Abstract
Pituitary adenomas were identified in 14 of 491 (2.9%) cynomolgus macaques evaluated from 1994 to 2004. Cases included male (8) and female (6) cynomolgus macaques ranging from 18 to 32 years of age. Seven of the pituitary adenomas caused gross enlargement of the pituitary gland that was visible on postmortem examination, whereas the remaining 7 were multifocal microadenomas identified on histologic examination. A total of 35 adenomas were identified in the 14 macaques, 6 of which were being treated for diabetes mellitus. Mean (± SD) pituitary weight was 0.31 ±.42 g, compared with 0.07 ±.02 g for 430 historical control animals (P < 0.0001). Immunohistochemical staining for follicle-stimulating hormone, luteinizing hormone, prolactin, human growth hormone, thyroid-stimulating hormone, and adrenocorticotropic hormone was applied to pituitary tissue from all cases. Immunostaining revealed 22 of 35 (62.9%) lactotroph adenomas, 5 of 35 (14.3%) plurihormonal cell adenomas, 3 of35 (8.6%) corticotroph adenomas, 2 of35 (5.7%) null cell adenomas, 1 of 35 (2.9%) somatotroph adenomas, 1 of 35 (2.9%) mixed corticotroph-somatotroph adenomas, 1 of 35 (2.9%) mixed lactotroph-corticotroph adenomas, 0 of 35 gonadotroph adenomas, and 0 of 35 thyrotroph adenomas. This study represents the first extensive retrospective case series performed to evaluate the histologic and immunohistochemical characteristics of pituitary adenomas in cynomolgus macaques. our findings indicated that macaque pituitary adenomas frequently had mixed histologic appearance and hormone expression, and that, similar to human pituitary adenomas, prolactin-secreting neoplasms were the most prevalent type.
Keywords
Pituitary adenomas have been reported in a wide variety of mammalian species and can be an important cause of age-related disease. Pituitary tumors are common in several domestic animal species, and are one of the most common types of human intracranial tumors; however, cases of pituitary tumors in nonhuman primates have only been sporadically reported. To the authors' knowledge, there are 11 previously reported cases of spontaneous pituitary tumors in nonhuman primates in literature. 3, 14, 16– 18, 20, 23, 26, 36, 37, 41 Of the reported cases, 7 were in Old World monkeys and 4 were in New World monkeys (Table 1). The frequency of these spontaneous neoplasms is likely higher than the reported rate owing to demographic and diagnostic factors, and may be similar to the rates reported for humans or other mammalian species.
Previously reported cases of spontaneous pituitary tumors in nonhuman primates.∗
OWM = Old World monkey, NWM = New World monkey, M = Male, F = Female, NR = not reported, y = years, m = months.
The prevalence of spontaneous pituitary adenomas in other animals and humans varies considerably depending on the study reviewed and the study population. In humans, pituitary adenomas have been identified in 2.7–27% of random autopsies, 4, 10, 27 whereas in domestic animals, reported prevalence of pituitary tumors includes approximately 40% in aged Beagles, 5, 19 9% in cats, 40 and 0.5% in horses. 8, 43 Among laboratory animals, spontaneous pituitary tumors are rare in most strains of mice, 30, 38 compared with those in rats, in which reported prevalence ranges up to 85% in some strains. 6, 13, 24, 28, 42 In general, the percentage of individual humans or animals with spontaneous pituitary tumors increases with age.
The classification of pituitary adenomas varies on the basis of histologic cell types and hormonal expression of the cells. Traditionally, pituitary adenomas were categorized into 3 basic cell types including acidophils, basophils, and chromophobes on the basis of the cell's differential staining properties as seen by light microscopy using HE staining. 4 Using immunohistochemical analysis, and in some instances, electron microscopy, pituitary adenomas are now routinely classified on the basis of their hormonal expression. 2, 4, 25, 31, 34 In general, acidophils include somatotrophs, which secrete somatostatin or growth hormone (GH), and lactotrophs, which secrete prolactin. Basophils include corticotrophs, which secrete pro-opiomelanocortin (a precursor molecule to adrenocorticotropic hormone [ACTH]), melanocyte-stimulating hormone, endorphins, and lipotropin; thyrotrophs, which secrete thyroid-stimulating hormone (TSH); and gonadotrophs, which secrete follicle-stimulating hormone (FSH) and luteinizing hormone (LH). Some sources indicate that chromophobes include corticotrophs, whereas other sources indicate that chromophobes may actually be degranulated acidophils and basophils. 11, 12, 22, 27 The classification of pituitary adenomas by hormonal expression is more specific than is the classic use of histologic cell type and is more clinically relevant.
The purposes of the retrospective study reported here were to evaluate the prevalence of spontaneous pituitary tumors in a large population of cynomolgus macaques and to characterize the features of these neoplasms histologically and immunohistochemically. This information is important for comparison of pituitary tumors in nonhuman primates with those of other mammalian species, and for improved recognition of this neoplasm in macaque populations.
