Abstract
Reports of primary nervous system tumors in wild raccoons are extremely rare. Olfactory tumors were diagnosed postmortem in 9 free-ranging raccoons from 4 contiguous counties in California and 1 raccoon from Oregon within a 26-month period between 2010 and 2012. We describe the geographic and temporal features of these 10 cases, including the laboratory diagnostic investigations and the neuropathologic, immunohistochemical, and ultrastructural characteristics of these tumors in the affected animals. All 9 raccoons from California were found within a localized geographic region of the San Francisco Bay Area (within a 44.13-km radius). The tight temporal and geographic clustering and consistent anatomic location in the olfactory system of tumor types not previously described in raccoons (malignant peripheral nerve sheath tumors and undifferentiated sarcomas) strongly suggest either a common cause or a precipitating factor leading to induction or potentiation of neuro-oncogenesis and so prompted an extensive diagnostic investigation to explore possible oncogenic infectious and/or toxic causes. By a consensus polymerase chain reaction strategy, a novel, recently reported polyomavirus called raccoon polyomavirus was identified in all 10 tumors but not in the normal brain tissue from the affected animals, suggesting that the virus might play a role in neuro-oncogenesis. In addition, expression of the viral protein T antigen was detected in all tumors containing the viral sequences. We discuss the potential role of raccoon polyomavirus as an oncogenic virus.
Keywords
Raccoons (Procyon lotor) are nocturnal omnivores native to, and widely distributed throughout, northern and central America. The high density of raccoons in urban and suburban areas reflects their robust adaptive ability. 51 Garbage is a major food source, and sewers are common as den sites; thus, the raccoon is an ideal sentinel for monitoring zoonotic diseases and toxicants in the environment. 3,4 Clinical neurologic signs in raccoons have been documented in infectious, 22,27,42 parasitic, 23,25,42 toxic, 16,19,42 and neoplastic diseases, 15,24,55 but reports of primary nervous system tumors in free-ranging raccoons are extremely rare. Only 1 astrocytoma was diagnosed in over 400 necropsies in 1 retrospective survey (1948–1977). 15 In subsequent reports, only an astrocytoma of the brainstem 24 and a renal lymphosarcoma with brain and leptomeningeal metastases were reported. 55
Within a period of 26 months between 2010 and 2012, 9 raccoons from 4 contiguous counties in Northern California were submitted for necropsy to the California Animal Health and Food Safety Laboratory System (CAHFS) or the Veterinary Medical Teaching Hospital, School of Veterinary Medicine, University of California, Davis, with pleomorphic tumors in the olfactory system (brain/olfactory bulb and/or olfactory nerve). Formalin-fixed tissue from a 10th raccoon, submitted to the Oregon State University Veterinary Diagnostic Laboratory (OSUVDL), was subsequently included in this study. The tight geographic and temporal clustering as well as the consistent anatomic location of the tumors prompted an extensive diagnostic investigation to explore possible oncogenic infectious and/or toxic causes. So far, a recently reported novel polyomavirus named raccoon polyomavirus (RacPyV), which was consistently associated with the tumor tissue in all 10 cases, is a likely candidate. 13 By whole genome nucleotide sequence alignment, RacPyV is in the genus Orthopolyomavirus and phylogenetically most closely related to the human Merkel cell polyomavirus (MCPyV). RacPyV genome organization is typical of known polyomaviruses, with a predicted early region encoding the T-antigen complex and late genes, including VPs1-3. Here we describe the geographic and temporal features of the 10 cases of raccoons with olfactory tumors, the diagnostic investigations, and neuropathologic, immunohistochemical, and ultrastructural characteristics of these tumors.
Materials and Methods
Case Material and Animal Location
Case databases of the CAHFS and the Pathology Service of the University of California–Davis Veterinary Medical Teaching Hospital were reviewed from January 2010 through May 2012 for all raccoons submitted for necropsy and those raccoons diagnosed with brain/olfactory tumors. The OSUVDL database was reviewed for similar data from July 2001 to December 2012. Relevant histories, including clinical signs and capture locations, were compiled for all raccoons diagnosed with brain tumors. The straight-line distances between tumor case locations were calculated using the proximity tools for ArcGIS 10 (ESRI, Redlands, CA).
