Abstract
A 9-month-old C57BL/6J mouse had progressive abdominal distension over a 1-week period, and a distended left renal capsule was discovered at postmortem examination. Incision of the capsule showed a tan, cloudy fluid that separated the renal capsule and the remnant left kidney. Microscopically, the capsule was significantly separated from the renal parenchyma by clear space and necrotic cellular debris. The majority of the lining of the renal capsule was composed of fibrous connective tissue and lacked an epithelial lining, consistent with a subcapsular perinephric pseudocyst. In addition, attached to intermittent portions of the renal capsule were thin rims of compressed cortical tissue lined by transitional epithelium. The finding of remnant cortical tissue lined by transitional epithelium is consistent with severe hydronephrosis and indicates that the hydronephrosis preceded the formation of the perinephric pseudocyst. To our knowledge, this is the first case report to characterize a perinephric pseudocyst secondary to severe hydronephrosis in a mouse.
Perinephric pseudocysts are locally extensive accumulations of fluid in a fibrous sac surrounding one or both kidneys. 8 The fluid accumulation may be extracapsular, but it is often subcapsular between the fibrous renal capsule and renal parenchyma. 1 Whereas a cyst is defined as a fluid filled cavity lined by epithelium, the term pseudocyst is more appropriate for this lesion, as it lacks an epithelial lining. 9 A review of the literature indicates multiple synonyms for this lesion, including capsulogenic renal cyst, capsular cyst, para-renal cyst, pararenal pseudocyst, capsular hydronephrosis, perirenal cyst, perirenal pseudocyst, and pseudohydronephrosis. 6, 8, 9 Perinephric pseudocysts are uncommon lesions in cats and are rare in dogs, ferrets, pigs, and humans. 4, 7, 11–13 This manuscript characterizes a unilateral perinephric pseudocyst secondary to hydronephrosis in a C57BL/6J mouse.
A 9-month-old female C57BL/6J mouse, part of a control group of an impending research study, showed progressive abdominal distension over 1 week's time. The mouse was euthanatized with carbon dioxide, and an incision of the abdomen showed a distended left kidney. The entire body was placed in 10% neutral-buffered formalin and submitted to Iowa State University's Surgical Pathology Service for histopathologic diagnosis. On examination, the renal capsule was greatly distended (3.4 × 2.5 × 2.2 cm) (Fig. 1) and encased a tan, turbid fluid. Cytospin preparations were non-diagnostic due to formalin fixation. Near the hilus of the capsule was an aggregate of tissue compatible with the remnant kidney, and the adjacent proximal ureter had small, pale serosal nodules (∼1 mm).

Kidneys. The left renal capsule is greatly distended and fluctuant. Compare this to the size of the normal right kidney (arrow). Bar = 1 cm.
The tissues were processed routinely and stained with hematoxylin and eosin (HE) for microscopic evaluation. The remnant left kidney was slightly shrunken (8 × 5 × 4 mm), and the capsule was elevated above the parenchyma by abundant clear space and some necrotic cellular material (Fig. 2). The renal capsule was thickened by increased fibrous connective tissue and lacked an epithelial lining. In a few intermittent foci, thin rims of compressed cortical tissue were attached to the renal capsule (Fig. 3). These were composed of sparse glomeruli and tubules, and transitional epithelium lined the medullary portion of the tissue. In the remnant kidney, glomeruli and tubules were effaced and replaced by interstitial fibrosis with lymphoplasmacytic aggregates and scattered tubular necrosis and hemorrhage. Epithelial cells of residual tubules contained small eosinophilic cytoplasmic droplets, and the tubules were dilated by eosinophilic cellular debris, degenerate neutrophils, macrophages, and small aggregates of eosinophilic crystalline material (cuboidal-to-spicule crystals) that were negative for periodic acid–Schiff stain. Special stains (Gomori methenamine silver and Gram's stain) for pathogenic organisms were negative. The proximal ureter wall was expanded by fibrosis and perivascular neutrophilic infiltration with multifocal aggregates of lymphocytes and plasma cells in the adventitia. The ureteral mucosa was hyperplastic, forming papillary projections into the lumen, which had small amounts of eosinophilic cellular debris, crystals, and degenerate neutrophils. Complete occlusion of the ureter was not evident. No significant gross or microscopic lesions were identified in the right kidney. All other tissues examined were within normal limits, except for the uterus, which had changes consistent with cystic endometrial hyperplasia.

