Abstract
The long-term administration of low doses of rotenone has been used to produce a model of Parkinson disease (PD) in rats. However, only about 50% of similarly treated rats develop the PD-like syndrome, with many dying during the first few days of treatment. The lesions in male Lewis rats that became moribund or died after short-term, low-dose rotenone administration are described. Dosed rats had fibrinoid change and acute hemorrhage involving small arteries and arterioles of the brain and lungs. The thalamus, hypothalamus, and medulla oblongata were most frequently and severely affected. Blood vessels in the brain of some male Lewis rats appeared acutely susceptible to the effects of rotenone. Understanding the selective nature of the fibrinoid change and hemorrhage might explain how rotenone produces PD-like signs and lesions in rats, and it might also provide the basis for a model of intraparenchymal hemorrhagic cerebrovascular disease (i.e., hemorrhagic strokes) in humans.
Rotenone has been used as a broad-spectrum insecticide and fish poison for many years. 13 It has 2 known modes of action in cells: the inhibition of complex I in the mitochondrial respiratory chain 3,12 and the depolymerization of microtubules. 6,19,26 A major function of mitochondria is the production of cellular energy, and one of the many functions of microtubules is the translocation of vesicles containing secretory products, including neurotransmitters, within neurons.
Parkinson disease (PD) is characterized by locomotor problems and selective degeneration of dopaminergic neurons in the substantia nigra of the brain in humans. 4,10,11,15,21 Long-term administration of low doses of rotenone has been used recently to create an experimental animal model of PD in rats that includes similar movement disorders and selective degeneration of dopaminergic neurons in the brain.∗ 1,4,6,7,8,14,15,16,17,21,25,29,30,33
Regrettably, in many of these experiments, acute death or illness necessitating premature euthanasia occurs in a substantial proportion of dosed rats. Additionally, variable results, including the failure to produce the lesions of interest, are often found in similarly treated rats that survive the acute phase of administration.† 1,2,3,4,5,8,14,15,16,17,20,21,25,30,33 Investigation into the nature of this unwanted morbidity and mortality is in the best interests of both researchers and laboratory animals raised for such experimentation. The purpose of the present study is to describe the lesions in male Lewis rats that became acutely moribund or died early after the administration of low doses of rotenone.
All procedures involving the use of animals received approval from the University Committee on Animal Care and Supply at the University of Saskatchewan. All animals were housed and cared for in an experimental animal facility at the University of Saskatchewan.
The rats described here were part of a larger experiment involving 54 male Lewis rats weighing approximately 400 g that were obtained from Charles River Canada (Saint Constant, QC, Canada). The design of the larger experiment was similar to those described by Greenamyre and colleagues, 2,5,23,30 Rajput and colleagues, 21,24 and others. 6,25,33 Briefly, osmotic minipumps (model 2ML4; Alzet Corp., Palo Alto, CA) were filled with either rotenone mixed in a delivery vehicle composed of equal volumes of dimethyl sulfoxide and propylene glycol or the delivery vehicle alone (all products from Sigma Chemical, St. Louis, MO). All filled pumps were placed in a sterile saline bath and warmed to 37°C prior to being surgically implanted subcutaneously over the back. The rats were anesthetized with inhaled halothane mixed in oxygen for the procedure. A total of 41 rats received minipumps containing rotenone mixed in the delivery vehicle, and 13 control rats received minipumps containing only the delivery vehicle. Dosed rats received 3.0 mg/kg rotenone per day calculated using a rat's weight at the time of implant surgery.
Animals were examined and weighed daily. Any animal that lost more than 5% of its initial weight was given 5–8 ml of a nutritional supplement (Ensure; Ross Laboratories, Columbus, OH) by gavage and 10 ml of lactated Ringer solution subcutaneously, twice per day. Any animal that was moribund to the point of being unable to ambulate was euthanatized by an intraperitoneal injection of sodium thiopental (Sodium Pentothal; Abbott Laboratories, Montreal, QC, Canada). Control rats were euthanatized by the same method.
Any dosed rats that died spontaneously or were euthanatized within 19 days of receiving rotenone, as well as 2 control rats that received the delivery vehicle only and remained healthy, were included in this study and examined postmortem. Tissues examined microscopically included brain, heart, lungs, liver, kidney, urinary bladder, stomach, small intestine, large intestine, pancreas, thymus, spleen, lymph nodes, adrenal glands, thyroid gland, salivary glands, and skeletal muscle. Tissues were prepared for microscopic evaluation using standard methods. Briefly, tissues were fixed in 10% neutral-buffered formalin, dehydrated, impregnated with and embedded in paraffin, sectioned at 4 μm, mounted on glass slides, and stained with hematoxylin and eosin. Selected sections of the brains also were stained with the Prussian blue (Perl) reaction to demonstrate iron salts, a silver (Holmes) stain to demonstrate axons, or both. Selected sections of pancreas also were stained with phosphotungstic acid hematoxylin (PTAH) to demonstrate fibrin.
This study involved 12 rats, including 10 dosed rats that died spontaneously or became moribund within 19 days of receiving rotenone (Table 1) and 2 control rats that were euthanatized after receiving the delivery vehicle only for 12 and 35 days, respectively. All dosed rats included in this study lost weight and received nutritional support as described above. In spite of the nutritional support, 2 dosed rats died spontaneously, and 8 rats were euthanatized because they continued to lose weight, were unable to ambulate, or both. In contrast, the 2 control rats that received only the delivery vehicle gained weight.
