Abstract
Neoplastic diseases in prosimians have been sporadically reported in the literature. To provide a comprehensive review of prosimian neoplasia, a retrospective evaluation of neoplasia in a large captive prosimian colony and an extensive literature review were performed. Primates that belong to the Order Primata, Suborder Prosimii with histologic evidence of neoplasia were included. One hundred twenty-three cases of spontaneous neoplasia were identified in 101 prosimians from the Duke Lemur Center, and 124 cases were reported in 116 prosimians in the literature. Overall, this review compiled a total of 247 neoplasms in 217 prosimians. Of the 217 affected animals, 88 of 217 were males (41%), 100 of 217 were females (46%), and sex was not reported in 29 of 217 (13%). Ages ranged from 2 days to 36 years. Prosimian families represented were Lemuridae (80/217 [37%]), Cheirogaleidae (61/217 [28%]), Galagidae (44/217 [20%]), Lorisidae (28/217 [13%]), and Indriidae (4/217 [2%]). The most commonly affected species were the gray mouse lemur (Microcebus murinus) (28/217 [13%]), thick-tailed greater bush baby (Otolemur crassicaudatus) (23/217 [11%]), and black lemur (Eulemur macaco) (19/217 [9%]). Organ systems affected, in order of descending occurrence, were digestive (75/247 [30%]), reproductive (40/247 [16%]), hematopoietic (34/247 [14%]), integumentary (28/247 [11%]), endocrine (26/247 [11%]), and urinary (17/247 [7%]). The respiratory, nervous, musculoskeletal, and cardiovascular systems were infrequently affected. The most common neoplasms were hepatocellular (32/247 [13%]), lymphoma and/or leukemia (29/247 [12%]), biliary (15/247 [6%]), and mammary neoplasms (12/247 [5%]). This article should serve as a valuable reference for the types and relative frequencies of neoplasms that occur in prosimian species.
Prosimians are considered to be “primitive” primates and belong to the Order Primata, Suborder Prosimii. The Suborder Prosimii contains 2 major Superfamilies: Loroidea, which includes lorises, pottos, and bush babies, and Lemuroidea, which includes lemurs, sifaka, indri, and aye-ayes. 34,38,74,86 Tarsiers, still considered by some taxonomists to belong to the Suborder Prosimii, although placed in their own infraorder (Tarsiiformes), 34,86 are in the midst of controversy over their appropriate phylogenetic classification, which arises from their possession of characteristics of both prosimian and anthropoid primates. 42,45,86,106 Tree shrews, previously thought to belong to the Suborder Prosimii, are now classified in the Order Scandentia, Family Tupaiidae, and, therefore, are not included in this article. Additional information on the current taxonomic classification of prosimian species is available in the literature.∗ 34,42,38, ,45,70,74,86,106
Prosimians are a unique group of primates that are less commonly recognized in the literature and in the scientific community compared with anthropoid primates, such as macaques. This understandably stems from the frequent use of anthropoid primate species in scientific research and the more frequent presence of anthropoid primates in zoologic collections. Few species of prosimians are used in laboratory research, and those that are used are typically limited to galagos and pottos. Many prosimian species are listed on the International Union for Conservation of Nature and Natural Resources Red List of Threatened Species as critically endangered, endangered, or vulnerable (http://www.iucnredlist.org). 70 The conservation status of prosimian species often prohibits their involvement in scientific research and limits their availability for acquisition by zoologic collections. A concerted effort to understand these species is crucial for species conservation and captive management.
To better understand any animal species, information regarding common diseases and conditions is critical. Knowledge of neoplastic diseases common to various species, especially primates, is important for the purpose of identifying appropriate animal models, which may provide valuable information about comparable human neoplastic diseases. In addition, this knowledge may provide assistance in species conservation efforts. Neoplastic diseases have been sporadically reported in prosimians throughout the years, often as individual case reports or as a small component of review articles on neoplasms in nonhuman primates. Many cases of prosimian neoplasia have repeatedly been reported in multiple articles, which leads to confusion as to the actual number of cases. Because of the relatively limited and sporadic literature available on prosimian neoplasia, there is a need for a more complete compilation of cases that would provide information on the prevalence of neoplasia in prosimian species.
The purpose of this article is to provide a comprehensive review of neoplasia in prosimians. This article will detail a large collection of prosimian neoplasia cases identified at the Duke Lemur Center (DLC), formerly known as the Duke University Primate Center, which have not previously been reported in the literature, including multiple tumor types that have not previously been reported in any prosimian species. In addition, an extensive literature review will serve to compile previously reported cases of prosimian neoplasia.
Materials and Methods
DLC case review
The surgical and necropsy pathology database at North Carolina State University (NCSU) College of Veterinary Medicine was searched for all cases of neoplasia in prosimian species submitted from the DLC from January 1991 through May 2008. In addition, pathology records at the DLC were searched to identify any cases of prosimian neoplasia submitted to institutions other than NCSU. Prosimian cases were included in this retrospective review if they were found to have histologic evidence of one or more neoplasms. Cases with exclusively hyperplastic or regenerative lesions were excluded.
For cases that met the inclusion criteria, the medical records, including abbreviated case records, postmortem examination data, and archived glass slides, were reviewed for relevant signalment, clinical data, and gross and histologic features of the identified neoplasms. Postmortem examinations were completed at the DLC, and selected tissues were submitted to NCSU College of Veterinary Medicine or another contract laboratory (Roche Biomedical Laboratories, Inc., LabCorp, Antech Diagnostic Laboratories, The Wildlife Conservation Society, or Philadelphia Zoo) for histologic examination. DLC case materials from Roche Biomedical Laboratories, Inc., and LabCorp, originally located in Burlington, North Carolina, were transferred to NCSU in 1998 and remain with the NCSU archives. Reviewed archived glass slides from NCSU contained tissues that previously had been fixed in 10% neutral-buffered formalin, paraffin-embedded, sectioned routinely at 4- to 6-µm thickness, and stained with hematoxylin and eosin stain. Glass slides archived at institutions other than NCSU were not reviewed, and the diagnoses provided in the pathology reports were used. Tumor types from the reviewed DLC cases were confirmed based on histologic examination. The compiled case data were organized in a tabular format. The reference population at the DLC varied from year to year. The DLC animal inventory data from mid-year counts (July or data from the closest available month) were used as a representation of each year's inventory. The incidence of neoplasia cases in the DLC population was calculated on a per year basis.
Literature review
Available literature was reviewed for published cases of spontaneous and experimentally induced neoplasia in prosimians. Database searches performed to identify cases were not limited by language, country of origin, or date so to be as inclusive as possible and reduce the number of missed cases. References in multiple languages, including German, French, and Japanese, were examined and translated by one of the coauthors or another individual in the veterinary pathology field. Cases of neoplasia were included if the primate belonged to the Order Primata, Suborder Prosimii. Previous reports were cross-referenced to avoid duplication of reports of the same case cited in multiple publications. The compiled case data were organized in tabular format. Cases in which the diagnosis of neoplasia was questioned by the investigators were excluded from the table s but included in the text for completeness.
