Abstract
A 14-year-old Haflinger gelding presented with a protruding mass involving the cornea of the right eye. The mass was resected and submitted for histopathologic and immunohistochemical examination. The preliminary diagnosis was corneal sarcoma, most likely fibrosarcoma. The immunohistochemical results confirmed the mesenchymal origin of the neoplastic cells, which were most consistent with a malignant peripheral nerve sheath tumor. Corneal mesenchymal neoplasms are extremely uncommon tumors in human beings and domestic animals. The cause for this tumor was not determined; infection with bovine papillomavirus was not detected.
Squamous cell carcinoma is the most common equine ocular tumor; it commonly arises at the limbus and originates from conjunctival or corneal epithelium. 3, 7 Angiosarcomas and hemangiosarcomas may occur at this location as well. 10 Melanoma has been reported in the human cornea 2 and in the eye of a horse. 14 Furthermore, a few cases of lymphoma at the temporal or ventrotemporal limbal region are described. 15 Equine sarcoid, attributed to infection with bovine papillomavirus (BPV) types 1 or 2, is a common skin tumor that, in the ocular region, usually involves the eyelids or periocular skin. 1, 13
A 14-year-old Haflinger gelding presented with a 2-year history of a protruding mass involving the cornea of the right eye. The history included repeated surgical removal of the mass. The previous histologic diagnosis for a biopsy specimen from the mass was granulomatous keratitis with fibrosis. Grossly, a protruding mass 3 cm in diameter involved about 90% of the right cornea. The tumor was a well-demarcated, hyperemic, soft mass that extended from the limbus to the center of the cornea, impairing closure of the eyelids. Corneal tissue adjacent to the tumor was opaque (Fig. 1).
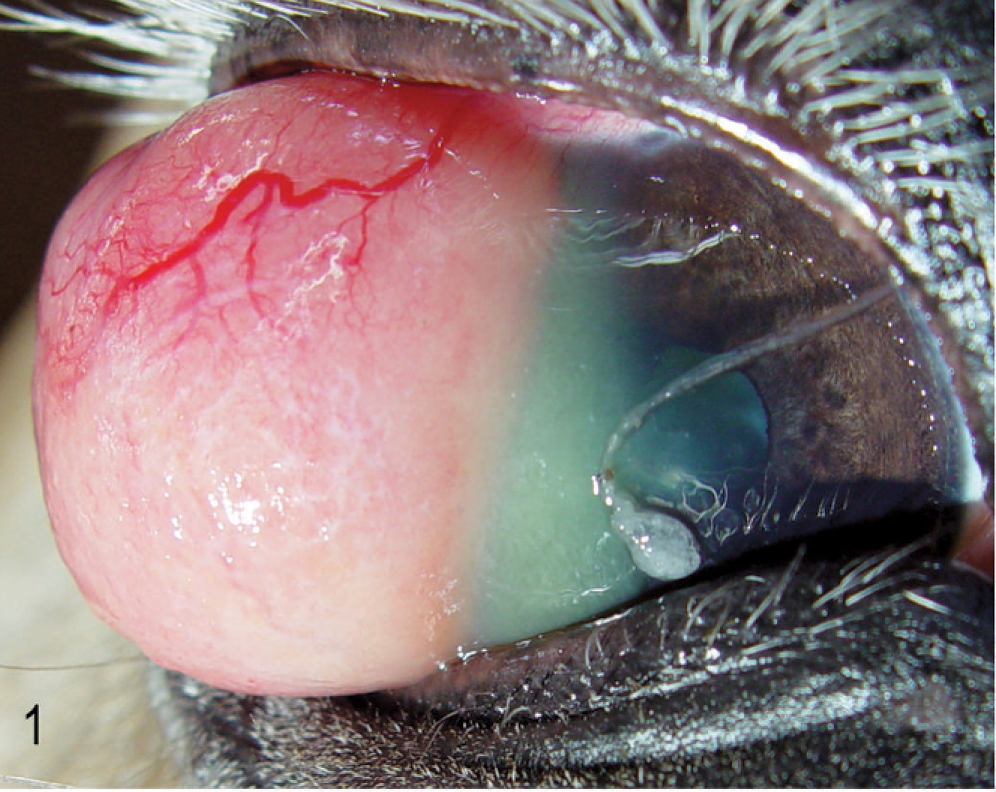
Right eye; horse. Clinical presentation before enucleation of the globe. Note the marked vascularization of the corneal tumor.