Materials and Methods
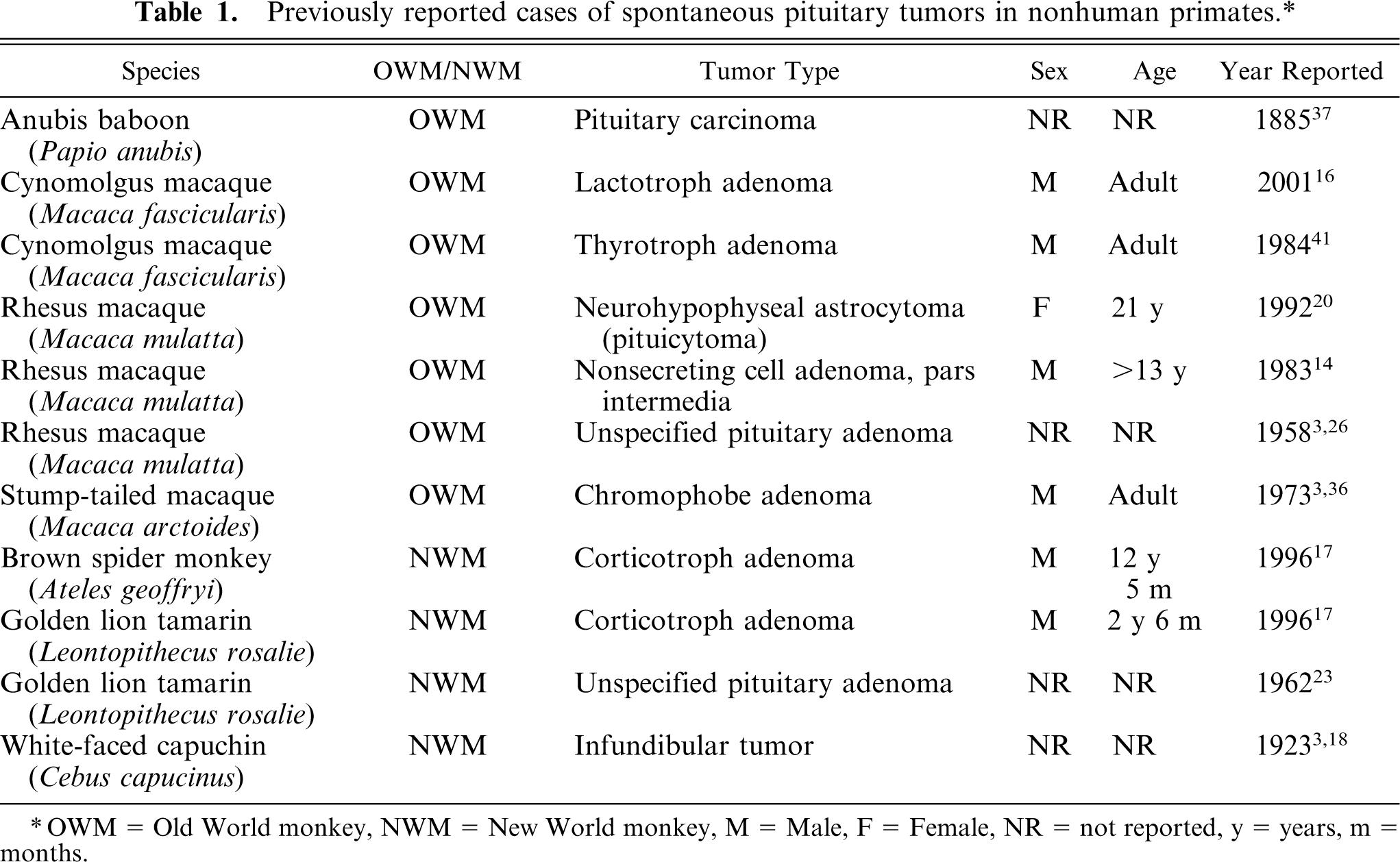
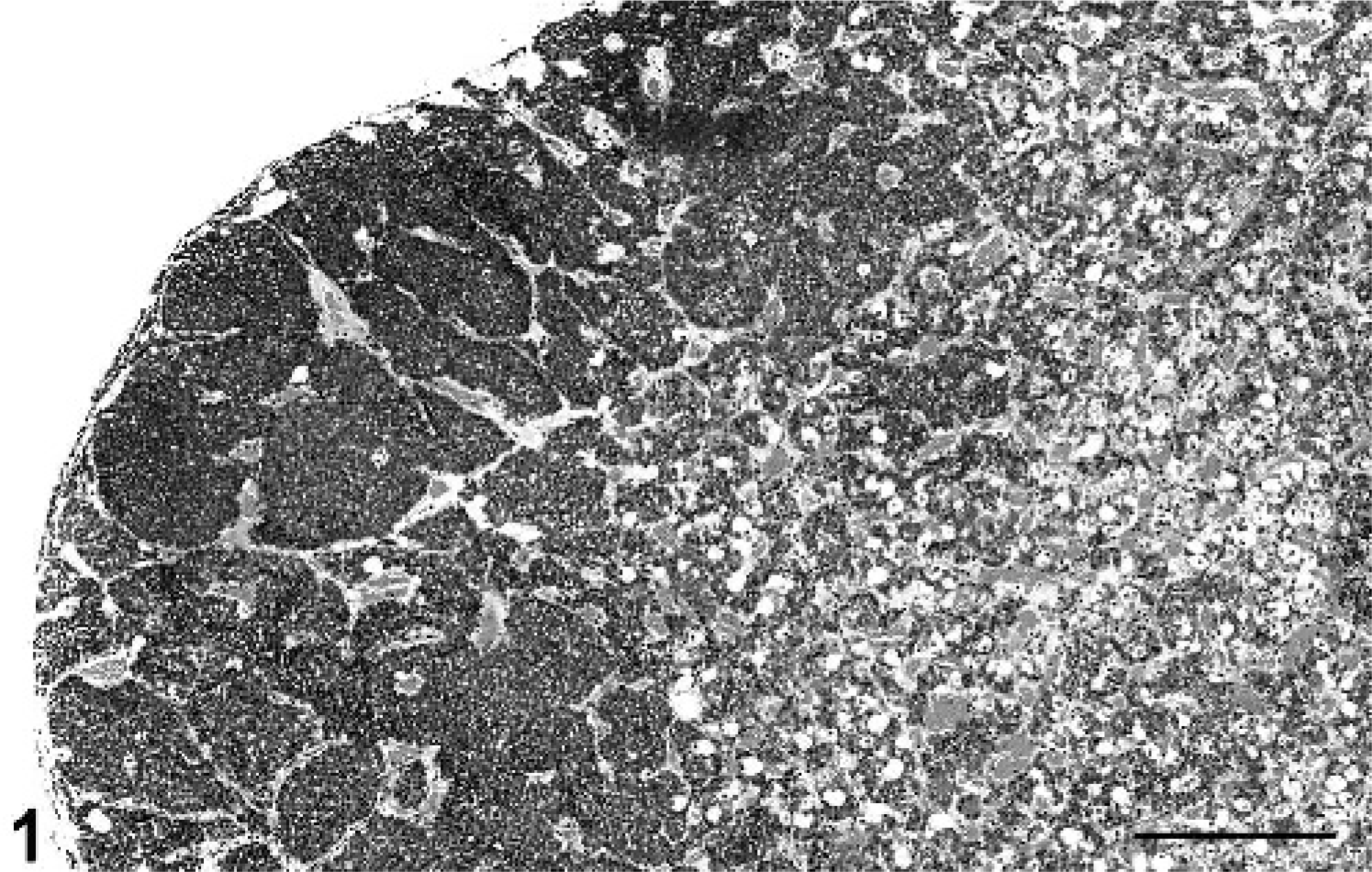
The nonhuman primate necropsy database at the Wake Forest University School of Medicine was searched for any cases of pituitary adenomas that were identified from 1994 to 2004. The reference population included a total of 491 primates: 373 cynomolgus macaques, 60 rhesus macaques, 41 African Green monkeys, and 17 stump-tailed macaques. The reference population included 209 males and 282 females. Primate cases were included in this retrospective study if they were found to have gross or histologic evidence, or both, of one or more pituitary adenomas. Cases with exclusively hyperplastic lesions, not forming discrete adenomas, were eliminated from the study (Fig. 1). Diagnosis of adenomas required the formation of discrete expansile foci with compression of adjacent parenchyma in the anterior pituitary (Fig. 2). Using this criterion, 14 cynomolgus macaques with pituitary adenomas were identified and included in this study. Seven of the macaques with pituitary adenomas also had some degree of pituitary hyperplasia.
