animals were cubs or juveniles (<1 year), but seven animals were subadults or adults ranging from 1 to 6 years of age (Table 1). Although 10 affected animals were from one genetic lineage, other lineages of the captive population were also represented. Overrepresentation of one lineage was likely influenced by mother to cub transmission.
The severity and unremitting nature of the dermatitis resulted in euthanasia of six cheetahs including one chronically affected animal that developed a cutaneous squamous cell carcinoma at the site of infection. Vaccination did not prevent the disease, and treatment was ineffective in most cases. All 20 cheetahs had been vaccinated with killed FHV as cubs (Fort Dodge Felovax PCT; 1 ml every 2–3 weeks from 6–8 weeks of age through 16 weeks of age) and then revaccinated annually. Advanced herpetic lesions were only minimally responsive to topical or systemic antiviral drugs, such as acyclovir, trifluridine, or idoxuridine, and only two animals improved on corticosteroids (Table 1). The most effective therapy was complete surgical excision, particularly when cryosurgery was used. The most effective preventive strategy was separating cubs from infected dams.
Most cutaneous lesions were preceded by clinical signs of upper respiratory tract infections or keratoconjunctivitis either in the affected animals or in the dams of affected cubs. The distribution of lesions in skin contacting ocular and salivary secretions implicates salivary and lacrimal glands as the source of virus infecting keratinocytes. Shedding of virus in saliva would also explain the resolution of lesions in cubs that were removed from infected dams. Although skin biopsies were the only tissues available from affected animals, herpes virus infection of salivary ducts has been noted in three other cheetahs (L. Munson, unpublished), indicating that these glands can be infected in cheetahs and providing support for a salivary gland origin of virus in the dermatitis cases. Cheetahs with herpesviral sialoadenitis had eosinophilic infiltrates associated with duct necrosis and intranuclear inclusions (Cowdry's type A) in the submandibular, lingual, and laryngeal salivary glands (Fig. 7).
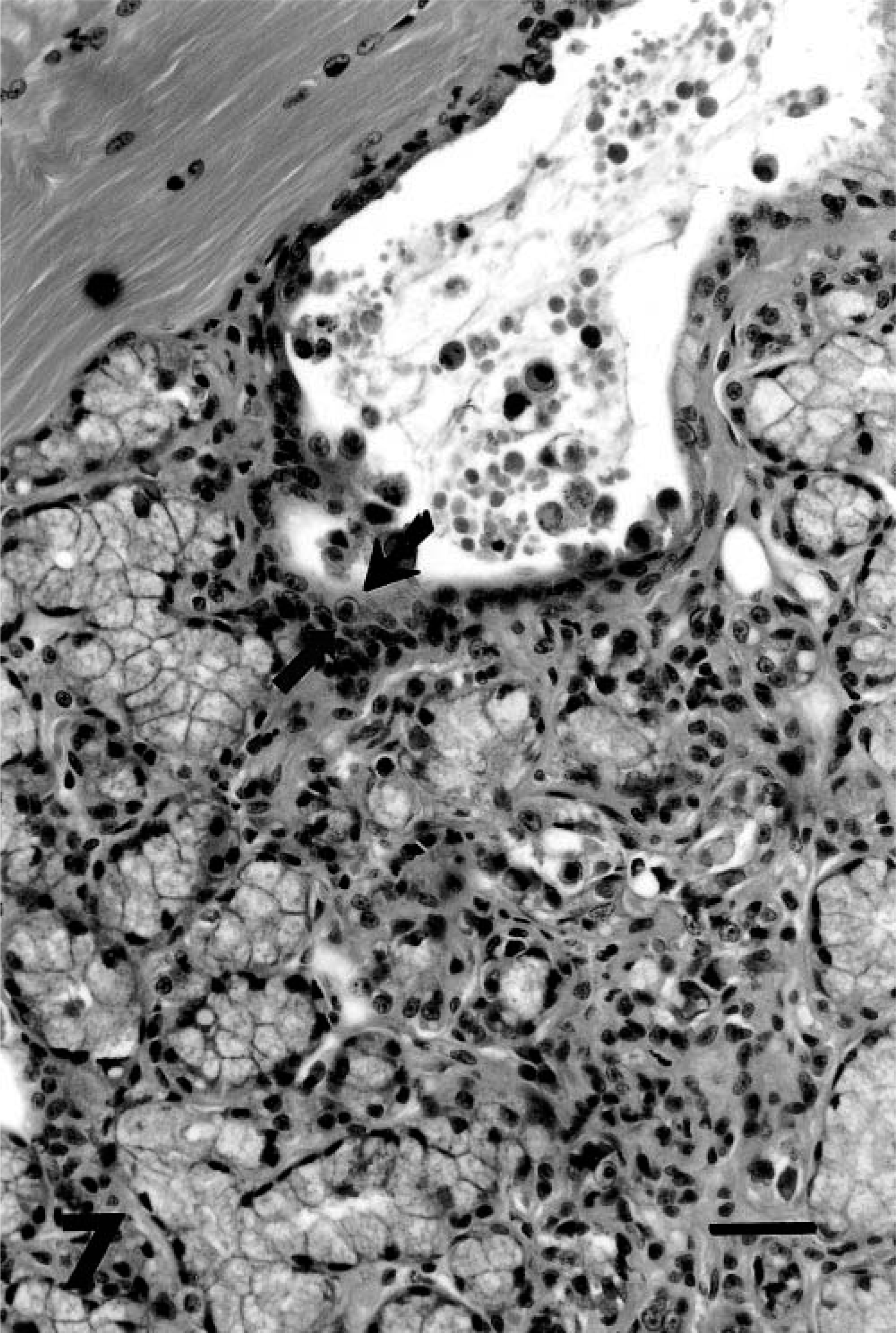
Salivary gland; cheetah. Necrotic duct and acinar epithelium have Cowdry's type A intranuclear inclusions (arrow) and mild eosinophilic infiltrates that extend into the stroma. HE. Bar = 50 µm.