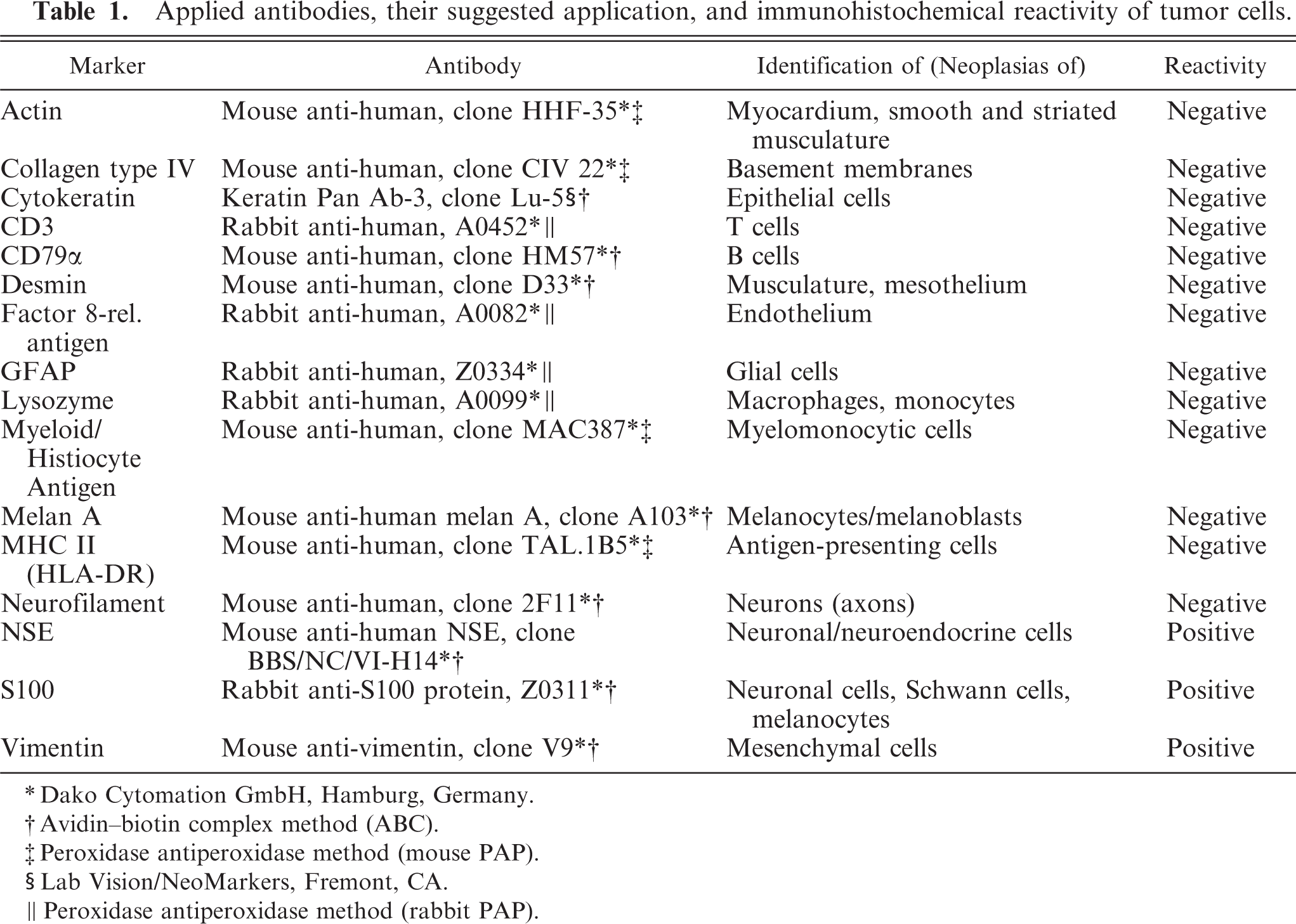
The enucleated eye was fixed in 10% neutral buffered formalin and processed routinely for histologic evaluation. Paraffin sections 3–5 µm thick were stained with HE, Goldner's trichrome stain, and periodic acid-Schiff (PAS). Immunohistochemical evaluation was performed using either the avidin–biotin complex method (Vectastatin ABC Kit, Vector Laboratories, Burlingame, CA, USA) or the peroxidase antiperoxidase (PAP) method (PAP mouse, Dianova GmbH, Hamburg, Germany; PAP rabbit, DAKO Cytomation GmbH, Hamburg, Germany). Applied primary antibodies are listed in Table 1.