Left remnant kidney and renal capsule. The elevated renal capsule is seen overlying the collapsed renal parenchyma. HE. Bar = 500 μm.
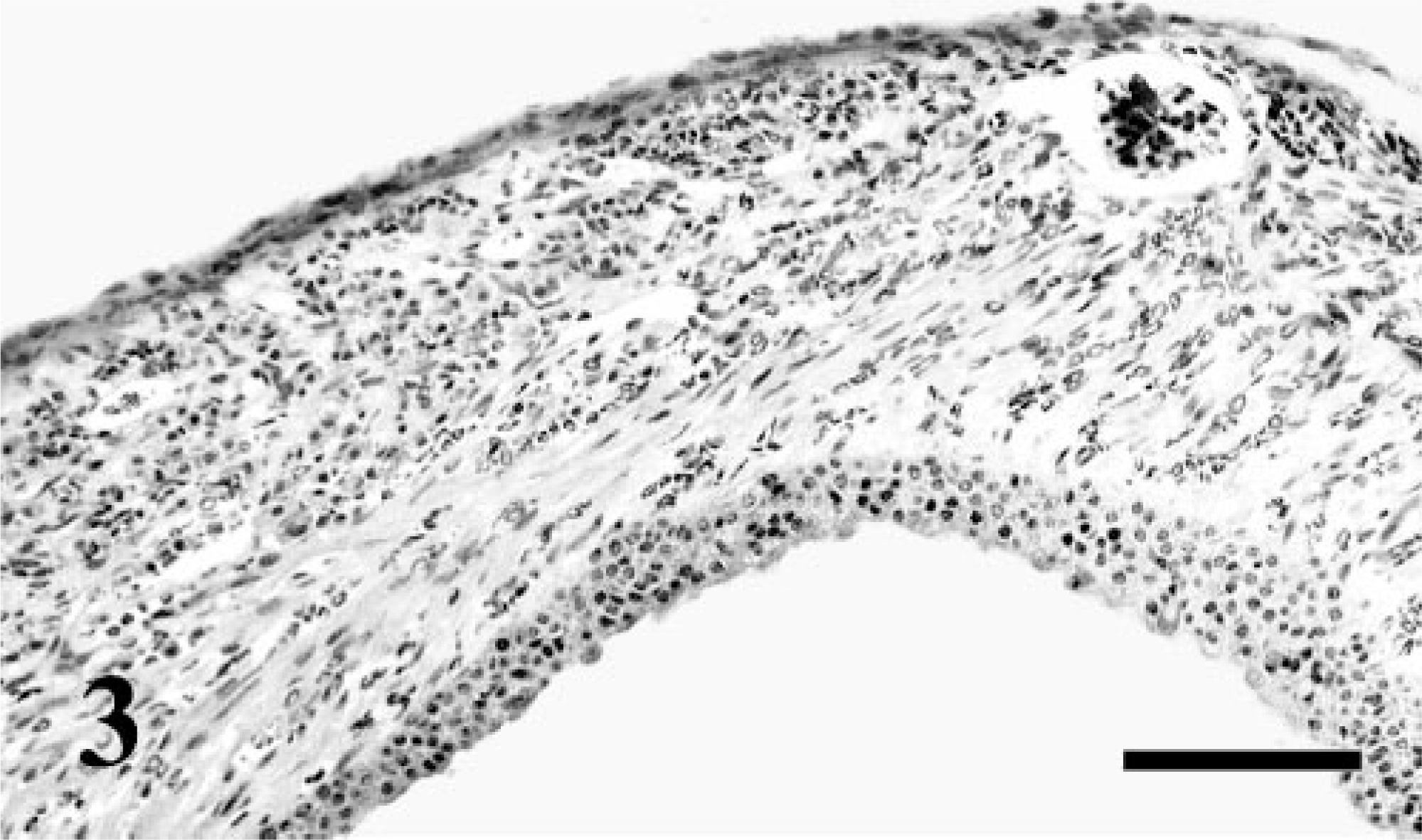
Left kidney, renal capsule. Attached to intermittent portions of the renal capsule are compressed rims of cortical tissue lined by transitional epithelium. HE. Bar = 80 μm.
