Abstract
Subcutaneous estrogen pellet implantation is commonly practiced to induce tumor promotion in nude (Nu-FOXnl nu ) mice for xenograft tumor modeling. Twelve of 23, 18-week-old nude mice developed dysuria after estrogen pellet implantation. Physical examination revealed variably distended abdomens with palpable distended bladders. All mice were euthanatized and postmortem examinations were made. Blood samples of the mice were collected, and serum estrogen concentration was measured. Postmortem examination revealed variable dilation of the urinary bladder and unilateral or bilateral dilations of ureters and kidneys. Microscopically, 12 of 23 mice and 13 of 23 mice showed dilated urinary bladder and hydronephrosis, respectively.
Background
Estrogens have been associated with tumor development in many experimental animals and humans. Estrogen acts as a tumor promoter in rat liver carcinogenesis 13 and spontaneous human endometrial 6 and mammary carcinomas. 10 Therefore, estrogen pellets are frequently used as a tumor promoter in animal models of human neoplasia. Besides being a tumor promoter, estrogen has been shown to increase urethral tone in mice. 2
Urogenital infections have been shown to flare up in elderly women. In postmenopausal women, estrogen therapy reduces vaginal atrophy, 12 which normalizes vaginal flora, thereby effectively reducing urinary tract infections. 11 On the contrary, the hydronephrotic effect of high-dose estrogen was observed in DDD (deutsche Maus at Denken) strain male mice. 9 In another study conducted by Buhl et al., 1 estrogen-treated adult male CF1 (Carworth Farms 1) mice showed an increase in urine volume. In both male and female gonadectomized DS (mutation disorganization) strain mice exposed to 17β-estradiol, significant urine retention and 100% hydronephrosis were observed. 7 An additional study found that 17β-estradiol treatment administered via subcutaneous pellets caused hydronephrosis and urine retention in female C57BL/6J mice. 8 The results observed in lab animals seem to be contradictory to the studies conducted in humans, possibly because of a different dose regimen of estrogen in lab animals and because postmenopausal women are deprived of estrogen for a very long time, which could trigger many pathways.
Estrogen implantation is commonly practiced to promote tumor growth in nude (Nu-FOXn1nu ) mice. After 1 week of estrogen implantation, 1 × 107 T47D human breast cancer cells were injected at the right inguinal region. Multiple mice developed dermatitis, bloated abdomens, or both after 4 weeks of estrogen implantation. The tumor growth study was terminated, and mice received supportive care before euthanasia.
In this case report, we describe similar clinical and pathologic conditions in estrogen-implanted nude mice as observed in the above mice studies; however, it reports for the first time that estrogen implantation is linked to urine retention and hydronephrosis in nude mice.
History, Gross Findings, and Laboratory Results
Twenty-five 4-week-old female Nu-FOXn1nu mice were obtained from Charles River Laboratories (Wilmington, MA). A 1.7-mg 17β-estradiol pellet (Innovative Research of America, Sarasota, FL) was subcutaneously implanted with a trochar in the subscapular region at 43 days of age. No inflammation or redness around the implantation area was observed after 2–3 days. Three weeks after estrogen implantation, pruritus and ulcerative dermatitis were observed in the perineum in some mice. The lesions then spread to the hindlegs, over the dorsum of the tail head, and, in 1 mouse, to the forelegs. After 4 weeks of estrogen implantation, mice looked bloated but otherwise healthy. On physical examination, in living mice, the urinary bladder appeared distended and was easily palpable.
The mice were euthanatized by CO2 asphyxiation and postmortem examinations were performed. Postmortem examination revealed a variably enlarged urinary bladder filled with clear yellow urine, mild to moderate hydroureter, and unilateral or bilateral hydronephrosis (Fig. 1). The majority of the mice had variable dilation of the uterus. After euthanasia, blood was collected via cardiocentesis from 14 mice and tested for serum estrogen levels. In the rest of the mice, blood was not collected. Two of 14 mice had no estrogen pellet and were found to be healthy (Fig. 2), having normal (3–9 pg/ml) serum estrogen levels. Six of 14 mice had increased estrogen levels (36–260 pg/ml).