Results
DLC case review results
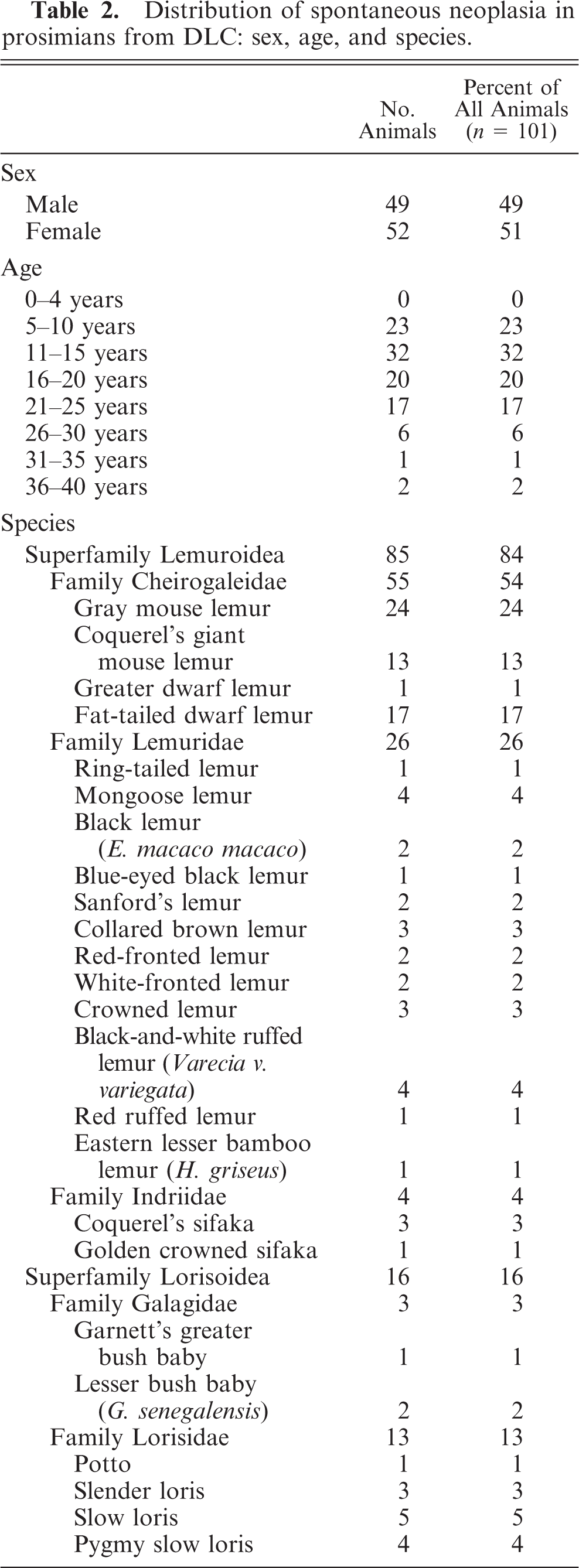
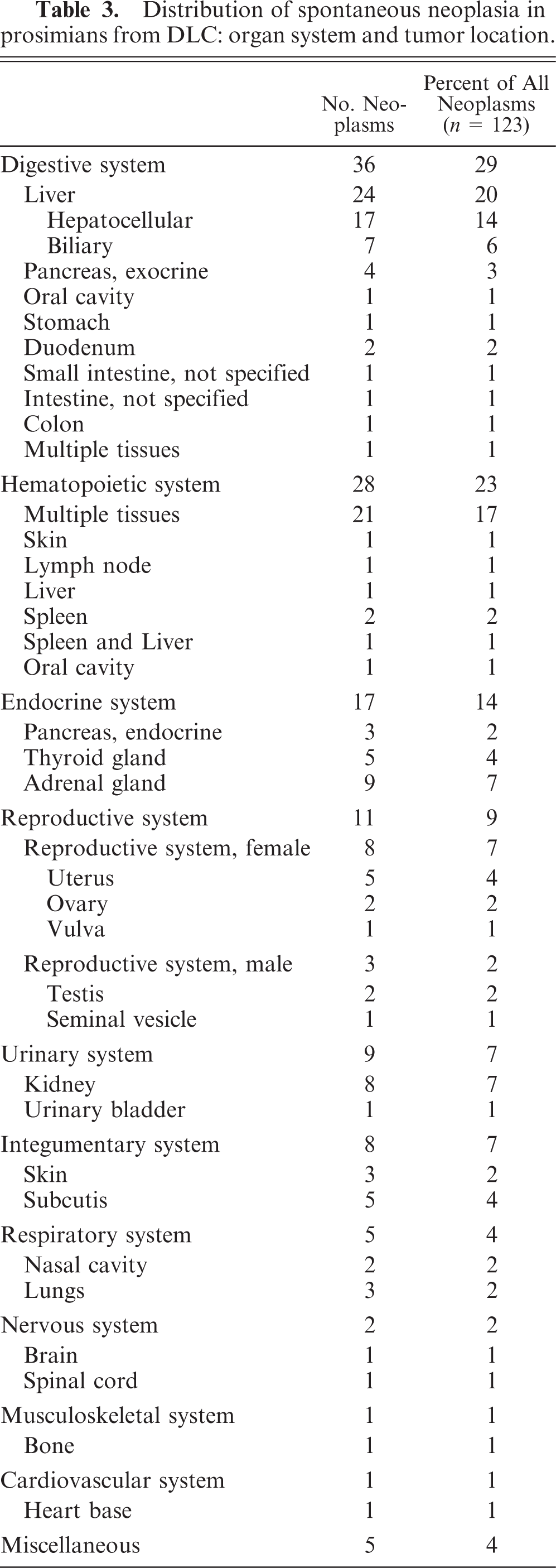
One hundred twenty-three neoplasms were identified in 101 prosimians from the DLC between January 1991 and May 2008. A summary of cases of spontaneous neoplasia in prosimians from the DLC arranged by organ system in order of decreasing occurrence and including detailed case information, such as organ system, tumor location, tumor type, species, age, and sex for each case in which such information was available, is provided in Table 1. Details of the sex, age, and species distribution are given in Table 2, and details of the organ system and tumor-location distribution of cases of spontaneous neoplasia in prosimians from the DLC are given in Table 3.
Summary of spontaneous neoplasia in prosimians from DLC.
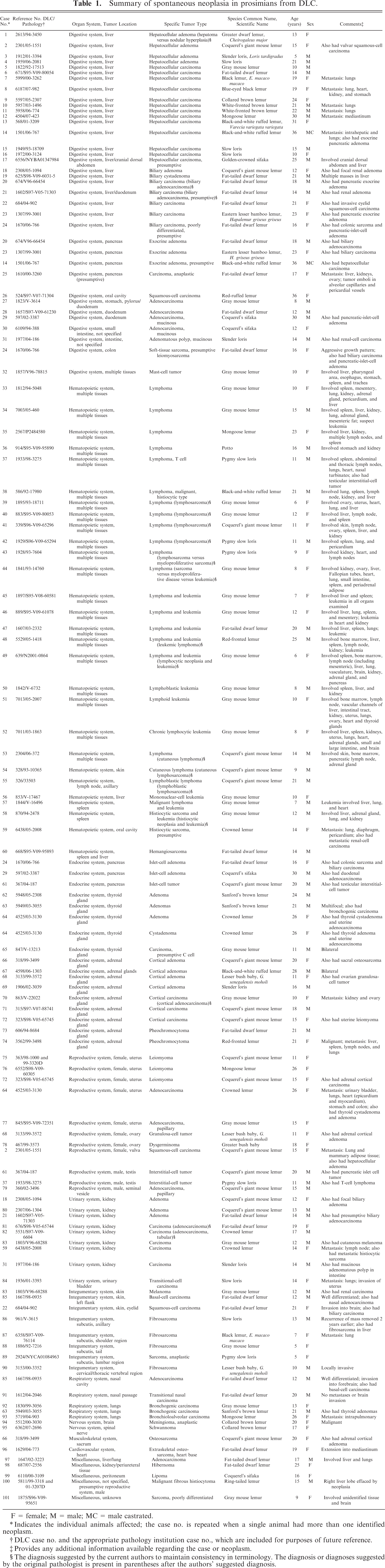
F = female; M = male; MC = male castrated.
∗Indicates the individual animals affected; the case no. is repeated when a single animal had more than one identified neoplasm.
†DLC case no. and the appropriate pathology institution case no., which are included for purposes of future reference.
‡Provides any additional information available regarding the case or neoplasm.
§The diagnosis suggested by the current authors to maintain consistency in terminology. The diagnosis or diagnoses suggested by the original pathologist is present in parentheses after the authors' suggested diagnosis.
Distribution of spontaneous neoplasia in prosimians from DLC: sex, age, and species.
Distribution of spontaneous neoplasia in prosimians from DLC: organ system and tumor location.
Identified cases occurred in lorises, pottos, bush babies, lemurs, and sifaka. No cases of neoplasia in aye-ayes or tarsiers were identified at the DLC. The majority of affected animals from the DLC were captive bred (80/101 [79%]), whereas the remaining animals (case Nos. 1, 4, 7, 13–15, 17, 26, 30, 36–38, 42, 43, 62, 63, 66, 71, 89, 93, and 99) were wild caught (21/101 [21%]). Most cases were prosimians that either died naturally or were euthanatized because of end-stage diseases and that underwent complete postmortem examination at the DLC. A small number of included cases were surgical biopsy submissions from animals that were part of the living collection at the DLC at the time of tissue biopsy. Eighty-one animals had a single identified neoplasm, and 20 animals had two or more identified neoplasms.
The incidence rate of neoplasia cases in prosimians in the DLC population was 7 of 523 (1.3%), 3 of 514 (0.6%), 5 of 501 (1.0%), 5 of 488 (1.0%), 7 of 496 (1.4%), 6 of 473 (1.3%), 7 of 429 (1.6%), 7 of 372 (1.9%), 7 of 326 (2.1%), 6 of 298 (2.0%), 5 of 278 (1.8%), 4 of 255 (1.6%), 5 of 251 (2.0%), 6 of 249 (2.4%), 8 of 253 (3.2%), 7 of 232 (3.0%), 5 of 224 (2.2%), and 1 of 207 (0.5%) in the years 1991 through 2008, respectively. The estimated cumulative incidence based on the number of prosimians identified at the DLC with neoplasms in a 17.5-year period between 1991 and May 2008 and when using the highest population count (from the year 1991) was 101 of 523 (19.3%).