A summary of the lesions present in 10 male Lewis rats dosed with approximately 3.0 mg/kg rotenone per day and dying or euthanatized after becoming moribund within 19 days.
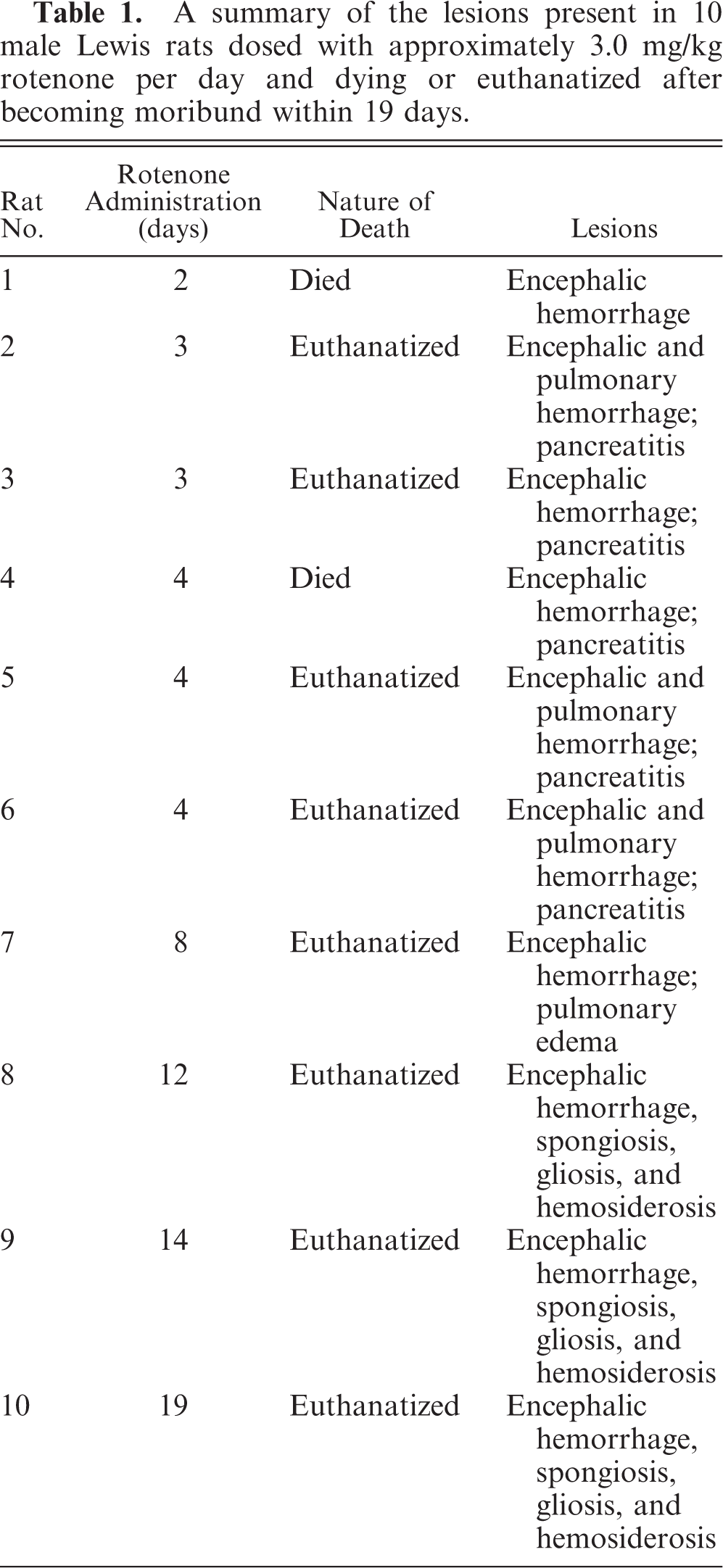
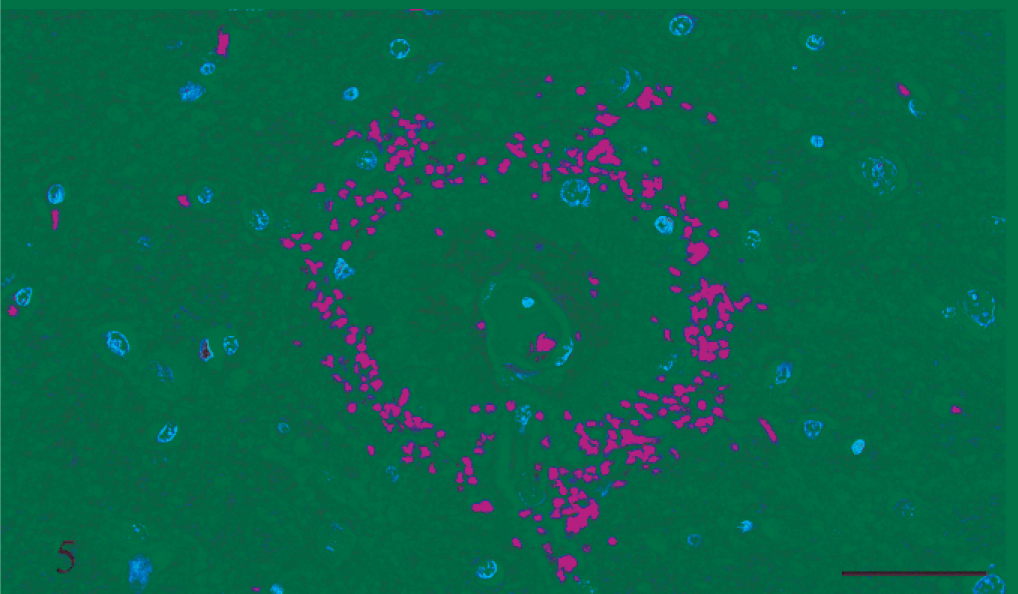
Gross and microscopic examinations did not reveal any lesions in the 2 control rats. Seven rats that received rotenone for between 2 and 8 days had many punctate foci of acute hemorrhage scattered throughout the brain and meninges. Hemorrhages occurred in all regions of the brain, but the thalamus, hypothalamus, and medulla oblongata were the most severely affected (Figs. 1, 2). Most of the hemorrhage appeared to be centered on or around small blood vessels interpreted to be small arteries and arterioles. The walls of several vessels had an amorphous, eosinophilic, hyaline appearance consistent with fibrinoid change 22,28 (Figs. 3, 4), and some of these vessels were surrounded by eosinophilic material (Fig. 5). These changes were interpreted as leakage of plasma proteins into the vascular walls and perivascular spaces. There were also instances of hemorrhage without vascular fibrinoid change (Fig. 6) and, conversely, vascular fibrinoid change with perivascular accumulation of