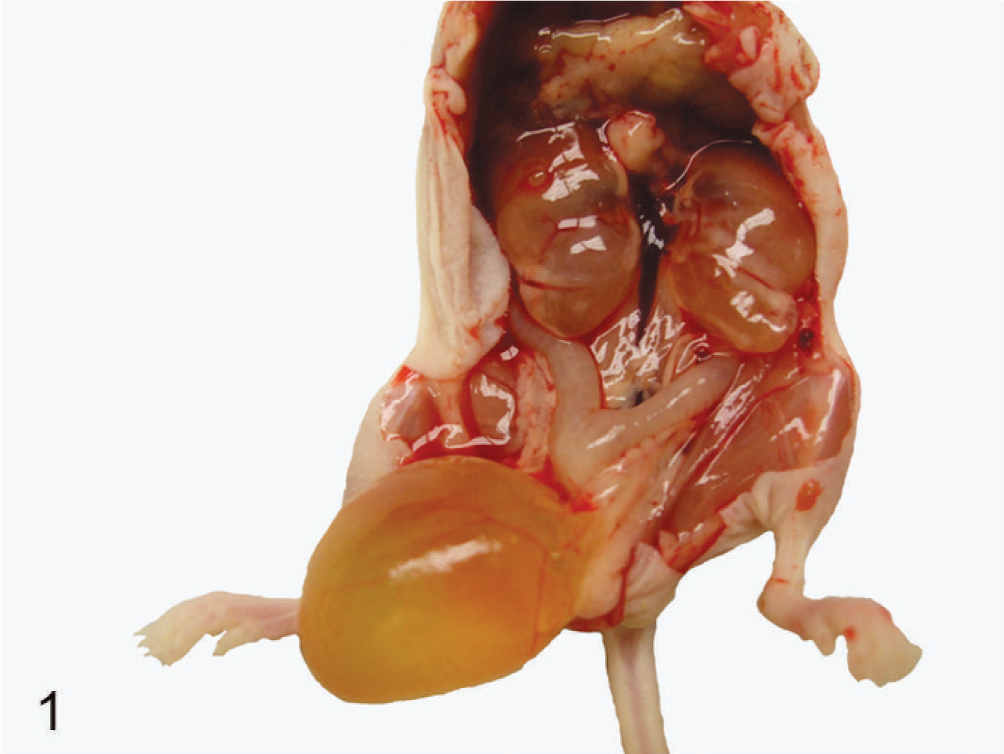
Urogenital tract of an affected nude mouse. Bilateral hydronephrosis and enlarged bladder filled with urine in a nude mouse.
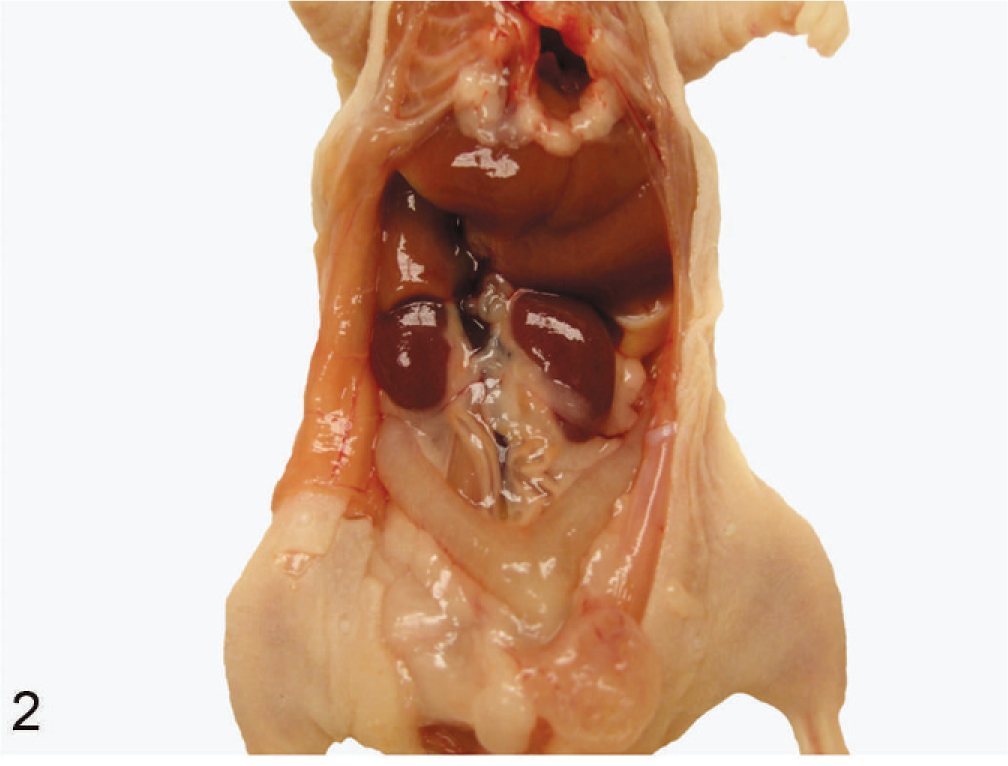
Urogenital tract of an unaffected nude mouse. Grossly, kidneys and bladder look normal.