Literature review results
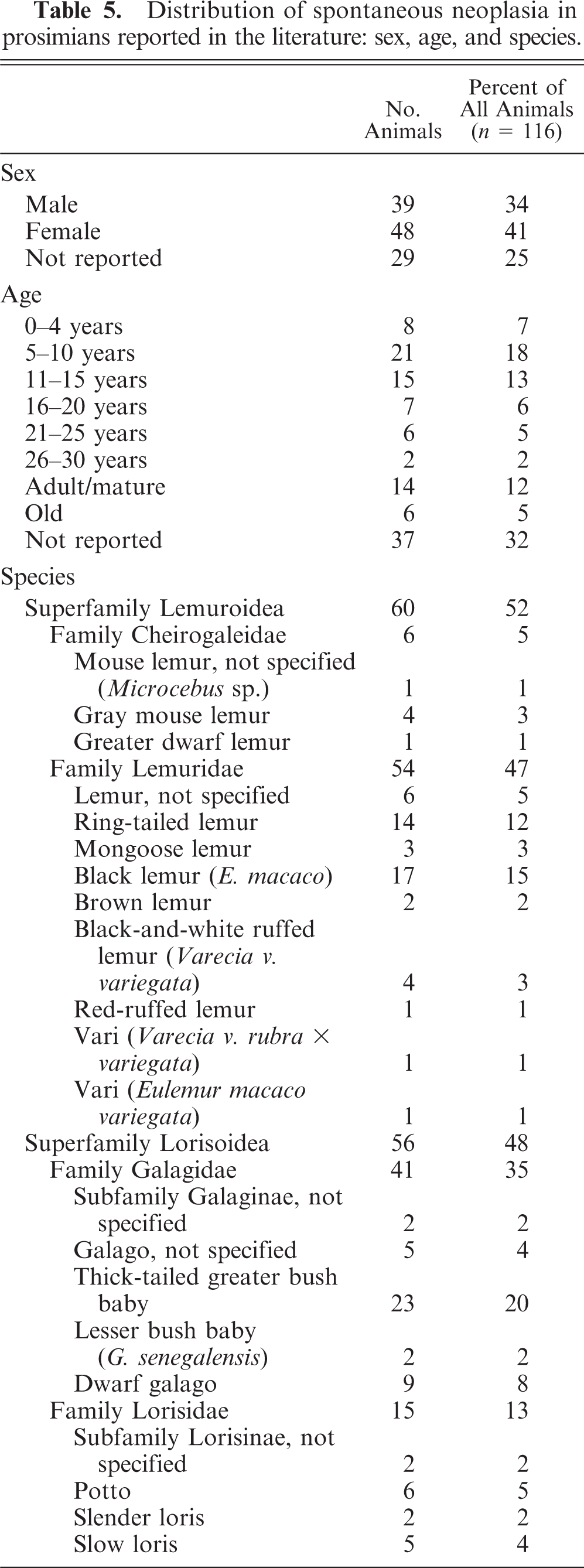
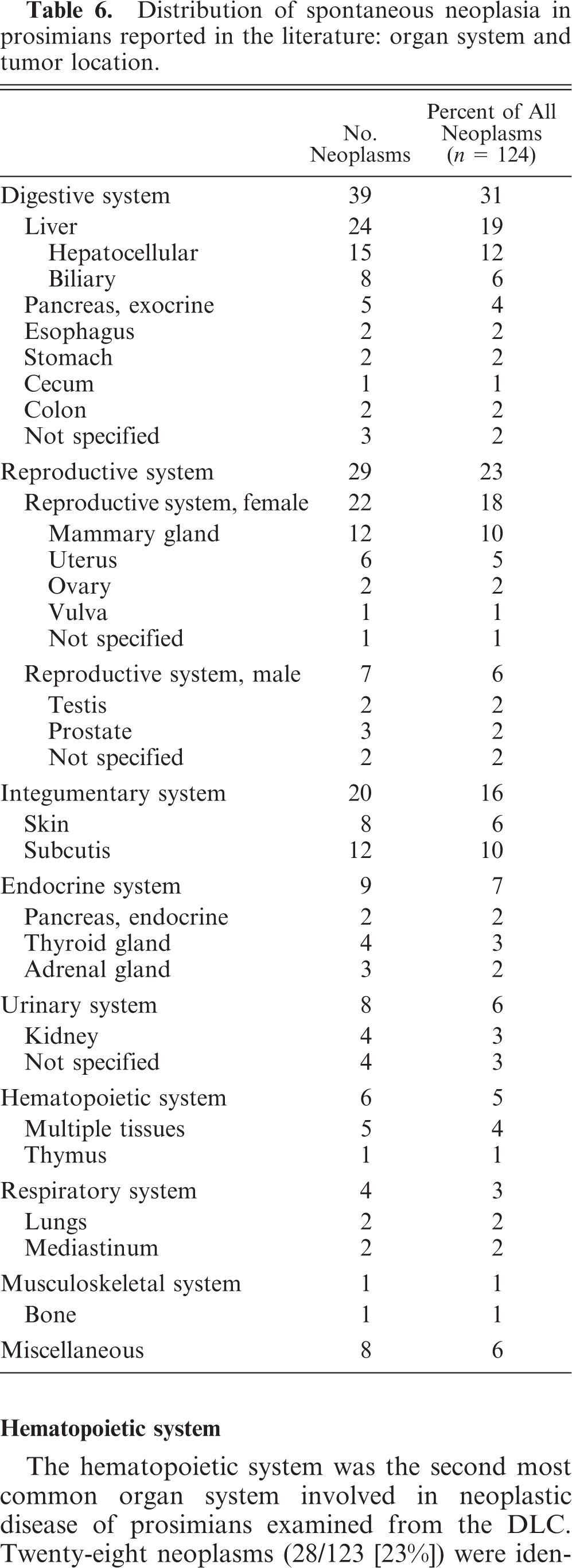
To the authors' knowledge, based on an extensive review of the literature, there are 124 reported cases of spontaneous neoplasia in 116 prosimians that date between 1910 and 2008. A summary of cases of spontaneous neoplasia in prosimians reported in the literature arranged by organ system in order of decreasing occurrence and including detailed case information, such as organ system, tumor location, tumor type, species, age, and sex for each case in which such information was available, is provided in Table 4. In some cases, the tumor location, tumor type, or signalment information was disputed by the author of a subsequent article (not to include the authors of the current article), and, in these cases, the alternatively suggested information was noted in parentheses, along with the associated reference. Details of the sex, age, and species distribution are given in Table 5, and details of the organ system and tumor-location distribution of cases of spontaneous neoplasia in prosimians reported in the literature are given in Table 6.
Summary of spontaneous neoplasia in prosimians reported in the literature.
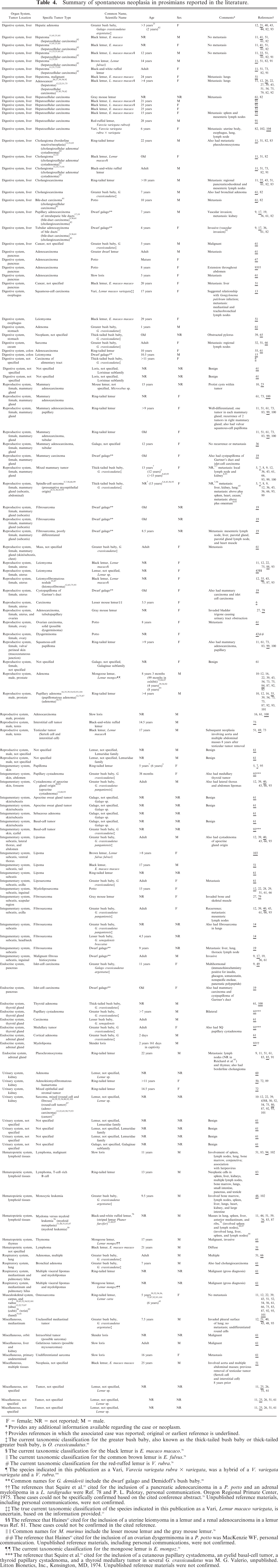
F = female; NR = not reported; M = male.
∗Provides any additional information available regarding the case or neoplasm.
†Provides references in which the associated case was reported; original or earliest reference is underlined.