Pituitary gland; cynomolgus macaque No. 7. Region of pituitary gland hyperplasia. HE. Bar = 0.625 mm.
Pituitary gland; cynomolgus macaque No. 1. Focal pituitary gland microadenoma. HE. Bar = 0.625 mm.
The complete medical records including postmortem examination data and archived glass slides were reviewed in each case for relevant signalment, clinical data, and gross and histologic features of the pituitary gland, other endocrine organs, and reproductive tissues. Reviewed archived glass slides contained tissues that previously had been fixed in neutral-buffered 10% formalin, paraffin embedded, sectioned in routine manner at 4- to 6-µm thickness, and stained with HE.
In preparation for immunohistochemical staining, the formalin-fixed, paraffin-embedded pituitary gland sections from all cases were sectioned and placed on positively charged glass slides. Immunohistochemical staining for FSH, LH, prolactin, human GH (HGH), TSH, and ACTH was performed on deparaffinized and rehydrated sections using a streptavidin/peroxidase/biotin/diaminobenzidine (DAB) chromogenic staining technique. Mouse antihuman β-FSH monoclonal antibody (dilution 1 ° 20,000, No. 0373), mouse antihuman β-LH monoclonal antibody (dilution 1 ° 40,000, No. 0374), and mouse antihuman β-TSH monoclonal antibody (dilution 1 ° 20,000, No. 0370) were obtained from Immunotech, Inc. (Westbrook, ME, USA). Rabbit antihuman prolactin antibody (prediluted, No. N1549), rabbit anti-HGH antibody (dilution 1 ° 1,600, No. N1561), and rabbit antihuman ACTH antibody (dilution 1 ° 8, No. N1531) were obtained from DAKO Corporation (Carpinteria, CA, USA). Appropriate positive- and negative-control tissues were stained with each batch. The positive-control tissue used was normal human pituitary gland. Each pituitary adenoma was classified according to its predominant hormone expression on the basis of immunohistochemical reactivity using a subjective scale of 0 (none), 1 (1–30%), 2 (30–60%), and 3 (60–100%) for number of immunopositive cells and a subjective scale of 0 (absent), 1 (mild), 2 (moderate), and 3 (marked) for staining intensity of the immunopositive cells to arrive at an overall assessment of each individual nodule.
Endocrine assays were performed in 7 macaques (Nos. 2–4, 8–10, and 14) to measure serum concentrations of prolactin, and in 3 macaques (Nos. 8, 10, and 14) to measure serum concentrations of growth hormone and cortisol. The endocrine assays were all performed by the Yerkes National Primate Research Center Endocrine Core Laboratory personnel using commercially prepared radioimmunoassay kits produced by Diagnostic Systems Laboratories (Webster, TX, USA). In all instances, the serum was retrieved from archived samples frozen at −80°C.
Results
Clinical features
The signalment, clinical signs of disease, and gross pathologic results are summarized in Table 2. Fourteen cases of pituitary adenoma in nonhuman primates were identified from a database search of 491 nonhuman primate cases, yielding an estimated prevalence of approximately 2.9% in the primate population examined. All primates either died naturally or were euthanized because of end-stage diseases at Wake Forest University School of Medicine and underwent complete postmortem examination and histologic evaluation of tissues between 1994 and 2004. All 14 cases were cynomolgus macaques, yielding an estimated prevalence of pituitary adenomas in cynomolgus macaques of approximately 3.8%. Of the 14 macaques, 8 were male (57.1%) and 6 were female (42.9%). This is compared with the reference population of 209 males and 282 females and the cynomolgus macaque reference population of 120 males and 253 females. The prevalence of pituitary adenomas in male cynomolgus macaques was 8 of 120 or 6.7%, and was 6 of 253 or 2.4% in female cynomolgus macaques. All animals were mature adults, ranging in age from 18 to 32 years, with mean (± SD) age of 23.65 ± 1.06 years (median, 23.4 years). This was significantly (
Pituitary adenomas in cynomolgus macaques (
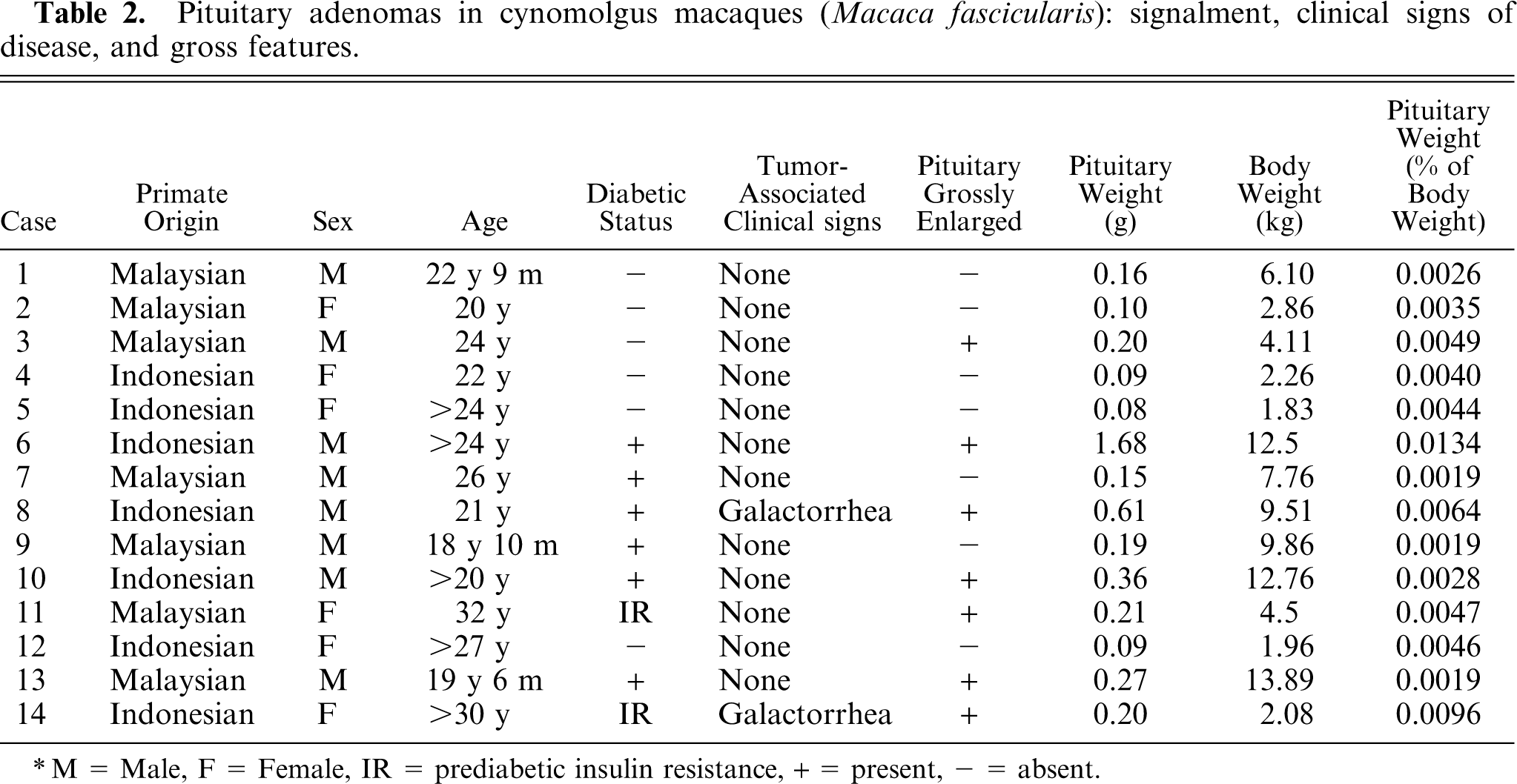
M = Male, F = Female, IR = prediabetic insulin resistance, + = present, − = absent.