proteinaceous fluid but no hemorrhage (Fig. 7). Around a few small blood vessels, there was perivascular accumulation of proteinaceous fluid (free of erythrocytes) between the vascular wall and a more peripheral ring of hemorrhage (Fig. 8A). Review of the Holmes-stained sections of these areas of brain revealed the presence of axons in the zone of proteinaceous fluid accumulation, and the lesion was interpreted to represent plasma within the neuropil between the affected vessel and a widening cone-shaped area of hemorrhage (Fig. 8B). The vascular lesions were occasionally associated with areas of spongiosis that were interpreted as being due to edema (Fig. 9).
Brain; rat No. 7. Serial transverse sections of the formalin-fixed brain of a male Lewis rat (No. 7) dosed with rotenone for 8 days prior to being euthanatized. There are many dark, punctate foci that represent acute hemorrhage in all regions of the brain, with the thalamus, hypothalamus, and medulla oblongata most severely affected.

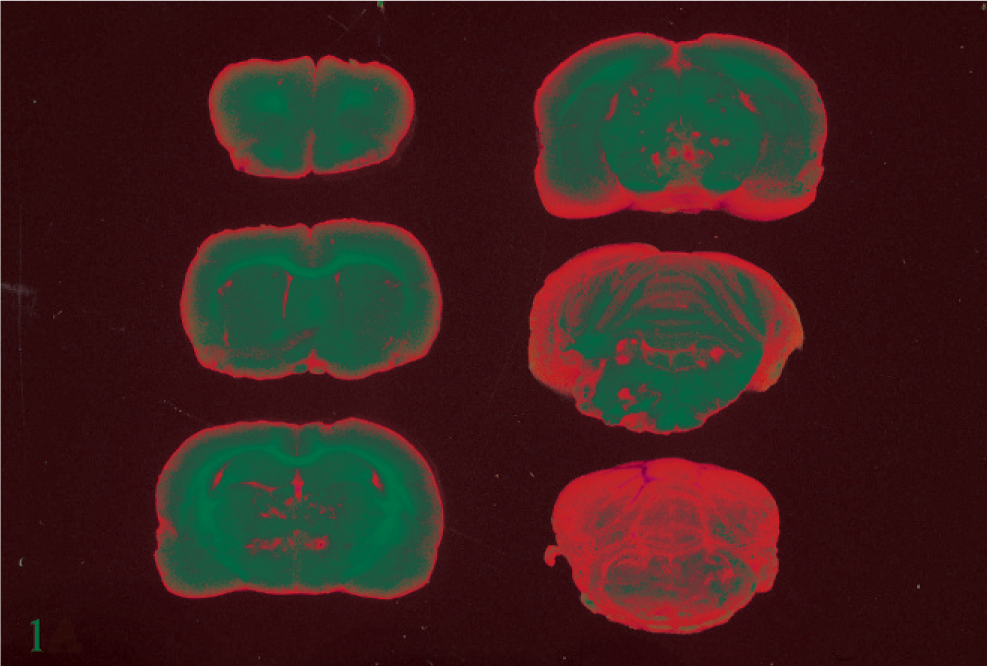
Thalamus; rat No. 3. Thalamus of a male Lewis rat (No. 3) dosed with rotenone for 3 days prior to being euthanatized. There are many foci of acute hemorrhage surrounding blood vessels. HE. Bar = 1 mm.
