Abstract
The morphology of a duplication phenomenon of the canine Descemet's membrane (DM) is described in relation to signalment, history, and ocular disease status. Sixty-six canine eyes from the Comparative Ocular Pathology Laboratory of Wisconsin archives between 2000 and 2007 were examined. All cases were stained with hematoxylin and eosin and Alcian blue periodic acid-Schiff (PAS), while 14 cases were additionally stained with Masson's trichrome, picrosirius red, cytokeratin AE1/AE3 (CK), vimentin, and α-smooth muscle actin (SMA). Transmission electron microscopy (TEM) examination was performed in 3 corneas and in 1 normal control eye. Alcian blue PAS staining and TEM confirmed the basement membrane nature of the abnormal secondary DM. The thickness of the first DM, referred to as the corneal layer (CL) and the second or anterior chamber layer (ACL), were nearly the same, with no significant difference seen (P = .93). In 39% (26/66) of the eyes, a fibrous, collagenous matrix component was present between the CL and ACL, which contains vimentin-positive and α-SMA-negative spindle cells (14/14).The corneal endothelial cells in 7/14 eyes stained weakly with CK and strongly in 2 additional eyes. The most frequent histopathologically confirmed, clinical ocular histories were chronic glaucoma in 76% (50/66) of eyes, previous intraocular surgery in 36% (24/66), lens luxation in 21% (4/66), and blunt trauma in 15% (10/66) of the cases. We speculate that activation and migration of endothelial cells, in association with trauma or lens contact, play a role in the pathogenesis of this phenomenon.
Introduction
Descemet's membrane (DM), the posterior limiting membrane of the cornea, was named after French physician Jean Descemet (1732–1810) and is the exaggerated basement membrane of the corneal endothelium (posterior corneal epithelium). 26 DM is composed of collagen components, such as I, III, IV, V, VI, and VIII, and noncollagenous components, including laminin, fibronectin, nidogen, heparin sulphate, dermatan sulphate, tenascin, and P component. 22, 28, 30 Electron microscopically, 2 zones were described: a fetal, banded zone adjacent to corneal stroma composed of irregularly patterned mainly type I collagen, and a postnatal, posterior nonbanded zone composed of homogenous, fibrogranular material. Observations in mammals suggest that only the posterior nonbanded zone is synthesized continuously throughout adult life. 18, 25 Three distinct zones of collagen distribution can be identified. There is a thin anterior area mainly composed of collagen V and VI, followed by a middle zone of collagen IV and VIII, and a posterior zone mainly composed of III and IV. 29 DM has a thickness of 10–15 μm in adult dogs and 30 μm in horses. 26 The normal human DM measures a thickness of 3–4 μm at birth, increasing to approximately 5 μm in children and 10–12 μm in adults. 16, 25
To the best of our knowledge, there are no morphometric data available that describe increasing canine DM thicknesses as a function of age. However, this can be observed empirically.
The corneal endothelial cell layer is a single layer of cuboidal cells of mesenchymal or neural crest origin. 8, 20 The wound healing capacity of the canine endothelial cell is thought to be limited, as cell numbers decrease significantly with increasing age as well as after intraocular surgery. 11, 12 Several studies have shown that adult human endothelial cells present in the G0/1 phase of the cell cycle do not demonstrate mitotic activity, and very few mitotic figures are found after surgical procedures. 13, 32 This is in contrast to endothelial cells of rabbits, which are mitotically active and easily turn in the G2 phase and M phase of mitosis, an ability inversely related to age. 23, 36
Experimental studies in young dogs show a limited presence of mitotically active endothelial cells. Thus, the primary wound healing mechanisms are found to be hypertrophy, migration, and cell division. 2, 37 Relative mechanistic contributions may be calculated by mathematical modeling in postnatal canine corneal endothelial cells, yielding a division of 86% hypertrophy and 14% mitosis. 1
Laceration of DM leads to curling at the free margins. In smaller breaks, endothelial cells can extend across the defect, synthesize new basement membrane, and fill the defect with proliferating stromal fibroblasts. 34, 39
The wound healing response of altered corneal endothelial cells can lead to retrocorneal fibrous membrane (RCFM) formation posterior to the preexisting DM. RCFM is thought to be an end-stage disease process of endothelial cells that underwent mesenchymal transformation. 21 Descemet's lesions can reflect diseases affecting the endothelial cells as well as the corneal stroma. 34
Guenther (1956) was the first author reporting a “duplication” of Descemet's membrane in 2 human eyes and postulated an abnormal regenerative process of unknown cause. 10 The same author was able to provoke similar features in a rabbit model 6 months after corneal alkali burns.
A multilamellar appearance, thickening, or abnormal formation of DM is recognized in the following human corneal diseases: blunt trauma, posterior keratokonus, syphilitic keratitis, congenital hereditary endothelial dystrophy, posterior polymorphous dystrophy, and after DM detachment as a complication of phacoemulsification. 9, 19, 31, 34, 39 Snip et al. described an overgrowth of basement membranes and endothelial cells as descemetization and endothelialization over the iris surface in the human vitreous touch syndrome. 33
To the best of our knowledge, only 3 previous reports describe thickening and a multilamellar appearance of DM in dogs. One case report is of a German Shepherd Dog with acute, bilateral corneal endothelial degeneration of unknown cause, and 1 experimental study is of endothelial cell cryodestruction in young dogs. 2, 5 In one review, DM thickening and duplication and other ocular features were catalogued in canine eyes removed after phacoemulsification. 24
The purpose of the present study is to describe the histopathologic and electron microscopic appearance of an unusual duplication of DM to determine if there is any correlation to clinical findings and possible causes for these abnormalities in a series of 66 dogs.
Materials and Methods
All 66 canine eyes diagnosed between 2000 and 2007 with duplication of DM were selected from the Comparative Ocular Pathology Laboratory of Wisconsin (COPLOW) database. The signalment, duration, and diagnosis of corneal disease, ocular disease status, and systemic history of each specimen had been recorded and were collected from the accompanying submission forms.
The first DM, adjacent to the corneal stroma, was defined as corneal layer (CL) and the second layer, resembling a posterior DM, as anterior chamber layer (ACL) in consideration of the anatomic location.
The histopathologic analysis was based on 4 main criteria: 1) description of DMs, including anatomic location and thickness of both DM layers relative to those measured from normal control dogs; 2) quality and extent of a fibrous matrix component between the CL and ACL; 3) quality of endothelial cells; and 4) presence of further corneal and ocular diseases
The study includes 66 paraffin-embedded globes; HE and Alcian blue PAS–stained 5-μm sections were reviewed in all cases.
A collection of HE and Alcian blue PAS sections from 8 normal dog eyes were evaluated as a control group. These purebred dogs, of 8 different breeds, ranged between 5 and 12 years of age, with a mean of 8.75 ± 2.37 years. Three thickness measurements, taken in the axial and peripheral cornea, were averaged. The same procedure was performed in the matching duplication area of both DMs. Measurement site choice was not random, as ACL domelike extension and tangentially sectioned foci were avoided.