Pathology and Immunohistochemistry
Raccoons had thorough postmortem examinations with selected tissues immersion fixed in 10% buffered neutral formalin, routinely embedded in paraffin, sectioned at 5 μm thick, and stained with hematoxylin and eosin (HE) for light microscopic evaluation. Slides of select tumor tissues were stained with Masson’s trichrome, Bielschowsky and Bodian stains, and Gordon and Sweet’s reticulin stains. Immunohistochemistry was done on selected tissues with antibodies to glial fibrillary acidic protein (GFAP; rabbit polyclonal, Dako, Carpinteria, CA, dilution 1:500), vimentin (clone 3B4, Dako, dilution 1:200), pancytokeratin (CK, clone Lu-5, Biocare Medical, Concord, CA, dilution 1:100), human von Willebrand factor VIII (rabbit polyclonal, Dako, dilution 1:2000) all with pretreatment with proteinase K, laminin (rabbit polyclonal PU078-UP, BioGenex, Fremont, CA, pretreatment: protease 37°C, 10 minutes, dilution 1:30), S-100 (rabbit polyclonal VP-276, Vector Labs, Burlingame, CA, dilution 1:400), alpha smooth muscle actin (α-SMA, clone 1A4, Biogenic Inc, Napa, CA, dilution 1:200), and CD20 (rabbit polyclonal, Lab Vision Corp., Fremont, CA, dilution 1:100) with no pretreatment, CD3 (rat monoclonal, clone CD3-12, Serotec Inc, Oxford, England, dilution 1:10), protein Ki-67 (clone MIB-1, Dako, dilution 1:50), synaptophysin (monoclonal, Dako, dilution 1:80), neuron-specific nuclear protein (Neu-N; clone A60, MAb377, Millipore Chemicon, Billerica, MA, dilution 1:500), Olig2 (rabbit polyclonal Ab9610, Millipore Chemicon, dilution 1:200), neuron-specific enolase (NSE; mouse monoclonal 18-0196, Invitrogen, Camarillo, CA, dilution 1:2000), pan-neurofilament (Pan-NF; mouse 18-0171, Zymed Inc, Oxnard, CA, dilution 1:100), neurofilament 200 (mouse monoclonal NCL-NF200, Novocastra Laboratories Ltd, Newcastle, England, dilution 1:50), chromogranin A (rabbit polyclonal NB 100-79914, Novus Biologicals, Littleton, CO, dilution 1:300), and desmin (mouse monoclonal D33 243M-16, Cell Marque, Rocklin, CA, dilution 1:100) all with antigen retrieval: HIER citrate buffer, following procedures as previously described. 14,35,44 In addition, we immunolabeled tumor sections from cases No. 1–8 with a mouse monoclonal antibody for the T antigen of simian vacuolating virus 40 (SV40), which cross-reacts with the T antigen of JCV (Clone PAb 416 -Ab2-, Millipore-Calbiochem, Billerica, MA; 1:100 dilution, antigen retrieval: HIER citrate buffer), also tested following a procedure previously described. 49 Canine distemper virus (CDV) immunohistochemistry was performed as previously reported on sections of the cerebellum and/or medulla oblongata from cases No. 3, 5, 7, and 8 using a mouse monoclonal anti-CDV nucleoprotein antibody (CDV-NP, VMRD Inc, Pullman, WA) at a dilution of 1:3000. 60 All the immunohistochemistry was performed using the avidin–biotin–peroxidase method according to manufacturer’s instructions (Vectastain Elite Kit, Vector Laboratories, Burlingame, CA). Positive immunoreactivity of all these antibodies was visualized using the chromogen 3-amino-9-ethylcarbazole (AEC K4001, Dako). For detection of West Nile virus (WNV), tissue sections from heart and kidney of case No. 4 were treated with a rabbit polyclonal anti-WNV primary antibody (BioReliance, Rockville, MD) at 1:2000 and results visualized with Nova Red chromogen (ImmPACT NovaRED, Peroxidase Substrate, Vector Lab, Burlingame, CA) following a procedure previously described. 48 Appropriate positive and negative controls were used for all the aforementioned antibodies. The proliferative index from Ki-67 immunoreactivity was expressed as percentage of the number of positively stained nuclei in a total of 2200 to 2900 tumor cells pooled from 3 fields examined at 600× from areas in which the nuclear immunoreactivity was most dense.
Laser Capture Microdissection JCV and Human Cytomegalovirus Polymerase Chain Reaction
Five-micrometer-thick formalin-fixed paraffin-embedded tumor sections from cases No. 1, 3, and 4 placed on glass slides were used for JCV polymerase chain reaction (PCR) amplification. Each section was deparaffinized in xylene, rehydrated through alcohol up to water, and air-dried for 24 hours. Afterward, laser capture microdissection (LCM) was performed under direct microscopic visualization by laser heating of a thermoplastic film mounted on optically transparent CapSure HS LCM caps (Arcturus Engineering Inc, Mountain View, CA). The PixCell II LCM System (Arcturus) was set to the following parameters: 15-μm spot size, 40-mW power, 3.0-ms duration. A total of 100 tumor cells were captured by focal melting of the membrane through pulse activation of a carbon dioxide laser. DNA isolation was performed using the Arcturus PicoPure DNA extraction kit according to the manufacturer’s instructions. DNA amplification was performed using a JCV early-region (T antigen) set of primers: Pep1 and Pep2 (nucleotides 4255-4274 and 4408-4427, respectively). Amplification was carried out in 50-μl reaction media containing 200 ng of template in Failsafe Buffer B, Failsafe Taq polymerase (Epicenter), and 500 nM of each primer. After denaturation at 94°C for 5 minutes, 35 cycles of denaturation at 94°C for 45 seconds, annealing for 45 seconds, and extension at 72°C for 45 seconds, a final extension step of 7 minutes at 72°C was allowed. Annealing temperatures were 55°C for Pep primers. Nucleotides homologous to the following JCV-specific sequences were used as probes: T-antigen probe (nucleotides 4303-4327). A plasmid containing the JCV early coding sequence was used as a positive control. For amplification of DNA from the housekeeping gene, GAPDH, the following primers were used: 5' TTC TCC CCATTC CGT CTT CC 3' and 3' GTA CAT GGT ATT CAC CAC CC 5'. 49 For human cytomegalovirus (HCMV), reactions were performed using Phusion polymerase (New England Biolabs), Phusion HF buffer (New England Biolabs), 250-µM dNTPs (Invitrogen), 50-µM primers (Invitrogen, USA), and DEPC-treated water (Ambion). Consensus-degenerate hybrid oligonucleotide primer PCR primers, namely SLYP1A and GDTDIB, were used as previously reported. 56 PCR conditions were as follows: 98°C for 0:10, 30 cycles (98°C for 0:10, 61°C for 0:40, 72°C for 0:30), 72°C for 10:00, and a 4°C hold.
Toxicology
Analyses for metals were performed on fresh liver samples from cases No. 1, 3–5, and 8 and formalin-fixed samples of liver, kidney, and brain from case No. 5. Samples were analyzed for lead, manganese, cadmium, copper, iron, zinc, molybdenum, arsenic, mercury, and selenium following procedures previously described. 41,62 All metal concentrations were estimated on a wet weight basis.