FHV1 was considered the cause of the dermatitis and not a secondary infection because FHV-infected cells were deep (and not superficial) in the epidermis, some lesions improved with acyclovir therapy, and most lesions resolved after infected tissues were removed by cryosurgery. If salivary or lacrimal antigens were the stimulus for the immune reaction, lesions would have recurred after cryosurgery.
Overall, histopathology was the most reliable diagnostic method. Viral culture of skin lesions using Crandall-Rees feline kidney cells also was useful and confirmed the presence of FHV1 in eight of 10 cheetahs (Table 1). The two negative cultures were likely due to sampling methods because infected keratinocytes in general were rare and typically found only deep in the hyperplastic epidermis. Three cases were examined by direct fluorescent antibody methods using anti-FHV1 antibodies, but herpesvirus infection was only confirmed in one case. Serum herpes virus titers measured by serum neutralization tests were negative in three of five animals, despite recent vaccination with killed feline herpesvirus vaccine, typical of the limited response of cheetahs to this vaccine. 18
The lesions in cheetahs were notable in intensity of the eosinophilic, mastocytic, and plasmacytic response and by the extent of epithelial proliferation. This dermatitis has been misinterpreted as insect bite hypersensitivity and eosinophilic granuloma complex because herpes viral–induced karyomegaly and nuclear inclusions were overlooked. Differentiating herpes virus dermatitis from these other conditions is crucial because corticosteroids used to treat these other conditions may worsen FHV1 infections.
The FHV1-associated eosinophilic dermatitis in cheetahs resembles the ulcerative facial and nasal dermatitis that rarely occurs in FHV1-infected domestic cats. 7,8 Although lesions in domestic cats are less widespread and milder than those in cheetahs, the histologic features were identical, including the florid eosinophilic infiltrates, epithelial hyperplasia, and intraepithelial nuclear viral inclusions. 8 This unusual response to herpes virus in cheetahs and domestic cats differs from the spontaneous epidermal herpes virus infections described previously in domestic cats in which the lesions were ulcerative rather than eosinophilic and less persistent. 6,9 Although a novel herpes virus was suspected in cheetahs and domestic cats with dermatitis, the causative virus was confirmed to be FHV1 by sequence analysis. 8,16
The prevalence of this rare manifestation of FHV1 infection in captive cheetahs suggests an aberrant immune response to this virus in this species. This florid inflammatory reaction is typical of this species' response to infectious agents that usually are of low pathogenicity, such as feline corona virus and Helicobacter. 4,5,12,13 It has been proposed that cheetahs may be immunecompromised because their major histocompatibility (MHC) genes lack heterogeneity, a feature that could limit their responsiveness to some antigens. 14 Although a reduced T-cell response to FHV1 was detected in some cheetahs in lymphocyte blastogenesis assays, this diminished response was overcome by interleukin 2 supplementation, suggesting that MHC restriction was not the cause. 3,11 A primary or viral-induced immunodeficiency is also unlikely because their inflammatory responses contain abundant lymphocytes and plasma cells. The intense plasmacytic and eosinophilic infiltrates were more characteristic of a Th2-dominant response or hypersensitivity reaction in other species, 3 which can be genetic in origin or arise from a corticosteroid-mediated Th1 to Th2 shift. 2,15
Stress has been linked to persistent FHV1 diseases in other species, including domestic cats with FHV1 dermatitis 6,8,9 and can cause hypercortisolemia. 1 Although cortisol levels were not measured in the cheetahs with herpesviral dermatitis, captive cheetahs in general have marked adrenal cortical hyperplasia 12 and mean corticoid levels four times greater than wild cheetahs, indicative of chronic stress. 17 If cheetahs with FHV1 dermatitis had persistent high corticoid levels and if cellular immunity in cheetahs is modulated by cortisol in a similar manner as in other mammals, 2,15 then a shift toward a local Th2 cytokine environment would account for the predominance of plasma cells and eosinophils in lesions. 3 Current investigations are characterizing the cytokine profiles in the unusual inflammatory lesions of cheetahs.
In summary, a severe persistent eosinophilic and plasmacytic dermatitis with marked epidermal proliferation and intrakeratinocyte herpes virus has affected approximately 5% of the captive NA cheetah population. Additional cheetahs with similar (but unconfirmed) lesions have been identified, suggesting that this condition may be even more prevalent in the captive NA population. Because of the progressive and debilitating nature of the disease, current recommendations are to remove lesions with cryosurgery, quarantine infected animals, and prevent maternal transmission by hand-rearing cubs from infected dams. Risk factors for this disease should be identified to reduce the prevalence in this endangered species.
Footnotes
Acknowledgements
We thank Dr. N. Lung (Fort Worth Zoo) and Dr. H. Weston (New England Zoo) for contributing cases with sialoadenitis to the American Zoo and Aquarium Association Cheetah Species Survival Plan pathology surveillance.