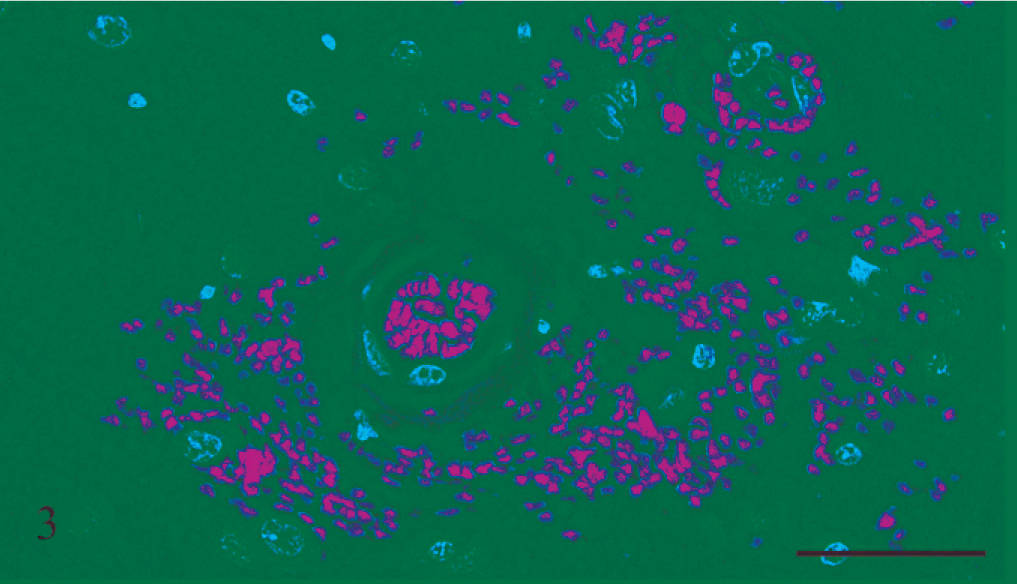
Thalamus; rat No. 2. Thalamus of a male Lewis rat (No. 2) dosed with rotenone for 3 days prior to being euthanatized. There is fibrinoid change of the blood vessel wall and associated pervascular hemorrhage. HE. Bar = 50 μm.
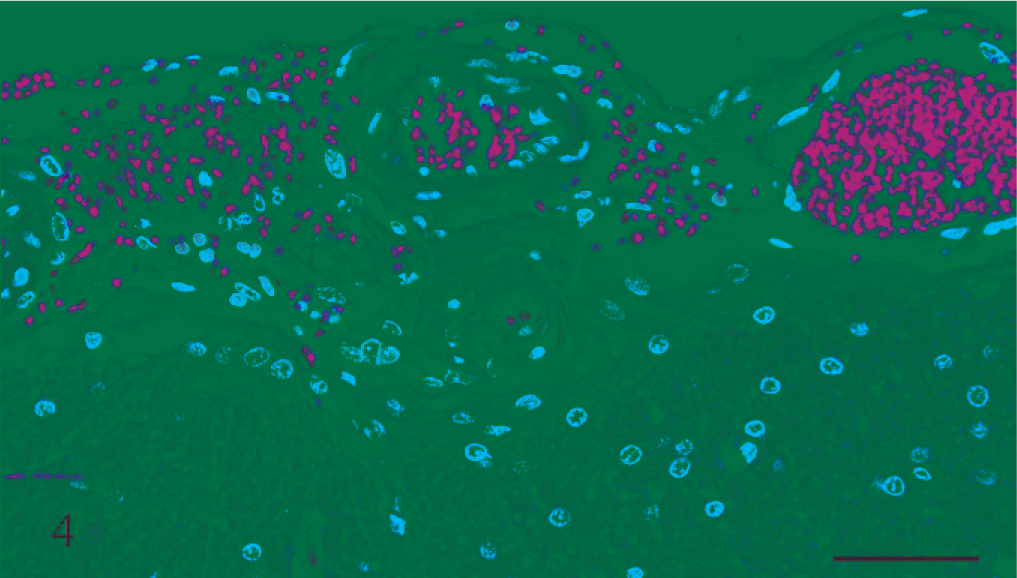
Caudatoputamen; rat No. 3. Leptomeninges at the level of the caudatoputamen of a male Lewis rat (No. 3) dosed with rotenone for 3 days prior to being euthanatized. There is hemorrhage and extravasation of proteinaceous fluid into the meningeal space and fibrinoid change of the smaller blood vessel. HE. Bar = 50 μm.
Thalamus; rat No. 2. Thalamus of a male Lewis rat (No. 2) dosed with rotenone for 3 days prior to being euthanatized. There is a zone of amorphous eosinophilic material between a small blood vessel and a circular area of hemorrhage. HE. Bar = 50 μm.
Medulla; rat No. 2. Medulla of a male Lewis rat (No. 2) dosed with rotenone for 3 days prior to being euthanatized. There is hemorrhage around a blood vessel without vascular fibrinoid change. HE. Bar = 100 μm.
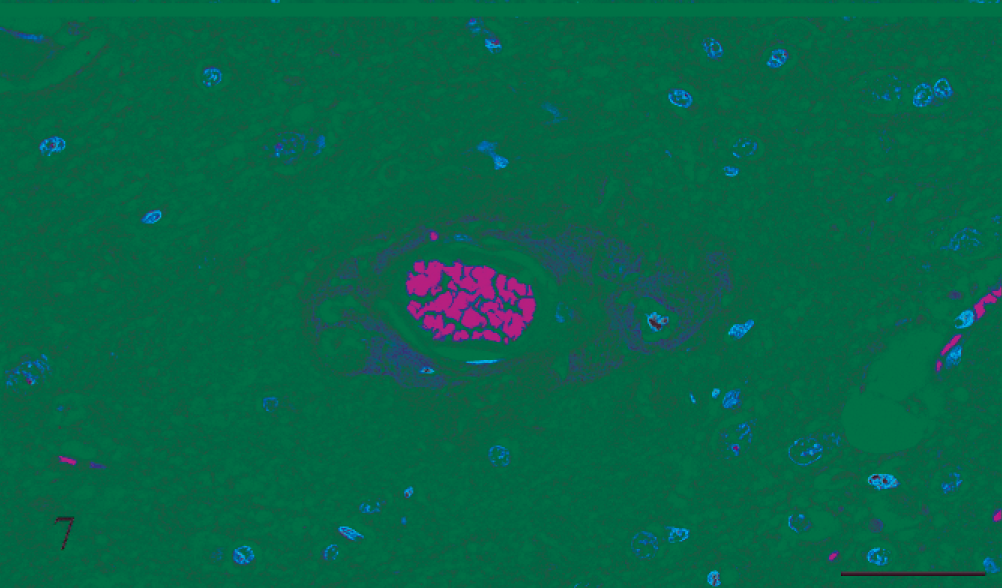
Thalamus; rat No. 2. Thalamus of a male Lewis rat (No. 2) dosed with rotenone for 3 days prior to being euthanatized. There is a focus of fibrinoid change in the blood vessel wall, and the vessel is surrounded by proteinaceous material, but there is no hemorrhage. HE. Bar = 50 μm.