Fresh liver tissue from case No. 4 was analyzed for carbamate and organophosphorus insecticides. Carbamate analysis was performed by liquid chromatography tandem mass spectrometry and included aldicarb sulfoxide, bendiocarb, carbaryl, carbofuran, 3-hydroxycarbofuran, methiocarb, methomyl, mexacarbate, oxamyl, and propoxur. The limit of quantitation was 0.10 mg/kg (wet weight). Organophosphorus insecticides were analyzed by gas chromatography with flame photometric detection and included acephate, azinphos methyl, carbophenothion, chlorfenvinphos, chlorpyrifos, coumaphos, crotoxyphos, crufomate, DDVP, DEF, demeton-O, demeton-P, diazinon, dicrotophos, dimethoate, dioxathion, disulfoton, EPN, ethion, ethoprop, famphur, fenamiphos, fensulfothion, fenthion, fonofos, isofenphos, malathion, methamidophos, methidathion, methyl parathion, mevinphos, monocrotophos, naled, parathion, phorate, phosalone, phosmet, phosphamidon, profenophos, propetamphos, ronnel, terbufos, tetrachlorvinphos, and triazophos. The limit of quantitation was 0.10 mg/kg (wet weight).
Liver samples from cases No. 5 and 10 were submitted for screening and quantification of 7 anticoagulant rodenticides (ARs) by liquid chromatography-tandem mass spectrometry for screening presence of ARs and high-performance liquid chromatography to quantitate positive samples. The AR compounds tested for included first-generation ARs (warfarin, diphacinone, chlorophacinone, and coumachlor) and second-generation ARs (brodifacoum, bromadiolone, and difethialone). The reporting limits (wet weight) were 0.01 mg/kg for brodifacoum; 0.05 mg/kg for warfarin, bromadiolone, and coumachlor; and 0.25 mg/kg for diphacinone, chlorophacinone, and difethialone. Detectable AR compound concentrations that were below quantitation limits were labeled as “trace” concentrations. All toxicology testing was done following CAHFS standardized operating procedures.
Electron Microscopy
Selected pieces of formalin-fixed brain tumor tissue from cases No. 1, 3, 4, 5, and 8 were postfixed in 2.0% glutaraldehyde and then routinely processed and embedded in epoxy resin (Eponate-12 epoxy resin Ted Pella Inc, Redding, CA), and selected thick sections were stained with toluidine blue as previously described. 67 Ultrathin sections from selected areas in the tumors were examined using a Zeiss 906E transmission electron microscope.
Feces from case No. 5 were suspended in distilled water, centrifuged, negatively stained with 2% phosphotungstate, and examined with an electron microscope (direct electron microscopy), as previously described. 68
Ancillary diagnostic testing
Fresh/frozen brain tissue samples from cases No. 1–3, 5, 6, and 8, including right and left cerebral cortex, brainstem, hippocampus, and cerebellum, were tested by the California Department of Public Health (cases No. 1–3, 5, and 8), or OSUVDL (case No. 6) for rabies virus (Lyssavirus, Rhabdoviridae) antigen detection by a direct fluorescent antibody test (FAT). Bacterial aerobic cultures (sheep blood agar, incubation at 35°–39°C in 5%–10% CO2 for 48 hours) were conducted on lung and liver tissue from cases No. 1–5 and 8 as well as brain swabs from cases No. 1, 2, and 8, and lymph node and meningeal swab from case No. 1. Salmonella sp. infection was tested by PCR on samples of intestine/feces from cases No. 1, 4, 5, and 8. Fungal culture (Inhibitory Mold Agar, incubated aerobically at 21°–25°C for 4 weeks) was performed on a brain swab from case No. 2.
Results
Case Material and Animal Location
Within a period of 26 months between late March 2010 and early May 2012, a total of 52 raccoon carcasses were submitted to CAHFS or the Pathology Service for full necropsy. Nine of these raccoons (cases No. 1–5 and 7–10) had tumors involving the nasal cavity, olfactory bulb, frontal lobe of the brain, and/or olfactory nerve, representing 17.3% (9 of 52) of all raccoon submissions during this period. A 10th case (case No. 6) was necropsied at the OSUVDL in March 2011, with tumor samples submitted to CAHFS for further testing. Between July 2001 and December 2012, a total of 144 raccoons were necropsied at OSUVLD, and no other brain tumors except that of case No. 6 were detected, representing 0.69% of the raccoons. All 10 raccoons in this series were free-ranging adults; 9 (cases No. 1–5 and 7–10) were in an urban/suburban environment within 4 contiguous counties in the San Francisco Bay Area in California (Marin, Contra Costa, San Francisco, and Sonoma), while 1 (case No. 6) was an isolated case from coastal Oregon (Curry County, approximately 650 km north) (Table 1). All 9 California raccoons were found in a localized geographical region with a radius of 44.13 km, and 5 of these 9 cases were found close together in a circular area of a 4.65-km radius (Fig. 1). Eight of the California raccoons (cases No. 1–5 and 8–10) had neurologic signs and were captured and subsequently housed at a local rehabilitation center (WildCare, San Rafael, CA). The other California raccoon (case No. 7) was found dead on a road in Contra Costa County.