Applied antibodies, their suggested application, and immunohistochemical reactivity of tumor cells.
∗Dako Cytomation GmbH, Hamburg, Germany.
†Avidin-biotin complex method (ABC).
‡Peroxidase antiperoxidase method (mouse PAP).
§Lab Vision/NeoMarkers, Fremont, CA.
∥Peroxidase antiperoxidase method (rabbit PAP).
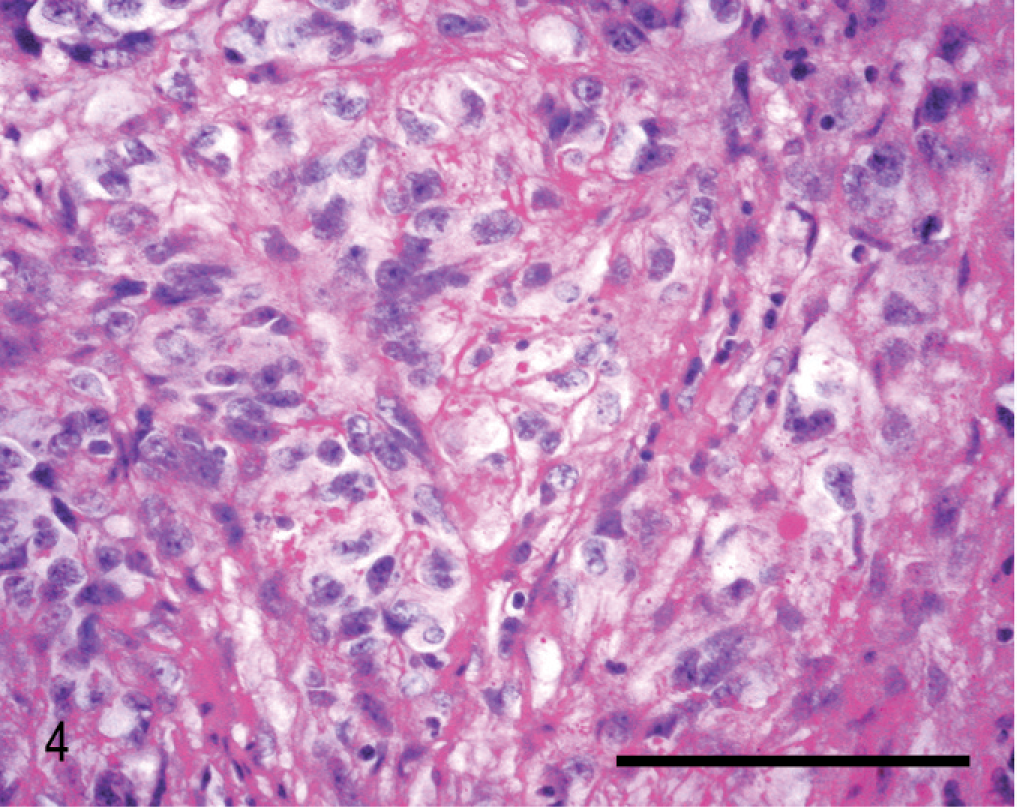
Histologically, almost the full thickness of the corneal stroma was infiltrated by an unencapsulated neoplasm composed of uniform, closely packed, large (up to 40 µm in maximal dimension), spindle-shaped cells. The tumor extended from the corneal center to the limbus (Fig. 2). Neoplastic cells were arranged in streams and bundles and separated by a moderate amount of fibrovascular stroma, admixed with dense aggregates of infiltrating CD3-positive lymphocytes. In some areas, CD79α-positive lymphocytes were arranged in follicle-like aggregates. Neoplastic cells had abundant homogeneous eosinophilic cytoplasm with indistinct cell borders. There was moderate anisocytosis and marked anisokaryosis. The nuclei were round to elongated with finely stippled chromatin and 1–3 prominent nucleoli. The mitotic rate was high (2–3 per high-power field). A few apoptotic cells and single multinucleated giant cells were scattered among the spindle cells (Fig. 3). Focally, the corneal epithelium was mildly hyperplastic. Single epithelial cells, especially in the center of the cornea, had hydropic degeneration; focal epithelial erosion was associated with superficial corneal infiltration by neutrophils. Descemet's membrane was not affected. Multifocally, PAS-positive, fibrillar to amorphic material was present between the tumor cells (Fig. 4). Collagenous fibers stained intensely green with Goldner's trichrome stain (Fig. 5).