Perinephric pseudocysts are generally characterized according to the type of fluid filling the cavity. At least four different classifications for the fluid types have been described: 1) uriniferous origin, 2) hematoma, 3) lymphoceole, and 4) those of undetermined origin. 7, 13 Perinephric pseudocysts of uriniferous origin are uniformly believed to be a result of trauma (e.g., obstruction) and require three separate events for the manifestation of the lesion: 1) a complete tear somewhere between the renal cortex and ureter, 2) a failure to seal the defect, and 3) urinary obstruction. 12 Perinephric hematomas are often associated with direct trauma or coagulopathy, leading to an accumulation of blood in the peri-nephric space. Perinephric lymph accumulation generally occurs secondary to conditions that obstruct lymphatic drainage, such as trauma, surgery, neoplasia, or inflammation. Perinephric fluid that does not fit into the above classification is termed a perinephric pseudocyst of unknown origin. 11
In cats with perinephric pseudocysts—the domestic species with the most-documented incidence of perinephric pseudocysts—the most common presenting clinical complaint is progressive abdominal distention. Antemortem diagnosis of perinephric pseudocysts is readily achieved in small animals by abdominal ultrasound. Ultrasonographic evaluation showed an accumulation of free fluid within the perirenal space, and in cats, fluid accumulation is often subcapsular. 1 Fluid analysis typically yields a transudate (e.g., uriniferous origin) or a modified transudate (e.g., hematoma). Urea nitrogen and creatinine concentrations are usually similar to those in serum for lymph and hematoma peri-nephric pseudocysts, but they are elevated compared to serum for those of uriniferous origin. 1, 3, 8 Cytologic examination yields various findings, depending on the origin of the fluid. Unfortunately, fluid analysis and cytologic examination in this particular case were not possible because of the formalin contamination of the cyst fluid.
In this instance, severe hydronephrosis was the major differential diagnosis at gross examination. Hydronephrosis is an uncommon condition in mice due to some degree of urinary obstruction, and certain strains of mice (e.g., C57BL/KsJ, C57BL/6J) are prone to spontaneous and hereditary hydronephrosis. 2, 5, 14, 15 In severe cases of murine hydronephrosis, a minimal amount of grossly identifiable renal tissue remains, and the atrophied renal cortex may be replaced by thin bands of connective tissue that are consistently lined on one side by the renal capsule and on the other side by atrophied-to-hyperplastic transitional epithelium. 14, 15 In the mouse of this study, the intermittent foci of compressed cortical tissue with transitional epithelium on the renal capsule were indicative of previous hydronephrosis. 15 However, the majority of the renal capsule lacked compressed cortical tissue and transitional epithelium. The large amount of remnant renal parenchyma remaining at the hilus is not consistent with severe hydronephrosis, but it is most compatible with perinephric pseudocyst formation secondary to hydronephrosis. 10, 14, 15 Hydronephrosis with secondary perinephric pseudocyst formation has been described in humans and cats. 9, 13 The suggested pathogenesis involves some degree of urinary obstruction leading to hydronephrosis (Fig. 4). At some point during the development of the hydronephrosis, the integrity of the lining wall is compromised, causing the rupture/leakage of urine into the subcapsular space that, in turn, forms a fluid-filled cavity (perinephric pseudocyst) between the renal capsule and the remnant kidney. 9, 10, 13 In the mouse of this study, the hydronephrosis had apparently progressed to a severe stage, as suggested by the thin rims of cortical tissue intermittently lining the renal capsule. We believe that, following the rupture/leakage into the subcapsular space, much of the injured renal parenchyma collapsed near the hilus, while small portions remained attached to the capsule. This is, to our knowledge, the first description of a peri-nephric pseudocyst secondary to hydronephrosis in the mouse and the first detailed veterinary description of the microscopic lesions associated with this dual condition.