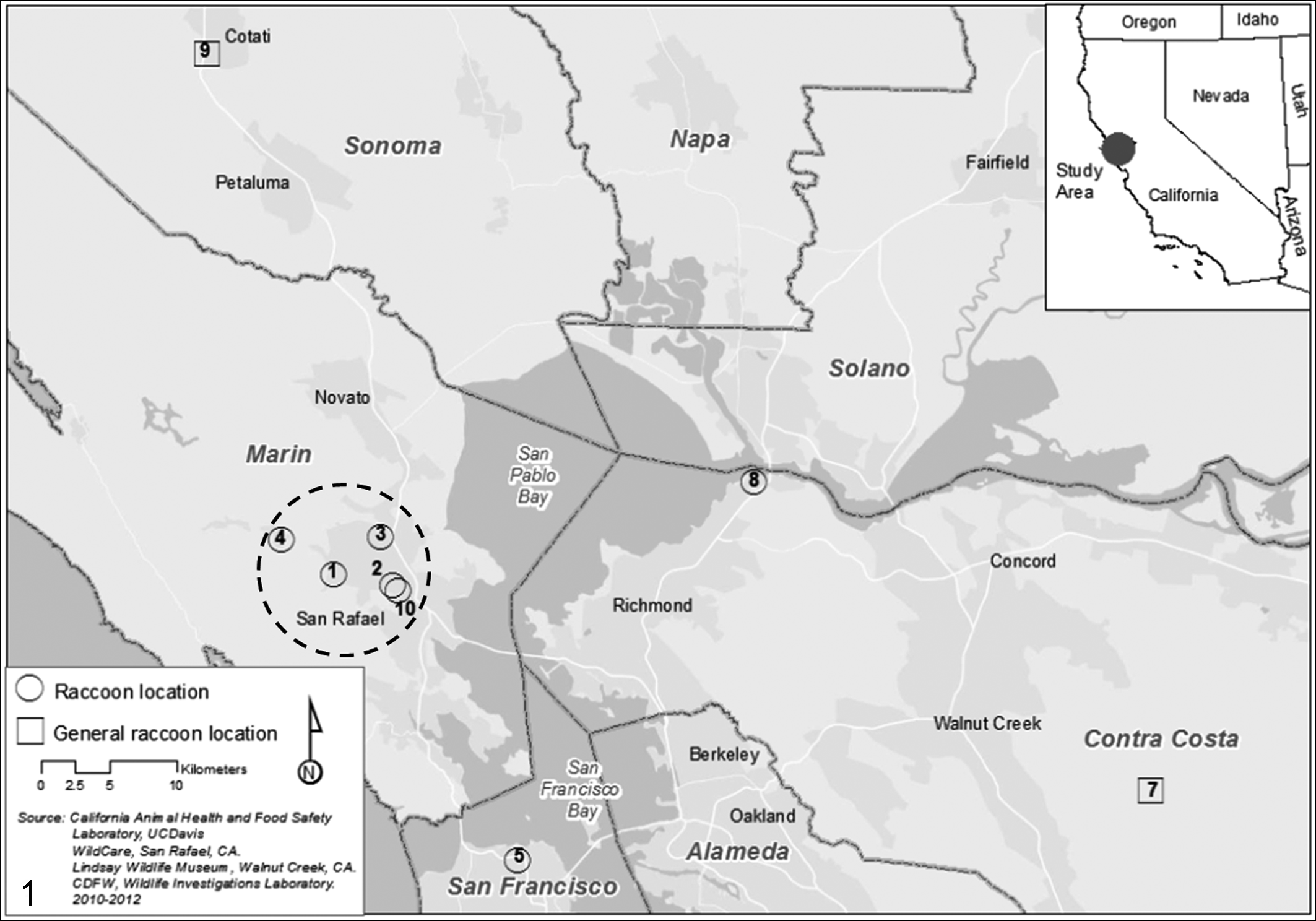
Map. San Francisco Bay Area. Capture locations of raccoons with olfactory and brain tumors in the northern San Francisco Bay Area, California. Open circles represent exact capture locations, while open squares represent the general area where 2 raccoons were found. Numbers indicate case numbers as in Table 1. Five of the 9 cases were found in Marin County within a circular area of a 4.65-km radius (encircled). The maximum distance between 2 locations (open squares) is 88.26 km, indicating marked geographical clustering. ArcGIS 10, Esri. Courtesy of Lora Konde.
Capture Location and Sex of Affected Raccoons and Anatomic Location and Histologic Classification of the Olfactory Tumors.
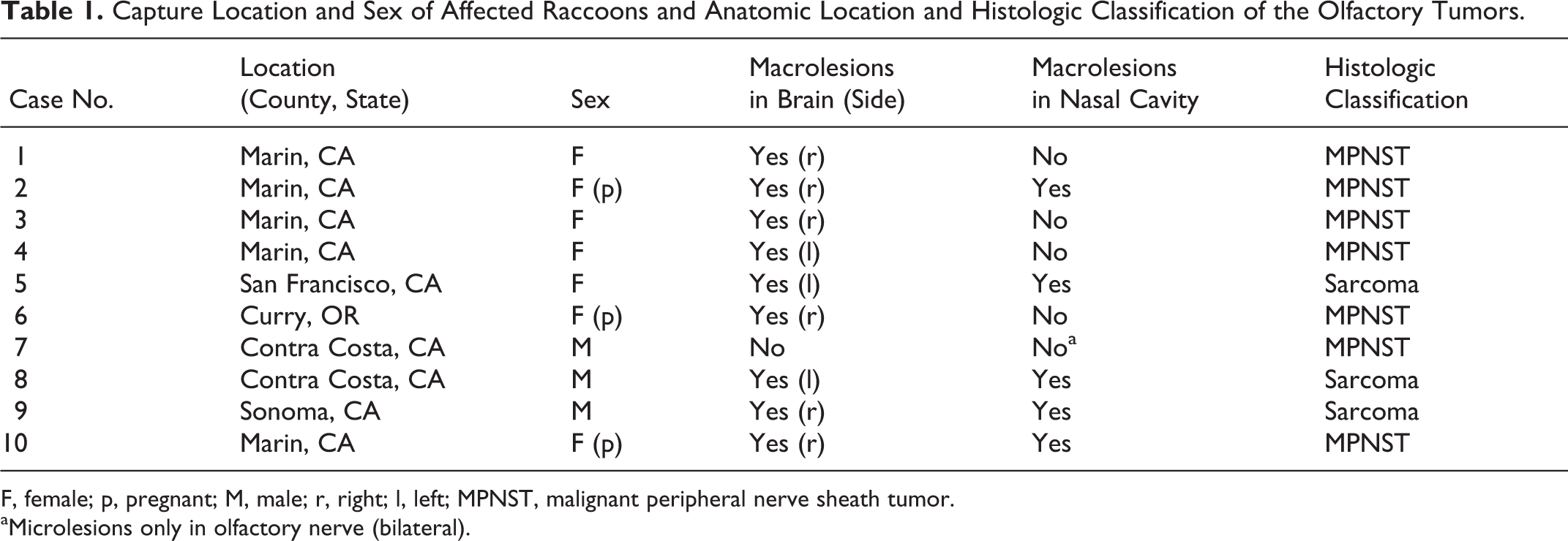
F, female; p, pregnant; M, male; r, right; l, left; MPNST, malignant peripheral nerve sheath tumor.
