Abstract
Ring hemorrhages are characteristic ring-shaped lesions in the parenchyma of the central nervous system (CNS) surrounding small blood vessels and are typically reported to occur in white matter of the brain. In humans, they are seen in various diseases, including pernicious anemia, cerebral fat embolism, and cerebral malaria. Ring hemorrhages are also sporadically seen in the CNS of animals and have been previously reported in 4 nonhuman primate (NHP) species with various forms of anemia. Here we present the results of 4 preclinical toxicity studies testing 4 drug candidates of different modalities, targets, mechanisms of action, and indications in NHPs (cynomolgus monkeys). Within each study, ring hemorrhages were observed associated with severe anemia in affected animals and were in line with those previously reported. They occurred in gray matter and occasionally white matter of the brain and spinal cord; in the brain, the thalamus, basal ganglia, and cerebellum were particularly affected. Lesions comprised a central eosinophilic core, sometimes with small blood vessels or eosinophilic to slightly basophilic amorphous material in the center, surrounded by red blood cells and/or microglial cells or microglia aggregates. Fibrin staining confirmed the presence of fibrin in the central core. No commonality in type/cause of anemia was noted; in one study, the anemia was considered a spontaneous (non-treatment-related) finding. A thorough examination of the brain is therefore recommended in the presence of anemic conditions in animals. Pathologists should be aware of this finding and its relationship with anemia when assessing associations with diseases or drug candidates.
Ring hemorrhages of the central nervous system (CNS) have been reported in the literature since the 1920s and have been observed in a wide variety of human diseases, such as pernicious anemia, 24 trauma, 22 overdose of certain drugs, 25 leukemia, 25 cerebral fat embolism, 13 extensive burns, 10 septicemia, 23 and cerebral malaria. 26 They are typically reported to occur in the white matter of the brain, but they have also been noted in the gray matter, as well as in the spinal cord.10,13 The classic type is described as circular lesions composed of red blood cells (RBCs) surrounding a pale perivascular focus of necrosis with a central capillary. The necrotic region contains fibrin and can transform into a homogeneous amorphic substance, or into a nodule of reactive cells. 4 Despite being noted in several human diseases, 4 ring hemorrhages were not reported in animal species until 1980, when they were observed in 4 species of monkey with various forms of anemia. 3 These species were the owl monkey (Aotus trivirgatus), Formosan macaque (Macaca cyclopis), capuchin (Cebus albifrons), and the crab-eating macaque, also known as the cynomolgus monkey (Macaca fascicularis).
Four histological types of ring hemorrhage lesions were observed in each of the 4 species of monkey, but most were of the classic type that were observed in humans with anemia and other diseases. 3 The suggested cause of these lesions was endothelial damage, likely at interendothelial tight junctions, caused by severely anemic conditions, resulting in leakage of RBCs and fibrinogen into the brain tissue. 3 Leaked RBCs then either remain as a circular lesion surrounding the vessel, or are pushed away from the leaking vessel to form a ring of RBCs, grossly observed as petechiae.
In humans, ring hemorrhages have recently been reported more frequently in the literature, predominantly in cases of cerebral malaria.8,11,16,26 However, this is likely due to the research emphasis placed on this area as malaria remains a major health care problem in some parts of the world. 19 In animals, ring hemorrhages have been rarely reported since their initial discovery,5,7,18,28 but they are now becoming increasingly recognized. 1 This may also be relevant for cynomolgus monkeys, as they are an accepted non-rodent species for preclinical toxicity testing by regulatory agencies, and they are commonly used in the development of biologics.15,17,20 Herein we report cases of ring hemorrhages in 4 separate toxicity studies in cynomolgus monkeys.
Materials and Methods
The results detailed herein are from 4 toxicity studies in cynomolgus monkeys sponsored by Roche Pharma Research and Early Development (F. Hoffmann-La Roche Ltd, Switzerland) and are summarized in Supplemental Table S1. Only the methods and results that are relevant for the presented lesions are reported. Studies were performed in laboratories approved by the Association for the Assessment and Accreditation of Laboratory Animal Care or in compliance with all applicable sections of the Final Rules of the Animal Welfare Act regulations (Code of Federal Regulations, Title 9), the Public Health Service Policy on Humane Care and Use of Laboratory Animals from the Office of Laboratory Animal Welfare, and the Guide for the Care and Use of Laboratory Animals from the National Research Council. Three studies were conducted in compliance with Good Laboratory Practice regulations. The fourth study was a non-regulatory study, but laboratory procedures were consistent with international standards of Good Laboratory Practice and in agreement with the study plan and subsequent amendments.
Compounds
All investigated compounds were in preclinical development at Roche Pharma Research and Early Development (F. Hoffmann-La Roche Ltd, Switzerland). Compounds 1, 2, and 3 were monoclonal antibodies, whereas compound 4 was a small molecule. Dosing of each compound was performed based on the anticipated pharmacokinetic/pharmacodynamic profile.
Cynomolgus Monkeys and In-Life Study Management
All animals were purpose-bred for these studies, and a health assessment was performed by the breeder before being transferred to the testing facility; no pre-existing conditions were noted. An additional health assessment was performed by a qualified member of the veterinary staff at the testing facility before the pre-dose phase to confirm the suitability of each animal for inclusion in the studies. Group assignments of the animals were, where possible, based on existing social groups and stratified body weights. All animals were acclimated to the test facility for at least 23 days prior to study initiation. In all studies, the animals were group-housed in stainless steel monkey cages; in 3 of the studies in accordance with European Union requirements (Directive 2010/63/EU). The animals were fed twice daily, supplemented with fresh fruit and water ad libitum. Animals were maintained under 12-hour light/dark cycles at room temperature (22–28°C) with a relative humidity of 30–75%. The total number of animals used, as well as group size and number of groups (Supplemental Table S1), were considered the minimum required to properly characterize the effects of each compound and were designed to not require an unnecessary number of animals to accomplish the study objectives. A concurrent control group was included in all 4 studies.