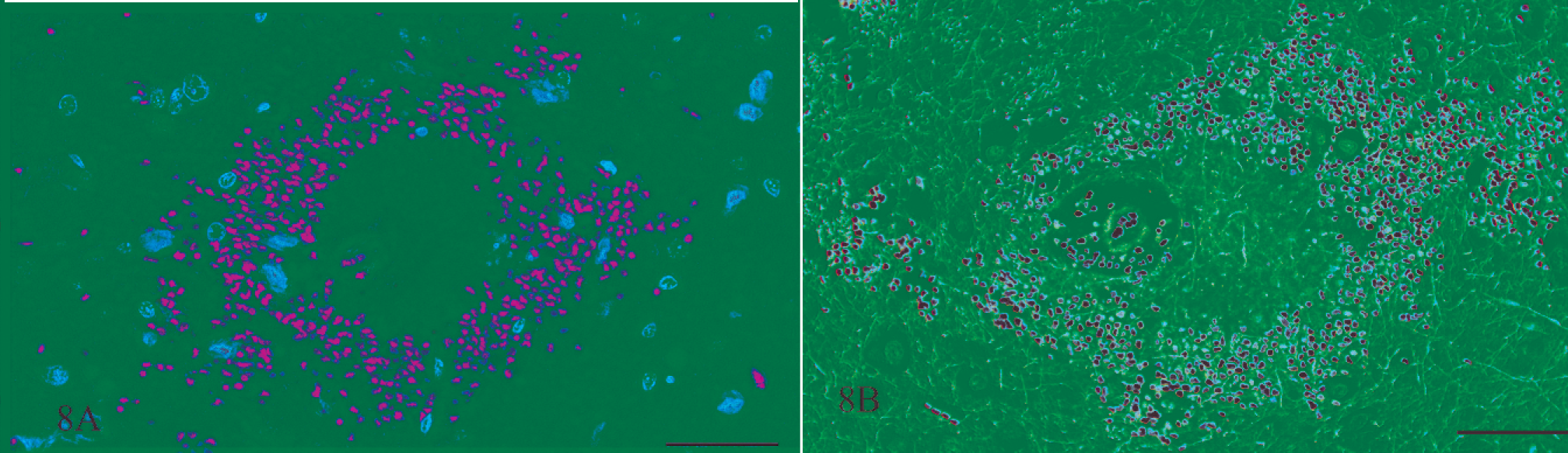
Thalamus; rat No. 2. Serial sections of the thalamus of a male Lewis rat (No. 2) dosed with rotenone for 3 days prior to being euthanatized. Axons are present in the zone of amorphous material between a small blood vessel and a circular area of hemorrhage. Fig. 8A. HE. Fig. 8B. Holmes silver stain. Bar = 50 μm.

Thalamus; rat No. 2. Thalamus of a male Lewis rat (No. 2) dosed with rotenone for 3 days prior to being euthanatized. There is an area of spongiosis, interpreted as edema, associated with perivascular hemorrhage. HE. Bar = 100 μm.
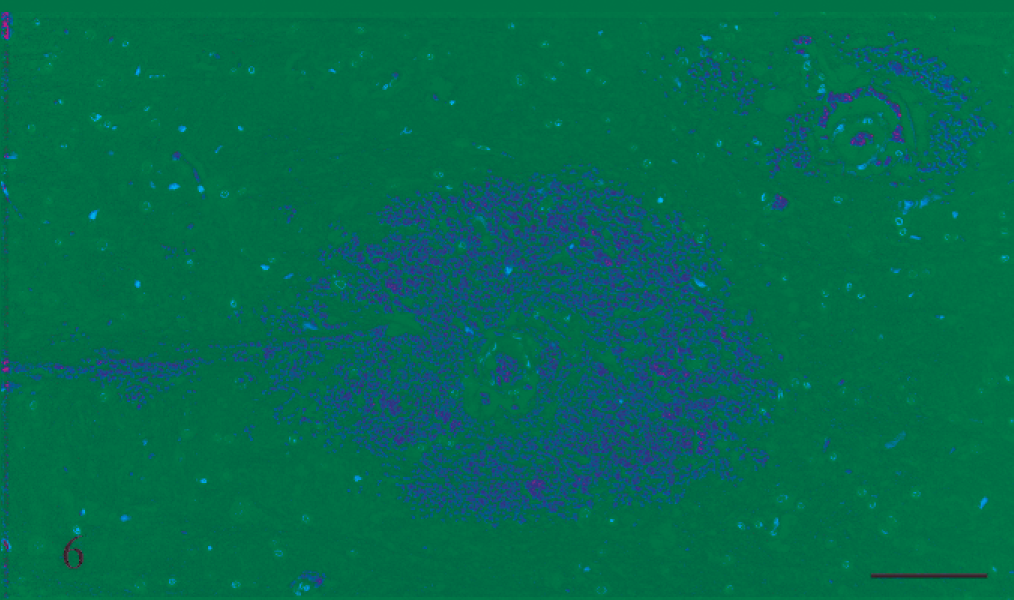
Three rats that received rotenone for between 12 and 19 days also had similar foci of acute hemorrhage, as well as evidence of more chronic and resolving hemorrhage in the form of hemosiderin-laden macrophages, edema indicated by spongiosis, and putative neuronal injury and loss recognized as astrocytosis and astrogliosis (Figs. 10–12).