aMicrolesions only in olfactory nerve (bilateral).
Nine of the 10 raccoons with tumors (8 from California and 1 from Oregon) were described by the local residents as unnaturally docile and fearless of people during daylight hours. Severity of clinical signs progressed to ataxia, blindness, lethargy, and/or circling, and all 9 were euthanized between 2 and 33 days postcapture due to poor prognosis.
Gross Pathology
Nine raccoons had single unilateral space-occupying intracranial masses in the region of the olfactory bulb and frontal lobe of 1 cerebral hemisphere (Table 1, Fig. 2a) that were irregularly shaped and involved the rostral aspect of the cribriform plate of the ethmoid bone. On transverse section, each intracranial mass was fairly sharply demarcated and varied in size up to 2.7 cm on cross-sectional area of the brain; each was also soft and yellow to tan resembling nervous tissue with areas of whitish yellow necrosis and hemorrhage, sometimes with small fluid-filled cavities (Fig. 2b). Associated left- or right-sided brain midline shifts with contralateral mass effects were seen in most cases. Five of these 9 cases (cases No. 2, 5, and 8–10) also had contiguous solid mass lesions within the caudal nasal cavity (Fig. 3). One case (No. 7) had no gross lesions in either location.
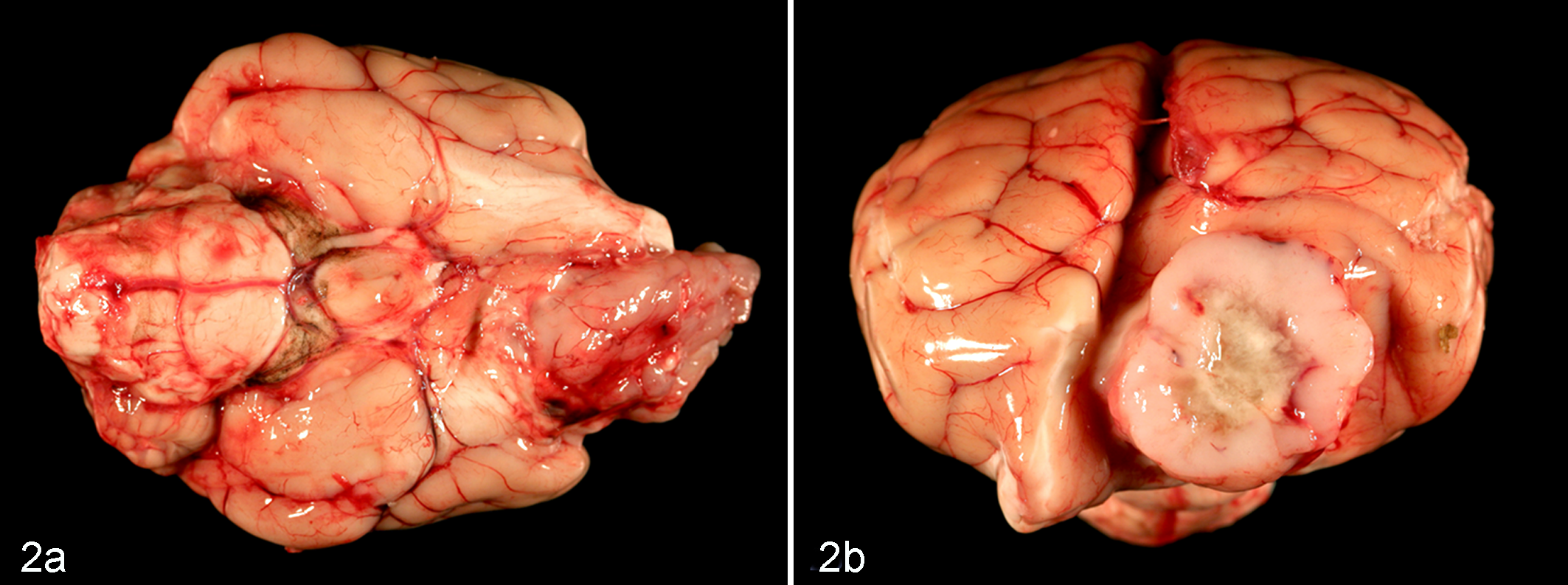
Brain; raccoon, case No. 4. (a) Ventral view. The malignant peripheral nerve sheath tumor (MPNST) expands the left olfactory bulb and tract unilaterally. (b) Transverse section through the frontal lobe of the brain through this MPNST. The tumor is 2.0 × 2.7 cm on cross section and has a focal, irregular, well-demarcated central area of necrosis and hemorrhage. Note the mass effect with right-sided midline displacement.