‡The current taxonomic classification for the greater bush baby, also known as the thick-tailed bush baby or thick-tailed greater bush baby, is O. crassicaudatus.86
§The current taxonomic classification for the black lemur is E. macaco macaco.70
∥The current taxonomic classification for the common brown lemur is E. fulvus. 70
#The current taxonomic classification for the red-ruffed lemur is V. rubra. 70
¶The species indicated in this publication as a Vari, Varecia variegata rubra X variegata, was a hybrid of a V. variegata variegata and a V. rubra. 104
∗∗Common names for G. demidovii include the dwarf galago and Demidoff's bush baby.86
††The references that Squire et al.93 cited for the inclusion of a pancreatic adenocarcinoma in a P. potto and an adrenal myelolipoma in a L. tardigradus were Ref. 78 and P. L. Palotay, personal communication. Oregon Regional Primate Center, 1975. These cases could not be specifically confirmed based on the cited conference abstract.78 Unpublished reference materials, including personal communications, were not confirmed.
‡‡The true current taxonomic classification of the species indicated in this publication as a Vari, Lemur macaco variegata, is uncertain, based on the information provided.13
§§The reference that Haines43 cited for the inclusion of a uterine leiomyoma in a lemur and a renal adenocarcinoma in a lemur was Ref. 81. These cases could not be confirmed in the cited reference.
∥∥Common names for M. murinus include the lesser mouse lemur and the gray mouse lemur.70
##The reference that Haines43 cited for the inclusion of an ovarian dysgerminoma in a P. potto was MacKenzie WF, personal communication. Unpublished reference materials, including personal communications, were not confirmed.
¶¶The current taxonomic classification for the mongoose lemur is E. mongoz. 70
∗∗∗The reference that Squire et al.93 cited for the inclusion of a cutaneous papillary cystadenoma, an eyelid basal-cell tumor, a thyroid papillary cystadenoma, and a thyroid medullary tumor in several G. crassicaudatus was M. G. Valerio, unpublished. Litton Bionetics, Inc., Kensington, MD, 1974. Unpublished reference materials were not confirmed.
Distribution of spontaneous neoplasia in prosimians reported in the literature: sex, age, and species.
Distribution of spontaneous neoplasia in prosimians reported in the literature: organ system and tumor location.
Identified cases included lorises, pottos, bush babies, and lemurs. No cases of neoplasia in sifaka, indri, aye-ayes, or tarsiers were identified in the literature. The majority of animals cited in the literature belonged to zoologic collections. The precise origin of these animals (captive bred or wild caught) was typically not provided. A total of 109 animals had a single identified neoplasm, and 7 animals had two or more identified neoplasms.
Five cases of spontaneous disease in prosimians reported in the literature were excluded from the summary of spontaneous neoplasia in prosimians; however, mention of these cases in this review was thought to provide valuable clarification. An orbital pseudotumor was reported in a 13- to 15-month-old female lesser bush baby (Galago senegalensis). 43,44,93 The pseudotumor was described as lymphoid, plasmacytic, and sclerosing. This case was excluded, because orbital pseudotumors are not neoplasms but rather growths of inflammatory origin. An unspecified cancer was reported in a lemur 39,92 ; however, this case was excluded, because the original cited reference 53 and a subsequent article 54 confirmed that this case was in a Mozambique monkey, which is a Cercopithecine species, and not in the prosimian suborder. Leukemia was reported in a Senegal galago (G. senegalensis), or lesser bush baby; 48,59,93 however, this case was excluded, because leukemia could not be confirmed in the originally cited reference, 46 in which the case was described as a galago with anemia and clumped leukocytes. A growth at the heart base in a ring-tailed lemur (Lemur catta) was reported in several published articles† and was listed in multiple articles as a case of spontaneous neoplasia.‡ This case was excluded, because the originally cited reference 90 diagnosed the lemur with tuberculosis. The lemur was originally described as having an early stage of tuberculosis, with a growth at the heart base that extended to the bronchial and intraclavicular glands (lymph nodes) and associated pulmonary edema. 90 Based on this information, the heart-base growth in this animal was likely related to the tuberculosis and was unrelated to any neoplastic process. Adenomatous hyperplasia of the bile ducts was reported in a Demidov's dwarf galago (Galagoides demidovii) as part of a review of neoplasia in nonhuman primates; 61 however, this case was excluded based on the diagnosis of hyperplasia and a lack of confirmation that this was a true neoplastic process.
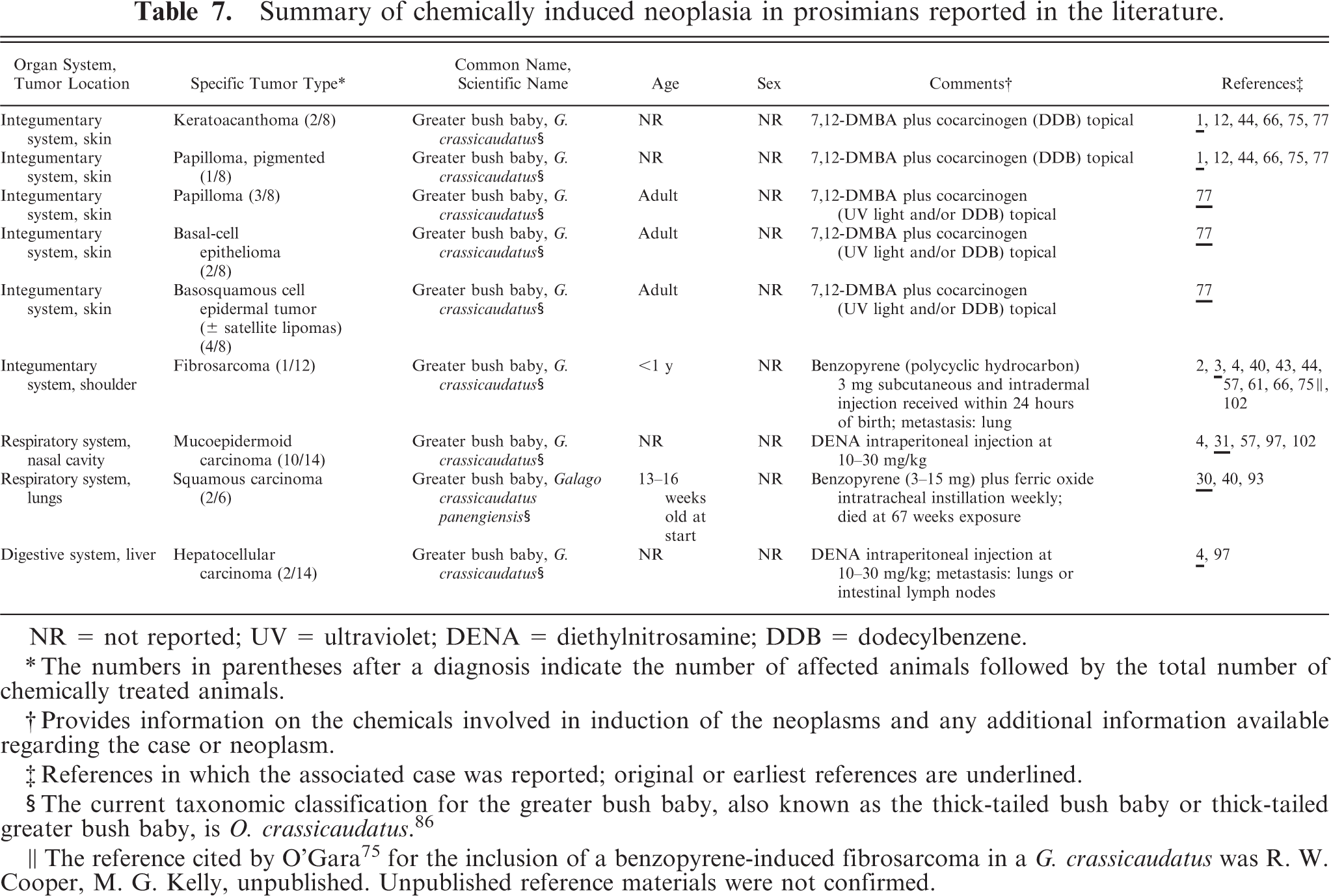
Although prosimians are infrequently used in scientific research, several studies that used galagos and pottos reported results with chemically induced neoplasms. A summary of cases of chemically induced neoplasia in prosimians reported in the literature, as well as detailed case information, including organ system, tumor location, tumor type, species, age, and sex, for each case in which such information was available is provided in Table 7.
Summary of chemically induced neoplasia in prosimians reported in the literature.
NR = not reported; UV = ultraviolet; DENA = diethylnitrosamine; DDB = dodecylbenzene.
∗The numbers in parentheses after a diagnosis indicate the number of affected animals followed by the total number of chemically treated animals.
†Provides information on the chemicals involved in induction of the neoplasms and any additional information available regarding the case or neoplasm.
‡References in which the associated case was reported; original or earliest references are underlined.