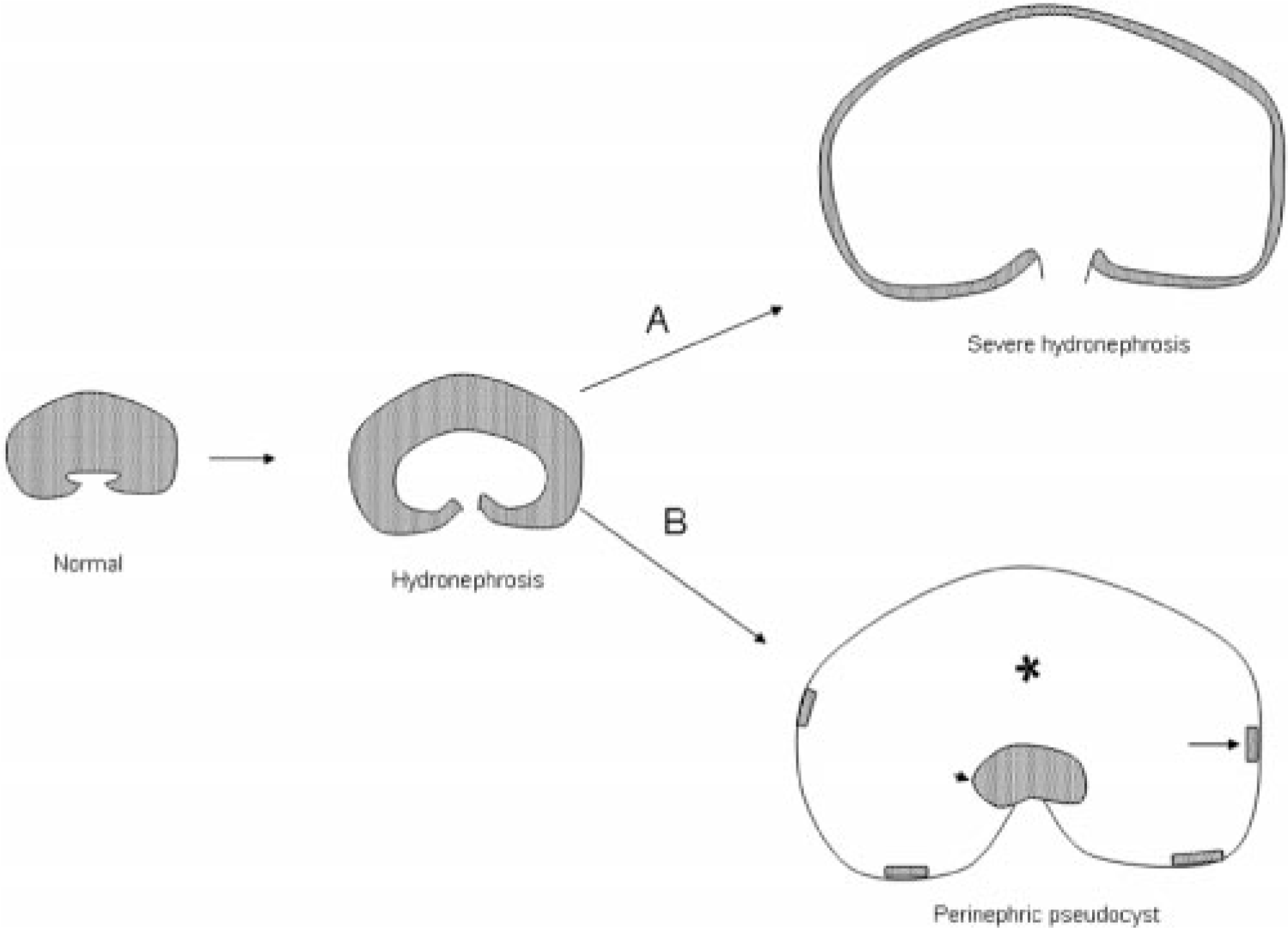
Contrast between severe hydronephrosis and peri-nephric pseudocyst secondary to hydronephrosis. Urinary obstruction leads to increased pressure and hydronephrosis. In severe cases of hydronephrosis (A), the renal parenchyma (cortical tissue lined by transitional epithelium) is uniformly compressed along the dilated capsule. Alternatively (B), during hydronephrosis development, rupture/leakage of fluid into the subcapsular space forms a fluid-filled cavity (∗) between the distended renal capsule and the collapsed renal tissue (arrowhead), with small foci of compressed renal parenchyma (arrows) remaining on the distended capsule.
Footnotes
Acknowledgements
We thank Dr. Kathy Mullin, Dr. Charles Brockus, and Dr. Vanessa Preast for technical assistance and Dr. Ronald K. Myers for reviewing this manuscript.