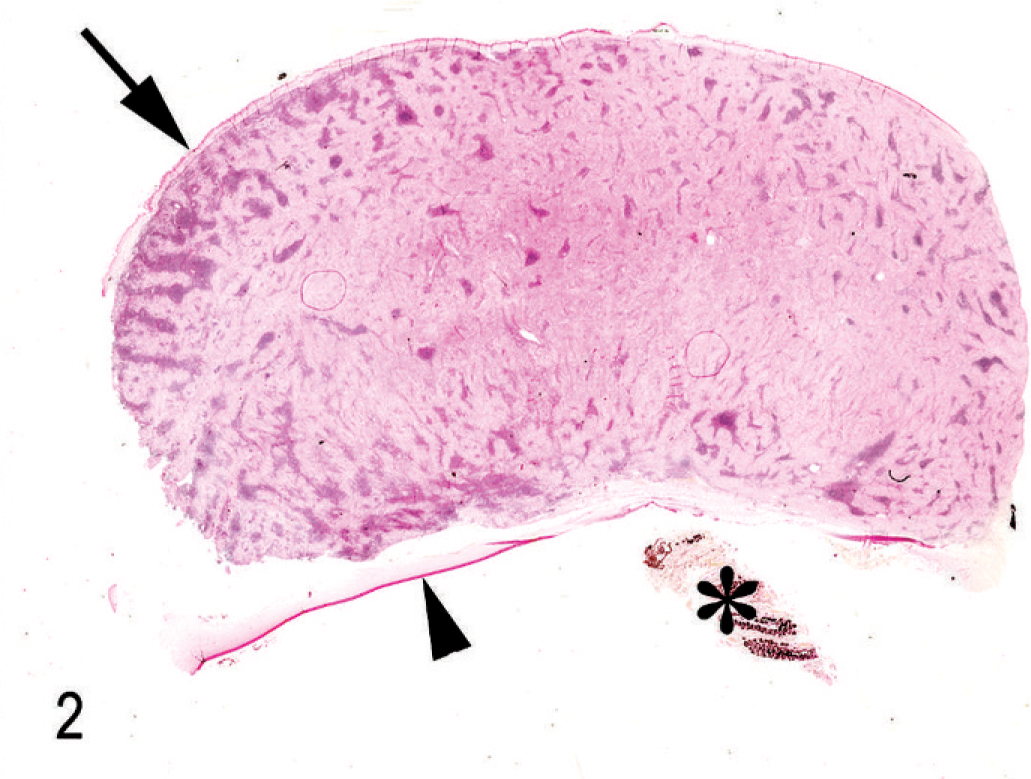
Cornea; horse. Subgross appearance of the corneal mass. Note the intact corneal epithelium (arrow), Descemets membrane (arrowhead) and remnants of the iris (asterisk). PAS method.
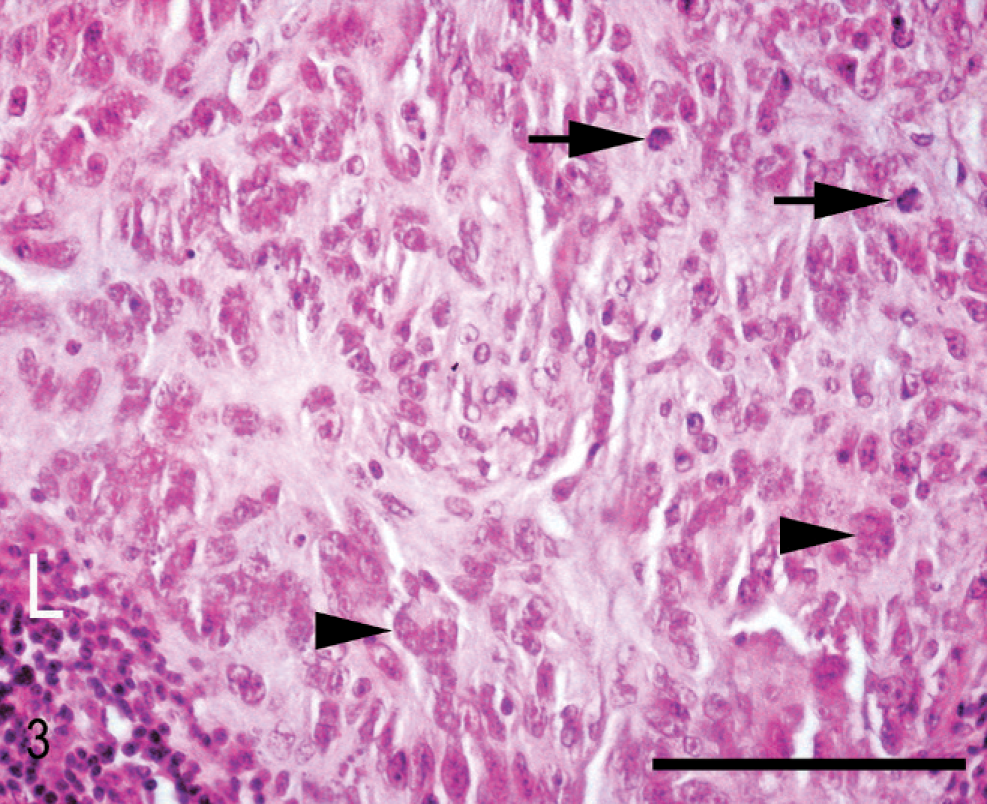
Cornea; horse. Neoplastic cells with mitotic figures (arrows) and multinucleated giant cells (arrowheads). Aggregates of lymphocytes (L) are interspersed in the tissue. HE stain. Bar = 100 µm.
Cornea; horse. Fibrillar, globular, or amorphous PAS-positive material between neoplastic cells. PAS method. Bar = 100 µm.
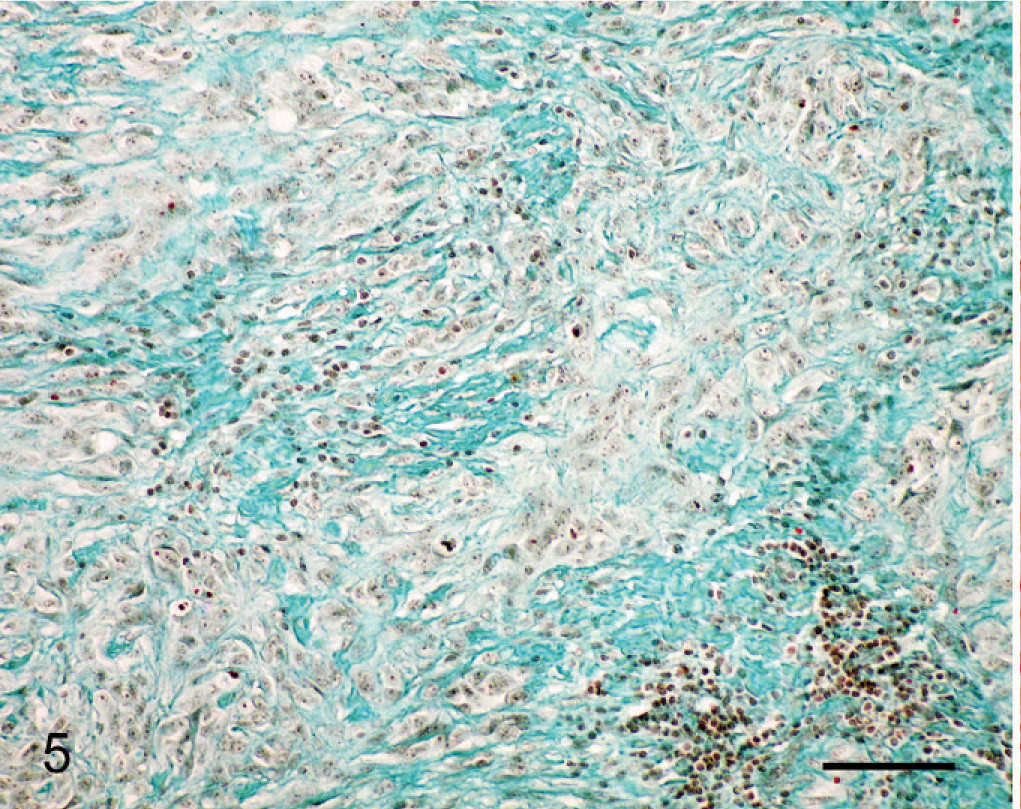
Cornea; horse. Collagen fibers in the stroma of the tumor. Goldner's trichrome stain. Bar = 100 µm.