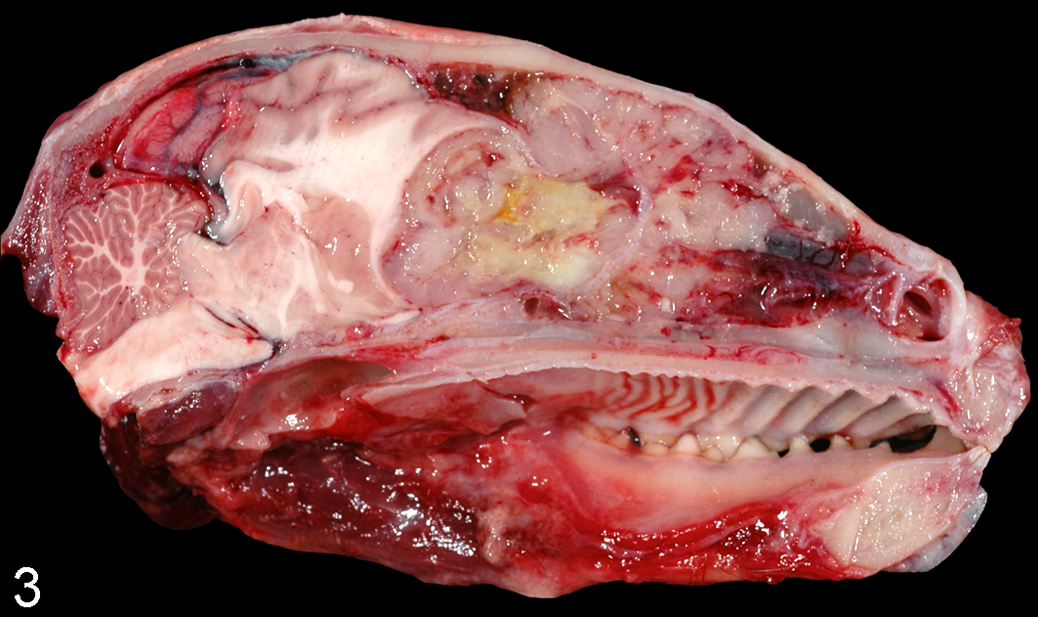
Head; raccoon, case No. 10. Right side—parasagittal section. A large intranasal malignant peripheral nerve sheath tumor (MPNST) occupies most of the nasal cavity and expands the cribriform plate, the right olfactory lobe, tract, and frontal lobe of the cerebral hemisphere. Caudally, the brain–tumor interface is well demarcated, indicative of an expansile pattern of growth. Areas of necrosis and hemorrhages occur in the intracranial and intranasal locations of the tumor. There is caudal herniation of the cerebellar vermis into the foramen magnum.
Histopathology and Immunohistochemistry
These 10 tumors exhibited some diversity of morphologic types and patterns within the nasal cavity and intracranially. The smallest tumors were found serendipitously in case No. 7 as multiple sites seen microscopically within some of the olfactory nerve branches just rostral to their entry through the cribriform plate. These proliferating spindloid cells, with slim elongate nuclei and bipolar tapering cytoplasm, were focally and bilaterally expanding the olfactory nerves (Fig. 4a). Clusters of these cells formed moderately cellular intraneural nodules that were well demarcated from surrounding tissue and had an individual pattern of pericellular reticulin staining also duplicated by immunoreactivity for laminin (Figs. 4b, 4c). No mitotic figures were observed, and no proliferating cells were detected with MIB-1 immunolabeling. The tumor cells forming these nodules were cytologically benign but focally infiltrated within olfactory nerve branches caudally through the cribriform plate into the cranial cavity. This tumor was diagnosed as a probable peripheral nerve sheath tumor (PNST) arising within the olfactory nerves but of unknown histogenesis.
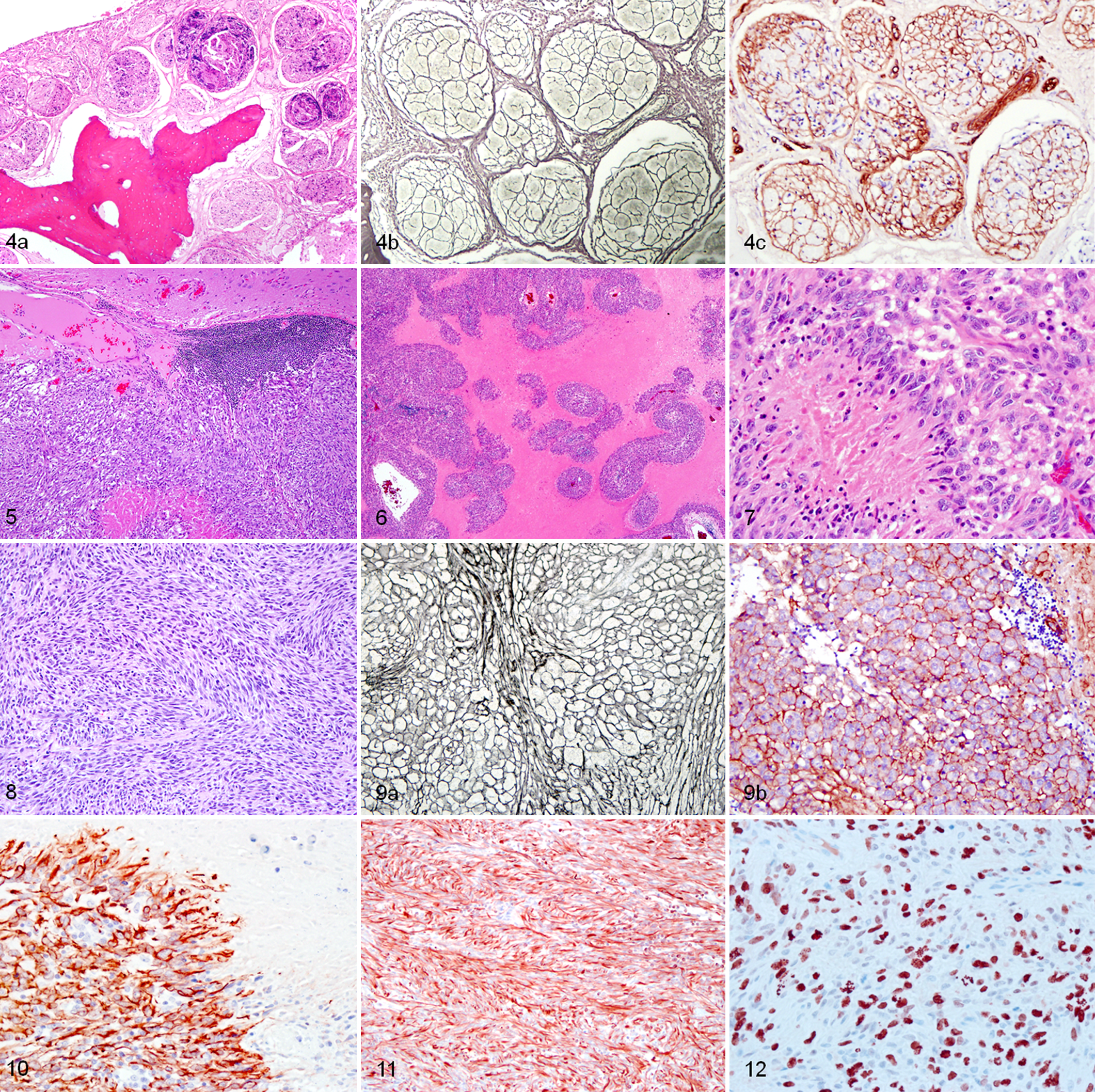
Nasal cavity; raccoon, case No. 7. Transverse sections. (a) Multiple well-demarcated intraneural nodular tumor proliferations of the olfactory nerve branches expand the nasal submucosa. HE. (b) Individual or small groups of neoplastic cells within the nodules are surrounded by an extracellular reticulin-positive meshwork. Gordon and Sweet’s reticulin stain. (c) Note the corresponding strong uniform extracellular immunoreactivity around each neoplastic cell with anti-laminin antibody. Immunohistochemistry with hematoxylin counterstain.