Clinical observations included health monitoring twice daily during the dosing and recovery phases for signs of ill health or overt toxicity. Postdosing observations were also conducted. Other examinations/observations included ophthalmic observations, body weight and food consumption recordings, and physical examinations. Scheduled and early euthanasia (if necessary) was performed with an intramuscular ketamine hydrochloride sedation (5–20 mg/kg) followed by an intravenous sodium pentobarbitone overdose (100–190 mg/kg) prior to exsanguination. All animals were subjected to a complete necropsy examination, which was performed by qualified personnel with appropriate training and experience in animal anatomy and gross pathology. A veterinary pathologist, or other suitably qualified person, was also available for consultation.
Hematology and Coagulation Profiles
Animals were fasted overnight prior to blood collection. Blood samples were collected by venipuncture and assessments included a complete blood cell count (RBC mass [RBC count, hemoglobin, and hematocrit], RBC indices, reticulocyte counts, platelet count, and white blood cell counts) and white blood cell differential cell counts (neutrophils, lymphocytes, monocytes, eosinophils, and basophils). Coagulation tests included activated partial thromboplastin time (aPTT) and prothrombin time (PT).
Tissue Collection and Preservation
Samples of all major organs and tissues were collected and preserved in 10% neutral-buffered formalin. Bone marrow smears were collected, allowed to air dry, and fixed with methanol prior to labeling and staining. Bone marrow cytology slides were evaluated by light microscopy for intact nucleated myeloid, erythroid, lymphoid, and other cells, if indicated (eg, macrophages, mast cells). Any notable cell abnormalities were recorded. Megakaryocyte numbers (eg, adequate, increased, or decreased) were estimated, and the myeloid to erythroid ratio was calculated based on the number of intact cells counted.
Histology and Immunohistochemistry
Tissues for examination were embedded in paraffin, sectioned, mounted on glass slides, and stained with hematoxylin and eosin. At least 5 sections of the brain were examined, as recommended by Bolon et al; 2 7 sections were examined in studies 1, 2, and 3, and 5 sections of the brain were examined in study 4, including the cerebral cortex, thalamus, midbrain, medulla, and cerebellum. Cross sections of the cervical, thoracic, and lumbar spinal cord were examined in all 4 studies. In addition, longitudinal sections of these spinal cord segments were evaluated in study 1. Additional special staining on individual animals and tissues included Martius scarlet blue for fibrin visualization, Congo red for amyloid, and Perls Prussian blue stain for iron associated with hemosiderin. Ziehl-Neelsen and Brown-Brenn bacteriological staining, and Grocott methenamine silver and periodic acid-Schiff staining for fungal organisms were also used.
Immunohistochemistry was performed for CD31 to confirm the presence of blood vessels within respective lesions and for the glial fibrillary acidic protein, which is found in astrocytes and glial cells of the CNS and is a biomarker for CNS injury and other neurological conditions. 27 Histological evaluations were performed by a trained veterinary pathologist with a contemporaneous peer review. For CD31, the mouse anti-CD31 antibody (ab9498; Abcam, Cambridge, UK) was diluted in Dako Antibody Diluent with Background-Reducing Components (S3022; Dako, California, USA) at 1:100. Signal detection was performed using the Ventana Omni anti-Mouse HRP kit and 3,3′-diaminobenzidine. The negative control antibody used was the mouse IgG control antibody (I-2000, VectorLabs, California, USA). For glial fibrillary acidic protein, the glial fibrillary acidic protein antibody (Z0334, Dako) was diluted in Dako Antibody Diluent with Background-Reducing Components at 1:2900. Signal detection was performed using the Dako EnVision+ System-HRP anti-Rabbit kit and 3,3′-diaminobenzidine. The negative control antibody used was the Rabbit IgG Control Antibody (I-1000, VectorLabs). For both CD31 and glial fibrillary acidic protein, internal positive controls (ie, known positive tissue parts) were used to validate the immunolabeling procedure and ensure the accuracy and reliability of the results.
Results
Study 1
In this study, compound 1 was generally not well tolerated due to severe anemia, which led to early euthanasia of 3/40 animals (Table 1). Clinical signs included hunched posture, decreased activity, and pale mucous membranes/gums. Altogether, 8/40 animals developed moderate-to-severe anemia, along with a moderate-to-severe decrease in reticulocyte counts, and a mild to severe decrease in mean corpuscular volume in 7/8 animals (Tables 1, 2). Onset of anemia development varied, occurring between 3 and 13 days before sacrifice. Moderate to severe thrombocytopenia in 6/8 anemic animals, moderate to severe neutropenia in 7/8 animals, and moderate lymphopenia in 3/8 animals was additionally noted (Table 2). Both aPTT and PT were within normal limits.
Overview of study results.
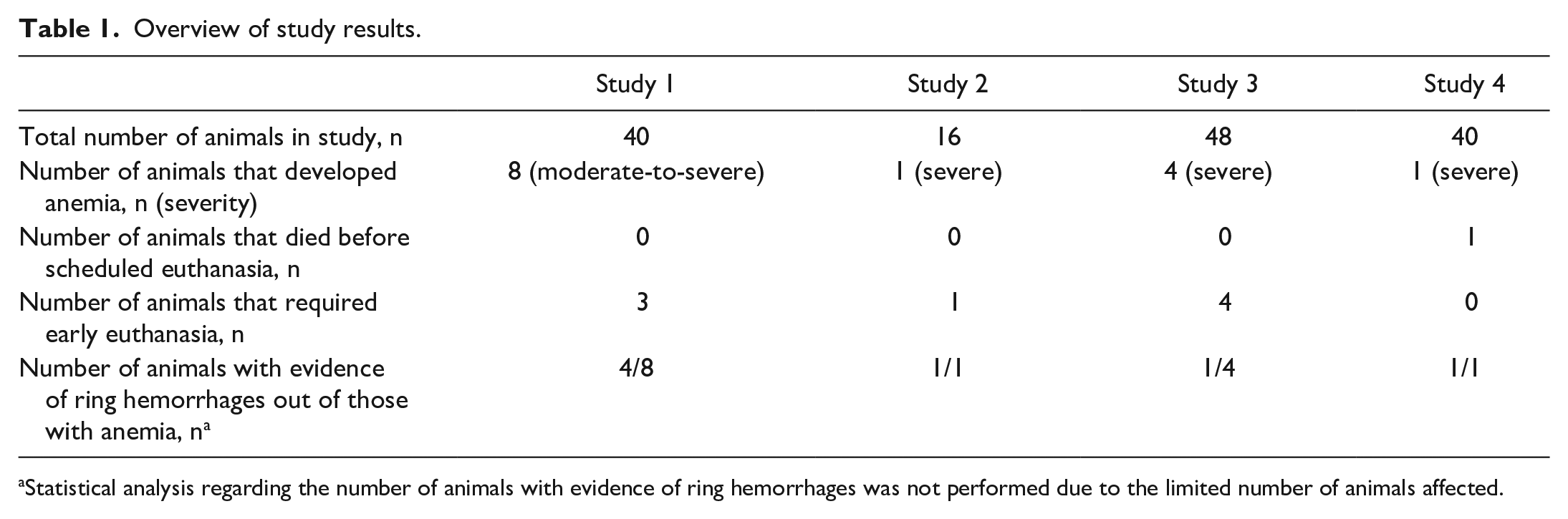
Statistical analysis regarding the number of animals with evidence of ring hemorrhages was not performed due to the limited number of animals affected.