Immunohistochemical details are shown in Table 1. The neoplastic cells were intensely positive for vimentin (Fig. 6) and negative for cytokeratin (Keratin Pan Ab-3), which was consistent with mesenchymal origin. Islands of cells expressed S100 protein (Fig. 7) and had diffuse cytoplasmic expression of neuron-specific enolase (NSE) (Fig. 8). Neoplastic cells were negative for von Willebrand factor–related antigen, neurofilaments, glial fibrillary acidic protein (GFAP), MHC-class II, MAC387, lysozyme and Melan A (MART-1). Collagen fibers were weakly positive for collagen type IV. No DNA of BPV type 1 or type 2 was amplified by the polymerase chain reaction (PCR). 19
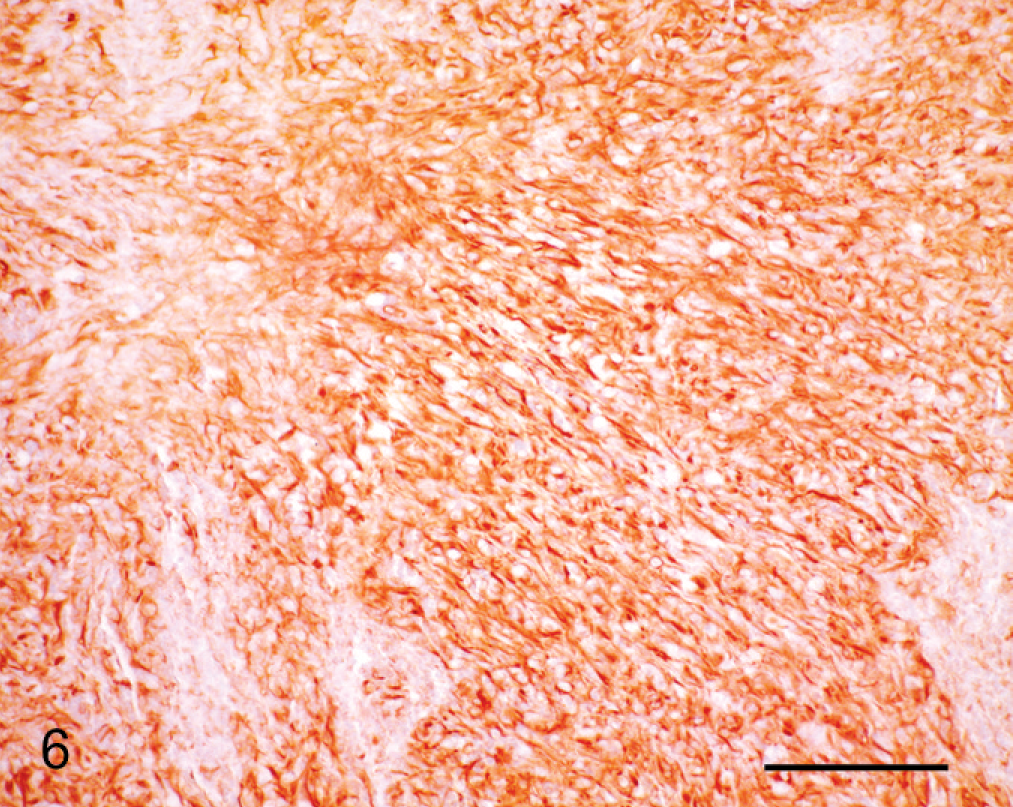
Cornea; horse. Neoplastic cells are arranged in bundles and express vimentin immunohistochemically. PAP method. Bar = 100 µm.