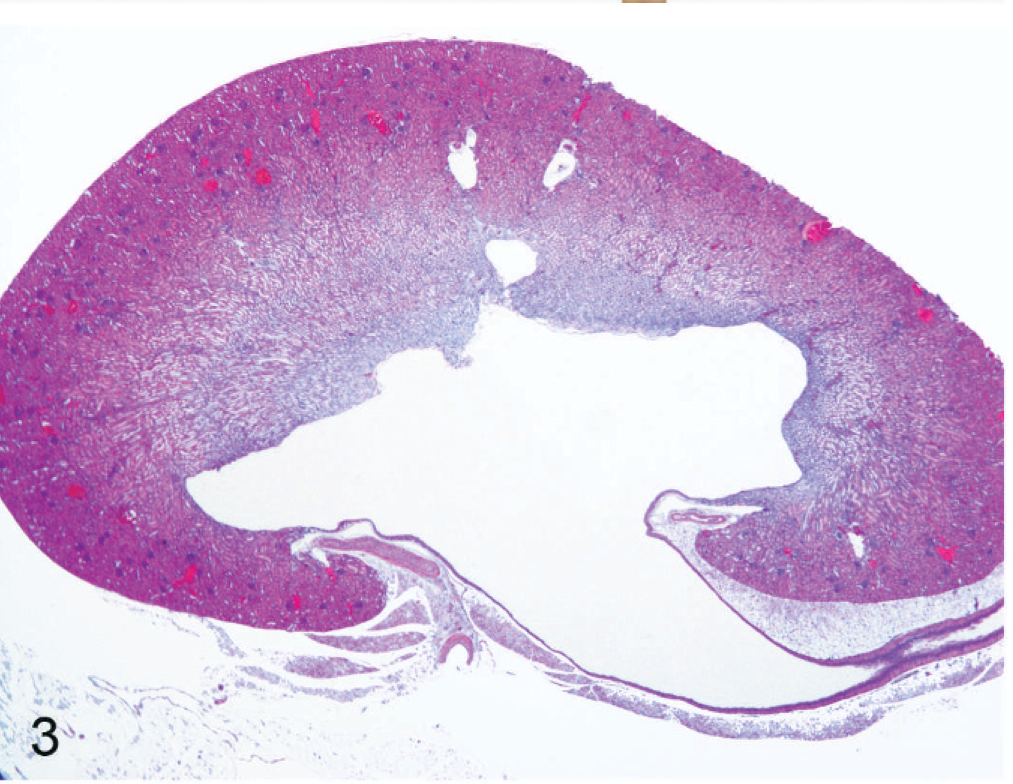
Kidney of an affected nude mouse. The renal pelvis is dilated; the ureter is moderately dilated. Note the flattened renal papillae. HE stain.
Differential Diagnosis
Differential diagnosis for hydronephrosis in animals includes anything that obstructs the urinary tract at the level of the urethra, urinary bladder, or ureters. Urinary calculi and severe cystitis are 2 of the common causes of urinary tract obstruction in domestic animals. Masses in the caudal abdomen or pelvis, such as abscesses, can cause external pressure to the urinary tract and cause a functional obstruction. In addition, displacement of the bladder through perineal hernias and acquired urethral or ureteral strictures can block the flow of urine.
