Abstract
Abnormal growths were observed on the lips and in the oral cavities of 2- and 3-year-old Chinook salmon (Oncorhynchus tshawytscha) maintained in one freshwater and one saltwater captive fish-rearing facility in the Columbia River (Pacific Northwest). Initially presenting as bilaterally symmetrical, red, irregular plaques on oral mucosal surfaces, the lesions developed progressively into large, disfiguring masses. Of the 502 natural parr collected for captive broodstock, 432 (86%) displayed these tumors, whereas cohort salmon (i.e., same year classes) in these same facilities remained unaffected. Morphologically similar neoplasms were collected occasionally from adult Chinook salmon that had returned to their natal streams. Histologic features of the tumors suggested that they were derived from the portion of dental lamina destined to form tooth root sulci; therefore, these neoplasms were diagnosed as ameloblastomas. The lesions also resembled archived specimens of Chinook salmon oral tumors, which had been described decades earlier. Etiologic investigations performed during the current outbreak included bacteriologic, virologic, genetic, ultrastructural analyses, and cohabitation exposure studies. Results of these efforts did not indicate an obvious genetic basis for this syndrome, attempts to isolate potentially causative viruses or bacteria were negative, and disease transmission to naïve fish was unsuccessful. A few intracytoplasmic hexagonal structures, possibly consistent with viral particles (∼100 nm), were observed ultrastructurally in a tumor cell from 1 of 6 specimens submitted for transmission electron microscopy. Although the presence of these particles does not constitute sufficient evidence for causality, an infectious or multifactorial etiology seems plausible.
Salmon populations on the west coast of the USA have experienced severe declines during the past several decades as a result of anthropogenic and natural factors. Contributing causes include habitat degradation and loss, hydropower development, overharvest, and the introduction of invasive species. 34 Chinook salmon (Oncorhynchus tshawytscha) populations, in particular, have diminished dramatically. Of 17 extant Chinook salmon evolutionary significant units (ESUs), 2 are currently listed as endangered under the US Endangered Species Act, 7 are listed as threatened, and 1 is considered to be a species of concern. 25 To prevent the continued decline and possible extinction of these distinct populations, conservation efforts have included captive broodstock propagation, dam removal or mitigation, and the restoration of degraded habitats. To maximize the effectiveness of any of these recovery efforts, it is necessary to identify and characterize diseases that might adversely impact hatchery-reared or wild populations. Of utmost concern is the potential for disease transmission from cultured to wild stocks, or vice versa.
In December 2002, abnormal growths were observed on the lips and in the oral cavities of several 2-year-old Chinook salmon that were being reared at Manchester Research Station in Port Orchard, Washington. These fish were originally collected in the summer of 2001 as wild parr from the upper Grande Ronde River as part of the Grande Ronde Basin Spring Chinook Salmon Captive Broodstock Program in northeast Oregon. After the initial collection, the cohort was divided into freshwater and saltwater rearing groups and cultured at the Bonneville Fish Hatchery in Oregon (freshwater component) and the Manchester Research Station in Washington (saltwater component). Sampling at the Bonneville Fish Hatchery in April 2003 revealed that the salmon raised in freshwater also had oral tumors. Other Grande Ronde River cohorts (i.e., year classes) of salmon in these same facilities remained unaffected. Meanwhile, during the summer of 2003, similar lesions were identified in the mouths of several adult Chinook salmon that were collected as they returned to spawn in their natal Oregon streams (Fig. 1). These adult salmon were captured at weirs in three different rivers in two separate watersheds, and at least one of these fish was considered to be of natural origin based on scale analysis, fin condition, and the absence of any implanted tags or external marks that are commonly used to identify hatchery origin fish. In addition, during the summers of 2004 and 2005, several natural origin and hatchery-raised adult Chinook salmon in Idaho presented with essentially identical oral lesions.
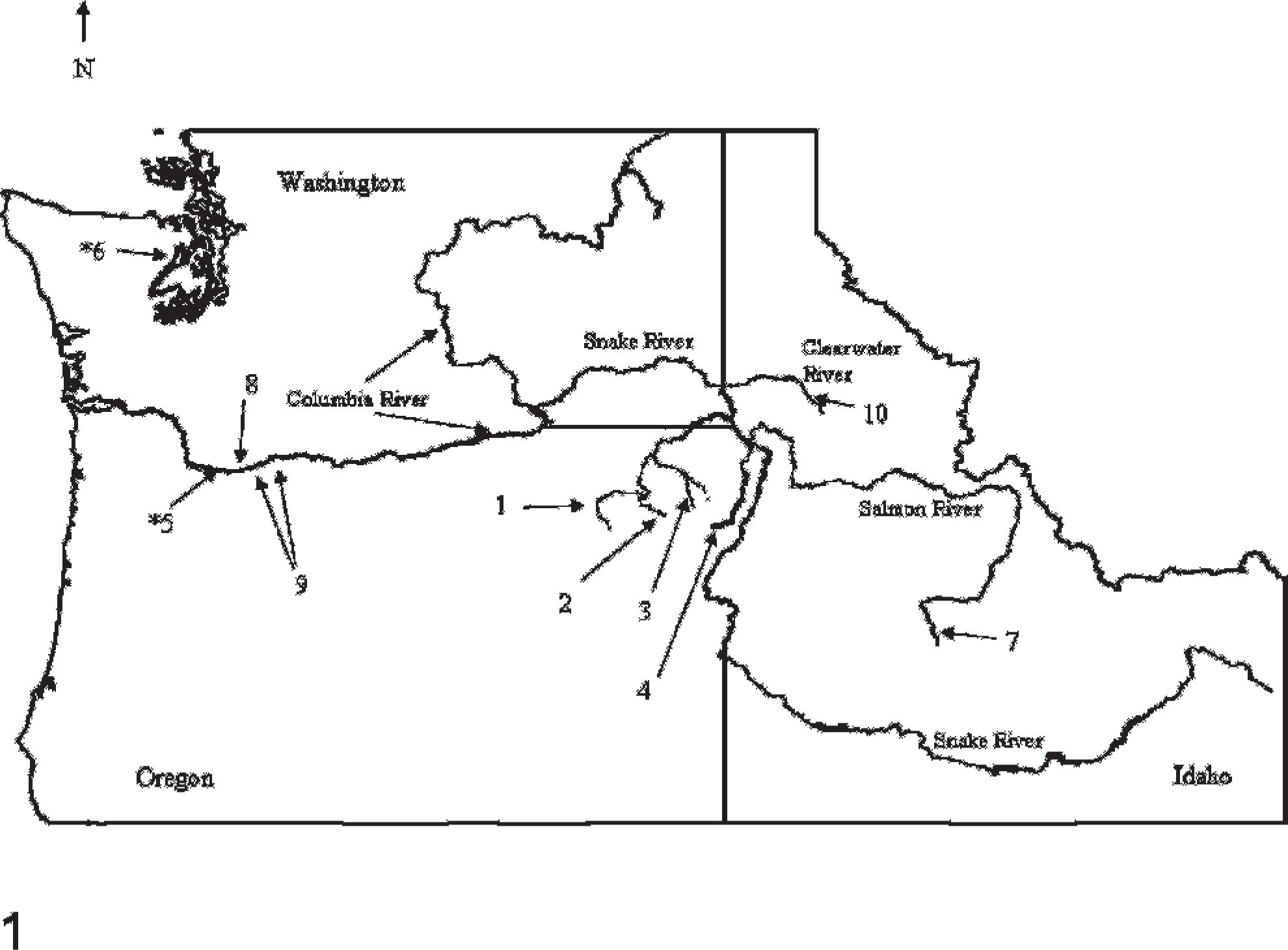
Geographic collection locations of Chinook salmon with tumors. (1) Grande Ronde River; (2) Catherine Creek; (3) Lostine River; (4) Imnaha River; (5) ∗Bonneville Hatchery (Rearing facility for Captive Broodstock Program); (6) ∗Manchester Research Station (Rearing facility for Captive Broodstock Program); (7) Sawtooth Hatchery; (8) Little White Salmon National Fish Hatchery; (9) Capture locations for two adult Chinook salmon with tumors in the lower Columbia River; (10) Kooskia National Fish Hatchery, Clearwater River, Idaho. ∗ (5 and 6) Bonneville Hatchery and Manchester Research Station are the rearing facilities for the Captive Broodstock Program. Tumors were first observed in the Captive Program Grande Ronde stock (these fish were collected as natural parr from the Grande Ronde River). All other designations refer to locations in which natural and hatchery returning adults were captured.
Described herein is the spectrum of macroscopic and microscopic features that characterize these neoplasms, the diagnostic procedures that have been performed to date, and possible etiologic origins of this epizootic. Identifying the underlying cause(s) of this condition would allow natural resource managers to recognize and evaluate factors that might contribute to tumor development and mitigate potential risks to wild Chinook salmon populations in these threatened ESUs.
Materials and Methods
Sampling/collection
Hatchery fish were examined by Oregon Department of Fish & Wildlife (ODFW) Fish Health Services personnel for grossly visible abnormalities at adult collection weirs, during all handling activities, and at spawning. While conducting routine sample collection of broodstock and fish, salmon with macroscopic lesions were euthanized by using MS-222 (Finquel) or Aqui-S followed by stunner machine or blunt trauma to the head. Oral cavities and extraoral surfaces were thoroughly examined. Suspect tumor tissue was photographed, excised, and placed in 10% neutral buffered formalin (pH 7.0) for histologic examination. Wild caught fish were similarly examined and sampled at one of several points of contact: when brought into the Captive Broodstock Program as wild parr, when collected at weir sites along rivers in northeastern Oregon, or when handled during spawning.