Seven macaques died spontaneously and 7 macaques were euthanized because of end-stage diseases. Clinical signs of disease attributable to a pituitary mass were seen in 2 macaques (Nos. 8 and 14) with galactorrhea: a male macaque and a postmenopausal female macaque. In all other animals, the pituitary adenomas were incidental findings discovered at postmortem examination or histologic evaluation of the pituitary gland. Six of the macaques were known to be type-2 diabetics (Nos. 6–10 and 13) and 2 additional animals had prediabetic insulin resistance (Nos. 11 and 14) (Table 2). The macaques were involved in a range of experimental studies over the duration of their life at the primate center. The studies included research on diabetes mellitus, lipid metabolism, major histocompatibility complex, women's health, and cognitive function.
Gross pathologic features
On postmortem examination of the macaques, the pituitary gland was enlarged in 7 of the macaques (Nos. 3, 6, 8, 10, 11, 13, and 14) (Table 2, Fig. 3). In macaque No. 3, the pituitary gland was enlarged, contained a 0.5-cm-diameter, raised, white focus, and weighed 0.20 g (0.0049% of body weight). In macaque No. 6, the pituitary gland was markedly enlarged, extended out of the sella turcica, measured 1.5 × 1.0 × 1.0 cm, and weighed 1.68 g (0.0134% of body weight). In macaque No. 8, the pituitary gland was enlarged, measured approximately 1 cm in diameter, contained a soft, tan 1.0 × 0.5 × 1.0-cm mass, and weighed 0.61 g (0.0064% of body weight). In macaque No. 10, the pituitary gland was markedly enlarged, measured 1.0 × 1.0 × 0.5 to 0.75 cm, contained multiple, raised, 2- to 3-mm-diameter, pale nodules, and weighed 0.36 g (0.0028% of body weight). In macaque No. 11, the pituitary gland was uniformly enlarged and weighed 0.21 g (0.0047% of body weight). In macaque No. 13, the pituitary gland was moderately enlarged by a pale-tan, poorly demarcated mass, and weighed 0.27 g (0.0019% of body weight). In macaque No. 14, the pituitary gland was enlarged to approximately 4 times normal size with most of the expansion occupying the right lobe, and weighed 0.20 g (0.0096% of body weight). Overall, the 7 macaques with grossly enlarged pituitary gland had a pituitary weight range of 0.20–1.68 g, with an average pituitary weight of 0.50 g. The pituitary-to-body weight percentage range was 0.0019–0.0134%, with an average pituitary-to-body weight percentage of 0.0062%. The remaining 7 macaques did not have any visible pituitary gland enlargement and had a pituitary weight range of 0.08–0.19 g, with an average pituitary weight of 0.12 g. The pituitary-to-body weight percentage range was 0.0019–0.0046%, with an average pituitary-to-body weight percentage of 0.0033%. The average normal pituitary weight in macaques was 0.07 ± 0.02 g on the basis of 430 historical control animals.
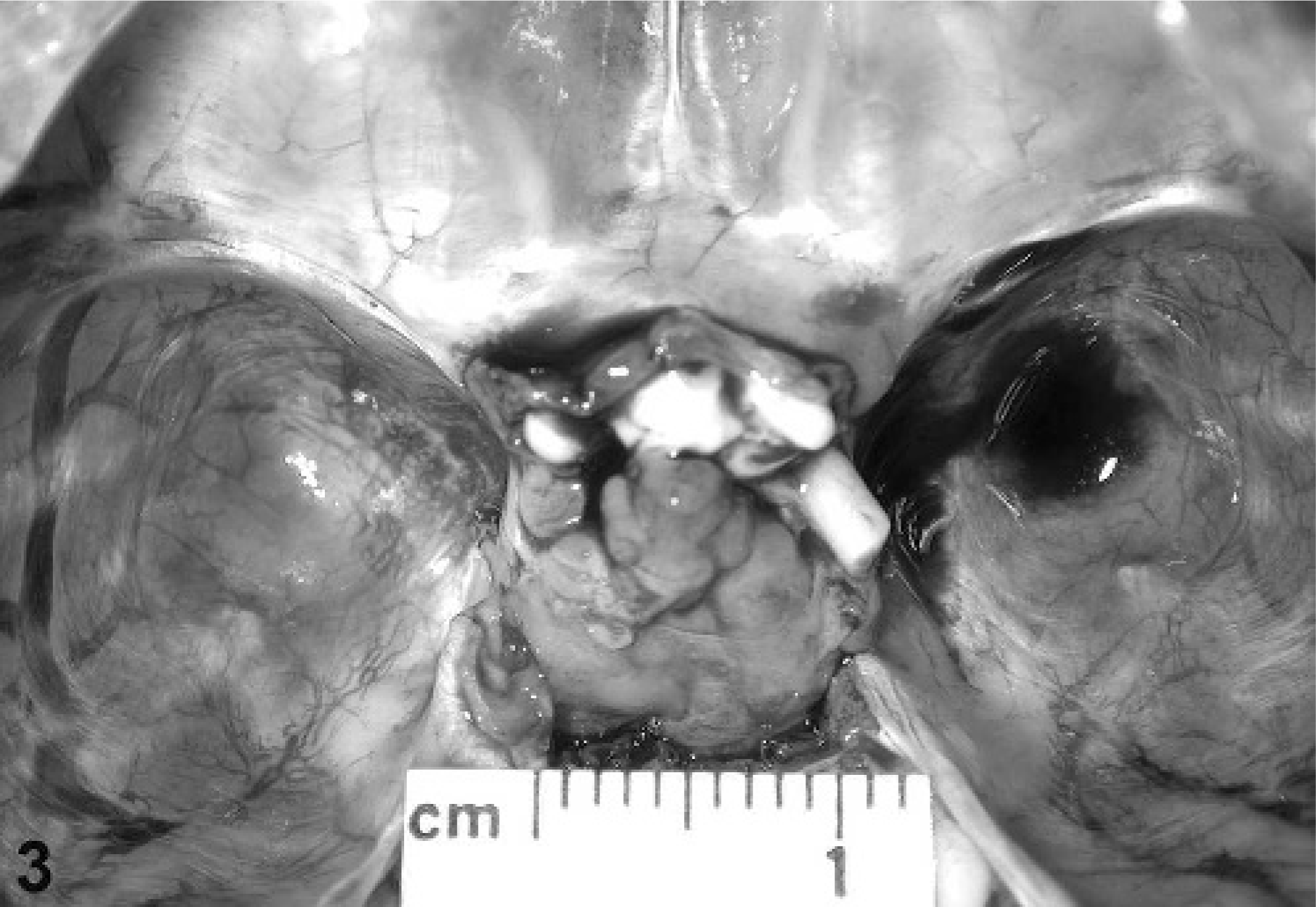
Pituitary gland; cynomolgus macaque No. 8. Photograph of grossly enlarged pituitary gland.