Ring hemorrhage findings and summary of hematology of animals with ring hemorrhages.
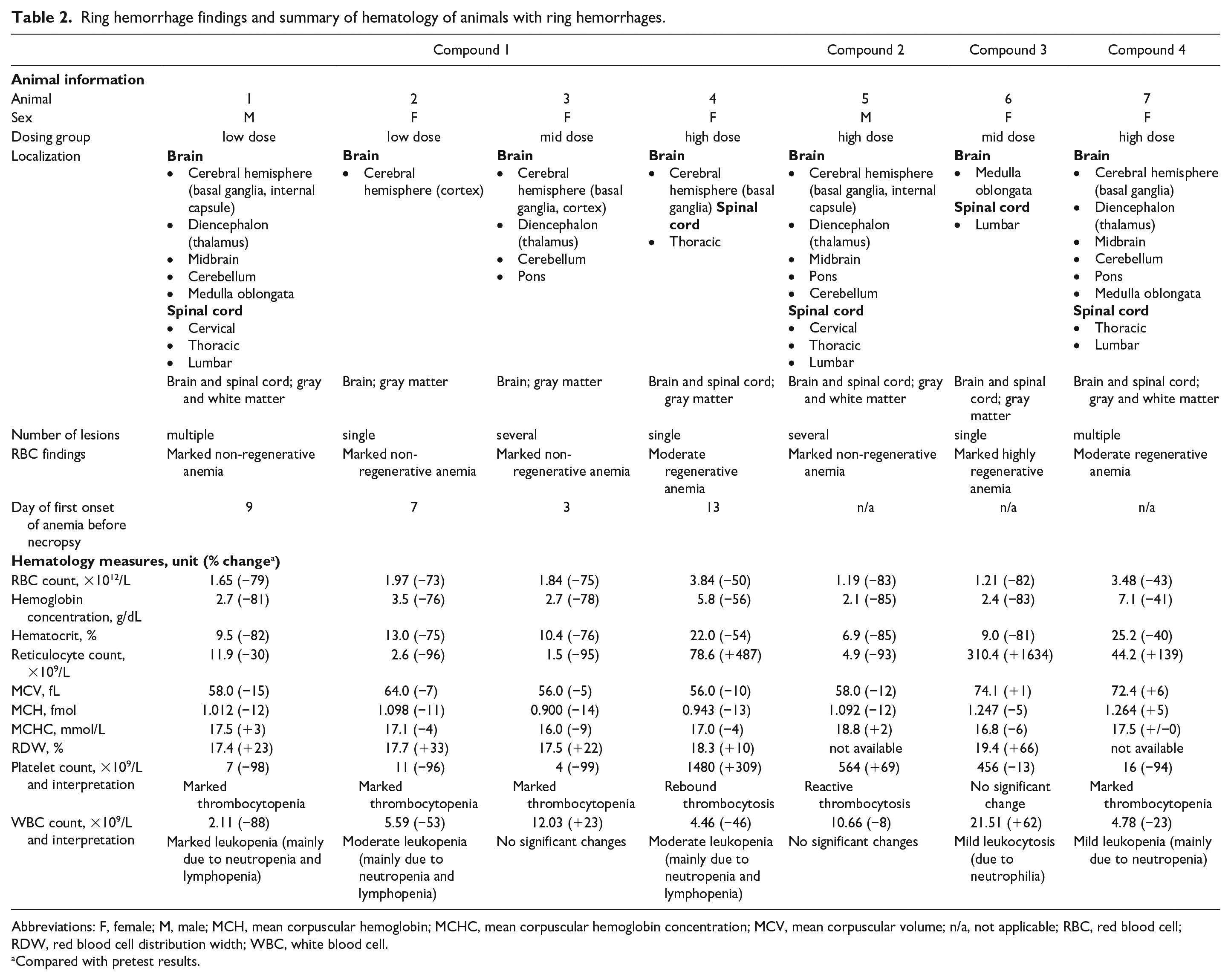
Abbreviations: F, female; M, male; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; n/a, not applicable; RBC, red blood cell; RDW, red blood cell distribution width; WBC, white blood cell.
Compared with pretest results.
At unscheduled necropsy, generalized or visceral organ pallor was noted due to the decreased RBC mass. Microscopic or macroscopic hemorrhages were noted in multiple tissues, including the brain, heart, and lung.