Histopathology
Between April 2003 and July 2006, the Registry of Tumors in Lower Animals (RTLA) in Sterling, Virginia, received donations of 27 Chinook salmon specimens in which one or more oral or facial neoplasms was present (Table 1). Formalin-fixed tissue specimens or histologic sections on glass slides were submitted from institutions in 3 northwestern American states and contributors included the ODFW and the Idaho Department of Fish and Game. Fixed tissues were trimmed for optimal sectioning, and then processed with an automatic processor (Tissue-Tek VIP; Sakura Finetek U.S.A., Inc., Torrance, CA, USA) and paraffin embedded according to routine histologic methods, microtomed to 4- to 6-µm thick sections, mounted on glass slides, and stained with HE (Hematoxylin 2 and Eosin-Y; Richard-Allan Scientific, Kalamazoo, MI, USA). Two American College of Veterinary Pathology-certified pathologists who specialize in fish histopathology examined the slides and arrived at a consensus diagnosis for each histologic section. During their evaluation, the pathologists had access to histologic sections from 2 prior cases of similar oral tumors in Chinook salmon that were archived in the RTLA collection (RTLA case Nos. 248 and 259).
Summary of Chinook salmon ameloblastoma cases submitted to the RTLA.∗
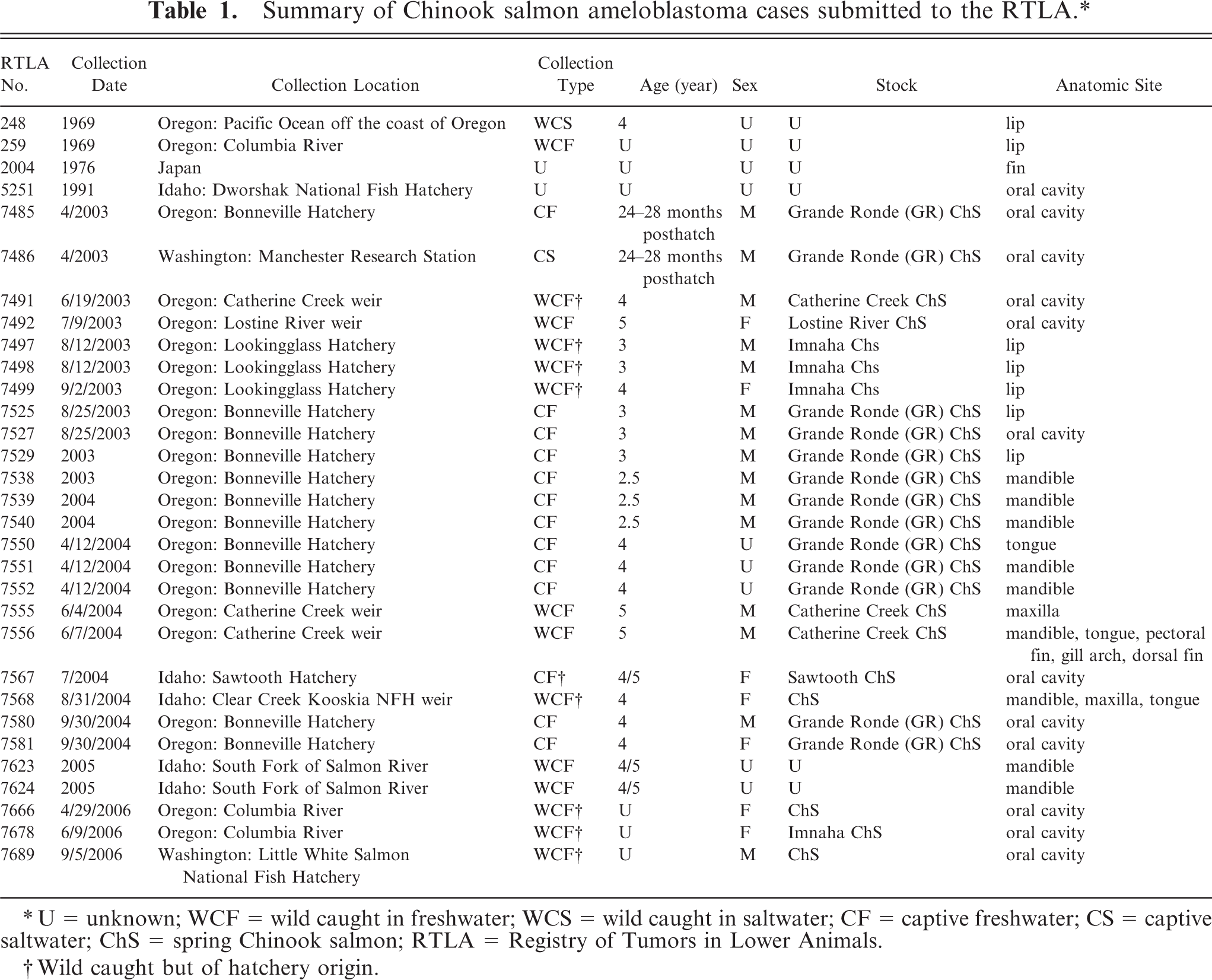
∗ U = unknown; WCF = wild caught in freshwater; WCS = wild caught in saltwater; CF = captive freshwater; CS = captive saltwater; ChS = spring Chinook salmon; RTLA = Registry of Tumors in Lower Animals.
† Wild caught but of hatchery origin.
Bacteriology
As part of a comprehensive evaluation of fish health, the kidneys of 3 tumor-bearing fish (RTLA case Nos. 7492, 7555, and 7556) were cultured aseptically. These fish had been collected as adults from streams in northeast Oregon and examined at the ODFW Fish Health Services Laboratory in La Grande. A flame-sterilized microbiology loop was used to obtain the fresh kidney specimens. Bacteriologic analysis was also performed on tumor samples from one of these fish (RTLA case No. 7492). A sharp, sterile scalpel was used to collect a clean tissue sample. All samples were streaked onto tryptone-yeast extract agar plates and incubated at 18°C. After 5–7 days, plates were examined for growth. Testing for the presence of Renibacterium salmoninarum, the causative agent of bacterial kidney disease (BKD), was performed by using an ELISA method, as previously described. 26 Testing for BKD was performed to determine whether it was a possible causative factor for tumor development.
Virology
Diagnostic procedures performed at ODFW to identify potential viral (and other) pathogens included: cultures from tumor samples and internal organs (including primary cultures of tumor tissue); transmission electron microscopic examination of tumor tissue, tumor cell culture, and internal organs; polymerase chain reaction (PCR) performed on tumor cell DNA extracts using primers designed to identify Oncorhynchus Masou virus (OMV) and Herpesvirus salmonis; injection of whole cell and cell filtrate tumor homogenates into spring Chinook fingerlings (6-month duration experiment); and cohabitation of spring Chinook fingerlings with tumor-laden adults (3-week exposure followed by a 6-month monitoring period).
Tissue culture
Diagnostic procedures for virus testing were performed at the ODFW Fish Health Services at Oregon State University in Corvallis, Oregon, and generally followed the guidelines in the Fish Health Section Blue Book. 1 Sampling for viruses by cell culture was performed using pyloric caeca/kidney/spleen and tumor tissue homogenates. Briefly, 6 1-cm3 samples of excised tumor tissue, and 6 kidney/spleen pools from affected individuals were homogenized separately in Hanks balanced salt solution (Sigma Chemical, St. Louis, MO, USA) supplemented with 0.1% fetal bovine serum and clarified by centrifugation at 2,000 × g for 10 minutes. The resulting supernatant was then diluted 1 : 2 in an antibiotic/antimycotic mixture containing 250 µg/ml gentimicin, 1,000 IU/ml penicillin, 1,000 µg/ml streptomycin, and 20 µg/ml amphotericin B. Sample solutions were stored at 4°C overnight. Replicate aliquots of 200 µl were then introduced onto fresh monolayers of epithelioma papulosum cyprini (EPC), 7 Chinook salmon embryo (CHSE-214), 8 rainbow trout gonad, 39 bluegill fin (BF-2) (American Type Culture Collection 1975), and fathead minnow 11 cells in 12-well tissue culture plates. Cell culture media consisting of minimum essential media (GIBCO-BRL, Bethesda, MD, USA) supplemented with 5% fetal bovine serum and containing antibiotic/antimycotic components described previously was added to the sample aliquots on the plates. Two sets of tissue culture plates were inoculated with replicate sample aliquots as described; one set was incubated at 15°C and the other set incubated at 8°C. Samples were examined at least twice weekly. All samples were subcultured onto fresh cells after 28 days and allowed to incubate an additional 14 days.
Primary cell culture
Two sections of exuberant tissue, approximately 1 cm3 each, were placed aseptically in Petri dishes with a small amount of cell culture medium (as described previously) and finely minced using sterile scalpel blades. Minced tumor pieces were then “planted” on the dry growth surfaces of two 25-cm3 tissue culture flasks and allowed to adhere for approximately 30 minutes. Cell culture medium was carefully introduced into the flasks so the explants would not become dislodged, and sufficient medium was used to completely submerse all planted tissue. Explant cultures were incubated at 15°C and fresh medium was added to the flasks as necessary.