Perineal ulcerative dermatitis preceded the clinical signs; therefore, an acquired stricture from scarring around the perineum as sequelae to dermatitis or a blockage from cystitis could not be ruled out with initial clinical presentation. Blockage because of scarring, cystitis, urinary calculi, or pressure from adjacent masses was not evident in any of the mice. Therefore, the most likely cause for the clinical findings was urine retention associated with estrogen administration.
Microscopic Findings
Microscopically, the distended urinary bladders had flattened epithelia and decreased thickness of the muscular layer (Fig. 4). All mice (12) that had distended abdomens had microscopically variable degrees of dilation of the urinary bladder, unilateral or bilateral dilation of the kidneys, and mild to moderate hydroureters. Six of the histologically affected mice had multifocal infiltrates with low to moderate numbers of plasma cells within the tunica muscularis (Fig. 4, inset). Hemorrhages or inflammatory cells were not evident within the bladder lumen in any of the mice examined. The mice with ulcerative dermatitis of the perineum had no histologic evidence of bacterial infection; however, lesions were not cultured.
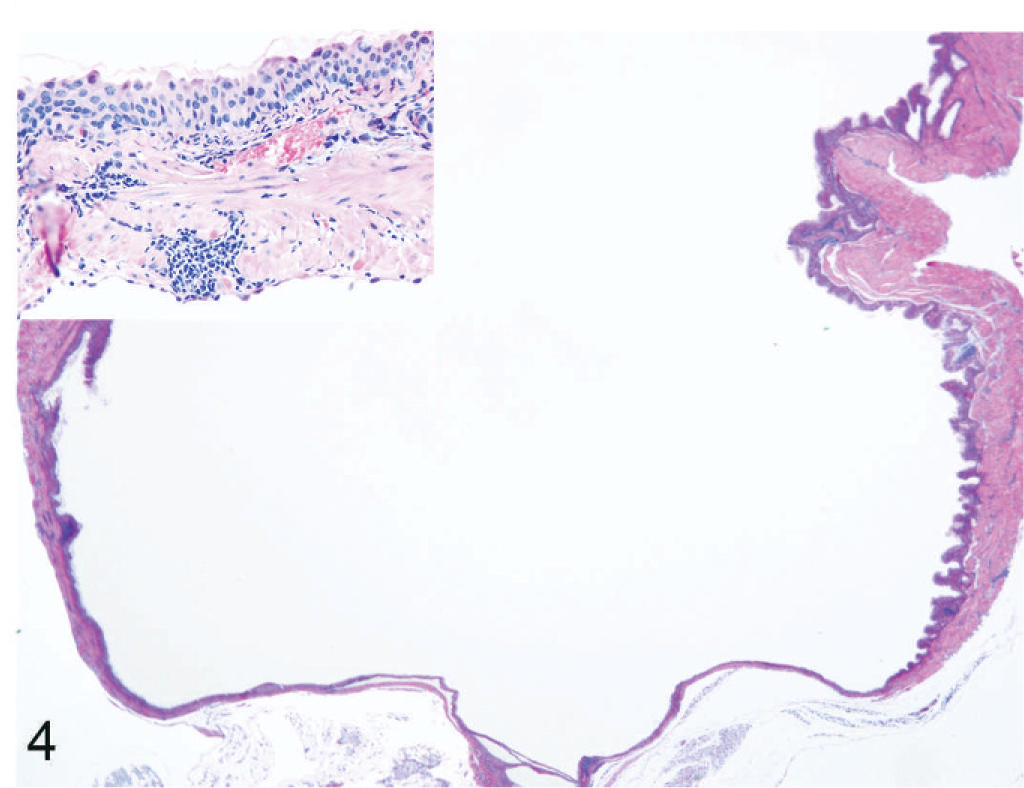
The urinary bladder is dilated with flattened mucosal epithelium and muscular layers. The lumen is free of inflammatory cells or hemorrhage. HE stain. The inset reveals multifocal infiltrates with low to moderate numbers of plasma cells within the muscular layer.
Microscopically, 10 of 23 mice having gross lesions of kidneys showed hydronephrosis. In addition, 3 mice with no gross lesions had similar histologic lesions. Five histologically affected mice that had hydronephrosis also had unilateral mild to marked dilation of the renal pelvis with flattening of the renal papillae (Fig. 3). The remaining (8) mice had mild to marked bilateral renal pelvis dilation. The proximal ureters were moderately to markedly dilated in all mice that had histologic evidence of hydronephrosis.