Histopathologic findings consistent with ring hemorrhages were observed in the brain of the 3 animals that were euthanized early, as well as in one female at the end of treatment. Changes in the bone marrow smears in these animals generally correlated with the changes observed in the peripheral blood and included alterations in the myeloid to erythroid ratio, maturation shifts toward immaturity of the myeloid and/or erythroid lineages, and increased numbers of macrophages, some of which were cytophagic or contained cellular debris and/or minor hemosiderin pigment. Respective bone marrow changes were noted in the histological sections of the 3 early-euthanized animals and consisted of decreases in erythroid and megakaryocytic lineages and increases in myeloid lineage cellularity, predominantly of immature myeloid precursors. Also noted was an increased presence of macrophages with/without cytoplasmic pigment and cell/cell debris or increased immature erythroid and myeloid lineages, composed predominantly of round immature precursor cells, decreased megakaryocytes, and increased macrophages, with/without cytoplasmic pigment and cell/cell debris. In the fourth animal, decreases in myeloid, erythroid, and megakaryocytic lineages were noted, and the myeloid lineage was predominately composed of immature precursors. In addition, increased macrophages and multiple areas of necrosis, characterized by foci of cell debris with phagocytosis by macrophages, were observed.
Lesions consistent with ring hemorrhages were characterized by focal or multifocal hemorrhages with a central eosinophilic core, neuronal degeneration, or areas of gliosis, some with small blood vessels or eosinophilic to slightly basophilic amorphous material in the center (Fig. 1). These lesions were particularly noted in the gray matter of the cerebral hemispheres and diencephalon, and, in more severe cases, also in the midbrain, cerebellum, and medulla oblongata. Some lesions were also noted in the white matter. The spinal cord was affected in 2/4 animals (Fig. 2, Table 2). Evidence for hypoxia was present in the liver of 1/4 animals in the form of marked centrilobular to bridging hepatocellular degeneration/necrosis.
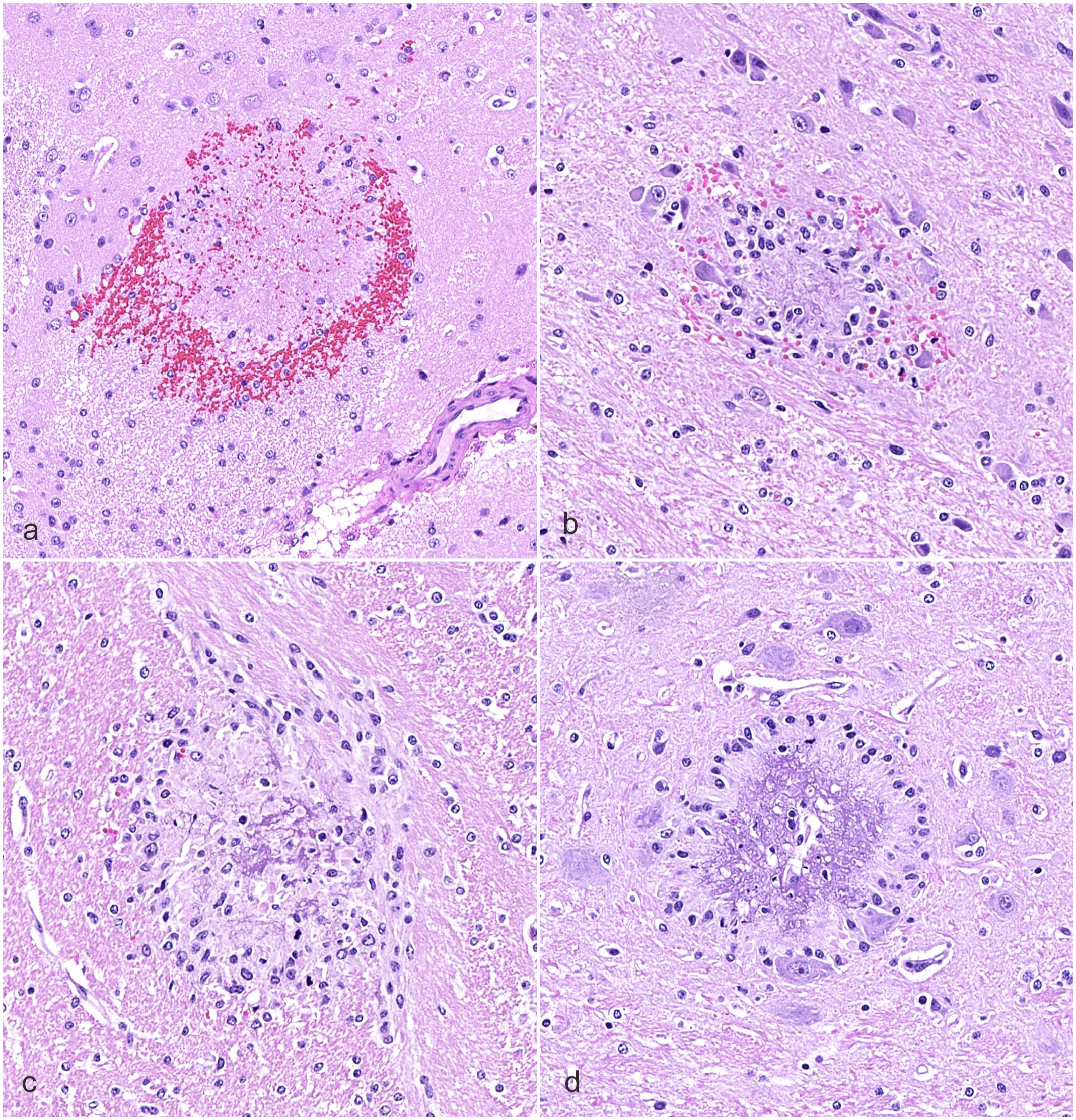
Ring hemorrhage lesions in gray and white matter of the brain from animals in study 1. Hematoxylin and eosin. (
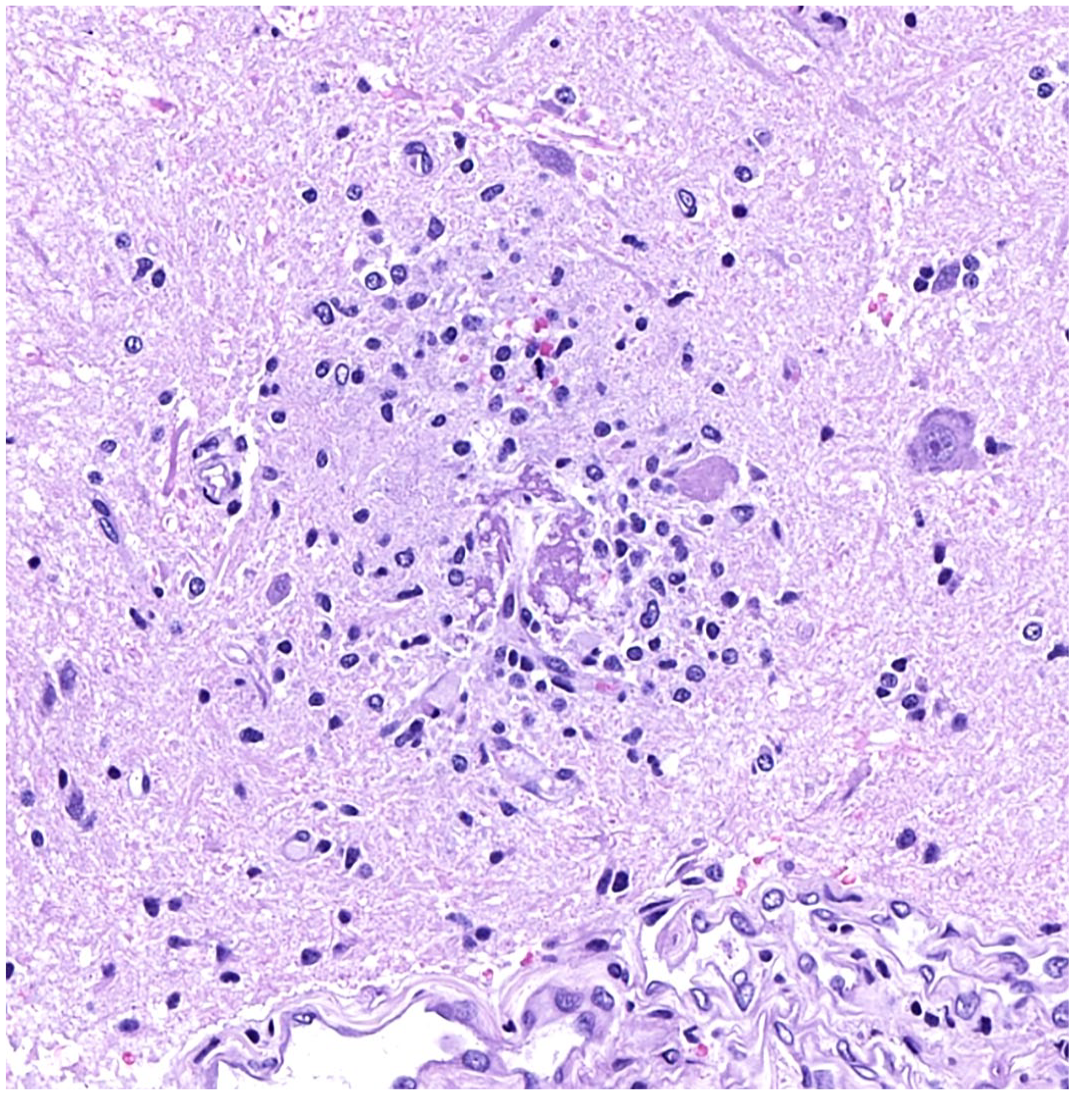