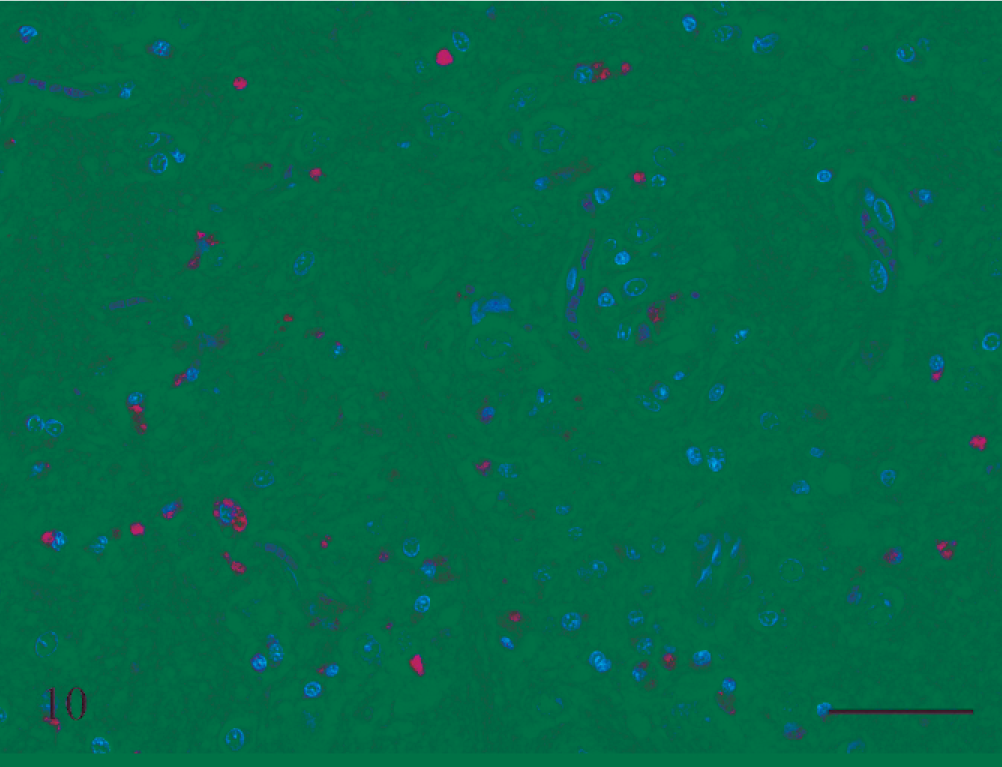
Medulla; rat No. 8. Medulla of a male Lewis rat (No. 8) dosed with rotenone for 12 days prior to being euthanatized. There are areas of astrocytosis and astrogliosis, consistent with neuronal injury and loss. There are also several cells, interpreted to be macrophages, that contain golden-brown pigment. Bar = 50 μm.

Medulla; rat No. 8. Medulla of a male Lewis rat (No. 8) dosed with rotenone for 12 days prior to being euthanatized. There are many macrophages that contain blue material, consistent with being hemosiderin using the Perl Prussian blue reaction, as well as a blood vessel with acute perivascular hemorrhage, interpreted as evidence of both chronic and ongoing hemorrhage, respectively. HE and Perl Prussian blue reaction. Bar = 100 μm.