Transmission electron microscopy—primary tumortissue
Transmission electron microscopy (TEM) was performed on two different groups of fish at two different facilities: 1) tumor tissues excised from 3 Chinook salmon (RTLA case Nos. 7550, 7551, and 7552) were submitted to the Laboratory for Advanced Electron and Light Microscopy (LAELOM–College of Veterinary Medicine, North Carolina State University, Raleigh, NC); and 2) tumor tissues from 2 additional Chinook salmon (RTLA case Nos. 7580 and 7581) were also examined by TEM at the Electron Microscope Service Laboratory (EMSL), Department of Botany and Plant Pathology at Oregon State University, Corvallis, Oregon. The fresh tissues submitted to LAELOM were trimmed to 1-mm3 cubes and placed in McDowell's and Trump's 4F : 1G fixative 20 for 1 hour at 4°C. After 2 rinses in 0.1 M sodium phosphate buffer (pH 7.2), samples were placed in 1% osmium tetroxide in the same buffer for 1 hour at room temperature. Samples were rinsed twice in distilled water and dehydrated in an ethanolic series culminating in 2 changes of 100% acetone. Tissues were then placed in a mixture of Spurr resin 32 and acetone (1 : 1) for 30 minutes, followed by 2 hours in 100% resin with 2 changes. Finally, samples were placed in fresh 100% resin within molds and polymerized at 70°C for 8 hours. Semithin (0.25–0.5 µm) sections were cut with glass knives and stained with 1% toluidine blue-O in 1% sodium borate. Ultrathin (70–90 nm) sections were cut with a diamond knife, stained with methanolic uranyl acetate, followed by lead citrate, and examined with TEM. The tumor tissues that were examined ultrastructurally at EMSL were processed in a similar fashion as previously, except that they were preserved initially in 4% gluteraldehyde fixative.
Transmission electron microscopy—tissue culture
TEM was performed on clarified supernatant pooled from tissue samples inoculated onto various cell lines (see Tissue culture section). After 42 days of incubation, 60 500-µl samples of supernatant were taken from the wells of tumor tissue preparation, inoculated onto the various fish cells, and pooled into 2 30-ml samples (one pool for each incubation temperature). Pools were clarified by centrifugation at 5,000 × g and this clarified supernatant was then centrifuged at 100,000 × g for 1 hour. The resulting pellets were negatively stained for examination by TEM. In addition, approximately 3 weeks after seeding primary culture tissue, morbid and dead cells were collected, pelleted by centrifugation, and prepared by thin section for examination under TEM.
Polymerase chain reaction assay
PCR was conducted on DNA extracts from 6 tumor tissues, 6 kidney/spleen pools, 1 primary cell culture debris pool, and a portion of each of 2 pellets of pooled supernatant from tumor tissue culture assays. DNA was extracted using Qiagen DNeasy Tissue Kit 69504, (Qiagen Sciences, Gaithersburg, MD, USA). The PCR protocol and primers were specific to H. salmonis and OMV and would generate products from those targets, if present. 40 The samples were amplified by using a Peltier Thermal Cycler PTC-200 (Bio-Rad, Hercules, CA, USA).
Exposure experiments
Three Chinook salmon exhibiting various stages of gross tumor development were transported from Bonneville Hatchery to the Center for Fish Disease Research, Oregon State University, Corvallis, Oregon. These fish were held in a 100-liter aquarium tank that was plumbed to divert all effluent to an adjacent 100-liter tank containing 100 spring fingerling Chinook salmon obtained from McKenzie Hatchery, Leaburg, Oregon. No additional water was supplied to the fingerlings. The fingerlings were exposed to effluent for 3 weeks, at which time the adults were removed, sacrificed, and examined. Postexposure, the fingerlings were monitored and examined for tumor induction for 6 months. Every month, 5 fish were sampled for tissue culture for virus production and visually examined for tumor growth. In addition to these effluent-exposed fish, 2 groups of 25 fingerling spring Chinook salmon were injected intraperitoneally with tumor tissue homogenate. One group received whole homogenate, the other group received homogenate passed through a 0.22-µm filter. Inoculated fish were also monitored for 6 months postexposure by tissue culture, and by visual examination of external and internal appearance.
Genetic analysis
To evaluate population genetic structure and diversity among affected and nonaffected individuals, variation was examined by using a suite of 8 putatively neutral microsatellite markers. 3 Genomic DNA was extracted and genotyped from a total of 228 individuals from the upper Grande Ronde River Captive Broodstock fish, including 192 individuals initially identified as tumor-affected and 36 unaffected individuals (see Results section for limitations in the use of the unaffected control group), as described by Banks et al. 2000. 3 Expected and observed heterozygosities were evaluated to estimate genetic diversity. 23 The number of genetically effective breeders (N b) that contributed to the sample of affected individuals was estimated 37,38 and compared to the minimum number of possible parents based on allele counts. Finally, because the control group was compromised (discussed later), the results were compared with similar studies of genetic diversity in other small Snake River Chinook salmon populations to determine whether 1 family, or even a small number of related individuals, might have produced all the tumor-affected individuals.
Results
Combining results from the freshwater and saltwater rearing facilities, the incidence of tumors from the Grande Ronde River cohort reached 86% (n = 432) of the 502 natural parr collected.
Macroscopic appearance and histopathology
Although the neoplasms in this study displayed variations in anatomic site, extent of growth, and precise growth pattern, they were united by a suite of common morphologic characteristics. In 21-month to 2-year-old hatchery salmon, the tumors initially presented macroscopically as multiple red, irregular, linear plaques along the inner buccal mucosae of the maxilla or mandible (Fig. 2). These initial lesions were often bilaterally symmetrical. Fish sacrificed at a later stage of disease progression had large, pink, nodular masses (Fig. 3) that caused disfigurement of the lips and oral cavity in advanced cases (Fig. 4). Rarely, single nodules were observed on the gill arch or pectoral fin, in addition to oral lesions. Affected wild-caught salmon generally had advanced tumors that were virtually indistinguishable from advanced lesions of Grande Ronde River Captive Broodstock fish.

Early ameloblastoma. Several pink-to-red linear plaques along the palate and lips of this Chinook salmon are seen, which are consistent with an early lesion. These plaques tended to occur in areas where the normal dental lamina are located.
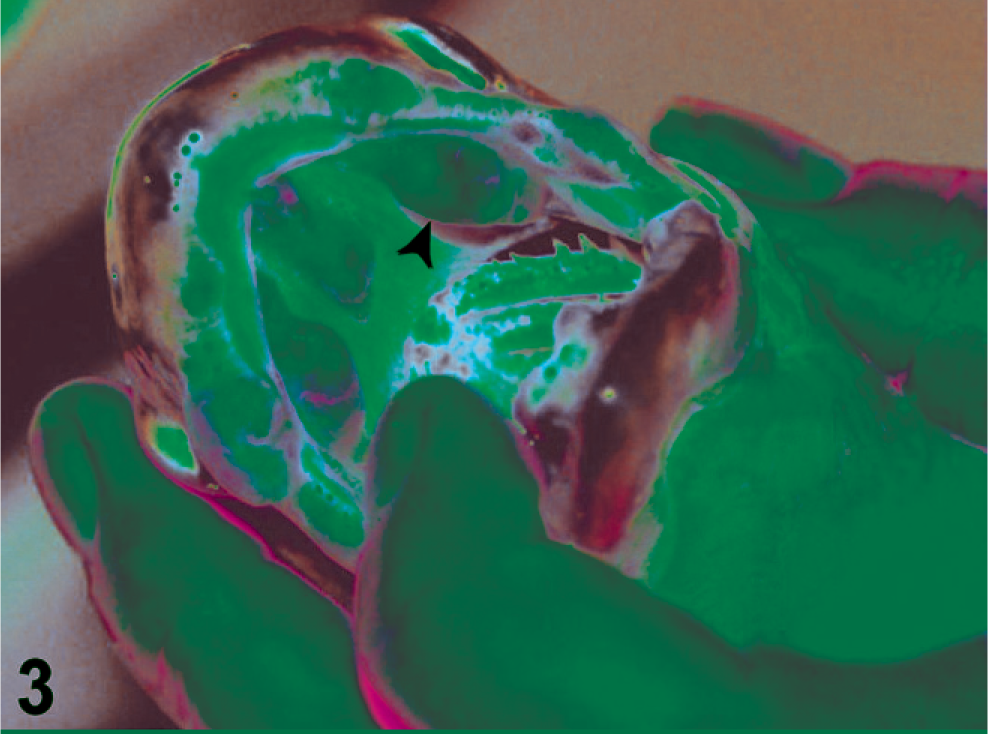
Moderately severe ameloblastoma. A Chinook salmon with large nodular masses, which are associated with a moderately severe case of ameloblastoma.