Tumors from 6 cases (Nos. 1–4, 6, and 10) were all similar solid intracranial tumors with a large intranasal component in cases No. 2, 4, and 10, with a sharply demarcated border at the tumor-brain interface (Fig. 5). Intracranially, they had a distinctive pattern of extensive focal or serpentine necrosis (Fig. 6) sometimes bordered by poorly defined cell palisading (Fig. 7). The spindloid tumor cells were arranged in extensive streaming bundles or swirling patterns and had a bipolar thin wispy cytoplasm and an elongate nucleus (Fig. 8), which was sometimes uniform but in other cases exhibited marked nuclear atypia. In multifocal patchy areas, individual or small groups of neoplastic cells were wrapped in a well-defined reticulin network around their cytoplasmic border that was immunoreactive for laminin (Fig. 9a, 9b) (cells in case No. 3 were supported by a reticulin meshwork that was negative for laminin and the intracranial component of case No. 2 was also negative for laminin). In all 6 cases, tumor cells were strongly immunoreactive with GFAP (Fig. 10) and vimentin (cases No. 1, 2 and 3 but not done in Nos. 4, 6, and 10) (Fig. 11) and showed variably (generally weak) immunoreactivity with S-100. They were uniformly negative for α-SMA, synaptophysin, Neu-N, Olig2, and CK. Mitotic figures were prominent in cases with nuclear atypia, and the MIB-1-derived proliferative index was up to 20% (Fig. 12). Case No. 4 also had similar well-demarcated multifocal sites of spindloid cells with similar characteristics proliferating in the olfactory nerve of the left nasal cavity and contiguous with the intracranial tumor. Based on these findings, these tumors were classified as probable malignant PNSTs (MPNSTs).
Undifferentiated sarcoma; raccoon, case No. 5. These images illustrate parasagittal sections of the ethmoid turbinates with the ethmoid bones (EB) lined by the respiratory and olfactory mucosa in the nasal cavity (NC). The nasal planum is located to the left and the cribriform plate and olfactory bulb to the right. (a) In the lamina propria, rostral branches of the normal olfactory nerve (arrowheads) transition caudally into nerve bundles that are hypercellular due to multifocal intraneural neoplastic spindle cell proliferation (arrows). HE. (b) Both normal and neoplastic foci in the olfactory nerve are strongly immunoreactive to GFAP and (c) S-100 antibodies. Immunohistochemistry with hematoxylin counterstain.