Two mice had unilateral suppurative nephritis. One of these had multifocal infiltration of large numbers of neutrophils within the tubules. The other mouse had multifocal randomly distributed infiltrates of neutrophils within the cortex and medulla, with intralesional colonies of gram-positive cocci confirmed by Gram's stain.
Discussion
The current report demonstrates that the subcutaneous implantation of 1.7-mg 17β-estradiol pellets resulted in severe urine retention in the bladder, with uni- or bilateral hydronephrosis in Nu-FOXn1nu . Hydronephrosis is distention of the kidney with urine, caused by the increased pressure in the renal pelvis when the flow of urine is obstructed. It can be due to any type of urinary obstruction that occurs at any level of the urinary tract, from the urethra to the renal pelvis. This obstruction can be complete or partial and can be acquired, inherited, or congenital. Certain inbred strains of mice, such as the C3H, C57L, and DDD mice, have been described to have a higher incidence of hydronephrosis, which is presumed to be inherited. 5 An autosomal recessive, progressive hydronephrosis has also been described in C57BL/6J mice. 4 Also, various transgenic mouse strains develop hydronephrosis because of their genetic alterations. In our study, we could not determine the cause of urine retention. However, bacterial infection was not evident in all histological examinations of kidney and bladder except in 1 kidney of a mouse that had intralesional colonies of gram-positive cocci. Struvite uroliathiasis in estrogen-treated ovariectomized female nude mice with Staphylococcus intermedius–induced cystitis was observed. 3 However, in our study, we did not observe any significant bacterial infection that could have been associated with urine retention. Ascending infections of the urinary tract was documented in female nude mice housed on nonautoclaved corncob bedding and implanted with 0.36-mg estrogen pellets for 9 weeks. 14 In this study, changing the corncob bedding to autoclaved paper bedding had no effect. The initial study was to generate tumor growth in Nu-FOXn1nu ; however, because of the adverse side effects of estrogen implantation, the study had to be terminated for the physical well being of the mice.
In all, 23 mice were assessed grossly or microscopically for urine retention and hydronephrosis. Two mice were not assessed because of untimely death. In 14 of 23 mice, blood was collected and serum estrogen level was measured. A normal serum estrogen level is 2–5 pg/ml in mice. Two of 14 mice had no estrogen pellet, were found to have normal serum estrogen levels (3–9 pg/ml), and were healthy until euthanasia with no gross or microscopic lesions in the kidney or bladder (Fig. 2). This suggests that estrogen implantation might have caused the urogenital effects observed in estrogen-implanted nude mice. Six of 14 mice had increased estrogen levels (36–260 pg/ml) and increased bladder volume either grossly or microscopically (Fig. 5). Four mice also had hydronephrosis. Out of these 6 mice, only 1 was as low as 36 pg/ml, whereas the others were in the 123–260 pg/ml range of serum estrogen levels. None of the mice with elevated estrogen levels developed hydronephrosis alone without urine retention in the bladder. This suggests that urine retention preceded hydronephrosis. The remaining 6 of 14 mice were unaffected, with high estrogen levels ranging from 43 to 220 pg/ml; however, only 1 animal was as high as 220 pg/ml, whereas the others were in the 43–102 pg/ml range of serum estrogen levels. It should be noted that the estrogen pellet was a 60-day release pellet. According to the manufacturer, estrogen levels should not be high after 60 days; however, estrogen levels were found high in the serum even after 105 days of implantation. Out of a total of 23 mice, 12 were grossly found to have increased urine retention, and 10 were grossly found to have either uni- or bilateral hydronephrosis (Fig. 6). Microscopically, 12 of 23 and 13 of 23 mice showed dilated bladder and hydronephrosis, respectively. In some mice, hydronephrosis was observed on microscopic examination.