Lumbar spinal cord. Ring hemorrhage lesion in the spinal cord of an animal in study 1. Type 3 lesion with an eosinophilic core and a central capillary surrounded by microglia cells. Single red blood cells are in periphery. Case 1. Hematoxylin and eosin.
Study 2
In study 2, compound 2−related changes at the mid and high dose consisted of decreases in RBC mass and indications of an acute phase response, resulting in dose-limiting, non-regenerative anemia in one animal, for which early euthanasia was required due to the severity of the anemia (Tables 1, 2), poor physical condition, and body weight loss. Decreased indicators of RBC mass on the day of euthanasia were accompanied by a severe decreased absolute reticulocyte count, which correlated with a markedly increased myeloid to erythroid ratio (33:1) and moderately decreased cellularity in the femoral and sternal bone marrow. Stainable bone marrow iron content was comparable with that of controls. The decrease in red cell mass occurred in the absence of coagulopathy (no concurrent prolongations in aPTT or PT). An increased platelet count was considered secondary to the changes in RBC mass indicators. The most recent blood sampling, 26 days before the sacrifice of the animal, did not show any significant anemia.
The microscopic findings were similar to those observed with compound 1. Multifocal lesions composed of a central eosinophilic core, sometimes with small blood vessels or eosinophilic amorphous material in the center, surrounded by RBCs and/or microglial cells or microglia aggregates were observed in the brain (Fig. 3) and spinal cord of the early-euthanized animal. Brain lesions were present in the gray and white matter of the cerebral hemispheres, thalamus, midbrain, cerebellum, and pons. In the spinal cord, gray/white matter of cervical, thoracic, and lumbar segments was affected. Special stains of the brain and spinal cord were negative for infectious organisms. Glial fibrillary acidic protein immunohistochemistry to assess glial cell reaction was mostly unremarkable, aside from occasional cells in some lesions in the brain and spinal cord. The eosinophilic core of the lesions did not show amyloid deposits when stained with Congo red. CD31 immunohistochemistry confirmed the presence of blood vessels within some lesions. Martius scarlet blue staining of brain and spinal cord sections showed positive staining of the eosinophilic core of the lesions, confirming the presence of fibrin (Fig. 3d).