§The current taxonomic classification for the greater bush baby, also known as the thick-tailed bush baby or thick-tailed greater bush baby, is O. crassicaudatus. 86
∥The reference cited by O'Gara75 for the inclusion of a benzopyrene-induced fibrosarcoma in a G. crassicaudatus was R. W. Cooper, M. G. Kelly, unpublished. Unpublished reference materials were not confirmed.
Twenty-seven individual chemically induced neoplasms in prosimians were reported in the literature. The sex was not reported for any of the cases. Nine cases (9/27 [33%]) were reported to be in adult prosimians, one case (1/27 [4%]) was reported in a prosimian less than 1 year of age, 2 cases (2/27 [7%]) were reported in prosimians that were 13 to 16 weeks old at the initiation of the experiment, and age was not reported in the remaining cases (15/27 [56%]). The only prosimian family represented in the chemically induced neoplasia cases was Galagidae. The only affected species was the thick-tailed greater bush baby (Otolemur crassicaudatus). The most commonly affected organ system was the integumentary system (13/27 [48%]), followed by the respiratory system (12/27 [44%]) and the digestive system (2/27 [7%]).
Fifteen cases of chemically induced disease in prosimians reported in the literature were excluded from the summary of chemically induced neoplasia in prosimians; however, mention of these cases in this review was thought to provide valuable clarification. Pseudoepitheliomatous (pseudocarcinomatous) hyperplasia of the skin was reported in 8 greater bush babies (Galago crassicaudatus, current taxonomic name O. crassicaudatus) and 7 pottos (Perodicticus potto) after topical administration of 7,12-dimethylbenz[a]anthracene (DMBA) plus a cocarcinogen (dodecylbenzene). 1,43,44 Although the chemical 7,12-DMBA plus a cocarcinogen was also shown to induce skin neoplasms in greater bush babies, 1,12,44,66,75,77 the cases of pseudoepitheliomatous hyperplasia were excluded based on the diagnosis of hyperplasia, which indicated that these lesions were not true neoplastic processes.
Overall
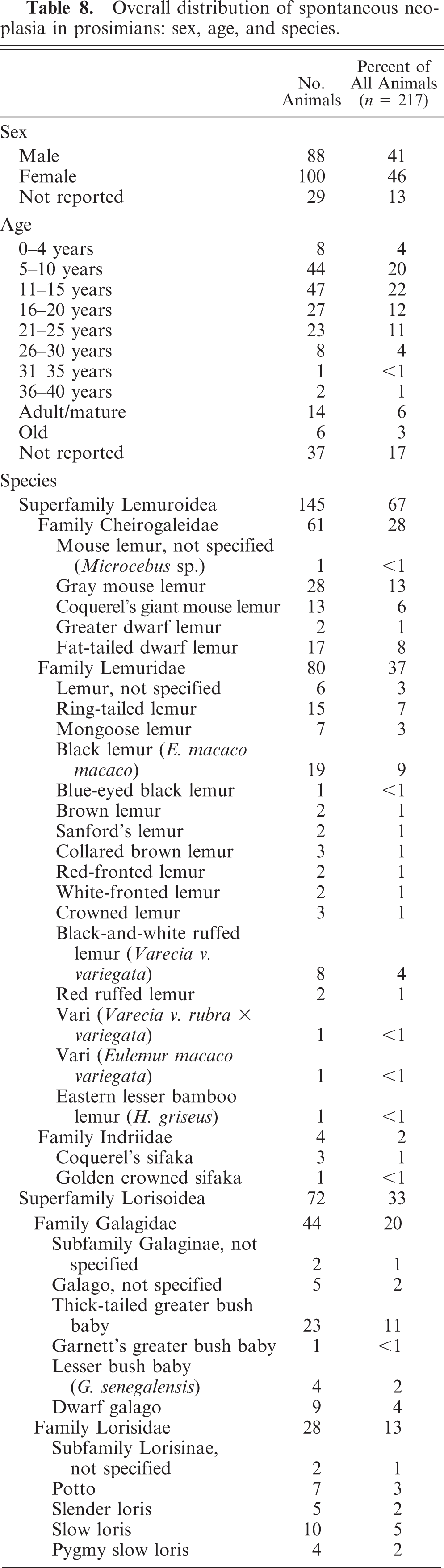
When all cases of neoplasia in prosimians reported in the literature and described in this article from the DLC were examined together, there were a total of 247 neoplasms identified in 217 prosimians. Details of the sex, age, and species distribution are given in Table 8, and details of the organ system and tumor location distribution of cases of spontaneous neoplasia overall are given in Table 9.
Overall distribution of spontaneous neoplasia in prosimians: sex, age, and species.
Overall distribution of spontaneous neoplasia in prosimians: organ system and tumor location.

Discussion
Epidemiology
The incidence of neoplasia cases in the DLC prosimian population was calculated on a per year basis, because the number of prosimians in the DLC collection varied from year to year, ranging from 207 to 523 total prosimians, depending on the year. The yearly incidence rates of neoplasia cases in prosimians in the DLC population ranged from a low of 0.6% in 1992 to a high of 3.2% in 2005. It should be noted that the incidence rate for 2008 was not included, because data for the year was incomplete and had only been collected through the month of May 2008 at the time of this writing. The estimated cumulative incidence based on the number of prosimians identified at the DLC with neoplasms in a 17.5-year period between 1991 and May 2008 and when using the highest population count (from 1991) was 101 of 523 (19.3%). The incidence of spontaneous neoplasia in prosimians reported in the literature could not be calculated, because the numbers of prosimians in the reference populations for the cases in the literature were not known or not provided.
Sex
The distribution of neoplasia cases identified at the DLC in male and female prosimians was approximately equal over all cases and within most organ systems, which supported a lack of sex predilection for most neoplasms. Reproductive-system neoplasms were more commonly identified in females than in males, with 8 females and 3 males affected with reproductive-system neoplasms. Nervous system neoplasms were only identified in female prosimians; however, the low number of cases (2) likely obscured the true sex distribution of these neoplasms. In addition, sex predilection could not be accurately determined for neoplasms of the musculoskeletal and cardiovascular systems, because only one case was identified for each of these organ systems.
Conclusions regarding the overall sex distribution of neoplasia cases in the literature or sex predilection for specific tumor types were influenced by the large percentage of cases for which sex was not reported and the lack of information regarding the reference populations from which these cases were obtained. Similar to the DLC cases, reproductive-system neoplasms were more commonly identified in females than males, with 22 female reproductive-system neoplasms and 7 male reproductive-system neoplasms. Whether reproductive-system neoplasms are actually more common in females than in males or whether these reproductive neoplasms typically produce more overt clinical signs in females and thus are more commonly recognized and reported cannot be determined. Of interest regarding cases in the literature, biliary-origin neoplasms were reported more frequently in males (6) than in females (2), exocrine and endocrine pancreatic neoplasms were reported more frequently in females (6) than in males (1), thyroid neoplasms were reported more frequently in males (3) than in females (1), adrenal neoplasms were reported more frequently in males (3) than in females (0), and hematopoietic system neoplasms were reported more frequently in males (6) than in females (0). None of these possible sex predilections were supported by the sex distribution of spontaneous neoplasia among different organ systems and tissues in the DLC cases.
Age
Of the affected animals from the DLC, ages ranged from 5 to 36 years of age. Of the cases of spontaneous neoplasia in prosimians in the literature, reported ages ranged from 2 days to 29 years of age. None of the DLC cases were reported to be in prosimians younger than 5 years of age, in contrast to the cases reported in the literature in which 8 of 116 of the cases (7%) were reported to be in prosimians younger than 5 years of age. Thirty-seven (37/116 [32%]) of the spontaneous neoplasms reported in the literature did not include information regarding the animal's age, which made the age distribution of cases in the literature difficult to accurately assess.
Species
Although the precise number of prosimians of each species present at the DLC varied from year to year, a relative species profile of the reference population was obtained by using the average number of each species present per year. The average yearly total number of prosimians in the reference population was 354: 87% (308/354) were in the Superfamily Lemuroidea and 13% (46/354) were in the Superfamily Lorisidae. When using the relative species profile information, the 5 most common species present in the reference population included the ring-tailed lemur, the mongoose lemur (Eulemur mongoz), the collared lemur (Eulemur collaris), the red-ruffed lemur (Varecia rubra), and the fat-tailed dwarf lemur (Cheirogaleus medius), which represented approximately 11% (39/354), 8% (29/354), 6% (21/354), 6% (21/354), and 6% (21/354) of the DLC prosimian population. Only one of the 3 most commonly affected species with neoplasms in this case survey (fat-tailed dwarf lemur) was in the 5 most common species present in the reference population. This information may be influenced by the decline in the number of animals of some species over the years examined in this study. For example, the gray mouse lemur (Microcebus murinus), the fat-tailed dwarf lemur, and the Coquerel's giant mouse lemur (Mirza coquereli) numbered 38, 35, and 36, respectively, in 1991, and 9, 4, and 1, respectively, in 2008. Notably, common species within the reference population, including the ring-tailed lemur, the mongoose lemur, the collared lemur, and the red-ruffed lemur, were infrequently affected by neoplasia, with only 1, 4, 3, and 1 cases of neoplasia identified for each of these species, respectively, in the 17.5-year period included in this survey.