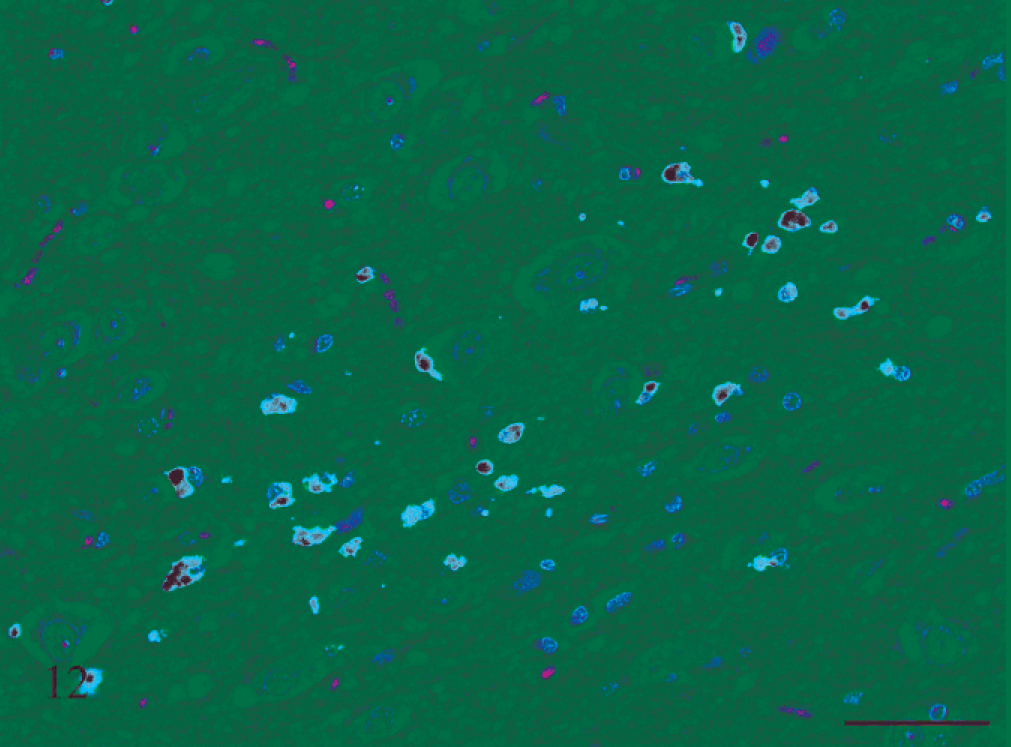
Higher-magnification image of Fig. 11. There are many hemosiderin-laden macrophages, mild spongiosis, and astrocytosis. HE and Perl Prussian blue reaction. Bar = 50 μm.
Fibrinoid change of blood vessels associated with hemorrhage or edema was also present in the lungs of 4 of the 7 rats examined that received rotenone for between 2 and 8 days. Within the areas of pulmonary hemorrhage were eosinophilic linear, rectangular, and rhomboid structures interpreted to be hemoglobin crystals 31,32 (Fig. 13). No pulmonary lesions were detected in the lungs of the 3 rats that received rotenone for between 12 and 19 days. The pancreas was inflamed in 5 rats that received rotenone for 3 to 4 days (Fig. 14).
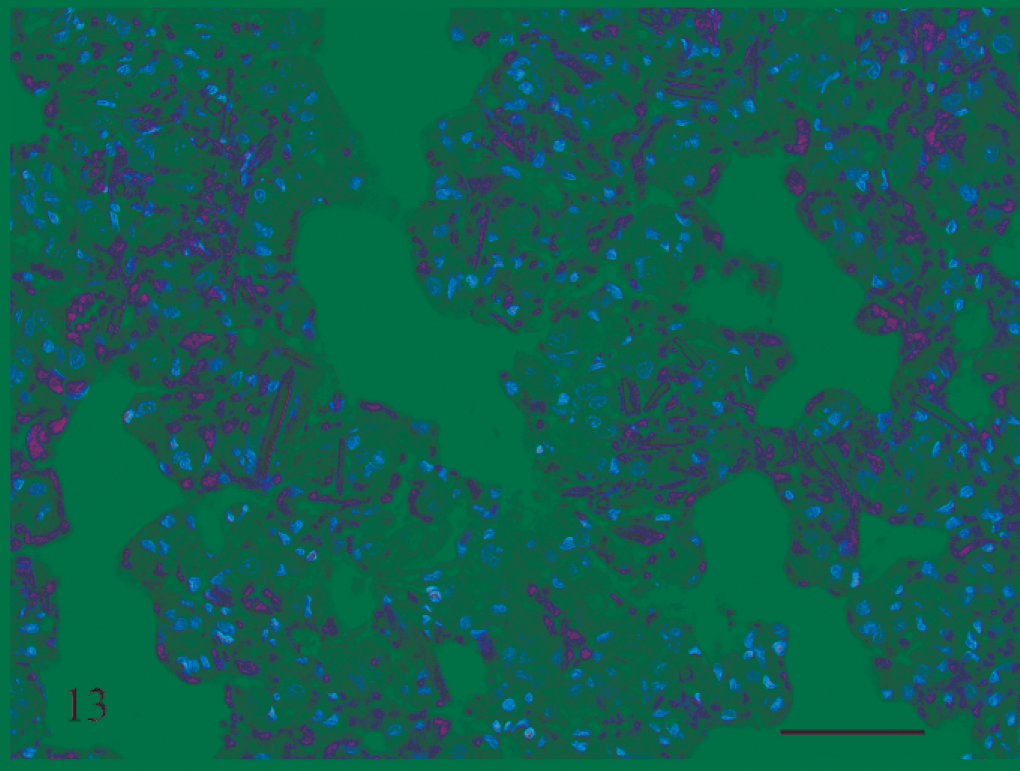
Lung; rat No. 2. Area of hemorrhage in the lung of a male Lewis rat (No. 2) dosed with rotenone for 3 days prior to being euthanatized. The linear, rectangular, and rhomboid eosinophilic structures are hemoglobin crystals. HE. Bar = 50 μm.