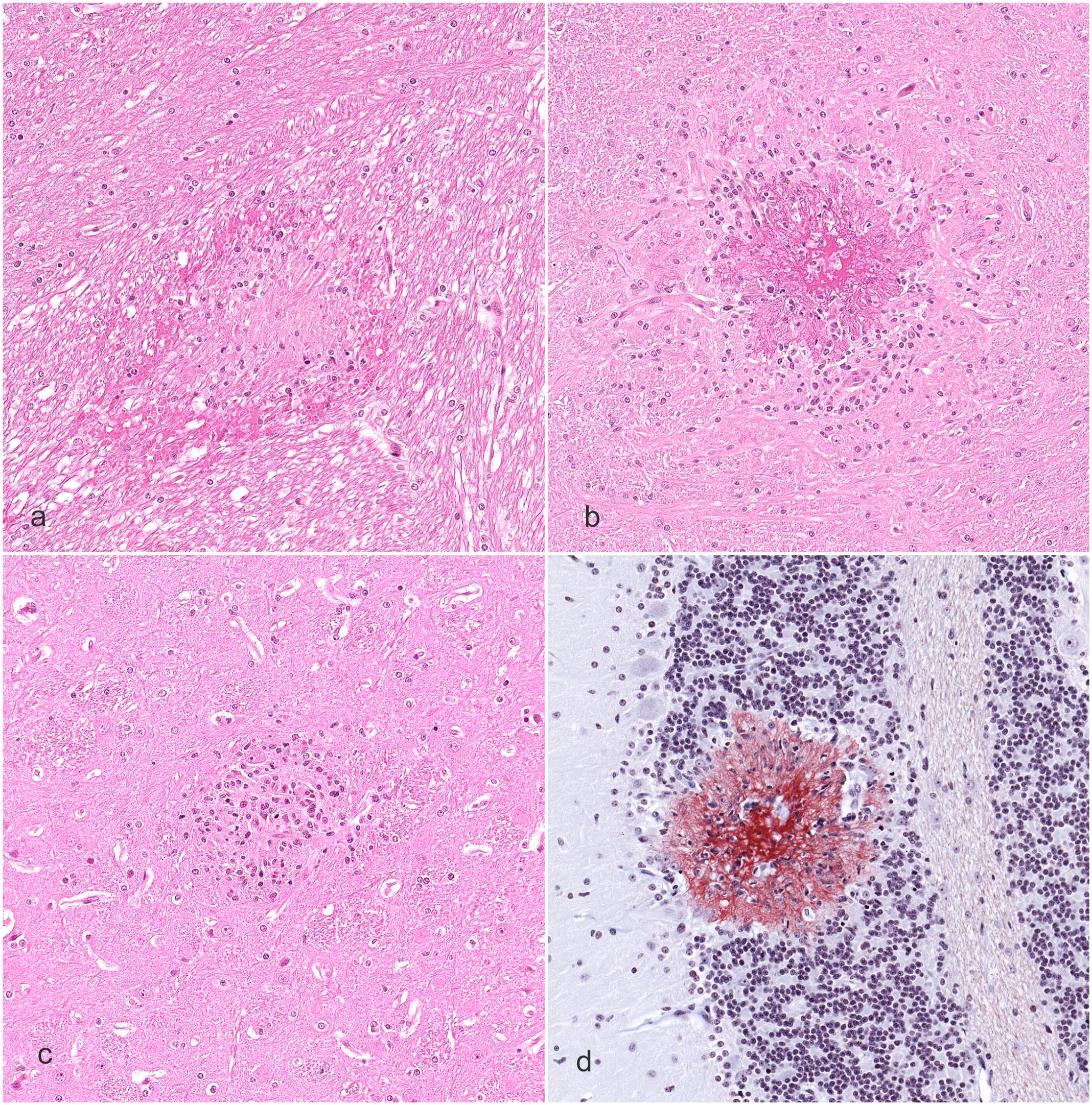
Ring hemorrhage lesions in gray and white matter of the brain from the animal in study 2. (
Study 3
In study 3, compound 3−related hematology findings were noted in treated animals and consisted of mildly decreased red cell mass, white blood cell count, and erythroid bone marrow precursors, suggesting decreased marrow production. Correlative microscopic findings included decreased microscopic cellularity of the sternal bone marrow.
All animals in the 4-week recovery group survived to the scheduled sacrifice date. Animals assigned to the 52-week recovery group underwent an early sacrifice and necropsy on days 351 (males) or 316 (females) due to low hematocrits and/or thin body conditions noted in 1 male and 2 females, and there was a moribund sacrifice of one female on day 307 due to general debilitation. Hematology findings in animals of the 52-week recovery period consisted of dose-independent, mild to moderately decreased red cell mass. Absolute reticulocyte counts were lower than expected for functional bone marrow responding to a decreased red cell mass, which supported a bone marrow effect and decreased bone marrow production of hematopoietic cells. Platelet counts, aPTT, and PT were unaffected.
The moribund sacrificed animal at day 307 presented with the most severe anemia, with a markedly decreased red cell mass and moderately increased absolute reticulocyte count (Table 2). A previous sampling for hematology at 279 days before sacrifice did not reveal any anemia. Histopathologically, this animal had one focus consistent with a ring hemorrhage in the brain (Fig. 4a) and one in the spinal cord. The foci were characterized by pale, slightly eosinophilic regions surrounding a small blood vessel. More peripherally, the foci had loss of normal neutrophil morphology and an increased number of microglial cells surrounded by a rim of RBCs.