Differences in species distribution of spontaneous neoplasia cases in the literature compared with the DLC may be a reflection of the species profile at this one specific primate center compared with a cross section of the multiple institutions and locations involved in the cases reported in the literature. Any apparent species predilections for specific tumor types were discussed under the organ system involved.
Neoplasms were identified in the following prosimian species in this review that have not been previously reported in the literature: the Coquerel's sifaka (Propithecus coquereli) and golden crowned sifaka (Propithecus tattersalli) from the Family Indriidae; the Coquerel's giant mouse lemur and the fat-tailed dwarf lemur from the Family Cheirogaleidae; the blue-eyed black lemur (Eulemur macaco flavifrons), Sanford's brown lemur (Eulemur sanfordi), collared brown lemur, red-fronted brown lemur (Eulemur rufus), white-fronted brown lemur (Eulemur albifrons), crowned lemur (Eulemur coronatus), and Eastern lesser bamboo lemur (Hapalemur griseus) from the Family Lemuridae; the Garnett's greater bush baby (Otolemur garnetti) from the Family Galagidae; and the pygmy slow loris (Nycticebus pygmaeus) from the Family Lorisidae.
Because of some changes in taxonomic classification of prosimian species with shifts of some subspecies to full species status, 70 it is difficult to accurately assess whether species generalizations may have been made when a more specific subspecies designation would have provided more detailed information about the species reported in the literature. The more general designation of black lemur (Eulemur macaco) may have been used without regard to the presence of 2 subspecies: the black lemur (Eulemur macaco macaco) and blue-eyed black lemur. In addition, the general designation of brown lemur (Eulemur fulvus) may have been used to include all 6 subspecies, now considered by some taxonomists to be separate full species: the common brown lemur, Sanford's brown lemur, collared brown lemur, white-collared brown lemur (Eulemur albocollaris), white-fronted brown lemur, and red-fronted brown lemur.
Digestive system
The digestive system was the most common organ system involved in neoplastic disease of prosimians examined from the DLC. Thirty-six neoplasms (36/123 [29%]) were identified in the digestive-system tissues of 32 prosimians (case Nos. 1–32). The majority of these neoplasms (24/36 [67%]) involved the liver and were either of hepatocellular (17/24 [71%]) or biliary (7/24 [29%]) origin. Four of the 17 hepatocellular neoplasms (4/17 [24%]) were considered benign (case Nos. 1–4). The remaining 13 hepatocellular neoplasms (13/17 [76%]) were diagnosed as hepatocellular carcinomas (case Nos. 5–17). Two of the 7 biliary neoplasms (2/7 [29%]) were considered benign (case Nos. 18 and 19). The remaining 5 biliary neoplasms (5/7 [71%]) were biliary carcinomas (case Nos. 20–24). Of the 7 biliary neoplasms, 5 neoplasms (5/7 [71%]) were present in fat-tailed dwarf lemurs (case Nos. 19–22 and 24). Additional digestive-system neoplasms involved the pancreas, oral cavity, stomach, and small and large intestine, and included a colonic sarcoma (presumptive leiomyosarcoma) in a fat-tailed dwarf lemur (case No. 24; Fig. 1). The 2 small-intestinal mucinous adenocarcinomas were both identified in Coquerel's sifakas (case Nos. 29 and 30).
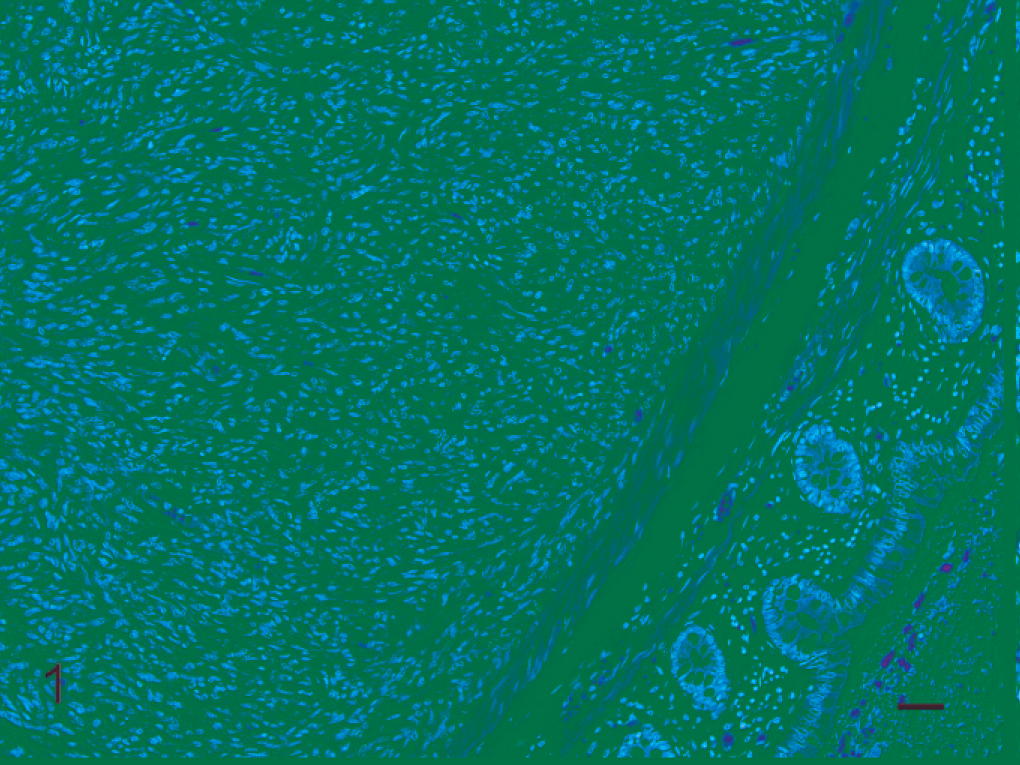
Colon; Fat-tailed dwarf lemur No. 24. Colonic sarcoma; histopathologic appearance. Effacing the muscular layer, there is a large mass composed of densely packed neoplastic spindle cells arranged in interlacing fascicles. HE. Bar = 20 µm.
The digestive system was also the most common organ system involved in spontaneous neoplastic disease of prosimians reported in the literature. Thirty-nine neoplasms (39/124 [31%]) were reported in the digestive system tissues of prosimians in the literature. The majority of these neoplasms (24/39 [62%]) involved the liver and were either of hepatocellular (15/24 [63%]) or biliary (8/24 [33%]) origin. Fourteen of the hepatocellular neoplasms (14/15 [93%]) were either originally diagnosed or later reclassified as hepatocellular carcinomas. Of the 15 hepatocellular neoplasms, 9 neoplasms (9/15 [60%]) were present in black lemurs (E. macaco). Three of the biliary neoplasms (3/8 [38%]) were considered benign, and the remaining 5 biliary neoplasms (5/8 [63%]) were classified as carcinomas.
This article provides the first reported prosimian cases of oral cavity neoplasms (squamous-cell carcinoma [case No. 26] and histiocytic sarcoma [case No. 59]); small-intestinal neoplasms (duodenal adenocarcinoma [case No. 28], duodenal mucinous adenocarcinoma [case No. 29], small-intestinal mucinous adenocarcinoma [case No. 30], and intestinal mucinous adenomatous polyp [case No. 31]); pancreatic exocrine adenomas (case Nos. 14, 20, and 23); and gastrointestinal mast cell tumor (case No. 32).
Hematopoietic system
The hematopoietic system was the second most common organ system involved in neoplastic disease of prosimians examined from the DLC. Twenty-eight neoplasms (28/123 [23%]) were identified in the hematopoietic system tissues of 28 prosimians (case Nos. 33–60). The majority of the hematopoietic system neoplasms were either lymphoma (14/28 [50%]) (case Nos. 33–43 and 53–55), leukemia (4/28 [14%]) (case Nos. 50–52 and 56; Fig. 2), or lymphoma with leukemia (6/28 [21%]) (case Nos. 45–49 and 57). Half of the lymphomas and leukemias (12/24 [50%]) occurred in the gray mouse lemur (case Nos. 33, 34, 39, 40, 45, 46, 49–52, 56, and 57). The strong species predilection of the gray mouse lemur in this population for the acquisition of lymphoid neoplasms and/or leukemia may indicate an underlying genetic predisposition or a potential infectious etiology, such as a viral agent to which the gray mouse lemur is vulnerable.