Histopathologic features
The histologic characteristics of the adenomas ranged from single to multiple microadenomas forming well-demarcated, unencapsulated, slightly expansile, densely cellular nodules with mild compression of the adjacent parenchyma. More advanced cases had large, poorly demarcated, expansile, densely cellular, often multinodular masses that effaced up to 90% of the normal pituitary gland architecture. A total of 35 adenomas of variable size were identified in the 14 pituitary glands included in the study. The number of adenomas per pituitary gland ranged from 1 to 20, with 3 macaques having more than 1 adenoma (Table 3). The tinctorial quality of the HE-stained adenomas varied: from pale eosinophilic to eosinophilic to pale basophilic. The neoplastic cells were arranged in densely packed cords and nests with a moderate amount of fine fibrovascular stroma. The neoplastic cells were polygonal with variably distinct cellular borders, moderate amounts of finely fibrillar cytoplasm, and central to paracentral, round to oval nuclei with finely stippled chromatin and single, variably prominent, basophilic nucleoli. There was minimal anisocytosis and anisokaryosis, and mitotic figures were rare to absent. There was no associated necrosis, hemorrhage, or inflammation in any of the adenomas. Acinar formation was not a feature in any of the adenomas examined. Several of the pituitary glands also had regions of hyperplasia, which were not assessed in this study.
Pituitary adenomas in cynomolgus macaques (
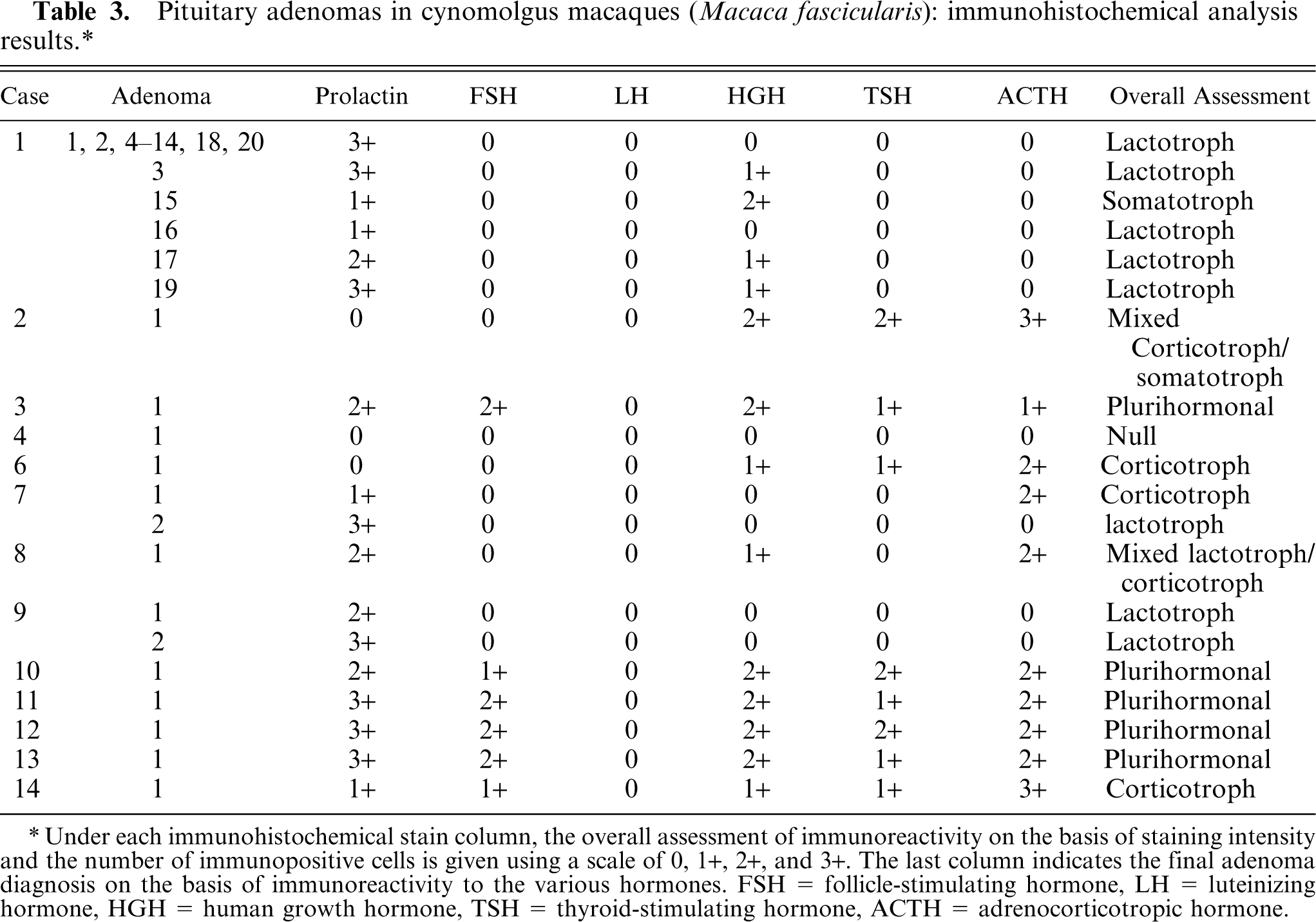
Under each immunohistochemical stain column, the overall assessment of immunoreactivity on the basis of staining intensity and the number of immunopositive cells is given using a scale of 0, 1+, 2+, and 3+. The last column indicates the final adenoma diagnosis on the basis of immunoreactivity to the various hormones. FSH = follicle-stimulating hormone, LH = luteinizing hormone, HGH = human growth hormone, TSH = thyroid-stimulating hormone, ACTH = adrenocorticotropic hormone.