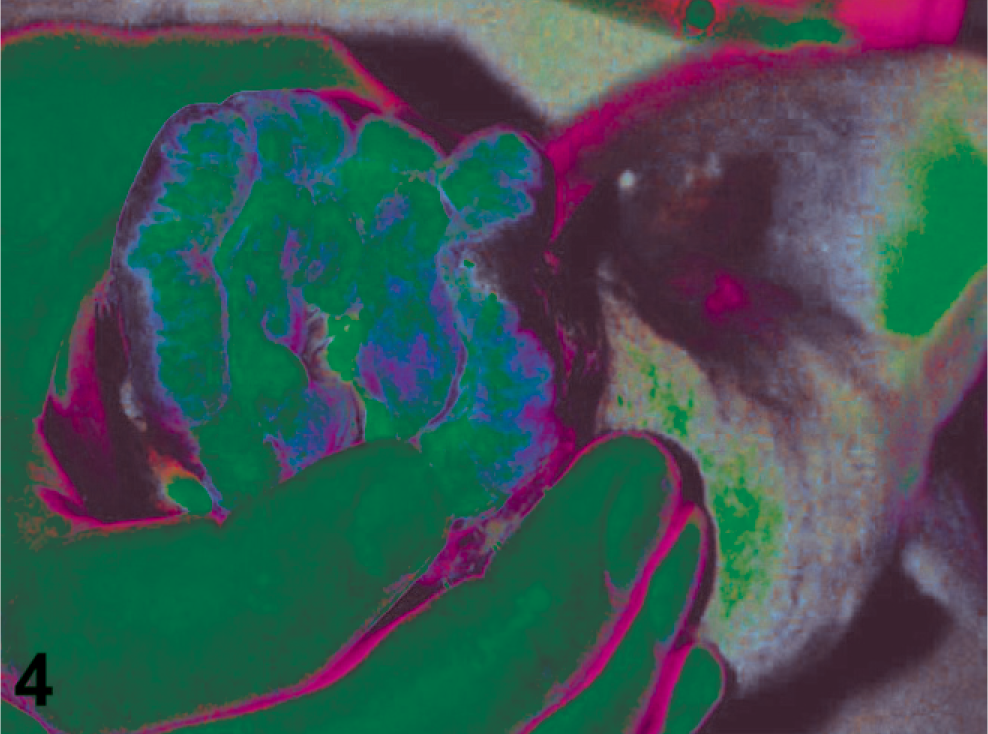
Severe ameloblastoma. The lips and palate of this Chinook salmon are markedly deformed by numerous, partially confluent tumors. Such large masses can disrupt normal feeding.
Microscopic changes in fish with very early proliferative disease were consistent with epithelial hyperplasia and mild inflammation (Figs. 5, 6). In such cases, the mucosal epithelium was thickened (with or without focal erosions), there was minimal if any cellular atypia, and the exuberant tissue did not extend beneath the epidermal or oral mucosal basement membrane. Conversely, lesions in moderately to severely affected fish consisted of solid islands and trabecular cords of neoplastic tissue that penetrated deep into the fibrovascular dermis or propria mucosa (Fig. 7). The tumor tissue consisted of pseudostratified columnar or stratified polygonal cells that palisaded along, or were constrained by, basement membranes. A universal feature of the tumors, to varying degrees, was a tendency for acantholytic-type cell dissociation and cleft formation to occur in the centers of the neoplastic cords and islands (Figs. 7, 9). These long, irregular clefts often appeared to connect with surface invaginations of the lip epidermis or the oral mucosa. Another recurring trait of these tumors was contiguity between the tumor cells and the basal layers of the epidermal or oral mucosal epithelium (Fig. 8). In addition, in sections that contained teeth, cords of tumor cells were occasionally observed to branch off tooth root sulci, and in such instances, the palisading tumor cells merged with ameloblasts of the outer dental lamina (Figs. 8, 4). Individual tumor cells had medium-to-large, oval or reniform nuclei, one or more small but distinct nucleoli, and moderate-to-abundant amounts of well-defined amphophilic cytoplasm. Cells situated along basement membranes tended to have apical nuclei. In the superficial areas of a few masses, the tumor tissue was chiefly comprised of mucus-containing goblet cells (Fig. 10). On average, there were 3–4 mitoses per microscopic field (40× objective). Intercellular bridges (artifact-induced indications of desmosomal attachments) were commonly observed (Fig. 11); however, there was never evidence of keratinization. With one possible exception, in which a deformed tooth was present within the tumor (Fig. 12), the neoplastic tissue was not observed to form hard tooth elements. Overall, the morphologic evidence suggested that these tumors were most likely derived from cells that line the tooth root sulcus, or their intramucosal primordia. Because such cells are considered to be components of the dental lamina, these neoplasms were diagnosed as ameloblastomas.
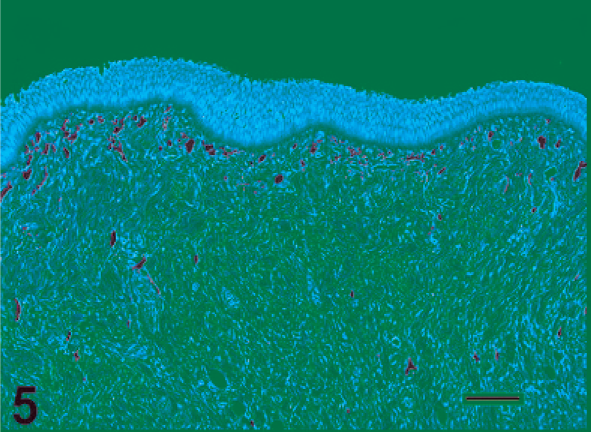
Normal oral mucosa from a Chinook salmon. HE. Bar = 100 µm.

Epithelial hyperplasia of the oral mucosa. The mucosal surface is markedly thickened and the stratum basale follows a tortuous path with projections into the submucosa. This type of focal proliferative change, which may be a precursor to tumor formation in some cases, was usually accompanied by only mild inflammation. HE. Bar = 100 µm.
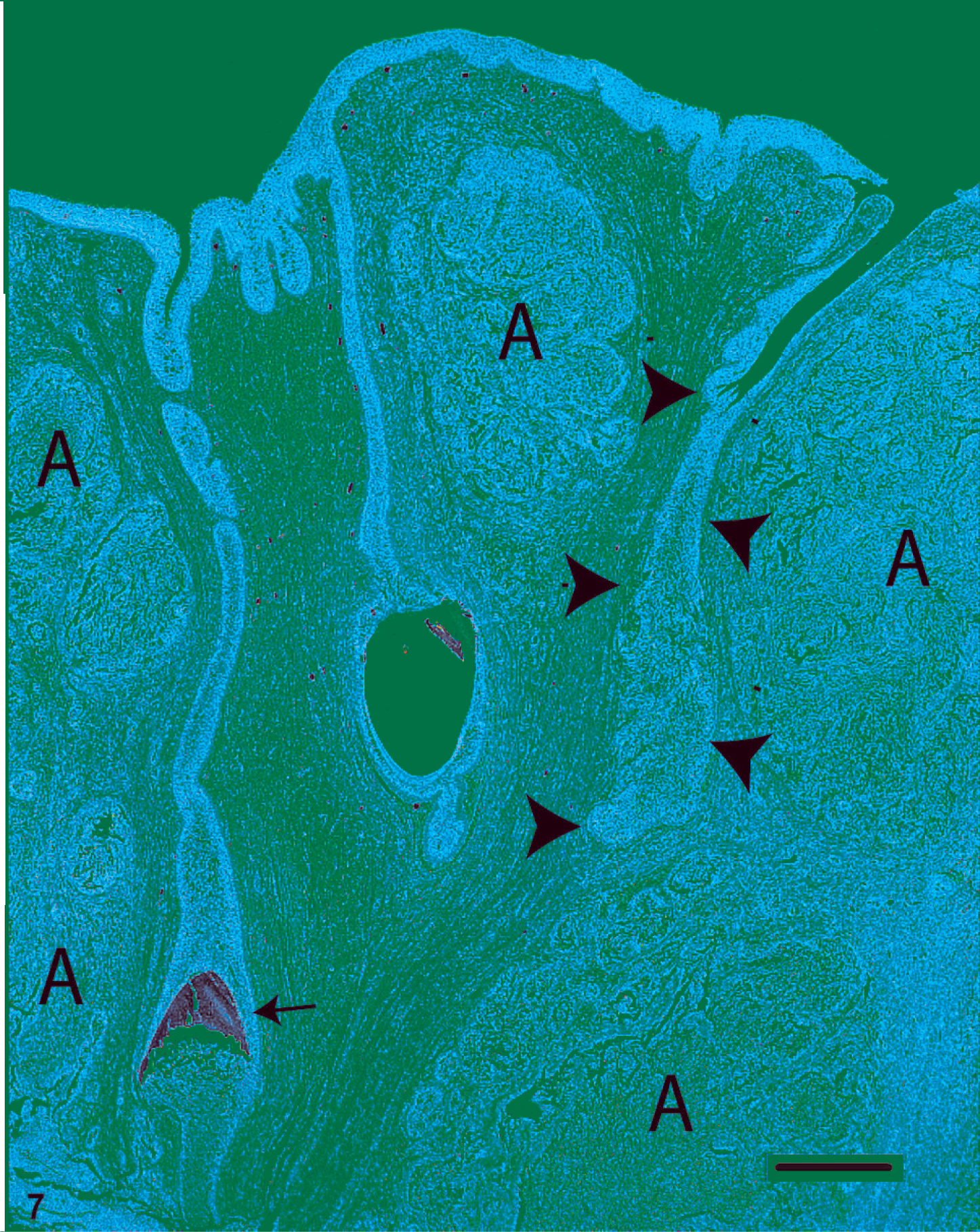
Ameloblastoma in the oral mucosa of a Chinook salmon. In this example, nodular islands of nonodontogenic ameloblastoma (A) are scattered throughout the propria mucosa. One deep invagination of the mucosal epithelium (arrowheads) merges with the tumor tissue. An unaffected portion of tooth (arrow) is surrounded by normal odontogenic epithelium. HE. Bar = 250 µm.