Liver; gray mouse lemur No. 52. Chronic lymphocytic leukemia; histopathologic appearance. Sheets of neoplastic round cells are present within the hepatic sinusoidal spaces and fill the lumen of the portal vein (top of figure). HE. Bar = 10 µm.
Among the cases of spontaneous neoplasia in prosimians reported in the literature, the hematopoietic system was relatively infrequently affected, with only 6 neoplasms (6/124 [5%]) reported. Five hematopoietic neoplasms were diagnosed as lymphoma and/or leukemia, and the one remaining neoplasm was a thymoma. None of the hematopoietic neoplasms reported in the literature occurred in the gray mouse lemur, further highlighting the cluster of hematopoietic neoplasms identified in the gray mouse lemur at the DLC.
Histiocytic neoplasms in prosimians have not previously been reported in the literature. Two histiocytic neoplasms were identified from prosimians at the DLC, including histiocytic sarcoma of the spleen with leukemia (case No. 58) and an oral-cavity histiocytic sarcoma (case No. 59). In addition, a hemangiosarcoma that involved the spleen and liver (case No. 60) represented the first reported case of hemangiosarcoma in a prosimian.
Endocrine system
The endocrine system was the third most common organ system involved in neoplastic disease of prosimians examined from the DLC. Seventeen neoplasms (17/123 [14%]) were identified in the endocrine system tissues of 16 prosimians (case Nos. 24, 29, and 61–74). The endocrine-system neoplasms most commonly involved the adrenal glands (9/17 [53%]), with 4 cortical adenomas (case Nos. 66–69), 3 cortical carcinomas (case Nos. 70–72), and 2 pheochromocytomas (case Nos. 73 and 74). Five endocrine neoplasms (5/17 [29%]) involved the thyroid glands and included 4 thyroid adenomas (case Nos. 62–64) and one thyroid carcinoma (case No. 65). The remaining 3 endocrine neoplasms involved the pancreatic islets (case Nos. 24, 29, and 61).
Nine spontaneous neoplasms (9/124 [7%]) in the endocrine system tissues of prosimians were reported in the literature. Reported in the literature are endocrine-system neoplasms that included 3 adrenal gland, 4 thyroid, and 2 pancreatic-islet neoplasms.
Neoplasms that involved the adrenal glands were relatively common and represented 53% (9/17) and 33% (3/9) of endocrine neoplasms in prosimians from the DLC and in the literature, respectively. Of interest, 33% (3/9) of the adrenal gland neoplasms identified in prosimians from the DLC were adrenal cortical carcinomas (case Nos. 70–72), which have not previously been reported in the literature. No neoplasms that involved the parathyroid glands or pituitary glands of prosimians were included in the DLC cases or reported in the literature. The absence of pituitary-gland neoplasms observed in prosimians at the DLC was influenced by the infrequent examination of the brain and pituitary gland during routine necropsy procedures.
Female reproductive system
Eight neoplasms (8/123 [7%]) were identified in the reproductive-system tissues of 8 female prosimians from the DLC (case Nos. 2, 64, 68, 72, and 75–78). Of the 52 female prosimians identified with neoplastic disease, 15% (8/52) had a neoplasm within the female reproductive system. Five of the reproductive neoplasms involved the uterus (5/8 [63%]): 3 leiomyomas (case Nos. 72, 75, and 76) and 2 adenocarcinomas (case Nos. 64 and 77). The remaining female reproductive system neoplasms included one ovarian granulosa-cell tumor (case No. 68; Fig. 3), one ovarian dysgerminoma (case No. 78; Fig. 4), and one vulvar squamous-cell carcinoma (case No. 2).

Ovary; lesser bush baby (Galago senegalensis moholi) No. 68. Granulosa-cell tumor; histopathologic appearance. Densely packed neoplastic granulosa cells are arranged in cords and nests separated by fine fibrovascular stroma. HE. Bar = 10 µm.

Ovary; Garnett's greater bush baby No. 78. Dysgerminoma; histopathologic appearance. The mass is composed of sheets and cords of neoplastic cells. Multinucleated tumor cells (arrow) and mitotic figures (arrowhead) are present. HE. Bar = 10 µm.
The reproductive system was the second most common organ system involved in neoplastic disease of prosimians reported in the literature. Twenty-two neoplasms (22/124 [18%]) in the reproductive system tissues of female prosimians were reported in the literature. In contrast to the DLC population in which mammary-gland neoplasms were not identified, the most commonly affected female reproductive tissue in the literature was the mammary gland (12/22 [55%]). Mammary-gland neoplasms included 6 carcinomas, one mixed mammary tumor, one spindle-cell sarcoma, 3 fibrosarcomas, and one unspecified mammary neoplasm. Six uterine neoplasms were reported with similar histopathologic types as the DLC cases, including 2 leiomyomas, one leiomyofibromatous nodule (leiomyofibroma), one cystopapilloma, one carcinoma, and one adenocarcinoma. Ovarian neoplasms included a solid ovarian carcinoma thought to be a possible dysgerminoma and one dysgerminoma. One vulvar squamous-cell papilloma was also reported. Of the ovarian neoplasms identified in prosimians from the DLC and reported in the literature, 3 neoplasms (3/4 [75%]) were ovarian dysgerminomas (case No. 78). The remaining ovarian neoplasm, identified in a prosimian from the DLC, was an ovarian granulosa-cell tumor (case No. 68), which has not been previously reported in prosimians in the literature.
Male reproductive system
Three neoplasms (3/123 [2%]) were identified in the reproductive tissues of 3 male prosimians from the DLC (case Nos. 37, 61, and 79). The 2 identified testicular neoplasms were both interstitial-cell neoplasms (case Nos. 37 and 61). One adenocarcinoma of the seminal vesicles was identified (case No. 79; Fig. 5). Two of the 3 male reproductive-system neoplasms were identified in Coquerel's giant mouse lemurs (case Nos. 61 and 79).

Seminal vesicle; Coquerel's giant mouse lemur No. 79. Papillary adenocarcinoma; histopathologic appearance. Long irregular fronds of neoplastic epithelial cells are supported by thin fibrous cores. Areas of necrosis and hemorrhage are present. HE. Bar = 20 µm.
Seven neoplasms in the reproductive tissues of male prosimians (7/124 [6%]) were reported in the literature. Two testicular neoplasms were identified: one interstitial-cell tumor and one mixed interstitial-cell and Sertoli-cell tumor. Based on the cases identified at the DLC and in the literature, the interstitial-cell tumor appears to be the most common testicular neoplasm in prosimians. Accessory sex gland neoplasms included 2 prostatic adenomas and one prostatic adenocarcinoma. Prostatic neoplasms were not identified in the DLC population. Neoplasms of the seminal vesicles in prosimians have not previously been reported in the literature. An adenocarcinoma of seminal vesicles was identified in a Coquerel's giant mouse lemur from the DLC (case No. 79). None of the male reproductive-system neoplasms published in the literature were reported in the Coquerel's giant mouse lemur.
Urinary system
Nine neoplasms (9/123 [7%]) were identified in the urinary-system tissues of 9 prosimians from the DLC (case Nos. 18, 21, 31, 59, and 80–84). The majority of the urinary-system neoplasms involved the kidneys (8/9 [89%]). Three of the renal neoplasms were considered benign (case Nos. 18, 21, and 80). Five of the renal neoplasms were diagnosed as carcinomas (case Nos. 31, 59, and 81–83). Eight neoplasms (8/124 [6%]) were reported in the urinary system of prosimians in the literature. Four of these neoplasms involved the kidneys. Neoplasms of the urinary bladder have not previously been reported in prosimians in the literature. A transitional-cell carcinoma of the urinary bladder was identified in a slow loris (Nycticebus coucang) from the DLC (case No. 84).
Integumentary system
Eight neoplasms (8/123 [7%]) were identified in the integumentary-system tissues of 8 prosimians from the DLC (case Nos. 22, 83, and 85–90). Subcutaneous fibrosarcoma was the most commonly identified integumentary system neoplasm (4/8 [50%]) (case Nos. 86–88, and 90). In addition, there was one subcutaneous anaplastic sarcoma (case No. 89).
The integumentary system was the third most common organ system involved in neoplastic disease of prosimians reported in the literature. Twenty neoplasms (20/124 [16%]) in the integumentary-system tissues of prosimians were reported in the literature. Similar to the DLC population, subcutaneous fibrosarcoma was one of the most commonly reported integumentary-system neoplasms (5/20 [25%]). In addition, lipomas and liposarcomas were commonly reported (5/20 [25%]).