Immunohistochemical analysis
The immunohistochemical results are summarized in Table 3. Of the 35 adenomas, 22 (62.9%) had predominantly prolactin expression and were considered lactotroph adenomas. Three adenomas (8.6%) expressed predominantly ACTH and were considered corticotroph adenomas. One adenoma (2.9%) predominantly expressed HGH and was considered a somatotroph adenoma. One adenoma (2.9%) expressed ACTH and HGH and was considered a mixed corticotroph-somatotroph adenoma. One adenoma (2.9%) expressed prolactin and ACTH and was considered a mixed lactotroph-corticotroph adenoma (Figs. 4, 5). Five adenomas (14.3%) expressed 3 or more hormones and were considered plurihormonal cell adenomas. Two adenomas (5.7%) did not express any hormone and were considered null cell adenomas. None of the adenomas expressed purely TSH, LH, or GH, although some of the plurihormonal adenomas expressed 1 or more of these hormones. In general, the grossly identified adenomas often expressed 2 or more hormones, whereas the smaller adenomas had a greater tendency to express a single predominant hormone.
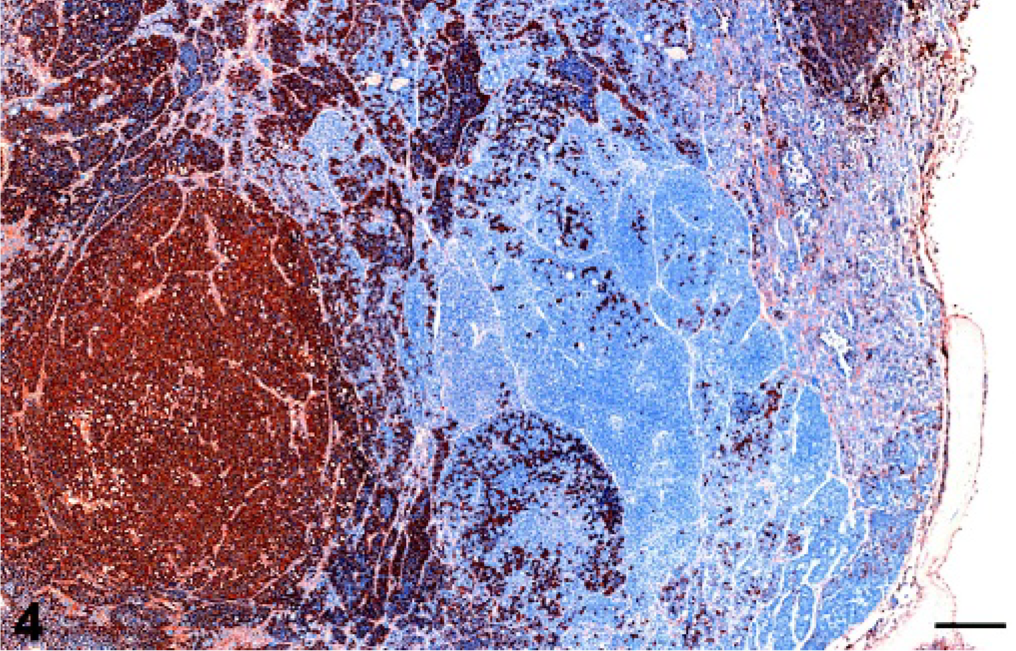
Pituitary gland; cynomolgus macaque No. 8. Multinodular pituitary gland macroadenoma with immunohistochemical staining demonstrating a mixed lactotroph-corticotroph adenoma using a streptavidin/peroxidase/biotin/diaminobenzidine (DAB) chromogenic staining technique. Bar = 0.3125 mm. Immunohistochemical staining for prolactin.
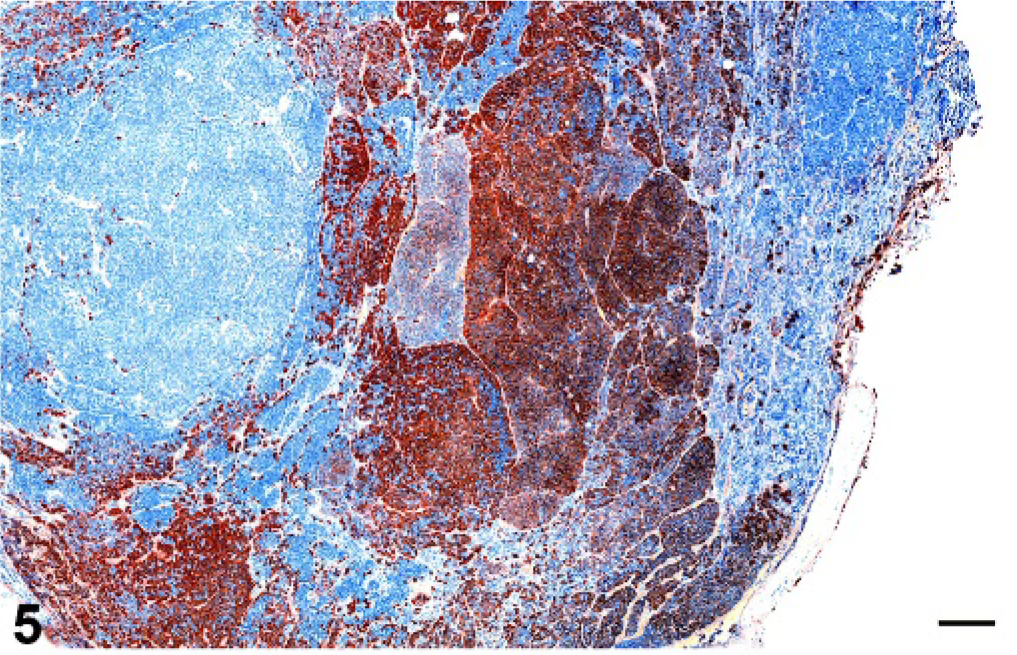
Pituitary gland; cynomolgus macaque No. 8. Multinodular pituitary gland macroadenoma with immunohistochemical staining demonstrating a mixed lactotroph-corticotroph adenoma using a streptavidin/peroxidase/biotin/diaminobenzidine (DAB) chromogenic staining technique. Bar = 0.3125 mm. Immunohistochemical staining for adrenocorticotropic hormone (ACTH).