Ameloblastoma arising from dental lamina. Cords of tumor cells (arrows) branch from, and are contiguous with, ameloblasts of an otherwise normal tooth (T). Note the solid nest of tumor tissue (A) in the upper right corner of the photomicrograph. HE. Bar = 100 µm.
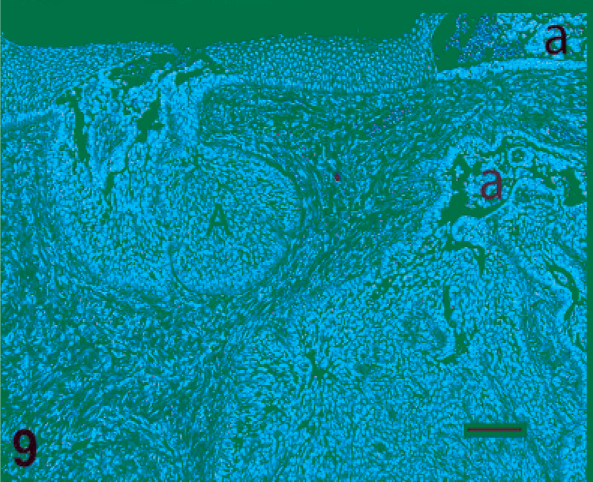
Ameloblastoma with acantholysis. This photomicrograph illustrates acantholysis (a) and cleft formation, which are characteristic features of the tumors in our series of Chinook salmons. Direct associations between the neoplastic tissue (A) and either the surface epithelium or the dental lamina are not always evident, presumably because such connections are out of the plane of tissue section. HE. Bar = 100 µm.
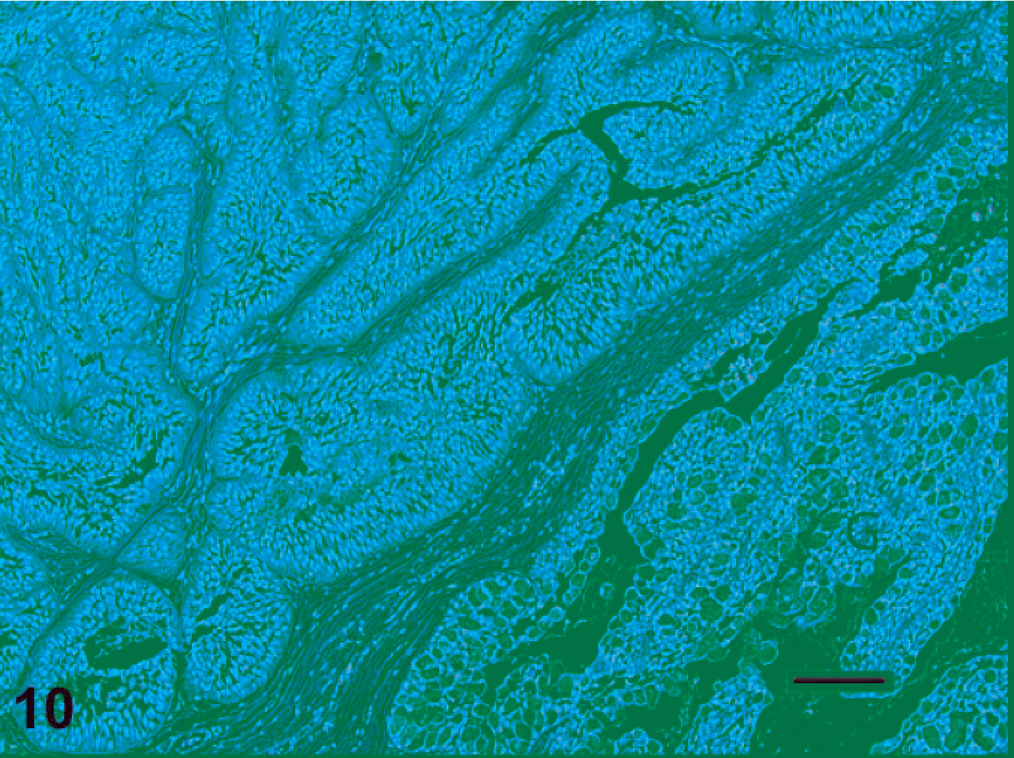
Ameloblastoma with mucus cell differentiation. There are numerous goblet cells (G), which suggests that this particular tumor may have originated from cells that were destined to reside in a superficial segment of the peridontal sulcus. Note the apical crowding of nuclei and cytoplasmic clearing of the basilar pole in these cells. HE. Bar = 100 µm.
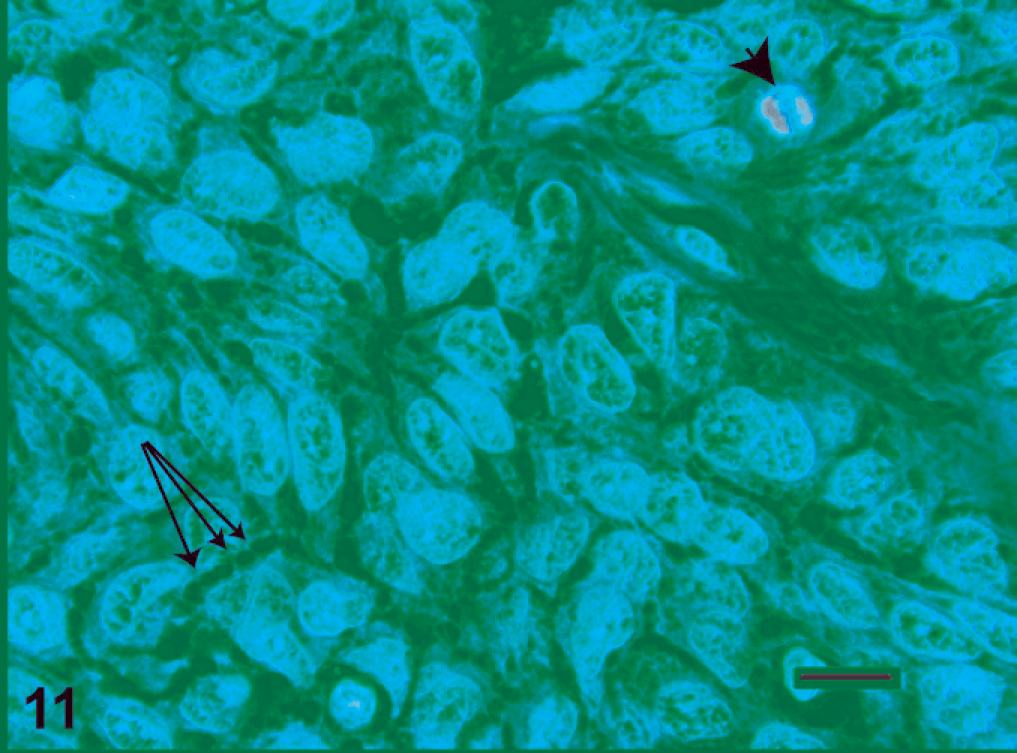
Ameloblastoma, high magnification. The columnar or polygonal tumor cells generally have ovoid to irregular nuclei, with distinct nucleoli. Intercellular bridges (arrows), consistent with desmosomal attachments, can be seen at this magnification. Mitotic figures (arrowhead) are occasionally evident. HE. Bar = 25 µm.
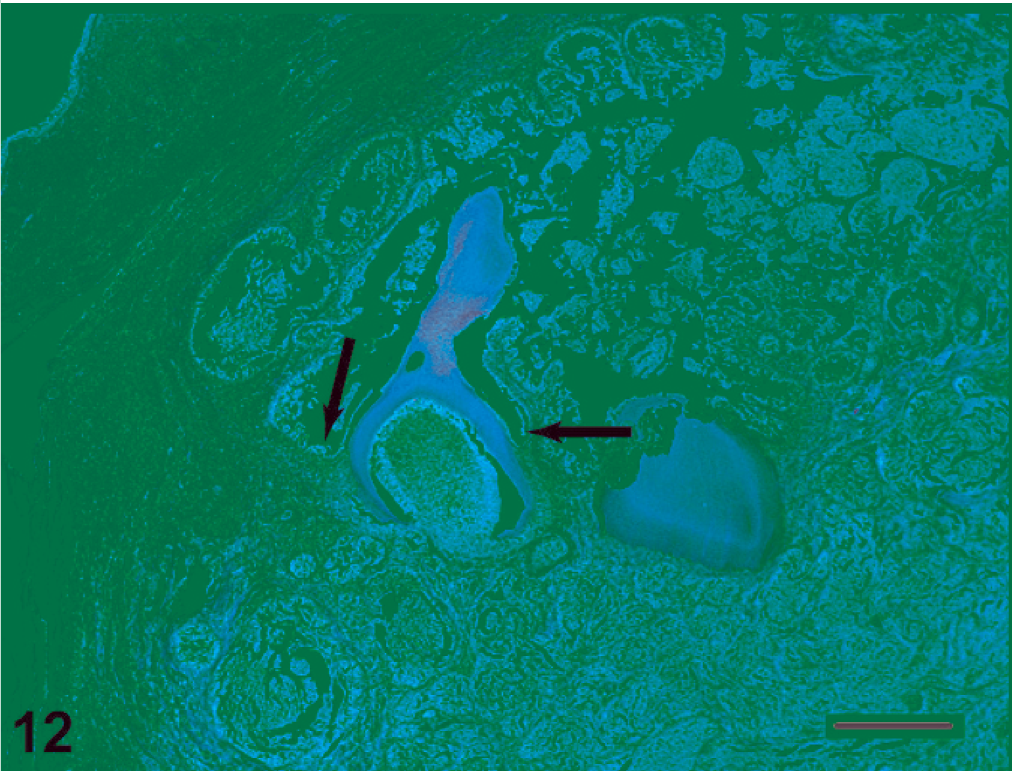
Ameloblastoma with incorporated tooth. A malformed tooth is surrounded by neoplastic tissue, which is continuous with ameloblasts of the dental lamina (arrows). Most of the tumors that we examined did not contain teeth, and of those that did, very few of the teeth were abnormal. HE. Bar = 500 µm.