For the transmission electron microscopic imaging (TEM), one 10% formaldehyde-fixed half of a globe was postfixed in 4% paraformaldehyde. The duplication of DM was observed under a dissecting microscope (Leica, M7 FLIII). The corneal region of interest was dissected in 3 individual 2 × 2-mm samples, postfixed a second time in osmium, and embedded in acrylic resin. The other half of the same globe was embedded in paraffin and stained with HE and Alcian blue PAS for further confirmation of the gross and TEM examination findings. In addition, 2 paraffin-embedded samples and 1 normal control eye were deparaffinized, and the same procedure for the TEM investigation was performed as described above. Ultrathin sections (90 nm) were cut, mounted on 200-mesh copper grids, and then stained with 5% methanolic uranyl acetate and Reynold's lead citrate. A Philips 410 TEM (Philips Medical System, Andover, MA) was used for the examination and imaging.
Of the series, 14 randomly selected cases were stained with Masson's trichrome (standard histologic procedure) and picrosirius red modified by Kalscheur (Comparative Orthopaedic Research Laboratory, UW-Wisconsin). 35
The following immunhistochemical stains were performed: cytokeratin (1 : 75, Clone AE1/AE3, No. M 3515, Dako Corp., Carpinteria, CA), vimentin (1 : 200, Clone V9, No. M 0725, Dako Corp.), were used in a streptavidin–biotin–peroxidase technique (Labeled Streptavidin–Biotin kit, Code K0690, Dako Corp.). Alpha-smooth muscle actin (1 : 500, Clone 1A4, No. A5228, Sigma, St. Louis, MO) was used in a biotin-peroxidase technique (Vector Laboratories, Burlingame, CA, BA-9200) in combination with Vector NovaRed for peroxidase (Vector Laboratories).
Statistical analyses of the DM thickness in the CL, ACL, and control groups were performed by multivariate analysis of variance and paired Student's t-tests using SAS statistic analysis (SAS Institute Inc., Cary, NC). A P value of less than .05 was considered to be statistically significant.
Results
A total of 11,482 canine submissions were evaluated at the Comparative Ocular Pathology Laboratories of Wisconsin between 2000 and 2007. Of those, 66 eyes (0.6 %) from 66 dogs were identified as meeting the criteria for inclusion in the present study.
Clinical information
The age of the dogs at the time of enucleation ranged between 3 and 14 years, with a mean age of 8.95 ± 2.92 years. There were 25 castrated males (38%), 11 intact males (17%), 26 spayed females (39%), and 3 intact females (6%); the sex of 1 dog was not recorded. Labrador Retriever was the most common breed (11%, 7/66), followed by Golden Retriever (9%, 6/66), Boston Terrier (8%, 5/66), Cocker Spaniel (6%, 4/66), Parson Russell Terrier (6%, 4/66), and Shih Tzu (6%, 4/66). However, 26 pure dog breeds were represented in the study as well as 3 mixed breed dogs. Thirty-seven (56%) left eyes (OS) and 29 (46%) right eyes (OD) were represented. The median duration of recorded clinical signs prior to enucleation was 17 months, with a range of 0.1–84 months.
A history of corneal disease was reported for 39 dogs, and the ocular history was provided in all 66 cases. The 7 most frequently described corneal abnormalities are summarized in Table 1. Other ocular abnormalities include inflammatory processes such as anterior uveitis in 11% (7/66), pigmentary uveitis in 9% (6/66), uveal neoplasm with glaucoma in 6% (4/66), endophthalmitis in 3% (2/66), anterior segment dysgenesis in 2% (1/66), and 1 case was diagnosed with persistent hyperplastic primary vitreous.
Seven most frequently clinical findings in patients with duplication of DM: corneal history (n = 36 cases) and ocular history (n = 66 cases) (number of patients in parentheses). ∗

∗CD = corneal degeneration (stromal scarring, Haab's striae, crystalline degeneration); PK = pigmentary keratitis; CU = corneal ulceration; CIK = chronic interstitial keratitis with corneal abscess; SCCED = spontaneous chronic corneal epithelial degeneration; DC = descemetocele with keratomalacia; p. io. = postintraocular; IOM/PS = intraocular membrane/posterior synechia.
†With chronic glaucoma.
Systemic diseases were present in 14% (9/66) of dogs, including systemic hypertension (5% (3/66)), uveodermatologic syndrome (3 % [2/66]), diabetes mellitus (2% [1/66]), Cushing's syndrome 2% ([1/66]), and chronic ehrlichia infection and lymphoma (2% [1/66]).
Histopathologic findings
The main histopathologic findings are listed in Table 2. The PAS-positive Descemet's membrane in all cases is doubled or layered. The 2 or more layers were either separated or nonseparated in varying degrees. The separated DMs showed cleftlike spaces; these were filled with a fibrous matrix component of varying thickness in which were found rare spindle cells and pigmented cells (Figs. 1,2). In other locations, there were localized areas with no separation between layers (Fig. 3). The DM closer to the posterior corneal layer (CL) has a smooth, regular appearance. In all cases, the endothelial layer was not in contact with the CL, but rather was in contact with the second DM (ACL). Single breaks of the CL in the peripheral as well as axial cornea were observed in 17 eyes. These do not include the limbal corneal incisions of patients that had previous intraocular surgery. The ACL is more irregularly formed, with breaks or broad, domelike extensions of the basement membrane. Less frequently, foci of 3 or more layers also were found. The ACL showed PAS-positive staining (Figs. 2,3). The extent of duplication of DMs varied in different anatomic locations of the cornea. The following locations were recorded: multifocal (peripheral and in the axial portion of the posterior cornea), peripheral, limbal, axial, and continuous (over the entire length of the posterior cornea). The mean thickness of both DM layers was approximately 14 μm (Table 2). The median value was 15 μm in the CL and 11.25 μm in the ACL. The thickness of the ACL showed a higher variability within each sample. Within the case series, the thickness ranged between 5–55 μm. The thicknesses of the CL and ACL were not statistically different (P = .93). The control group had thicknesses of 18.13 ± 4.67 μm in the peripheral and 16.9 ± 5.35 μm in the axial cornea, which were not statistically different (P = .62). The thickness of the CL to the peripheral (P = .13) versus axial thickness (P = .32) of the control group also was not statistically different.