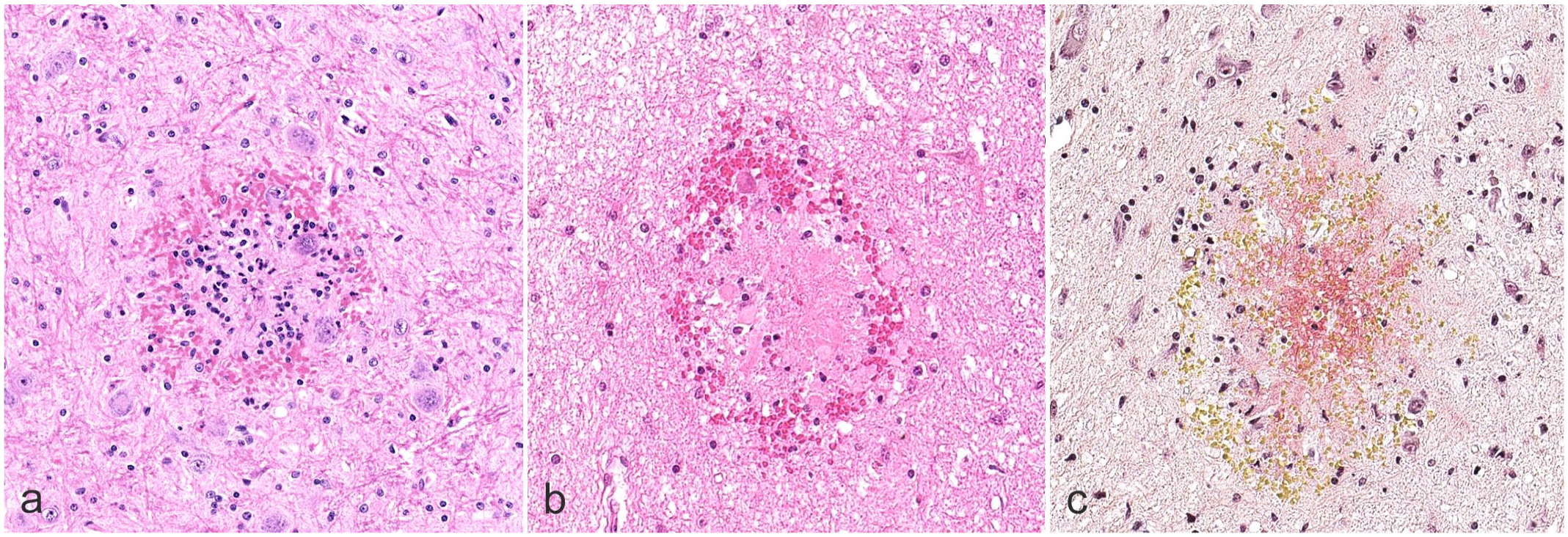
Ring hemorrhage lesions in the brain of animals in studies 3 and 4. (
Study 4
In study 4, there were no compound-related adverse effects. However, one female died before the scheduled date of euthanasia (Table 1); this death was not considered to be related to compound 4 but due to spontaneous severe megakaryopoietic hypoplasia and dysplasia resulting in severe thrombocytopenia and hemorrhages, the latter leading to severe anemia. On the day prior to death, the animal appeared slightly pale, cold, and collapsed. A hematoma was observed on the left side of the head from days 107 to 111, and strong menstrual bleeding was observed until day 110. The last sample for hematology was collected on day 87; changes consisted of moderate anemia, marked thrombocytopenia, and a moderate increase in reticulocyte counts (Table 2). Both aPTT and PT were unaffected. No earlier samples were taken during the treatment period.
Circulatory failure, following marked regenerative anemia and severe thrombocytopenia, associated with heavy menstrual bleeding, was considered as the cause of death in this one animal; additional findings in the brain and spinal cord might have contributed to the fatal outcome. Excessive bleeding was considered to be associated with low platelet and megakaryocyte counts and was associated with abnormal megakaryocyte morphology. Microscopic evaluation of the sternal bone marrow and the bone marrow myelogram showed a severe reduction of megakaryocytes with abnormal morphology, correlating with marked thrombocytopenia. The thrombocytopenia was considered responsible for the heavy and prolonged menstrual bleeding and the multifocal hemorrhages observed in the heart and brain. Microscopic findings included multiple foci of hemorrhage, and lesions consistent with ring hemorrhages in the gray and white matter of the brain (Fig. 4b). Lesions were especially observed in the thalamic region but were also in the cerebral hemisphere, midbrain, cerebellum, pons, and medulla oblongata.
The spinal cord (cervical and lumbar segments) also showed parenchymal hemorrhage and ring hemorrhage lesions in the gray and white matter. Martius scarlet blue staining confirmed presence of fibrin in lesions with an eosinophilic core (Fig. 4c). Some minor Perls Prussian blue iron staining for hemosiderin was present in areas of hemorrhage in the thalamus, whereas periodic acid-Schiff reaction for fungi was negative.
Discussion
In the 4 toxicity studies presented herein, brain and spinal cord ring hemorrhages were observed in 7 animals. These lesions have been observed in humans in a variety of diseases and various underlying mechanisms have been proposed. It has been hypothesized that ring hemorrhages were the result of vasoparalysis, agonal blood pressure changes, or prolonged hypoxemia,9,21,25 but the exact pathophysiological mechanism for their development remains elusive. 14 There are only a few reports of ring hemorrhages in animals.3,5,7,18 They have been described in Thoroughbred horses with experimentally induced endotoxemia following exposure to endotoxin, and lesions were similar to those reported in humans with septic shock. 18 Another case was reported in a harbor seal with a West Nile virus infection. 7 In monkeys, ring hemorrhages have been reported in 4 species with different types of anemia.3,5 Ring hemorrhages in association with anemic conditions have also been reported in humans, in association with aplastic anemia and pernicious anemia. 4 In the cases presented herein, the only consistent finding associated with ring hemorrhages was the presence of moderate to marked anemia; there were no indications of previous trauma, infections, or metabolic disorders, which can present with similar lesions.
The relationship between anemia and ring hemorrhages in the CNS has been previously proposed by Bronson et al, 3 who reported light microscopic findings of 4 types of ring hemorrhage. Type 1 is described as a ball hemorrhage that consists of red cells and strands of fibrin surrounding a central capillary. Type 2 ring hemorrhages have pale homogeneous eosinophilic centers with microglia and polymorphonuclear leukocytes surrounded by a ring of erythrocytes. Type 3 ring hemorrhages are similar to type 2, with a pale core and some polymorphic leukocytes or microglia, although the ring of RBCs is absent. Type 4 ring hemorrhages are composed of aggregates of microglia and macrophages without eosinophilic cores, and without evidence of hemorrhage or hemosiderin. These different types are thought to evolve sequentially, although type 3 might also develop independently without previous RBC leakage. 3 The pale ring hemorrhage cores were ultrastructurally identified as degenerating neurites with interstitial fibrin deposition. 3 Erythrocytes at the lesion periphery were located in intercellular spaces or were phagocytosed. Based on these morphological findings, the primary mechanism of ring hemorrhage formation proposed by Bronson et al 3 was considered to be endothelial tight junction damage, with the resulting fibrinous exudation leading to neuritic degeneration.