Bacteriology
In adult tumor-bearing Chinook salmon returning to northeast Oregon streams, no substantial bacterial growth was detected from the kidney samples. Low-level growth of mixed bacteria developed from the 1 tumor that was directly sampled. ELISA values for BKD from the 4 sampled fish were 0.104 OD units or less, a value that is lower than the threshold indication for active or recent R. salmoninarum infection. Although there was no indication that these 4 fish had BKD, the disease was a significant problem in the tumor-laden Grande Ronde River 2000 cohort, as evidenced by gross signs of BKD (e.g., swollen, gray kidneys with small white foci) and ELISA values ≥0.800 OD units detected in 187/502 (37.2%) of the fish (see mortality table for 2004 annual report). 5
Virology
Tissue culture
Five different cell lines at 2 different incubation temperatures were used in an attempt to isolate the virus from homogenized tumor and organ tissues. EPC and CHSE-214 cells are known to support the growth of several aquatic viruses that affect salmonids and other teleosts. 1 Rainbow trout gonad cells are known to support the growth of OMV, a known oncogenic herpesvirus of salmonids, and H. salmonis. 40 Fathead minnow and Bluegill fin (BF-2) cells can support the growth of the iridovirus, which causes lymphocystis disease. 41 Twenty-eight–day incubation and subsequent 14-day subcultures of all tissue homogenates failed to produce notable cytopathic changes in any of the inoculated cell lines.
Primary cell culture
Primary cell monolayers from explanted tumor tissue were grown in culture and observed for atypical growth or cytopathic changes induced by endogenous virus. No evidence of viral pathogenicity was noted during a 3-week period, after which monolayer growth had subsided in both flasks and viability was no longer apparent. Attempts to subculture the primary cells into new flasks were unsuccessful. Cellular debris was harvested from the primary flasks and examined by electron microscopy and PCR.
Transmission electron microscopy—primary tumor tissue
Ultrastructurally, the 2 tumor tissue samples submitted to LAELOM contained a monomorphic population of cells that had lobed nuclei and scarce heterochromatin, interdigitating borders, perinuclear accumulations of mitochondria, prominent rough endoplasmic reticulum, and bundles of cytoplasmic filaments that resembled tonofilaments, which occasionally formed clumps. Desmosome-type structures were evident, consistent with the suspected epithelial cell origin of the tumor. Adjacent to the nucleus in the cytoplasm of a single tumor cell were a few hexagonal structures (∼100 nm) that may be of viral etiology (Fig. 13), although it is also possible that these structures are cytoplasmic vesicles of host origin. These particles were discovered serendipitously, as the primary goal of the TEM evaluation was to further characterize the tumor cells. Virus-like particles were not evident in any of the other evaluated tumor specimens.
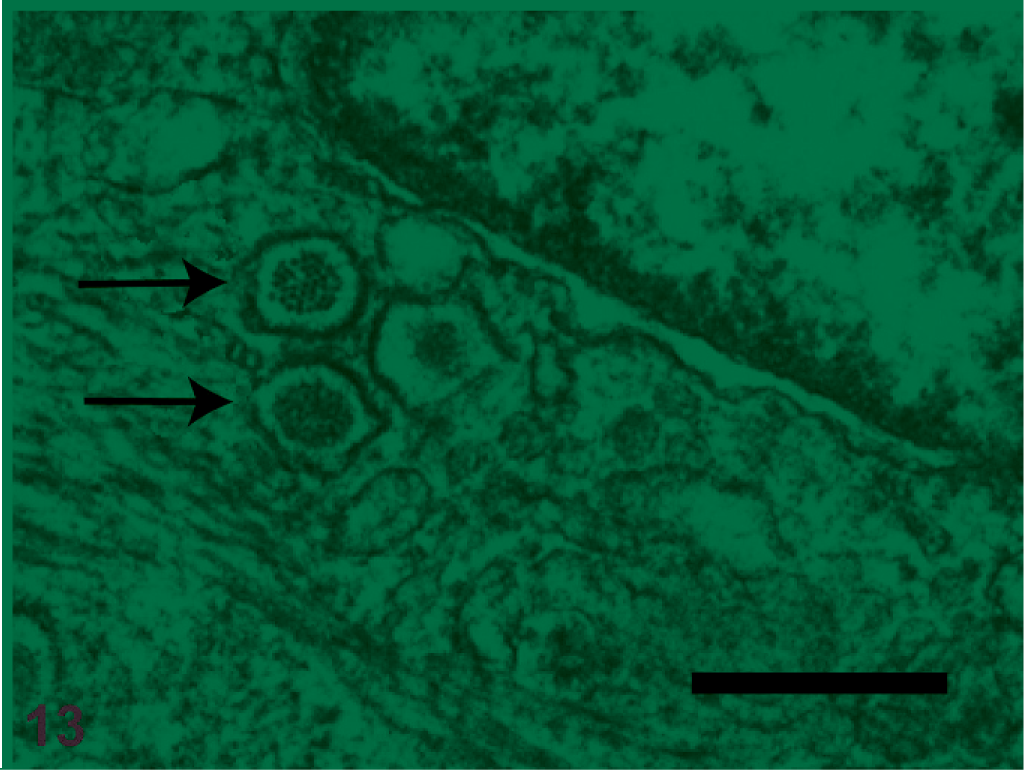
Suspected viruslike particles in an ameloblastoma cell. Several hexagonal, cytoplasmic particles (arrows) are present just adjacent to the cell nucleus. Electron micrograph, bar = 300 nm.
Transmission electron microscopy—tissue culture
Cell culture supernatant and pellets collected from the pooled tissue culture were examined by negative stain. No evidence of a viral agent was found in thin sections prepared and examined from the cellular debris of the primary culture flasks.
Polymerase chain reaction assay
DNA extracts from all sample sources were evaluated simultaneously along with positive and negative controls. The positive control consisted of H. salmonis DNA extracted from tissue culture supernatant, which produced an amplicon product of approximately 800 base pairs. OMV, if present, produces an amplicon of approximately 439 base pairs. None of the DNA extracts from any of the sample sources produced an amplicon of either molecular weight, indicating that H. salmonis or OMV was not present in tumor, kidney or splenic tissues from infected fish, tissue culture supernatant, or cultured primary cells at the level of detection for this PCR assay, which is approximately 6.3 TCID50/ml. 40
Exposure experiments
No mortality occurred in the 100 fingerling Chinook salmon exposed to effluent from an aquarium housing 3 tumor-laden adults. A total of 30 fingerlings were examined during the 6 months after the exposure period. No evidence of tumor growth or virus infection was found in any sampled fish. At termination, all remaining fingerlings were examined visually for tumors and subjected to viral tissue culture. No evidence of tumor formation or viral infection was observed in any fish.
During the 6 months after the injection of the tumor homogenates, a total of 7 mortalities occurred from both groups combined. Three dead fish from the whole cell-injected group and 4 from the filtered homogenate group were examined visually and by tissue culture methods using EPC and CHSE-214 cell lines. No evidence of a viral pathogen was found. At the end of the investigation, all remaining fish were sacrificed and examined. No tumor growth was noted on any examined fish, and there was no evidence of viral infection.
Genetic analysis
By the time genetic data were available, many individuals in the control, nonaffected group had developed oral tumors, thus compromising a straightforward comparison between groups. Nevertheless, when average heterozygosity among individual fish in the tumor-affected group was compared with values obtained from large, outbred, natural and hatchery populations (data not shown), there was no obvious depression of diversity that would be consistent with a limitation of the tumor phenotype to one or a few families. Admittedly, heterozygosity is relatively insensitive to population bottlenecks; nevertheless, expected heterozygosities were nearly identical between tumor and nontumor samples and were similar to values obtained in studies of these and geographically proximate Chinook salmon populations (Table 2 21 ). Very conservative inferences of minimum parents based on allele counts were also inconsistent with single-family origin. Twenty-four unique alleles were observed at the Ots104 locus alone, indicating that the absolute minimum number of parents contributing to the sample of tumor-affected individuals was 12 unrelated individuals (not sharing either parent).
Genetic diversity (H S) and genetically effective number of breeders (N b estimated from linkage disequilibrium) contributing to the sample of 192 tumoraffected fish compared with other samples of Snake River Chinook salmon.