Pathohistomorphologic characteristics of duplication of DM. ∗
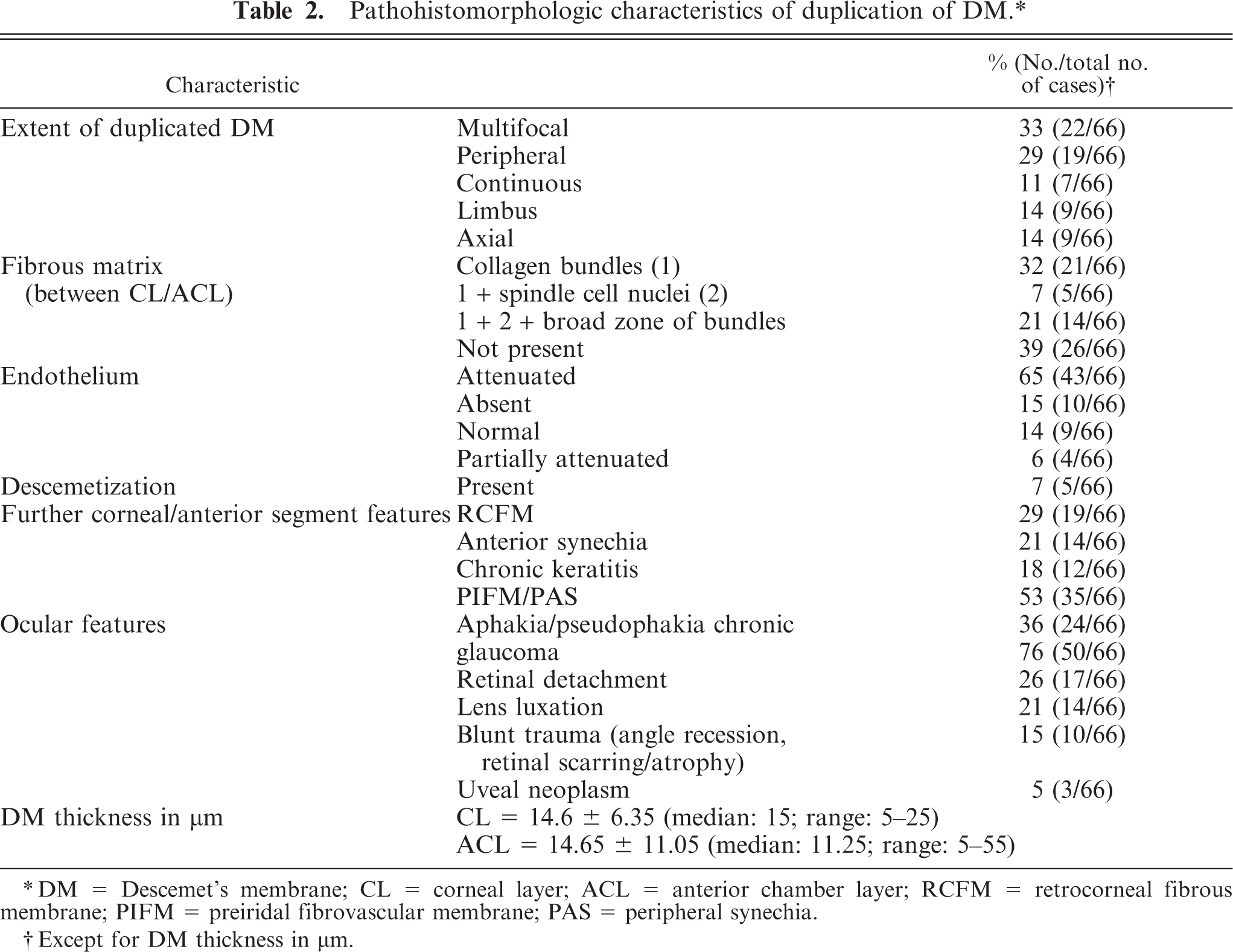
∗DM = Descemet's membrane; CL = corneal layer; ACL = anterior chamber layer; RCFM = retrocorneal fibrous membrane; PIFM = preiridal fibrovascular membrane; PAS = peripheral synechia.
†Except for DM thickness in μm.
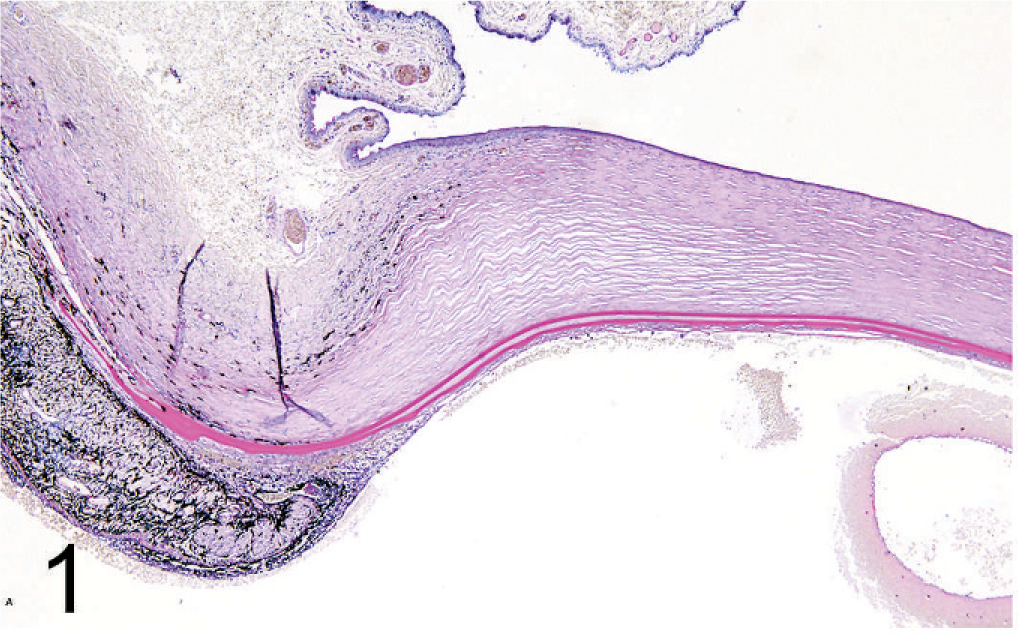
Histopathologic appearance of canine duplication of DM. Photomicrograph of the limbus, peripheral cornea with duplication of DM, anterior synechia and retrocorneal membrane, and hemorrhage and proteinous material in the anterior chamber. Alcian blue PAS (200×).
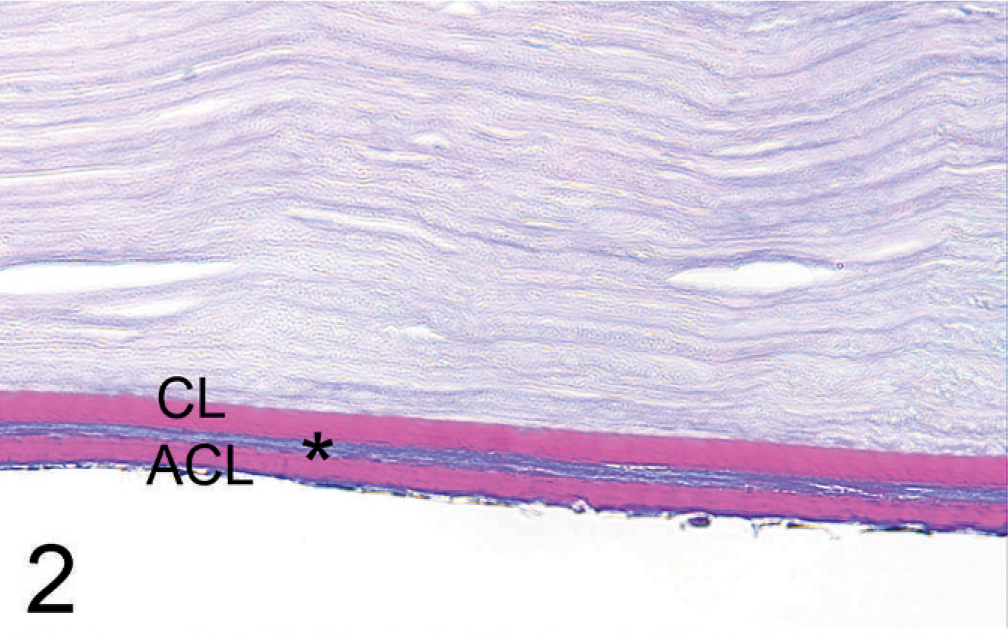
Detail image of the CL, the ACL, and a zone of fibrous matrix in between (∗).