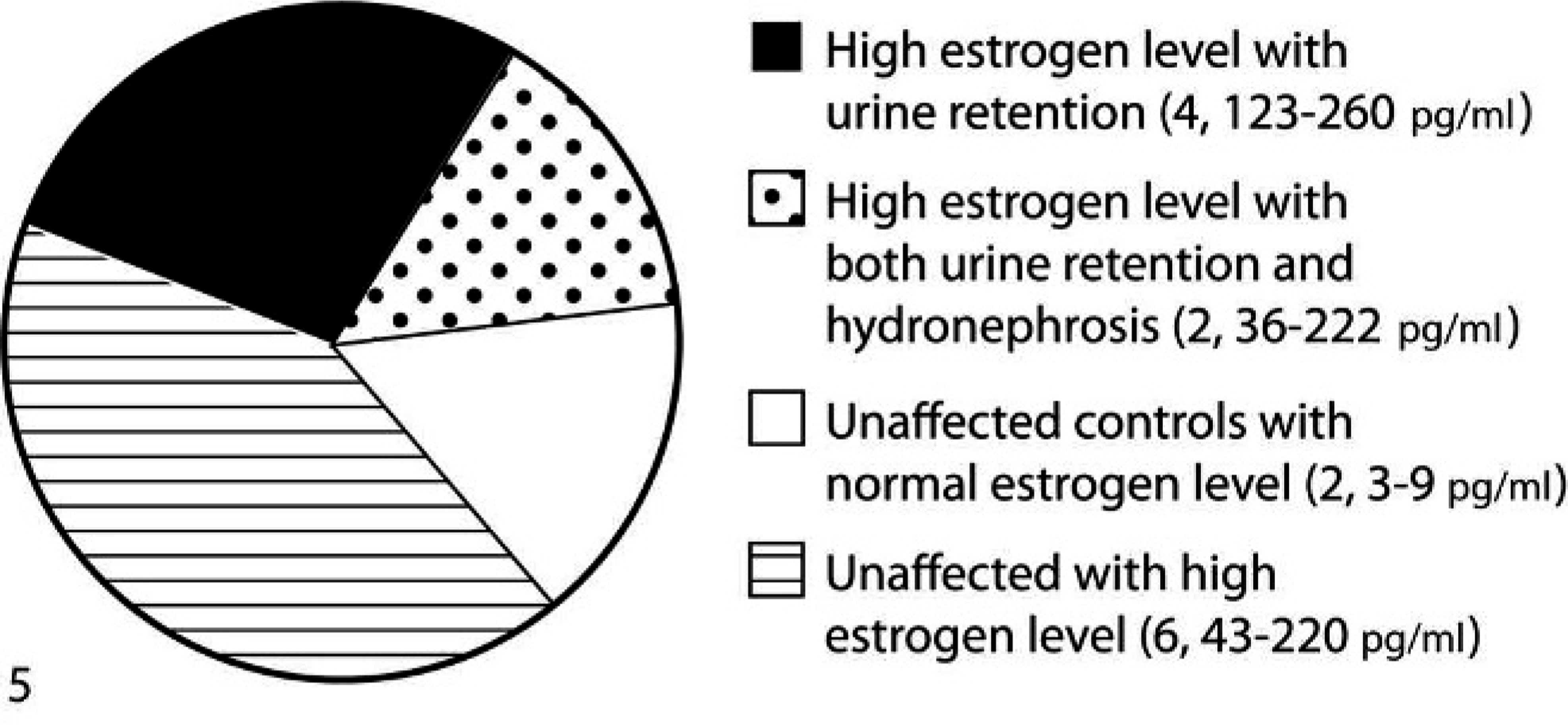
Pie chart showing hydronephrosis, urine retention, in mice in which the estrogen level was measured. Urine retention was seen in 6 mice, out of which 4 also exhibited hydronephrosis. Six mice were unaffected, but high serum estrogen levels were observed. Two control animals with normal estrogen levels also were found to be unaffected. Blood serum from 14 mice was used for the estrogen level measurement. Values within parentheses are the number of mice in each group followed by their serum estrogen levels (pg/ml). n = 14.