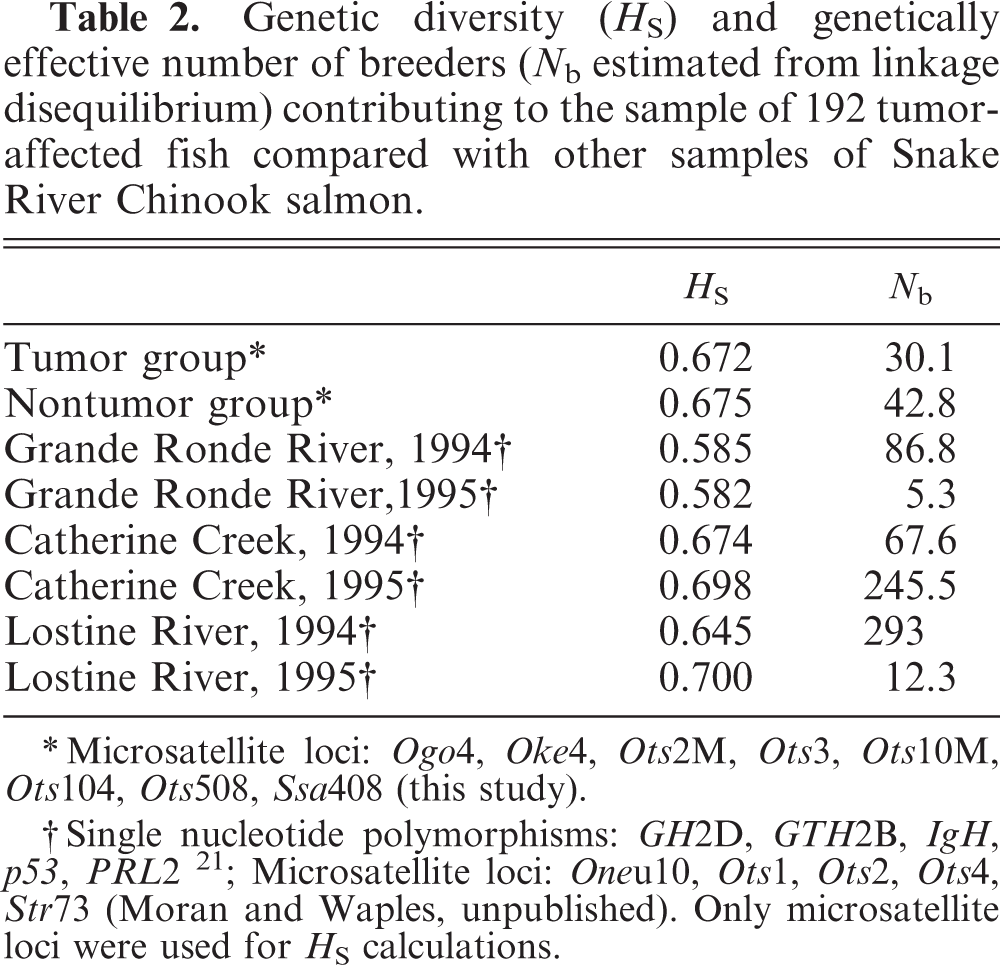
∗ Microsatellite loci: Ogo4, Oke4, Ots2M, Ots3, Ots10M, Ots104, Ots508, Str408 (this study).
† Single nucleotide polymorphisms: GH2D, GTH2B, IgH, p53, PRL2 21; Microsatellite loci: Oneu10, Ots1, Ots2, Ots4, Str73 (Moran and Waples, unpublished). Only microsatellite loci were used for H S calculations.
The distribution of the disease among families, and more recently among populations (including wild fish in other river systems), is not consistent with this condition being a transmissible genetic trait that is limited to a single or few families. The genetically effective number of breeders, N b, was estimated to be 30.1 (parametric 95% confidence interval [CI] 26.2–34.6). To minimize bias in estimating N b while retaining maximum information, alleles less frequent than 0.02 were eliminated from consideration. 37 Even a more conservative lower allele frequency cutoff of 0.05 produced a 95% CI of 12.2–18.9. These are small values but not exceptionally small for Snake River Chinook salmon populations, in general, and certainly not for the upper Grande Ronde River population, in particular. N b estimates from brood years 1993 and 1994 in the upper Grande Ronde River were 86.8 and 5.3, respectively (Moran and Waples, unpublished). The median estimate of N b for Lostine River, Catherine Creek, and upper Grande Ronde River in those years was 77, whereas the harmonic mean was only 20 (P. Moran and R. Waples, unpublished). A similar pattern obtained when results for tumor-affected fish were compared with N b estimates for populations in the Salmon River, another major tributary of the Snake River (M. Baird, unpublished). Again, the number of breeders that contributed to the sample of tumor-affected fish was small, but not smaller than estimates obtained in the mid-1990s or other Snake River spring Chinook salmon populations from the early 2000s. Comparison of N b is useful; however, the results suggest that the tumors were manifest in more than a single family.
Discussion
We tend to think of cancer as something that afflicts individuals rather than populations. This perception is understandable given the complex interplay of molecular and biochemical events considered requisite for the formation of a malignant neoplasm. 12 Opposing this perception are various examples of tumor epizootics, defined as more than three cases of neoplasia that originate from a specific cell lineage in a defined geographic area, 13 in mammals and other vertebrates. Common causality is a major reason why certain neoplasms are exceptionally prevalent in subpopulations that are defined by particular geographic, social, environmental, or genetic factors. In mammals, known causes of cancer include certain viral infections, hereditable predispositions, exposure to carcinogenic compounds or ionizing radiation, and idiosyncratic reactions to chronic irritation or trauma. A similar list of causes can be applied to fish, and classic examples include virus-induced dermal sarcomas in walleye 19 and neurofibromas in bicolor damselfish, 31 hereditable melanomas in Xiphophorus hybrids, 9 and chemical carcinogenesis in Puget Sound flatfishes 33 and Elizabeth river mummichog. 4,35
It is certain that the outbreak described herein more than meets the criteria to be considered an epizootic. Individual cases of ameloblastomas have been reported previously for several fish species, including Chinook salmon. 14,30 This single previous salmon report was published more than 30 years ago, and the original histologic sections that were the basis for that report (RTLA case Nos. 248 and 259) were reviewed for the present study (Table 1). Notably, the appearance of those early ameloblastomas was found to be similar to that of the more recent cases. Although the initial salmon neoplasms were actually designated as “fibroameloblastomas,” a recent review of the original specimens by these investigators revealed that the fibrous tissue component was not especially prominent in any of these lesions, and that the component did not appear to be neoplastic per se; therefore, the “fibro” prefix was considered superfluous. Differential diagnoses that were contemplated included squamous cell carcinoma and basal cell carcinoma; however, certain morphologic features of the salmon oral tumors indicated that the diagnosis of ameloblastoma was more appropriate (see additional discussion to follow).
To facilitate further discussion, a brief review of normal odontogenesis is presented here. Tooth primordia in the jaws of teleosts are located within the mucosal epithelium as linear arrays that run parallel to the oral margin. During early tooth development, these primordial cells form discrete condensations known individually as dental placodes, and collectively as the discontinuous dental lamina. Each tooth-forming unit (the dental organ) initially develops as a focal invagination of a placode into subjacent mesenchymal tissue. As tooth development progresses through the classic bud, cap, and bell stages, a sheet of odontogenic epithelial cells folds around the conical dental papilla. This folding creates 2 closely apposed cell layers: the inner dental epithelium (IDE) and outer dental epithelium (ODE). Destined to become the enameloid-generating ameloblasts, the IDE remains intimately apposed to the ODE until the onset of tooth rupture. The process of tooth rupture involves a separation of the ODE from the IDE, and detachment of cellular interdigitations among ODE cells that are superficial to the apex of the tooth. 17 It is likely that this loss of cohesion among ODE cells would appear as acantholysis in histologic sections.
To provide a starting point for elucidating the pathogenesis of Chinook salmon ameloblastomas, we propose a working hypothesis of tumor development. Because it is based almost entirely on morphologic observations, the model requires confirmation by further experimentation before it can be considered robust. Taken as a whole, our findings suggest that the salmon tumors arose from the epithelium of the dental lamina, and that in most cases the cell of origin was probably a precursor of the ODE. This theory is supported by the following observations: 1) macroscopically, the shape and orientation of many hyperplastic oral lesions and early tumors are consistent with the location of the dental lamina; 2) microscopically, the tumor cells closely resemble those of the normal odontogenic or mucosal epithelium; 3) ultrastructurally, the neoplastic cells have epithelial cell hallmarks such as desmosomal attachments and tonofilaments, as would be expected in cells of ameloblastic or squamous epithelial origin, for example; 4) the tumors are spatially associated with, and often contiguous with, either the mucosal or odontogenic epithelium; 5) consistent features of these neoplasms include acantholysis and a tendency to form crevices or clefts, many of which are extensions of oral mucosal crypts; 6) in the vast majority of these tumors, there was little to no evidence of hard tissue odontogenesis, such as the development of dental or cemental matrices or enameloid.