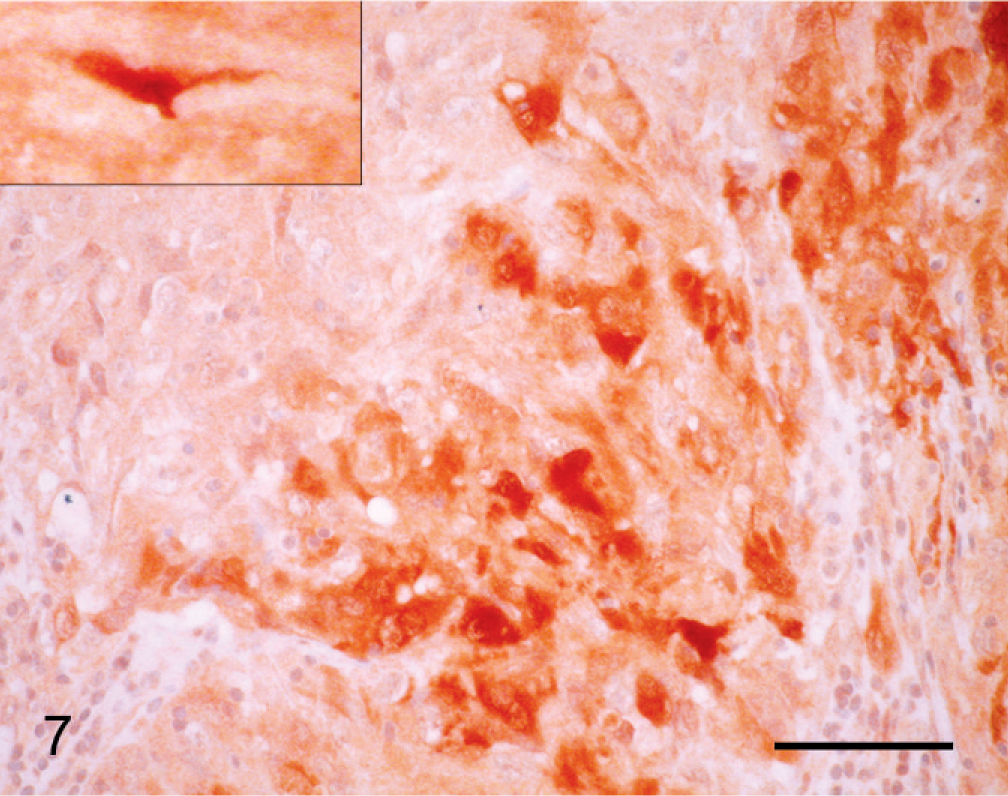
Cornea; horse. Individual and groups of neoplastic cells express S100 protein. Inset: Fibroblast of adjacent corneal stroma expresses S100 protein. PAP method. Bar = 100 µm.
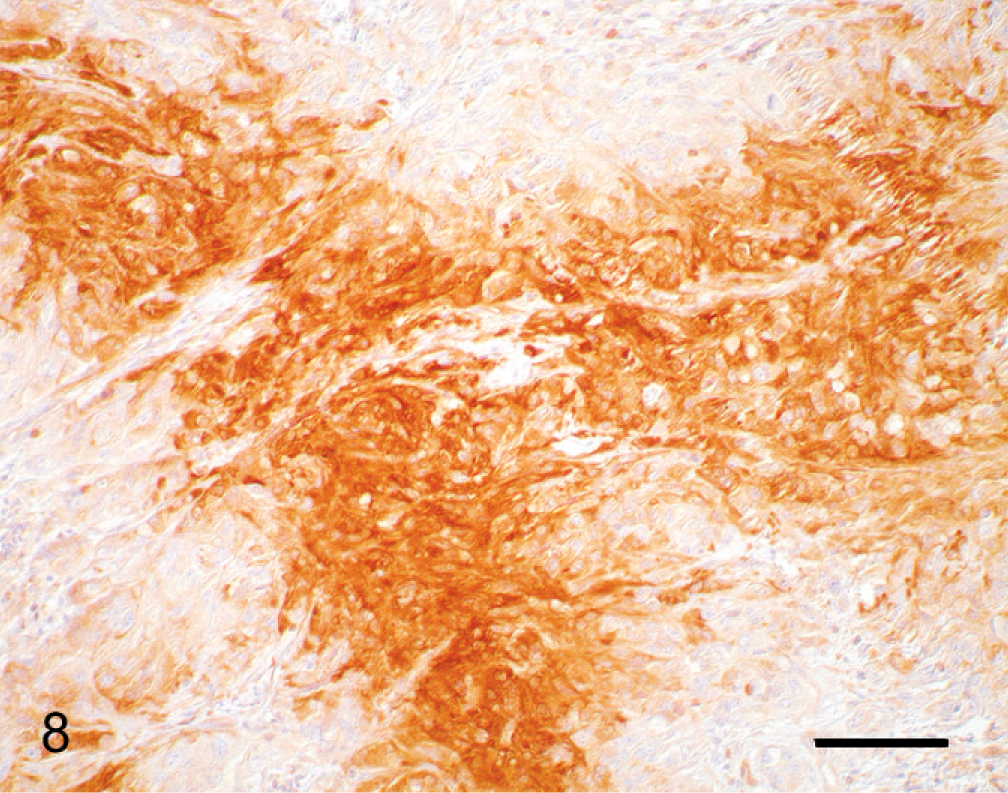
Cornea; horse. Neoplastic cells express NSE immunohistochemically. PAP method. Bar = 50 µm.