The lesions noted in our studies were similar in nature to those described in the medical literature, as well as exhibiting features of those reported in 4 species of monkey with different types of anemia (aplastic, hemolytic, and hemorrhagic). 3 For example, Figs. 1a, 3a, 4b, and 4c show lesions similar to that of the type 1 lesions, those in Figs. 1b–d, 2, 3b, 3d, and 4a are similar to the type 2 or type 3 lesions, and the lesion in Fig. 3c is consistent with the type 4 lesions. 3 Of note, Bronson et al described the absence of RBCs in type 3 lesions, but in some of our lesions categorized as type 3 lesions, single RBCs were present in the periphery. In the brain, the thalamus, basal ganglia, and cerebellum were particularly affected, suggesting these regions may be predisposed to hemorrhage. However, due to the limited number of animals in which this was observed in these studies, a definitive conclusion on hemorrhage localization patterns in relation to the proposed mechanisms of endothelial damage and anemia cannot be drawn.
In our studies, there was no commonality in the type and cause of anemia, nor in the absence or presence of thrombocytopenia or coagulopathies. This is in line with previous reports in the literature. In 3 of the cases, the anemia was considered to be induced by the administered compound; in one study, it was considered a spontaneous finding.
Bronson et al 3 also reported a relationship between the degree of anemia and the presence of ring hemorrhage lesions, which was also noted in the studies presented herein. Endothelial damage was suggested to occur when hemoglobin concentrations fall below 5 g/dL, causing fibrin plaques and subsequent leakage of RBCs and fibrin. 3 The presence of fibrin prevents diffusion of the intercellular fluid, leading to cell degeneration and infiltration of inflammatory cells. 3 . However, not all of the cases reported here had hemoglobin concentrations below the suggested 5 g/dL threshold; one case (study 4) involved an animal that was sacrificed only 25 days after the last blood sample was collected and based on the clinical history of this animal, a more severe form of anemia would have been expected at the time of sacrifice. In another case (study 1), the anemia developed over approximately 13 days, and at the time of sacrifice, the hemoglobin concentration was still above the reported threshold of 5 g/dL (5.8 g/dL). This animal had only 2 ring hemorrhage lesions in the brain and spinal cord, whereas other individuals with multiple ring hemorrhages in various lesions developed more severe anemia over a shorter period of time. Therefore, it seems likely that in addition to the severity of anemia, the duration and time course of anemia development may play a role in not only the presence or absence of ring hemorrhages, but also in the number of ring hemorrhage lesions. Furthermore, other unknown predisposing or contributing factors are likely to play a role, especially considering that in study 1, with frequent blood sampling prior to sacrifice, animals with comparable severity and time course of anemia did not show any evidence of ring hemorrhages. This is in line with reports from humans, where patients with severe malarial anemia also did not show any evidence of ring hemorrhages. 16
In the literature, ring hemorrhages are typically reported to occur in the white matter, although several authors also mention the presence of lesions in gray matter10,13 Interestingly, in our studies, the ring hemorrhages were mainly localized in the gray matter. This may be explained by the severity of the anemia, which resulted in the early sacrifice or even death of most animals, and the higher susceptibility of the gray matter to hypoxic-ischemic injury. 12
In addition to the ring hemorrhages described in the toxicity studies presented, one animal within study 1 also showed centrilobular to bridging hepatocellular degeneration and necrosis in the liver. This is similar to the findings reported in owl monkeys with hemolytic anemia. 5 In the owl monkeys, centrilobular necrosis in the liver, extramedullary hematopoiesis in the liver and spleen, and hemoglobin casts in kidney tubules were prominent histopathologic features of hemolytic anemia, and these monkeys also had microscopic brain lesions consistent with ring hemorrhages. 5 It is well known that centrilobular hepatocellular degeneration and necrosis can develop as a consequence of hypoxia, 6 and the presence of hepatocellular degeneration and necrosis in one monkey in our study supports the theory that the brain lesions were a consequence of hypoxia secondary to severe anemia. The microglia accumulation and hemosiderin reported herein indicate that these lesions were due to a prolonged course of anemia and exclude agonal change as a cause of the lesions. According to Bronson et al, 3 hemosiderin deposits were not a feature in their cases of ring hemorrhages. In our studies, however, hemosiderin was occasionally observed, although not as a prominent feature. Hemosiderin deposits have also been described in some cases of ring hemorrhages in humans, 13 and it can be assumed that there is some interindividual variation depending on the duration and the severity of the underlying disease. Multifocal hemorrhages in the CNS without features of ring hemorrhages, and extra-CNS hemorrhages in some animals, were considered likely due to the marked thrombocytopenia observed.
In summary, we have reported the occurrence of ring hemorrhages in the CNS of cynomolgus monkeys in 4 separate preclinical toxicity studies that tested 4 drug candidates of different modalities, targets, mechanism of actions, and indications. The ring hemorrhages were all associated with moderate to marked anemia and were consistent with lesions previously described in the literature. An association between anemia and ring hemorrhages in the CNS has been suggested previously, and it is recommended that a thorough examination of the brain be performed in the presence of anemic conditions in animals. Pathologists should be aware of this finding and its relationship with anemia when assessing associations with diseases or drug candidates.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858251343012 – Supplemental material for Ring hemorrhages in the central nervous system of severely anemic cynomolgus monkeys
Supplemental material, sj-pdf-1-vet-10.1177_03009858251343012 for Ring hemorrhages in the central nervous system of severely anemic cynomolgus monkeys by Barbara Lenz, Martina Stirn, Elke-Astrid Atzpodien, Annamaria Braendli-Baiocco, Anna Maria Giusti, Kerstin Hahn and Vanessa Schumacher in Veterinary Pathology
Footnotes
Acknowledgements
We would like to thank Ben McDermott of the Bioscript Group, for providing medical writing assistance, which was funded by F. Hoffmann-La Roche Ltd.
Authors’ Note
This manuscript has been prepared in the Uniform Requirements format.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of F. Hoffmann-La Roche Ltd. B.L., M.S., V.S, A.M.G., A.B.-B., and E.-A.A. are shareholders of F. Hoffmann-La Roche Ltd.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Roche Pharma Research and Early Development, F. Hoffmann-La Roche Ltd., Basel, Switzerland.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