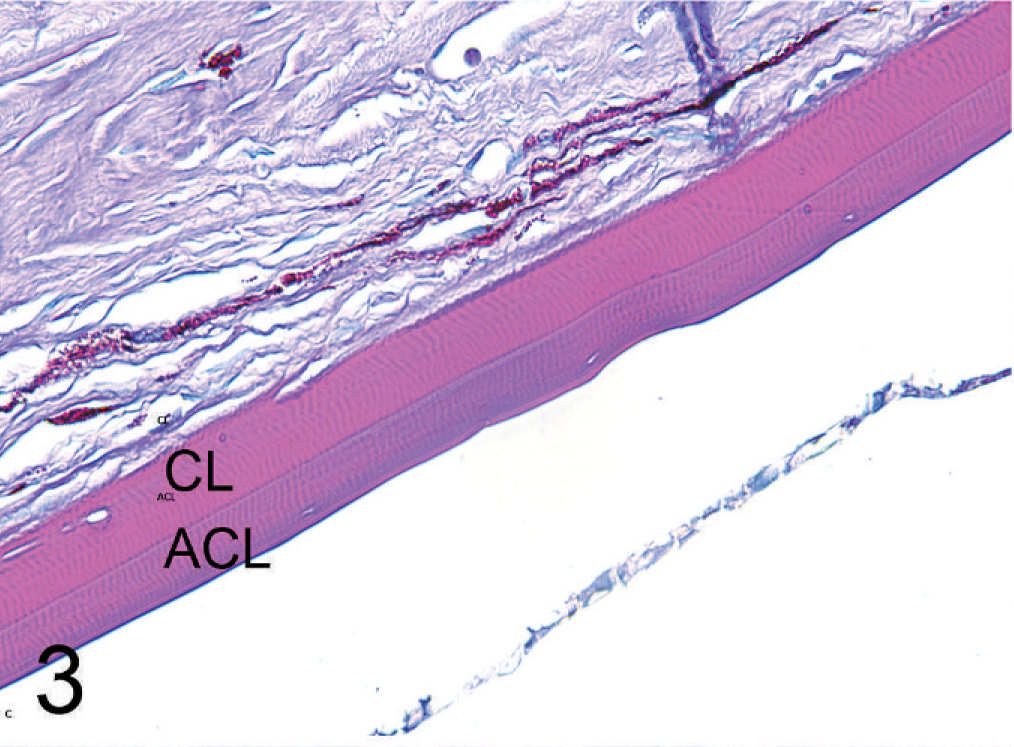
Detail image of a nonseparated duplication of DM with a distinct junction between the CL and ACL. The corneal endothelium is detached. Alcian blue PAS (600×).
The fibrous matrix component between the 2 layers was present as a dense, avascular, fibrous connective tissue and was observed in 61% of the cases (40/66). All fibers stained blue with Masson's trichrome stain, suggesting collagen. Furthermore, all picrosirius red–stained samples showed enhanced birefringent characteristics of collagen in polarizing light (Fig. 4). There were rare scattered spindle cells embedded in the fibrous tissue between the 2 layers, which stained negative for CK and SMA but positive for vimentin. Histologically, endothelial cells were not recognizable in the fibrous intermatrix component. The endothelium was attenuated in the majority of the cases but appeared normal in 14% (9/66). The CK stain was weakly positive in <50% of the cells in 7/14 cases, markedly positive in >50% in 2/14 cases, and negative in >90% of cells in 5/14 cases (Fig. 5). In 5 samples, additional “descemetization” with “endothelialization” was found. 19 This phenomenon is a continuous overgrowth of varying extent of PAS-positive basement membrane arising from the DM, carpeting intraocular structures such as the drainage angle and anterior iris leaflet (Fig. 6). In 1 of the 5 cases, the endothelium adherent to the Descemetization showed strong CK-positive staining. In 2 of the 5 cases, the basement membrane extended over a paraxial anterior synechia.
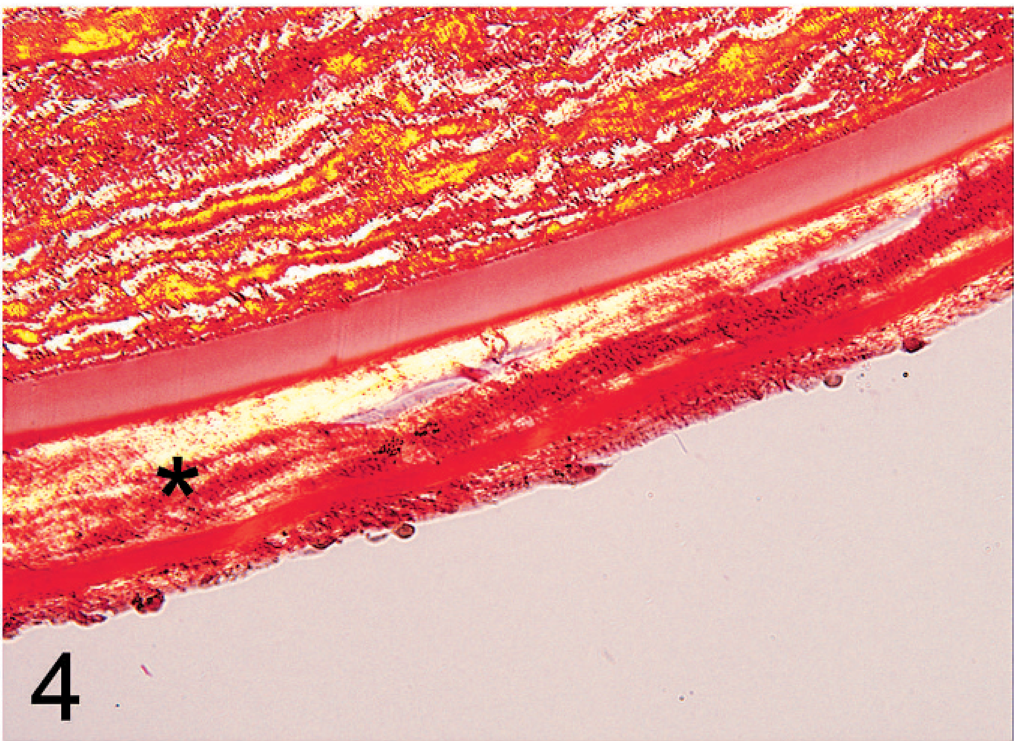
Detail image highlights the birefringent capacity of collagen fibers of the fibrous matrix (∗). Picrosirius red stain in polarizing light (600×).

Photomicrograph of weakly cytokeratin-positive stained corneal endothelial cells. Immunhistochemical cytokeratin stain (AE1/AE3) (600×).