Endocrine assays
Endocrine assays for serum prolactin, GH, and cortisol concentrations were performed in a subset of cases. Results of the endocrine hormone assays are summarized in Table 4. Estimated reference intervals for prolactin, GH, and cortisol concentrations in macaques were based on information available in scientific literature. The reference interval for serum prolactin concentration is 1–7 ng/ml in premenopausal and ovariectomized macaques. 21, 32, 35 The pulse reference interval for serum GH concentration in aged macaques is 1–10 ng/ml, 45 and in young macaques is 1–20 ng/ml, 45 whereas the baseline serum GH concentration in ovariectomized macaques is 1–4 ng/ml. 35 The reference interval for serum cortisol concentration is 10–25 µg/ml in premenopausal macaques and 15–40 µg/ml in ovariectomized macaques. 46 Serum prolactin concentration was markedly increased in macaque No. 8 (243.76 and 308.20 ng/ml), moderately increased in macaque Nos. 2 (42.91 ng/ml), 10 (32.98 and 17.73 ng/ml), and 14 (50.32 ng/ml), and minimally increased in macaque No. 4 (8.79 ng/ml). Serum prolactin concentration was within the reference interval in macaque Nos. 3 (4.26 ng/ml) and 9 (5.44 ng/ml). Serum GH concentration was mildly increased in macaque No. 14 (12.91 ng/ml) depending on the range used for comparison, and was below the reference interval in macaque Nos. 8 (<0.10 ng/ml) and 10 (0.17 ng/ml). Serum cortisol concentration was minimally increased to within the reference interval in macaques 8 (40.45 µg/dl), 10 (21.61 µg/dl), and 14 (19.08 µg/ml).
Hormone assay results for serum prolactin, growth hormone (GH), and cortisol in selected cynomolgus macaques (
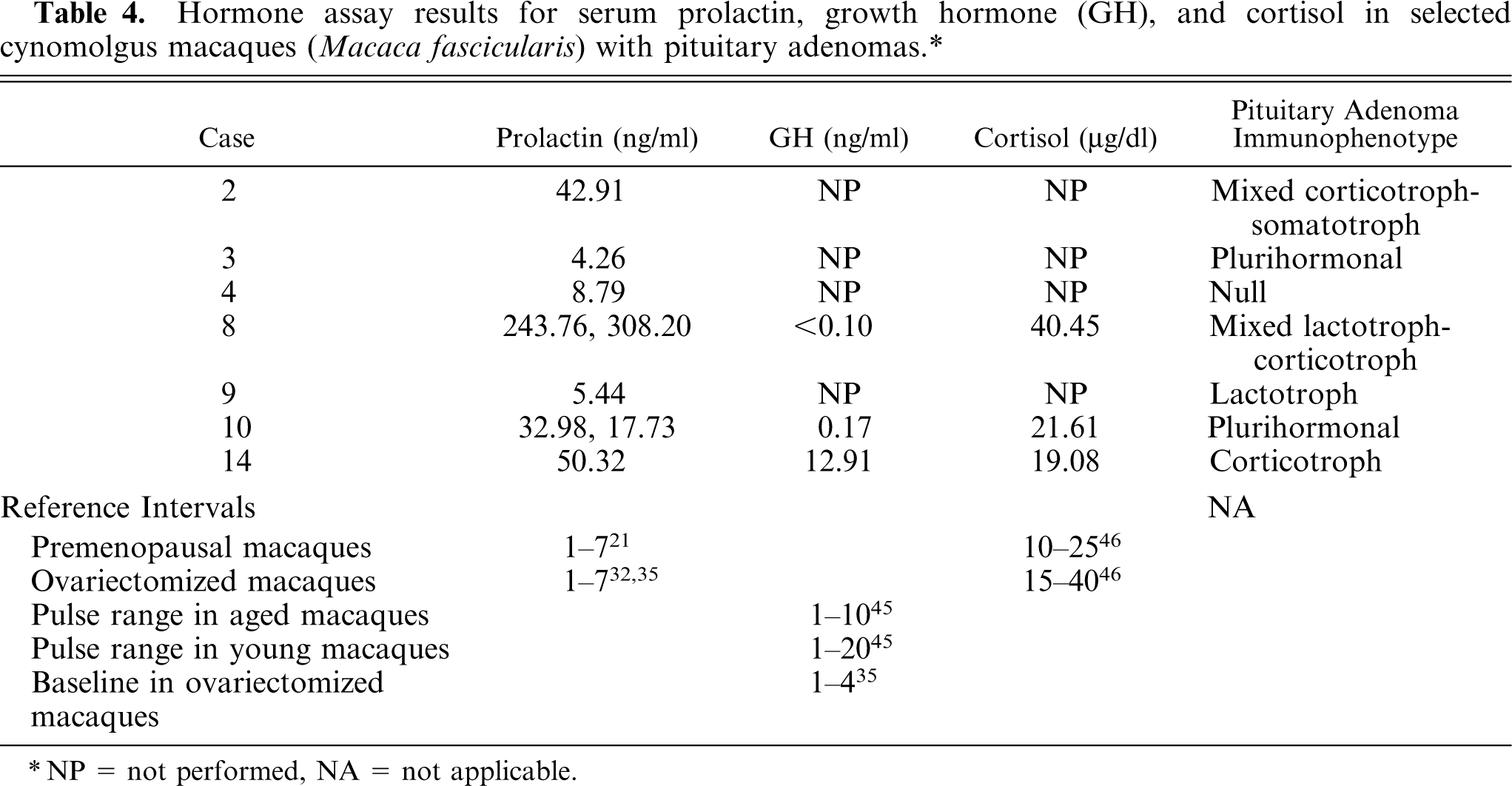
NP = not performed, NA = not applicable.