Although the gross and microscopic appearance of these neoplasms was generally quite similar from case to case, there were minor variations in epithelial cell morphology within and among the different cases (e.g., from columnar cells to oval basaloid cells to polyhedral cells to goblet cells). It is possible that this morphologic variability is related to the precise microanatomic destiny of the progenitor cell(s) that were responsible for each tumor. For example, tumors derived from cells destined to reside within the IDE would likely have a columnar appearance, whereas tumors of cells slated to become ODE might have a more basaloid appearance. Alternatively, if some of its cells represent the superficial ODE, the resulting tumor might have areas consisting of polygonal or even goblet cells. This scenario could be compared with an analogous situation involving the adnexal differentiation of hair follicle neoplasms in dogs, in which the cellular origins of 3 types of tumors (trichoepithelioma, tricholemmoma, and trichoblastoma) are thought to correspond to 3 different hair follicle layers. 10 The histologic appearance of ameloblastomas in mammals tends to be less variable than what we have observed in the Chinook salmon, possibly because mammalian tumors tend to arise within the IDE, which has a more uniform cellular constituency than the ODE.
The majority of the current and previous Chinook salmon ameloblastomas were located on or near tooth-bearing surfaces. 30 An interesting finding was the rare occurrence of these neoplasms in nondental sites, such as the tongue and gill arch, or even extraoral locations, such as the pectoral fin (Table 1). Although it might seem incongruous for a tumor of dental origin to be found outside the mouth, this phenomenon is not unprecedented, as extraoral ameloblastomas (and ectopic teeth) have been reported to occur in humans and other vertebrates. 29 As previously postulated, it is likely that at least some extraoral tumors represent neoplastic proliferations of embryonic “rests” rather than metastases from primary tumor sites. 29
The inciting cause(s) of these salmon ameloblastomas have not been determined and it is likely multifactorial. Given that the epizootic in hatchery-reared captive broodstock fish was confined to a certain age class of salmon, which were acquired as offspring from a particular seasonal run and a single watershed, in combination with the fact that the salmon were housed together in close proximity after collection, the most likely potential etiologies were initially thought to include genetic predisposition, toxic/carcinogenic insult, and viral induction. A less commonly considered cause could be direct animal-to-animal spread of the neoplasms, as observed in the transmissible venereal tumors of dogs 22 (Sticker's sarcomas) and possibly the facial tumors of Tasmanian devils. 15
Because retention of genetic diversity is an explicit goal of captive broodstock propagation, it is important to understand genetic relationships among diseased individuals. Using neutral microsatellite markers we sought to determine whether the tumors were limited to a single family of fish, perhaps reflecting a strong genetic basis and reason for caution in captive broodstock propagation. The genetic analysis ruled out a single-family origin for predisposition to developing ameloblastomas. There were relatively few spawning adults in the Grande Ronde River in 2000 that gave rise to the 2001 parr collection. The small number of parents combined with initial restriction of the tumors to the Grande Ronde River system raised the possibility that a spontaneous mutation in the germ line of a single individual might have given rise to the tumor-affected individuals. The results of the analyses strongly refute the single-family hypothesis indicating that more than a dozen genetically diverse parents contributed to the sample of tumor-affected individuals. The actual number of genetically diverse parents was likely more than four dozen given the conservative assumptions of our model, which eliminates rare alleles. In addition, census size in breeding populations of Pacific salmon is generally 2.5–5 times larger than the genetically effective number of breeders. 36 The genetic study, however, has not entirely ruled out a potential genetic basis for susceptibility to the disease. Genetic drift associated with small population size in the upper Grande Ronde River may have resulted in a loss of genetic resistance and a high prevalence of disease in this population. Although heterozygosity was not depressed in the affected fish, earlier Grande Ronde River samples show low heterozygosity, and it is worth noting that the heterozygosity of the Chinook salmon population in the upper Grande Ronde River has been the most depressed of the 3 northeast Oregon captive broodstocks. 16 This study was not designed a priori to evaluate the genetics of disease resistance within or among populations, nor was it designed to rule out potential genetic mutation. The loss of genetic diversity or disease resistance as a result of genetic drift would almost certainly be a stochastic event, and thus we cannot meaningfully evaluate this hypothesis by using the current neutral microsatellite data. Rather, we can only say that extreme drift is no more likely in this population than in similar populations in the same region. Accurate description of the genetic basis for susceptibility to developing ameloblastomas would require controlled breeding experiments combined with disease challenge. However, uncertainty in terms of causality significantly complicates such a design. It will require substantial additional work to fully describe the selection coefficients and heritabilities associated with the development of ameloblastomas.
Exposure to environmental carcinogens has not been completely dismissed as an etiology for these neoplasms, given that toxicologic analyses were not performed. It has been suggested that there is an increasing number of tumor epizootics in fish of the United States, 13 and that the majority of neoplasms associated with environmental chemical contamination are of epithelial origin, with liver and skin as the primary affected organs. 4 Examples of toxin-induced tumors in fish include a strong relationship between polyaromatic hydrocarbons and skin tumors in brown bullheads (Ameiurus nebulosus), and chlorinated wastewater effluent associated skin tumors in black bullheads (A. melas), white suckers (Catostomus commersoni), and white croakers (Genyonemus lineatus). 4 Concerning the captive subpopulation, at least two of the original brood collection sites, Lostine River and Catherine Creek, are considered to be more pristine than the Grande Ronde River, and captive cohorts from those areas did not develop ameloblastomas. Aside from the occurrence of tumors in the Captive Broodstock Program, ameloblastomas have been documented in returning adult hatchery fish and a few fish of natural origin. The affected returning adults all migrated through mainstem Columbia River and Snake River corridors as juvenile fish, and again as returning adults, where some sections may harbor high levels of contaminants. One such example is the Hanford Nuclear Power Plant, a superfund site on the Columbia River. Although the captive Grande Ronde River cohort did not travel through these sites, they are offspring of parents that migrated through the corridor. Effects from exposure to some carcinogens can be transferred to, and manifest in, subsequent generations through epigenetic mechanisms (e.g., DNA methylation), as has been demonstrated with endocrine active substances such as diethylstilbestrol. 24 Given that subpopulations of Chinook salmon were differentially susceptible to developing ameloblastomas, it is reasonable to assume that common environmental stressors, such as contaminants, could contribute directly to the formation of this tumor, or possibly to diminished immunocompetence of affected salmon.
Whereas certain characteristics of these tumors do not suggest an infectious etiology (e.g., bilateral symmetry), one plausible theory is that the ameloblastomas were induced by an infectious agent such as a virus. If this were the case, it is reasonable to surmise that the virus might be endemic in regional Chinook salmon, and that low natural tumor prevalence would result in the type of sporadic disease that has been observed in wild fish to date. Alternatively, the collection and artificial confinement of parr in a group that included one or more virus-infected fish could lead to concentration of the agent. Reduced immunocompetence due to shipment stress combined with higher exposure rates to infectious agents could result in a high prevalence of tumors in the captive subpopulation. As previously mentioned, several viruses have been implicated in epizootic tumor induction in fish, and oral epithelial neoplasms in coho salmon have been associated with viral infection. 2 In humans, viral elements have been identified in ameloblastomas. 6 and evidence has been presented that links human ameloblastomas to papillomaviruses 28 and Epstein-Barr virus. 18 In the current situation, we present subcellular structures that are possible viral particles; expert opinions that we have solicited vary widely. If these are viral particles, presence in ameloblastic cells certainly does not indicate causality. On the other hand, it is somewhat remarkable that any viruslike particles were observed, as only 6 well-preserved specimens were submitted for TEM, and only a small number of cells were examined in total. Other results neither support nor necessarily refute a viral etiology. There were no viruses isolated from this tumor using standard cell culture methods, and PCR screens for certain known fish pathogens were negative. In addition, injection of tumor tissue homogenates into naïve fish, and communal housing of affected and unaffected fish did not result in tumor formation. One potential explanation for the lack of viral recovery in adult fish, and the lack of disease transmission in the exposure experiments, is that viral-induced tumor initiation occurred in young fish, and progression of the lesions continued long after viral replication had ceased. It is also possible that a causative virus might replicate intermittently in a seasonal pattern, as is the case with walleye dermal sarcoma virus. 27
To date, 3 natural origin (wild caught in freshwater as adults [WCF]) fish had oral lesions that were diagnosed as ameloblastomas by RTLA pathologists. The extent to which these large, disfiguring oral tumors might adversely affect individual salmon, in terms of general health and reproduction, is unknown. Potentially, an increase in the incidence of ameloblastomas could pose a survival threat to both wild and hatchery-raised Chinook salmon populations, and it would be easy to foresee negative impacts on both the economic value of these fish and the economic health of the fishery. In light of the fragile status of certain wild Chinook salmon populations, further investigation into potential etiologies, including infectious and toxic causes, is warranted.
Footnotes
Acknowledgements
This project was performed, in part, by using the services provided by the National Cancer Institute's Registry of Tumors in Lower Animals, operated under contract by Experimental Pathology Laboratories, Inc., N02-CB-27034. We also thank the contributions and efforts of Phyllis Spero (RTLA) and Debbie Haegele (RTLA) for organizing slide materials. We thank Dr. Richard R. Dubielzig, DVM, for aiding in the diagnosis of these tumors, Dr. Jerry R. Heidel, DVM, for histopathologic work on tumor tissues, Dr. Lee Harrell, DVM, for tissue collection and input on managing this problem in the Captive Broodstock Program. We also thank Dr. James Casey for conducting viral-probe testing, Marla Chaney for assistance with tumor sampling and taking care of these fish, Sharon Vendshus for performing numerous fish necropsies, and Glenda O'Connor for sampling and necropsying the fish from Northeast Oregon. This work was supported in part by Bonneville Power Administration project 198909600.
1 The views presented are the professional opinions of the authors and do not necessarily reflect the official positions of the government agencies involved.