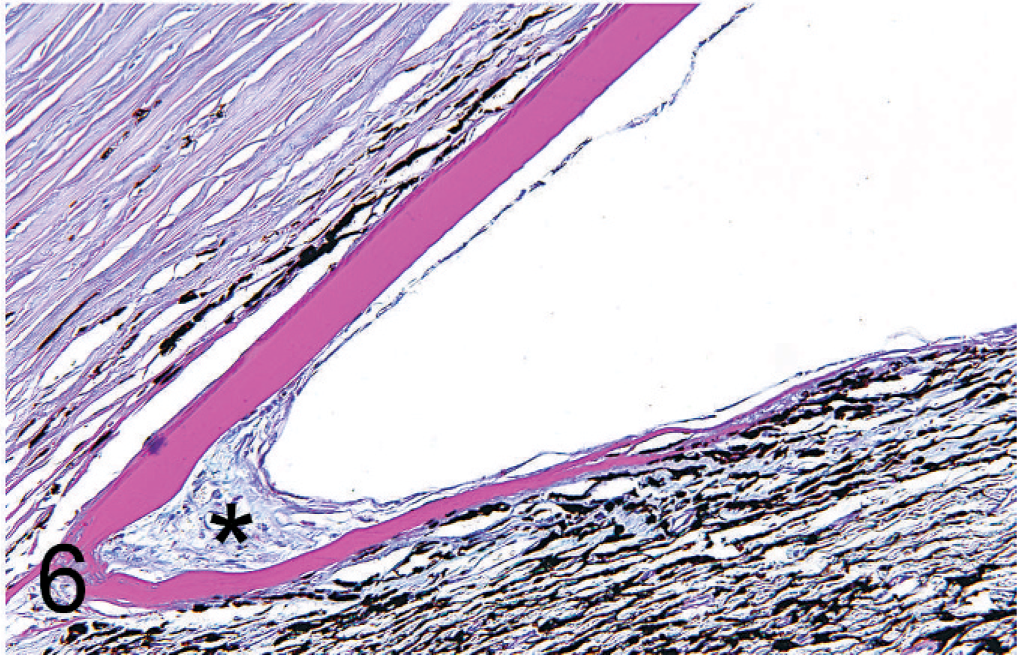
Detail image of Descemetization and endothelialization over the drainage angle, with underlying fibrous connective tissue (∗). Alcian blue PAS (600×).
Further corneal alterations are summarized in Table 2. The majority of the histopathologic examinations confirmed the clinical diagnosis. Additional observations included retrocorneal membrane formation in 29% (19/66), a broad PAS-positive epithelial basement membrane with severe chronic stromal fibrosis and pigmentary keratitis in 7% (4/66), perforation in 5% (3/66), Haab's striae in 5% (3/66), band keratopathy in 2% (1/66) as well as epithelial dysplasia, stromal thinning, and stromal dystrophy. There was no corneal history reported in 34 submissions; out of these, no corneal histopathologic alteration was found in 20% (13/66) of the cases. Further ocular histopathologic observations were mostly consistent with the history reported (Table 2). However, blunt trauma was reported in only 8% (5/66) but was diagnosed histopathologically in 15% of the cases (10/66) based on the presence of iridociliary angle recession, severe regional retinal scarring, and atrophy.
Transmission electron microscopic findings
TEM of 3 affected eyes confirmed the histopathologic finding of DM duplication with the formation of a second basement membrane. There were no major differences observed in comparison between the CL and the normal DM of the control dogs (Figs. 7,8). The original, native DM or CL is homogeneous, with uniformly distributed bridges interdigitating into the intact posterior corneal lamellae without specific attachment elements, also named interfacial matrix zone. 22 The banded and posterior nonbanded zone can be differentiated. The fibrous intermatrix component includes irregular multilamellated basement membrane components and irregularly arranged dense fibers of lower electron density. The anterior junction of the ACL to the matrix component is irregularly formed, containing horizontal and vertical basement membrane extensions (Fig. 9a). In contrast, the CL/posterior corneal “interfacial matrix zone,” or stroma junction, appeared straight and regular (Fig. 9b). The anterior banded zone and the posterior unbanded zone were not observable in the ACL (Fig. 9a, b). These ACLs can be separated with or without a fibrous intermatrix component or nonseparated, which can be differentiated by a fine junctional line (Fig. 11). Some irregularities were observed in the mid to posterior, nonhomogenous ACL. These consisted of irregular longitudinal and transversal translucent zones, inclusions of cell organelle remnants, and broad-banded linear collagen filaments (Figs. 10,11).
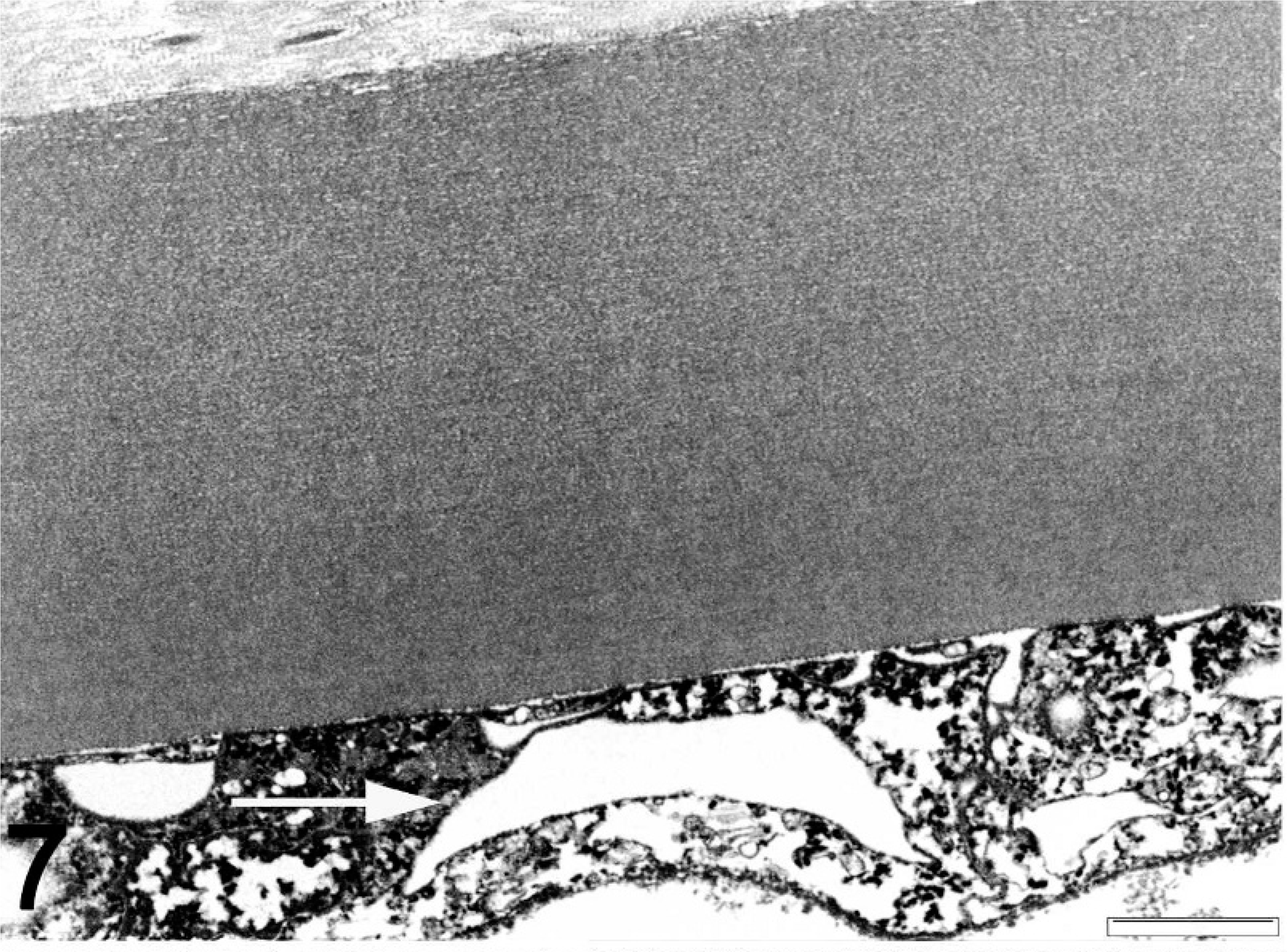
TEM image of deparaffinized normal globe. DM of a normal dog; the endothelial cells are artifactually vacuolated (→). Bar = 2 μm.