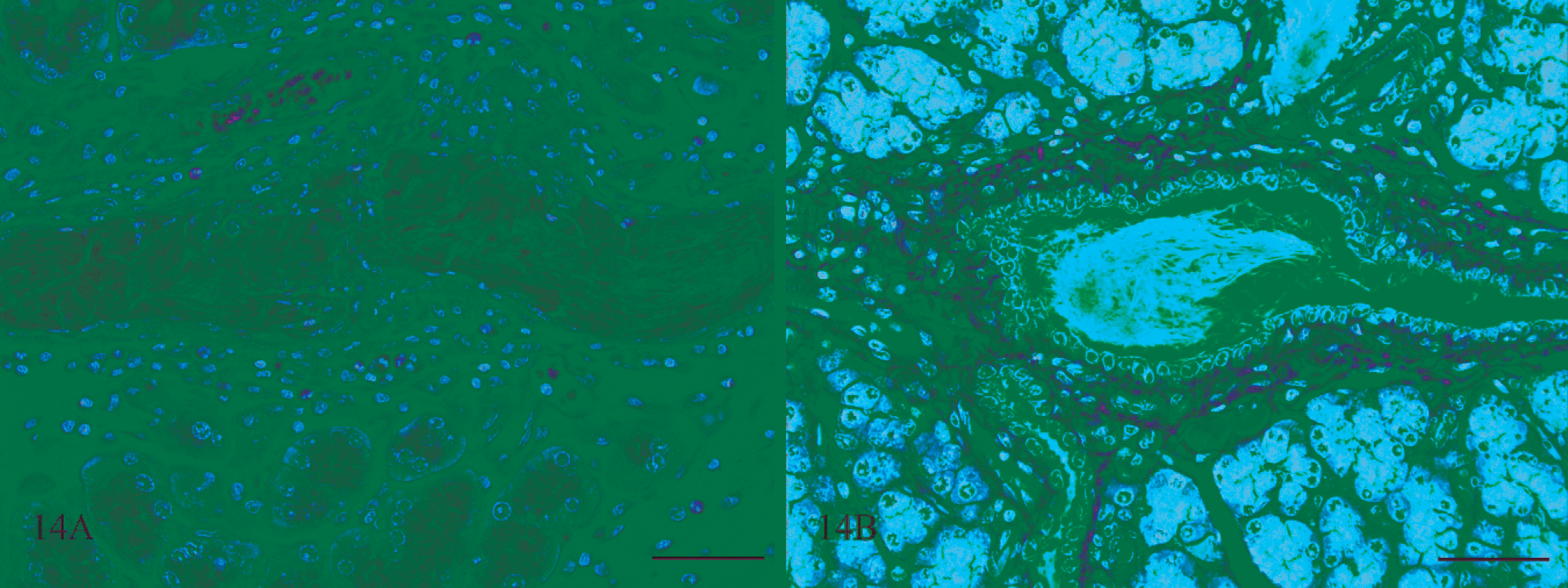
Pancreas; rat No. 3. Pancreas of a male Lewis rat (No. 3) dosed with rotenone for 3 days prior to being euthanatized. Fig. 14A. There are a moderate number of inflammatory cells, mostly macrophages and a few eosinophils, surrounding a ductule filled with fibrillar eosinophilic material HE. Bar = 50 μm. Fig. 14B. The material within the ductule stains blue, consistent with being fibrin, using the PTAH method. Bar = 50 μm.
The chronic parenteral administration of relatively low doses of rotenone in rats has gained popularity as a laboratory animal model for the study of PD. 3,10,15,18 Unfortunately, rats treated in the same way do not uniformly develop the desired clinical signs and brain pathology. In fact, a large proportion of dosed rats will become moribund or die within the first few days to weeks of administration. The very different response to rotenone administration among a genetically similar population of rats is intriguing and not understood. Work by Phinney et al. 25 suggested that some of the difference might be explained by the age of the dosed rats.
The cause of the clinical signs and death of acutely affected rats in this study appeared to be related to blood vessel damage and hemorrhage within the brain, particularly the thalamus, hypothalamus, and medulla oblongata. We surmise that blood vessels in the brain of male Lewis rats, particularly in the diencephalon and brain stem, are either different from blood vessels in other organs and tissues in the body or are more susceptible to the systemic effects of rotenone. 9 A similar situation must also exist with regard to pulmonary vessels because there was fibrinoid change associated with hemorrhage and edema in the lungs of 4 of the 7 rats examined that received rotenone for between 2 and 8 days.
The lesions in the brain (and lung) reflect vascular injury, but whether the nature and cause of this injury are relevant to the rotenone model of PD has not been investigated. Any attempt to elucidate the mechanism of acute cerebrovascular injury in the rats in this study would be speculative. Nevertheless, disruption of the blood–brain barrier with transmural vascular and perivascular leakage of proteinaceous fluid, along with hemorrhages affecting small arteries and arterioles, are characteristic of hypertensive brain injury. 27 It remains to be determined whether rotenone administration leads to hypertension and acute cerebrovascular injury or whether some other mechanism is involved.
Because rotenone has 2 known modes of action at the cellular level, understanding the pathogenesis of the selective nature of the hemorrhage might provide insights into how rotenone produces PD-like behavioral changes and brain lesions in some rats. Such understanding might be useful in determining how findings in this PD-like animal model resemble PD in humans.
Similarly, an understanding of why certain vessels in the brain of rats are more susceptible to rotenone and develop fibrinoid change and hemorrhage might provide a basis for developing a model for the intraparenchymal hemorrhagic cerebrovascular disease (i.e., hemorrhagic strokes) of humans.
Footnotes
Acknowledgements
This work was supported by the Print West Golf Classic for Parkinson's Disease and the Regina Curling Classic for Parkinson's Research. We wish to thank Mohammed Khysar Pasha, Hélèlene Philibert, and Lothar Resch for their technical assistance and valuable contributions.