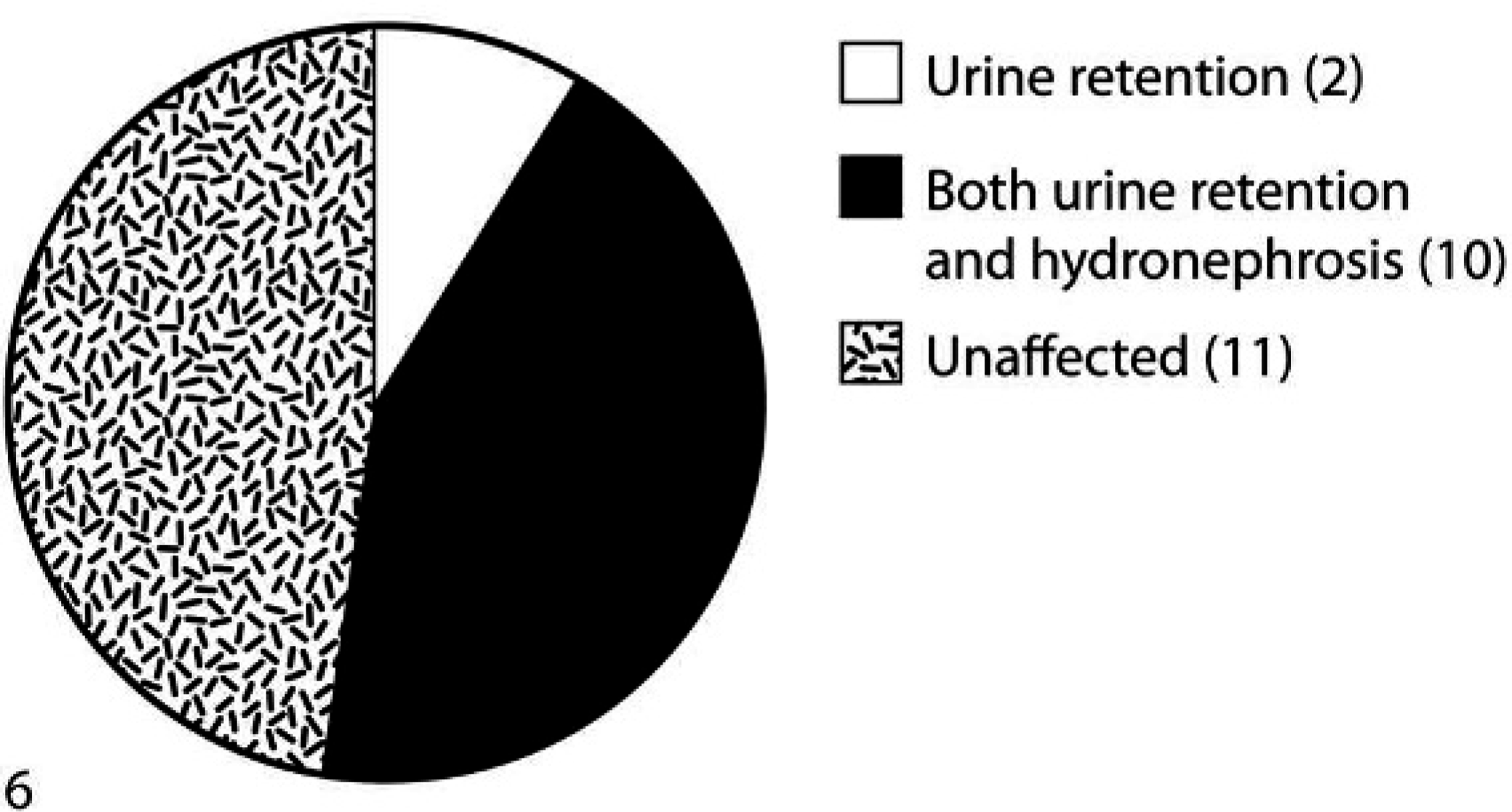
Pie chart showing hydronephrosis and urine retention in all 23 mice. The pie chart is based on the gross examination. Urine retention was observed in 12 mice, out of which 10 mice also showed hydronephrosis. Eleven mice were unaffected and were healthy. Values within parentheses are the number of mice in each group. n = 23.
Estrogen implantation can cause urine retention with uni- or bilateral hydronephrosis in nude mice. Further studies need to be done to fully understand the relationship of estrogen implantation to urine retention. An effective safer dose regimen of estrogen needs to be established in nude mice.
Footnotes
Acknowledgements
We gratefully acknowledge financial support from the National Institutes of Health (COBRE, P-20 RR017686) and Kansas State University, Center for Basic Cancer Research.