TEM image of deparaffinized affected globe. Higher magnification of the CL with a homogenous appearance. Bar = 2 μm.
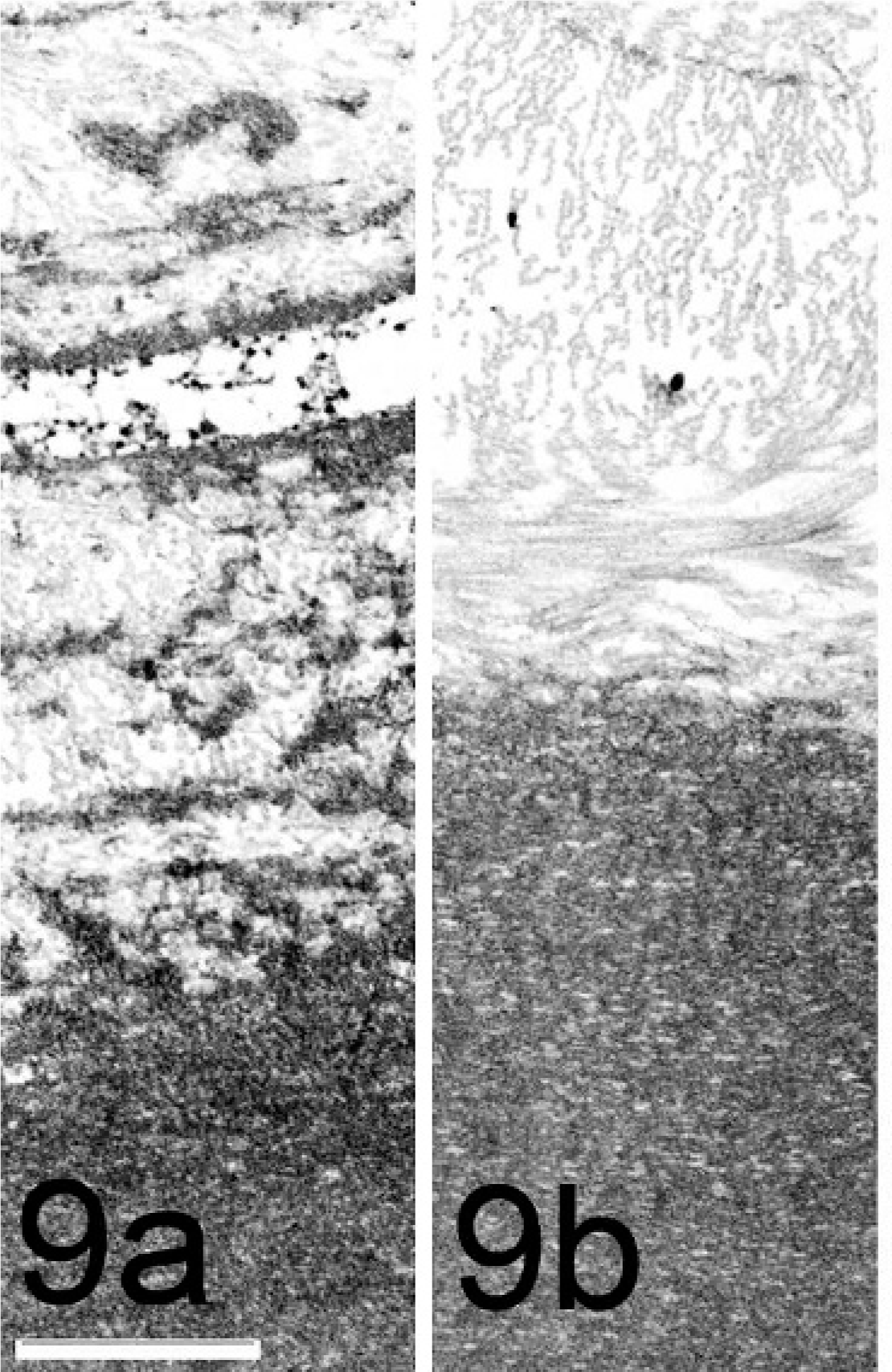
TEM image of deparaffinized affected globe. Note the irregular interdigitating, electron-dense basement membrane extensions of the ACL to the intermatrix component. Bar = 1 μm. Fig. 9b. Note the regular interfacial matrix of the CL connecting to the posterior stromal lamellae. Bar = 1 μm.