Histologic and immunohistochemical features of this corneal tumor were those of a malignant mesenchymal neoplasm. Because of its infiltrative growth and pleomorphism, the neoplasm was classified as a pleomorphic sarcoma. The differential diagnosis included fibrosarcoma and malignant peripheral nerve sheath tumor. The cell of origin may have been the keratocyte, a special type of fibrocyte. 17 Keratocytes produce collagen fibrils and intercellular matrix and, if injured, can transform into fibroblasts. 11 Corneal stromal fibroblasts produce extracellular matrix, especially collagen, which is highly condensed in Descemet's membrane. 17 In this tumor, collagen fibers and PAS-positive matrix were found between tumor cells.
Reports of primary mesenchymal corneal tumors are extremely rare in human beings or animals. Experimentally, corneal sarcomas were induced in opossums (Monodelphis domestica) by ultraviolet (UV) radiation. 5, 6, 8 Fibrosarcomas were most common, followed by malignant fibrous histiocytoma, hemangiosarcoma, and fibrosarcoma with concurrent squamous cell carcinoma; the preneoplastic lesions in these cases were corneal neovascularization and fibroplasia. 5, 6, 8 In UV irradiation–induced corneal fibrosarcomas of opossums, proliferating fibroblasts were proposed to originate from resident corneal stromal fibroblasts or from immigrated, potentially vessel-associated, mesenchymal cells. 5 The horse of this report had had a previous biopsy diagnosis of granulomatous keratitis with fibrosis. It is possible that the neoplasm arose in the inflamed corneal tissue.
Immunohistochemical expression of S100 protein and NSE by the neoplastic cells prompted consideration of peripheral nerve sheath tumor in the differential diagnosis. Fibroblasts of the corneal stroma adjacent to the neoplasm had cytoplasmic immunoreactivity for S100 protein (Fig. 7, inset). In contrast, corneal fibroblasts of control tissue (normal equine cornea) did not express S100 protein. Stromal fibroblasts that are activated by removal of the corneal epithelium express S100A4, but the protein is hardly detectable in clinically normal limbal or corneal fibroblasts. 16 Expression of S100 protein by adjacent stromal and neoplastic cells could be a reaction to neoplastic proliferation, to infiltration by lymphocytes, or to the secondary erosive keratitis. Epidemiologic studies support the hypothesis that chronic inflammation may be associated with an increased risk of cancer; carcinogenesis is a multistep process to which leukocytes and a network of signaling molecules may contribute. 9
The corneal stroma contains numerous myelinated sensory nerves that enter the subepithelial basement membrane and branch between epithelial cells with unsheathed ends. 12, 17 Neoplastic cells can originate from perineural fibroblasts or Schwann cells. 4 A periocular neurofibrosarcoma has been reported in a Thoroughbred mare 18 ; however, such neoplasms have not been documented within the corneal stroma. In the authors' opinion, because of the positivity for S100 protein and NSE, a malignant peripheral nerve sheath tumor is a relevant alternative diagnosis. Malignant peripheral nerve sheath tumors are spindle cell sarcomas closely associated with nerves. These tumors can originate from Schwann cells, perineural cells, fibroblasts, or any combination of these cells. 4
Papillomavirus infection was ruled out as a cause of the tumor in this horse because no fragments of the BPV genome were detected by PCR amplification and the lesion was not histologically consistent with equine sarcoid. Nevertheless, a prior infection or a traumatic insult could have initiated a chronic inflammatory process that may have resulted in tumor development. An actinic cause, similar to that in corneal sarcomas in the opossum, is likewise conceivable. 8
To the authors' knowledge, this is the first report of a malignant mesenchymal neoplasm of the equine cornea. Histologic and immunohistochemical features of the tumor were most consistent with a diagnosis of malignant peripheral nerve sheath tumor. Mesenchymal corneal neoplasms originating from keratocytes or perineural cells should be included in the differential diagnosis for ocular tumors.