Of the cutaneous neoplasms identified in prosimians at the DLC, melanoma (case No. 83) has not previously been reported in prosimians. Although cutaneous squamous-cell carcinoma has not previously been reported in prosimians, 3 squamous-cell carcinomas (oral cavity [case No. 26], cutaneous [case No. 22], and vulvar [case No. 2]) were identified in the DLC prosimians, and a vulvar squamous-cell papilloma and an esophageal squamous-cell carcinoma have been reported in the literature.
Respiratory system
Five neoplasms (5/123 [4%]) were identified in the respiratory-system tissues of 5 prosimians from the DLC (case Nos. 63, 85, and 91–93). Three primary pulmonary neoplasms were identified, including 2 bronchogenic carcinomas (case Nos. 63 and 92) and one bronchioloalveolar carcinoma (case No. 93). Four neoplasms (4/124 [3%]) in the respiratory-system tissues of prosimians were reported in the literature. Unlike the DLC cases in which all 3 primary pulmonary neoplasms were considered malignant, the 2 primary pulmonary neoplasms reported in the literature were diagnosed as adenomas.
Two nasal-cavity neoplasms, one adenocarcinoma and one transitional nasal carcinoma, which have not previously been reported in prosimians, were included in the prosimian cases from the DLC (case Nos. 85 and 91; Fig. 6). Both of the nasal-cavity neoplasms were identified in fat-tailed dwarf lemurs, a species in which no cases of neoplasia have been previously reported.

Nasal cavity; fat-tailed dwarf lemur No. 91. Transitional carcinoma; histopathologic appearance. Large cuboidal to columnar neoplastic epithelial cells are arranged in pseudostratified layers often palisading around protein-filled microcysts. HE. Bar = 10 µm.
Nervous system
Two neoplasms (2/123 [2%]) were identified in the nervous-system tissues of 2 prosimians from the DLC (case Nos. 94 and 95). One cerebral meningioma (case No. 94) and one spinal-cord schwannoma (case No. 95) were identified. Both neoplasms were identified in female collared brown lemurs. There were no neoplasms that involved the nervous-system tissues of prosimians reported in the literature.
Musculoskeletal system
One neoplasm (1/123 [1%]) was identified in the musculoskeletal system tissues of one prosimian from the DLC. The identified musculoskeletal-system neoplasm was an osteosarcoma of the sacrum in a Coquerel's giant mouse lemur (case No. 66). Similar to that identified in the DLC population, one osteosarcoma was reported in the literature. In this case, the osteosarcoma involved the carpus and radius of a ring-tailed lemur.
Cardiovascular system
Only one neoplasm (1/123 [1%]) was identified in the cardiovascular-system tissue of one prosimian from the DLC. This unusual neoplasm was an extraskeletal osteosarcoma of the heart base in a fat-tailed dwarf lemur (case No. 96; Fig. 7). There were no neoplasms that involved the cardiovascular system tissues of prosimians reported in the literature.

Heart base; fat-tailed dwarf lemur No. 96. Extraskeletal osteosarcoma; histopathologic appearance. Plump neoplastic spindle cells surround irregular islands of eosinophilic osteoid material. Scattered multinucleated tumor cells (arrows) are present, and a great vessel is visible (top of figure). HE. Bar = 20 µm.
Miscellaneous
Five neoplasms (5/123 [4%]) were identified in miscellaneous tissues of 5 prosimians from the DLC, which could not be easily classified into any one specific organ system (case Nos. 97–101). The identified miscellaneous neoplasms included one hibernoma in the region of the kidney and periureteral tissue (case No. 98; Fig. 8), one peritoneal lipoma (case No. 99), one adenocarcinoma of undetermined origin that involved the liver and lungs (case No. 97), one poorly differentiated sarcoma that involved an unidentified section of tissue and the brain (case No. 101), and one malignant fibrous histiocytoma that effaced the right liver lobe and was also presumed to involve the male reproductive-tract tissues (case No. 100).

Periureteral tissue; fat-tailed dwarf lemur No. 98. Hibernoma; histopathologic appearance. A multilobulated mass adjacent to the ureter (top right of figure) is composed of large neoplastic cells with abundant finely vacuolated eosinophilic cytoplasm. HE. Bar = 20 µm.
Eight neoplasms (8/124 [6%]) were reported in miscellaneous tissues of prosimians in the literature, which could not easily be classified into any one specific organ system. The miscellaneous neoplasms included one unclassified mediastinal neoplasm, one intraorbital tumor classified as a possible sarcoma, one gelatinous tumor of the liver and kidney classified as a possible myxosarcoma, one undifferentiated sarcoma of undetermined primary location, one unspecified neoplasm that involved multiple tissues, and 3 additional unspecified tumors. Of interest is the recognition of a hibernoma in the region of the kidney and periureteral tissue in a fat-tailed dwarf lemur (case No. 98; Fig. 8). Hibernomas are rare tumors of brown fat and have not previously been reported in prosimians.
Terminology
Given that the literature review included in this article covered cases of prosimian neoplasia over an extended period of time (1910 to 2008), it is understandable that changes in the diagnostic terminology used over time have occurred. In some cases, the tumor diagnosis was disputed by the author of a subsequent article, and, in these cases, the alternatively suggested diagnosis was noted in parentheses in Table 4, along with the associated reference. The current authors elected not to make further changes to the diagnoses provided in the literature, because the original case materials were not available for examination. Of note is the use of the term hepatoma. In veterinary pathology, hepatoma implies a benign neoplasm of the liver; however, in human pathology, hepatoma implies a malignant neoplasm of the liver. To make a recommendation for changing the diagnosis to either hepatocellular adenoma, as may be suggested in veterinary pathology, or hepatocellular carcinoma, as may be suggested in human pathology, one would need to review the original case materials. For the DLC cases, the authors suggested the use of more current terminology after review of the cases; however, the original diagnoses were noted in parentheses in Table 1 for informational purposes. The authors agreed to maintain the original pathologists' diagnoses when possible.
It addition to changes in current terminology, it also should be recognized that data compiled from the literature and from the DLC involved diagnoses made by numerous institutions and pathologists, which inherently leads to inconsistent usage of diagnostic terminology.
Comparative review
A thorough review of neoplasms identified in anthropoid nonhuman primate species for use in comparison with prosimian neoplasms recognized by this review is beyond the scope of this article. There are multiple case reports or restricted case series of spontaneous neoplasms in nonhuman primates available in the literature. Complete review articles on primate neoplasia are relatively limited, and reported reviews often relate to cases observed in specific zoologic collections.
Of interest is an article that contains an in-depth review of spontaneous neoplasia in baboons. 24 In this review, neoplasms in baboons were most commonly observed in the hematopoietic system, and lymphoma was the most common tumor type, with mention of a large cluster of lymphoma cases in baboons at one particular institution. The outbreak of lymphomas in baboons was linked to interspecies transmission of simian T-cell lymphotropic virus type 1 from rhesus macaques. As previously mentioned, a cluster of lymphoma or leukemia cases was also observed in the gray mouse lemur at the DLC. A potential viral etiology associated with these cases was also suspected but has not yet been confirmed.
Selected published reviews on primate neoplasia are available that may be of some interest for interspecies comparison of the types and relative incidences of neoplasms.§ More focused system- or organ-specific reviews in nonhuman primates are also available, including reviews of gastrointestinal-, 15,32,55,98 hematopoietic-, 47,59,64 reproductive-, 62,71,96,105 urinary-, 37,50 and respiratory-system neoplasms. 18
This article provides a comprehensive review of spontaneous neoplasia in prosimians reported in the literature plus the invaluable addition of an almost equal number of cases from prosimians maintained at the DLC. This review should serve as a valuable reference for the types and relative frequencies of neoplasms that occur in prosimian species. In addition, this review provides new information on neoplasia in prosimians, including cases in several prosimian species in which neoplasms have not been previously reported and a large number of neoplasms that have not previously been reported in prosimians. Further studies on common prosimian diseases, potential species predilections for specific conditions, and possible etiologies of the most common neoplasms, such as hepatocellular neoplasms and hematopoietic tumors, would be valuable additions to the existing literature.
Footnotes
Acknowledgements
We thank the staff at the DLC for their dedication to the conservation of prosimian species. We thank the myriad pathologists and pathology residents at NCSU, Roche Biomedical Laboratories, Inc., LabCorp, Antech Diagnostic Laboratories, The Wildlife Conservation Society, and Philadelphia Zoo who were involved in the pathological diagnosis of the multiple prosimian cases from the DLC over the years. In addition, we thank the Histology laboratory staff at NCSU for their technical assistance and Cynthia Swanson at WIL-Biotechnics, LLC, for her assistance with the images used in this article.