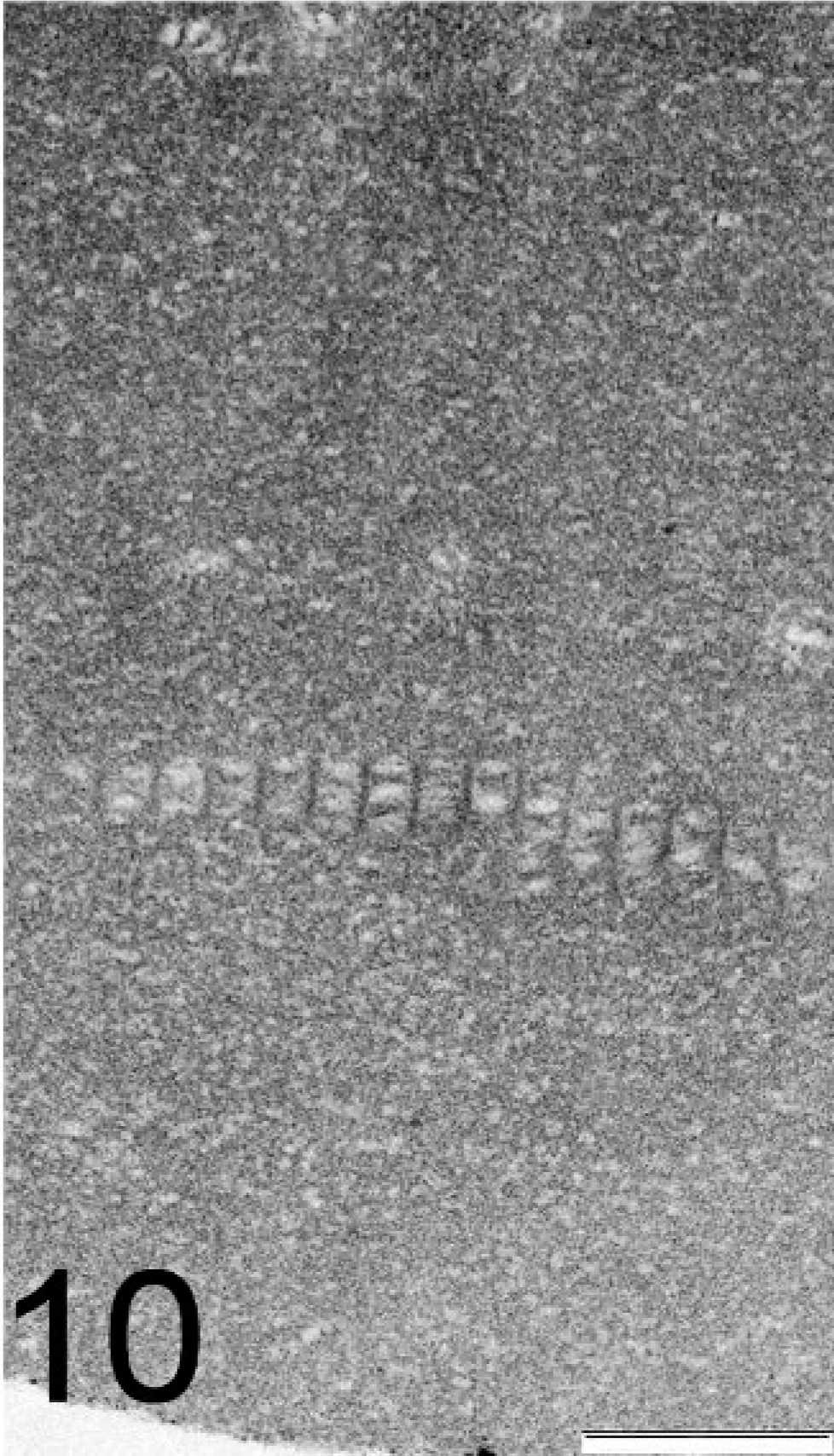
TEM image of deparaffinized affected globe. Note the broad banded zone in the ACL. Bar = 500 nm.
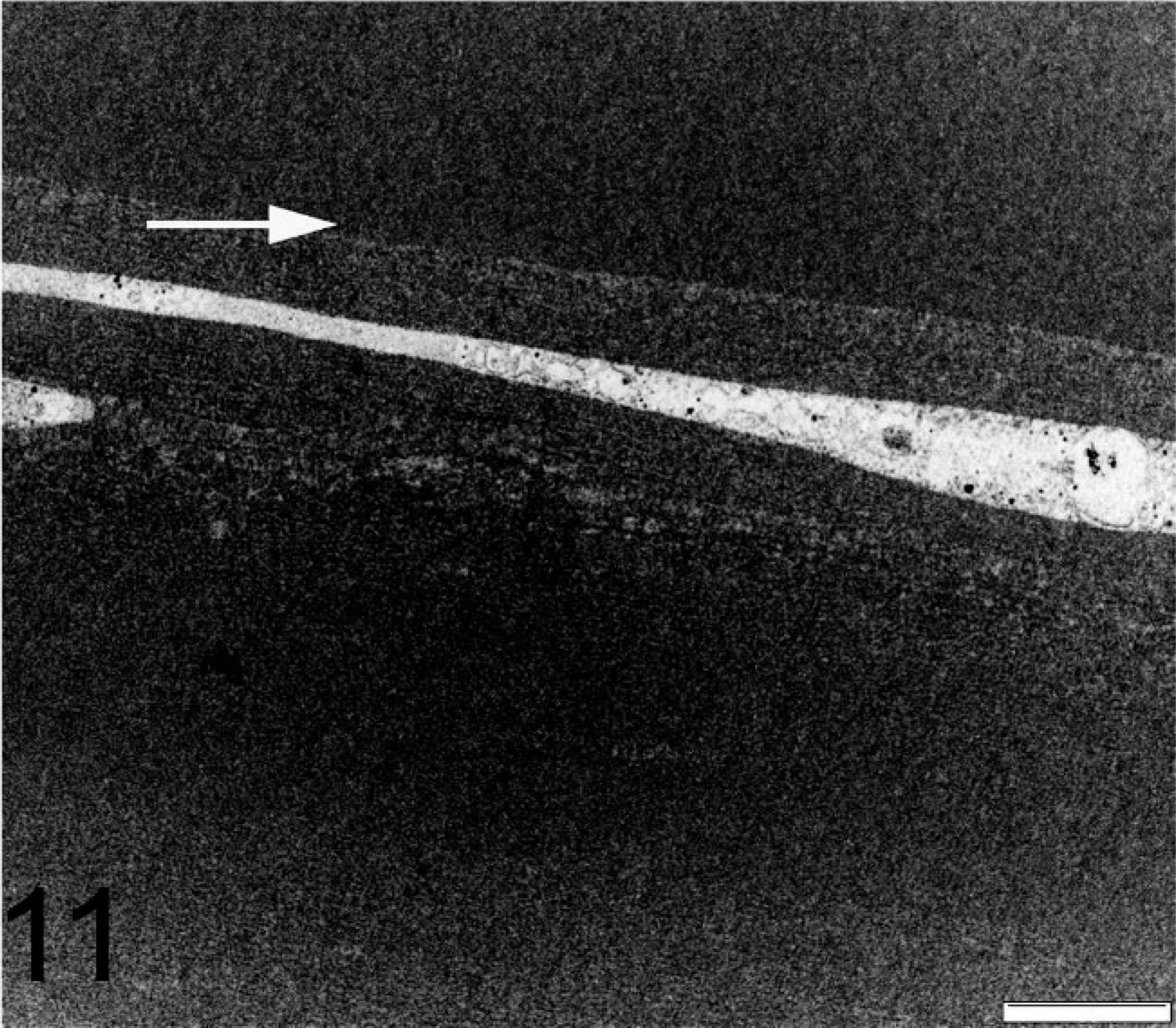
TEM image of deparaffinized affected globe. Note the junction between the nonseparated CL and ACL and cell organelle remnants in the ACL (→). Bar = 2 μm.