Discussion
In this study, we performed a retrospective case series evaluation of spontaneous pituitary adenomas in cynomolgus macaques. Previous case reports in literature have been sporadic, with only 11 reported cases of pituitary neoplasms in nonhuman primates dating back to 1885. Many pituitary tumors in nonhuman primates appear to be incidental findings, so it is likely that the prevalence of these tumors is higher than the reported rate. In this retrospective review, 14 cynomolgus macaques with spontaneous pituitary adenomas were identified in a population size of 491 macaques, yielding a prevalence of 2.9% in the population examined, and a prevalence of 3.8% in the cynomolgus macaques examined. Although this prevalence is lower than that published for spontaneous pituitary neoplasms in humans, useful correlations are still possible on the basis of the hormonal expression by the neoplastic cell populations.
The pituitary adenomas in the population examined developed only in cynomolgus macaques. Nonhuman primates used at this institute and included in the reference population were cynomolgus macaques, rhesus macaques, African Green monkeys, and stump-tailed macaques; however, the reference population is not balanced, and thus, it is difficult to evaluate possible species differences. There was some evidence for a sex predilection for males versus females, with an estimated prevalence of pituitary adenomas in male cynomolgus macaques of 6.7% versus 2.4% in female cynomolgus macaques. The pituitary tumors tended to develop in older animals. The population examined by necropsy at the institute is biased toward an older group of primates because the animals are necropsied only after spontaneous death or after humane euthanasia because of a life-threatening condition or poor quality of life. However, compared with reference population of diagnostic necropsy cases examined, the macaques with pituitary adenomas were significantly older than the reference population.
In this study, 6 of the cynomolgus macaques were known to be type-2 diabetics, with 2 additional macaques manifesting prediabetic insulin resistance. Type-2 diabetes mellitus is a spontaneously developing disease in aging cynomolgus macaques. 44 A variety of substances can cause insulin antagonism or resistance in animals, including excess GH or glucocorticoids. Among multiple other causes, excess GH or glucocorticoid concentration can result from a functional somatotroph pituitary adenoma or functional ACTH-secreting corticotroph pituitary adenoma, respectively. Both hormones can contribute to insulin resistance. Growth hormone can act as an insulin antagonist, 33 and glucocorticoids can inhibit cell receptor affinity for insulin and inhibit insulin action at a postreceptor level. 33 The development of spontaneous type-2 diabetes mellitus and pituitary adenomas in cynomolgus macaques provokes consideration as to an association between the 2 conditions.
The primates included in this study either had a grossly enlarged pituitary gland or single or multiple pituitary microadenomas identified on histologic examination. In human literature, a macroadenoma is defined as a pituitary adenoma >1 cm in diameter, and a microadenoma is a pituitary adenoma <1 cm in diameter. 7, 27 Although there is considerable variation in the body size of cynomolgus macaques, compared with that of humans, the pituitary adenomas identified in this study could be similarly categorized. The tumors could be considered macroadenomas if the pituitary gland was grossly enlarged, since the gland was typically enlarged to a size that exceeded 1 cm in diameter. The tumors could be considered microadenomas if they were identified on histologic examination only, since all the microscopically identified pituitary adenomas measured <1 cm in diameter. The classification is arbitrary, and its relevance in terms of clinical outcome or functionality of the tumor was not well examined. In this study, the macroadenomas typically manifested mixed or plurihormonal expression on the basis of results of immunohistochemical analysis, whereas each individual microadenoma typically expressed one predominant hormone. It can be speculated that the macroadenomas were composed of multiple microadenomas that coalesced over time.
The hormone expression patterns between spontaneous human and macaque pituitary adenomas had several similarities. Of spontaneous pituitary adenomas in humans, approximately 20–30% are lactotroph adenomas, 20% are null cell adenomas, 15% are plurihormonal cell adenomas, 10–15% are corticotroph adenomas, 10–15% are gonadotroph adenomas, 5% are somatotroph adenomas, 5% are mixed somatotroph-lactotroph adenomas, and 1% are thyrotroph adenomas. 4, 9, 15 In humans and the cynomolgus macaques of this study, lactotroph adenomas were the predominant type of pituitary adenoma. Plurihormonal cell adenomas are common in both species. Mixed cell adenomas develop in humans and cynomolgus macaques, although different combinations of hormones secreted were associated with somatotroph-lactotroph adenomas in humans and with corticotroph-somatotroph and lactotroph-corticotroph adenomas identified in the macaques. Although not mentioned previously, corticotroph-somatotroph adenomas 1, 39 and lactotroph-corticotroph adenomas 29 have also been sporadically identified in humans.
Serum hormone concentrations and hormone expression of the neoplastic cells that was based on immunohistochemical analysis were not closely associated in this study. Two macaques with serum prolactin concentration within the reference interval (Nos. 3 and 8) had 2+ or 3+ prolactin expression on immunohistochemical analysis, whereas other macaques with moderately high serum prolactin concentration (e.g., Nos. 2 and 14) had none to mild 1+ prolactin expression on immunohistochemical analysis. All 3 macaques in which serum GH concentration was measured had values below reference interval or only minimally increased serum values and either 1+ or 2+ GH expression on immunohistochemical analysis. Similarly, all 3 macaques, in which measured serum cortisol concentration had minimally increased to within reference interval values, had 2+ or 3+ ACTH expression on immunohistochemical analysis. These results suggest that serum hormone concentrations are not a reliable indicator of hormone expression of the neoplastic cells when it is based on immunohistochemical analysis. Discordance between serum hormone concentration and pituitary tumor immunostaining could be attributable to improper secretion of hormones or secretion of abnormal hormones that are not recognized by antibodies used in serum hormone assays. 2
In conclusion, we have described a series of spontaneous pituitary adenomas in nonhuman primates that includes the first reported cases of a somatotroph adenoma, mixed hormonal cell adenomas, and plurihormonal cell adenomas in nonhuman primates. The study indicated that, similar to humans, cynomolgus macaques develop spontaneous microadenomas and macroadenomas of the pituitary gland; that lactotroph adenomas are the predominant type; and that both species can develop mixed and plurihormonal cell pituitary adenomas. These similarities suggest that cynomolgus macaques may provide a potential model for the study of certain pituitary disorders in humans. Further studies of pituitary lesions in nonhuman primates, including various primate species, would be necessary for clarifying species variation in prevalence and for evaluating the comparative biology of these neoplasms.
Footnotes
Acknowledgements
We thank Jean Gardin, Kathy Kaplan, Beth Phifer, and Hermina Borgerink at Wake Forest University for their valuable technical assistance. We also thank Dr. A. Julian Garvin, MD, PhD at Wake Forest University for his assistance in interpretation of pituitary lesions. We thank Beth Mahler with Experimental Pathology Laboratories, Inc., and National Institute of Environmental Health Sciences for her expertise in preparation of the digital images. This work was supported by the National Institutes of Health, National Center for Research Resources grant T32 RR07009.