Discussion
Reports describing alteration of DM in dogs with similar features observed in the present study are few. Little is known about canine endothelial cell response to ocular trauma or chronic ocular disease. 2, 5, 24, 29 Classic thinking holds that the endothelium has a limited ability to respond to injury, but we observed a very intense response of endothelial cells forming a second basement membrane. Furthermore, in a limited number of cases, Descemetization and endothelialization of the iridocorneal angle and anterior iris leaflet were observed. We hypothesize that endothelial cells are activated and proliferate following appropriate stimuli, considering the long distances of newly formed basement membranes lining surfaces in the eye. However, the classical response of endothelial reaction is limited to elongation and migration. 29 The phenomenon of Descemetization and endothelialization, also known as ectopic surface proliferation, is reported in dogs for the first time, while it is known to occur in humans following trauma or inflammation. Proliferated endothelial cells grow along surfaces such as the trabecular meshwork, iris, lens, ciliary body, and vitreous, producing a Descemet's-like membrane. In humans, this is more commonly found in children after penetrating or blunt trauma. 27, 34 This is contrary to clinical and laboratory evidence suggesting that endothelial cells do not normally proliferate. 33, 34 In the present series, dogs with Descemetization were relatively young, in the age range of 3–5 years compared with the mean age of 8.75 years for the entire group.
In the remaining cases, we suspect an initial activation of endothelial cells is followed by duplication of the DM. The duplication phenomenon is rarely seen in the human eye, although less organized multilayering or formation of new basement membrane is well known. 9, 10, 19, 27, 31, 34, 38
The phenomenon of duplication of DM in the COPLOW database has a moderate frequency. It is seen in cats but not in horses or birds. Clinically, the canine duplication of DM was not recognized. This might be partially explained by the fact that in approximately 80% of the eyes, there were corneal alterations that prohibited an accurate posterior corneal examination. In the remaining cases, the duplication phenomenon might have been obscured by stromal edema or misinterpreted as posterior scar formation. Similar limitations are present in human patients with retrocorneal fibrous membranes, which were clinically diagnosed in only 1 of 11 patients. 7
The median time interval between the recognized clinical onset of ocular problems and time of enucleation was 17 months, with a wide range of 0.1–84 months. This means that the ACL grew to the same thickness as the CL in a very short period of time.
The PAS-positive staining and TEM investigations imply that the ACL maintains typical morphology for DM. By TEM, the CL appeared as the unaltered original DM. Interestingly, there were several alterations and irregularities found only in the ACL. First, localized broad-based, dome-shape extensions were seen in otherwise parallel membranes. These extensions were not typical of guttata formations, which are often seen in humans with Fuchs' corneal dystrophy. In Fuchs' dystrophy, it is assumed that endothelial cells undergo epithelial metaplasia and that the altered DM forms warts of altered basement membrane, which is thought to reflect a slow degeneration of individual endothelial cells. 4, 14 Our findings show more similarity to Hassall-Henle warts, found in the peripheral cornea of elderly patients with an idiopathic pathogenesis. 39
The presence of broad-banded zones in the ACL (Fig. 10) could refer to deposits composed mainly of collagen VIII. In normal DMs of older patients, in Fuchs' dystrophy and in humans with iridocorneal-epithelial syndrome, a similar wide-spaced collagen VIII of unknown significance could be found. 25 Collagen VI shows similar periodic banding of filaments and fibrils demonstrated in fibroblast cultures. 6 Scattered areas of cell components were included in the ACL (Fig. 11). However, because of the relatively poor quality of the processing, no further interpretation of the cellular origin could be made. The most common finding of the ACL was the irregular appearance seen in most of the membrane, which was interpreted by experts consulted as “stress collagen.”
Surprisingly, the mean thicknesses of both layers is approximately equal and not statistically different. An observer influence is present because measurements of focal thicknesses on folds were avoided. However, the ACL thickness varies much more than the CL. The median thickness for the ACL was lower (11.25 μm) and had a large range of 5–55 μm. The median thickness of 15 μm in the CL was slightly higher but still relatively similar. This led to the suggestion that there is a programmed thickness of DM related to the age. One can assume that under stress conditions (trauma), endothelial cell mechanisms are activated and a second DM of similar thickness is synthesized.
The sum of the CL and ACL thicknesses was statistically higher than the peripheral DM (P = .0097) and the axial DM (P = .0043) of the control group. This supports our observation that the ACL is a second, newly formed basement membrane and not a separation or splitting within the DM. A splitting appearance can be seen at the surgical incision, but this duplication phenomenon is found in patients without intraocular surgeries as well. Furthermore, the duplication was restricted to the axial cornea, in a marked distance from the corneal incision site, or duplication could be seen continuously over the entire length of the posterior cornea.
The fibrous component between the CL and ACL is composed of irregularly arranged collagen bundles intermixed with basement membrane–like strands, which are mainly located at the junction of the ACL (Fig. 9a). Rare spindle cells were found, which occasionally contained pigmented granules. The immunhistochemical staining suggests that these are fibroblasts.
There are similar features to RCFMs, despite the demonstration of myofibroblasts, which could be found in 8/11 human eyes and are thought to be involved in the corneal wound healing mechanism. 7
In the RCFM, the corneal endothelium undergoes endothelial mesenchymal transformation by the action of fibroblast growth factor 2 and a protein factor released by polymorphonuclear leukocytes. The transformed corneal endothelial cells (fibroblasts) can produce both basement membrane and fibrillar matrix. 21, 28 In the present case series, the origin of the fibrous matrix component could not be determined. It might arise from the posterior corneal stroma growing through breaks in the CL or from transformed endothelial cells, as would be the case in RCFMs. The latter is unlikely, because there are several cases that show a fibrous matrix component with an underlying intact corneal endothelium attached to the ACL.
The endothelium, when present, was mostly attenuated and was absent in 15% of the cases. The absence of the endothelium might be artifactual. Endothelial cells were evaluated for the possibility of epithelial transformation in HE sections. A transformed epithelial phenotype with abnormal DM production is a pathognomonic feature of human posterior polymorphous dystrophy and iridocorneal endothelial syndrome (ICE syndrome), which were not recognized in this series. 3, 13 However, the CK AE1/AE3 staining was weakly positive in 7 cases and strongly positive in 2 more. TEM images failed to demonstrate microvilli or cytokeratin fibrils. 3 In conclusion, the immunohistochemistry may hint that endothelial cells have undergone incomplete epithelial transformation. The endothelial layer was in contact with the ACL or lining the inner aspect of the RCFM. One possible pathogenic mechanism might be a trauma-related (blunt, surgery-related, lens luxation) temporary detachment of the native endothelial cell layer, which reattaches to the DM in a slightly different position, causing activation of the endothelial cells. Endothelial cells without lateral contact inhibition were activated in vitro and entered in the G1 phase of the cell cycle. A similar mechanism might occur if basolateral contact is lost temporarily after trauma. 17
The phenomenon of Descemet's membrane doubling is interesting to both clinicians and basic scientists. It has important implications for the understanding of the cell biology of the corneal endothelial cell and its basement membrane. Clinically, duplication of DM leads to corneal opacity of the cornea and reduces the visual outcome after phacoemulsification. Furthermore, Descemetization could lead to secondary glaucoma. In summary, we have described a canine DM duplication phenomenon for the first time. Further research is required to clarify the origin of the duplication phenomenon and to understand the causative mechanism.
Footnotes
Acknowledgements
We thank Dr. Ivan Schwab, Dr. William H. Spencer, and Dr. Daniel Albert for expert consultations. The authors also would like to thank Doris Dubielzig, Melanie Rawlings, and Lea Loehndorf for their helpful advice.