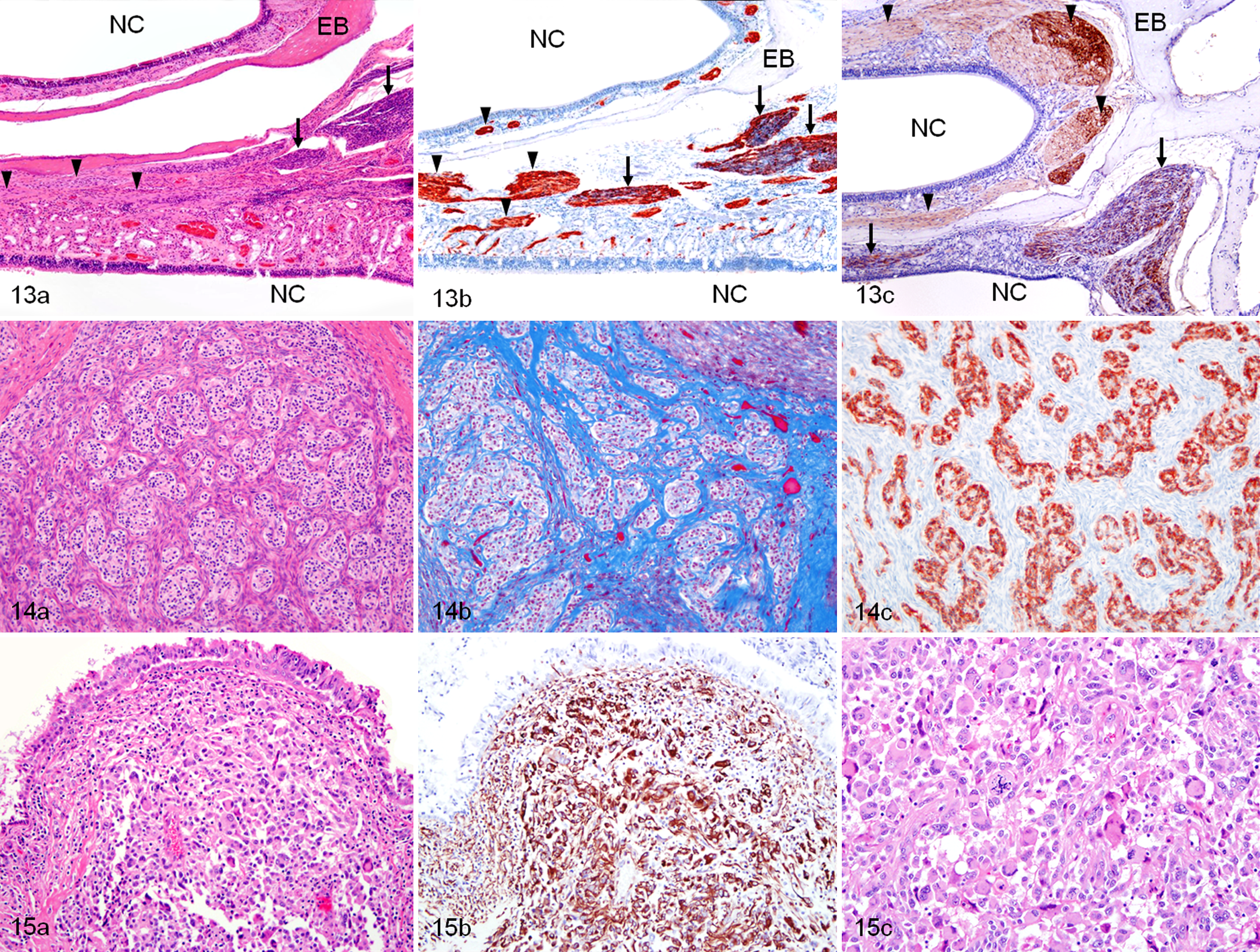
Case No. 5 was an anomalous tumor in that, although it exhibited a typical nasal component involving the olfactory nerves, it also had a unique intracranial component consisting of several different histologic patterns of unidentified cells embedded in collagenous spindloid cells. In the nasal cavity, normal olfactory nerve branches had multifocal intraneural transitional areas thickened with a neoplastic spindle cell proliferation (Fig. 13a) that was strongly immunoreactive for GFAP (Fig. 13b) and S-100 (Fig. 13c). Caudally, there was a uniform tumor cell thickening of these nerves, which penetrated intracranially through the cribriform plate. The atypical clusters or islands of unidentified cells embedded in a collagenous fibrous stroma (Fig. 14a, 14b) were strongly immunoreactive for GFAP (Fig. 14c). These cellular islands were Olig2, synaptophysin, NSE, chromogranin A, von Willebrand factor VIII, and laminin immunonegative. Bielschowsky stain, Pan-NF, and NF-200 immunolabeling did not reveal any neuronal processes associated with these cells. Further caudally there was a monomorphic population of spindloid cells with areas of classical serpentine necrosis but which were surprisingly GFAP immunonegative, but were invested with an individual reticulin pattern that was also laminin immunoreactive. This intracranial tumor was classified as a pleomorphic sarcoma with focal differentiation of unknown cell type, although the intranasal component was consistent with a MPNST.
Case No. 8 had both substantial intracranial and intranasal tumor cell invasion. In both locations, tumor cells were extremely pleomorphic, with marked nuclear atypia and a large amount of cytoplasm, often multinucleate, and at their borders were highly infiltrative (Fig. 15a). The cells were strongly immunoreactive for vimentin (Fig. 15b) and weakly immunoreactive for GFAP but had a minimal reticulin fibril component and were also immunonegative for laminin, CK, desmin, and α-SMA. There were prominent normal and abnormal mitotic figures (Fig. 15c), and the proliferative index was up to 15%. This tumor was classified as an anaplastic sarcoma of unknown origin.
In case No. 9, the intracranial tumor had gross and microscopic similarities to the MPNSTs except that the elongate cells were vimentin immunoreactive but GFAP and CK immunonegative, and their reticulin fibril component was minimal. This tumor was classified as a spindle cell sarcoma.
Infiltrates of moderate numbers of lymphocytes with fewer plasma cells, eosinophils, and Mott cells were distributed multifocally within and around the periphery of the tumors, usually in perivascular locations. Immunohistochemistry demonstrated approximately equal numbers of B cells (CD20) and T cells (CD3).
Electron Microscopy
Ultrastructural features of the brain tumors of cases No. 1, 3, 4, 5, and 8 were similar: tumors were densely cellular, and closely opposed cells had a high nuclear:cytoplasmic ratio. Large pleomorphic nuclei had clumped and marginated chromatin often containing amorphous inclusions (case No. 1). No virus particles were evident in these inclusions or in any other cells in the tumors from the 5 animals examined ultrastructurally. Cytoplasm of occasional cells had patchy, randomly distributed, loose arrays of intermediate filaments, abundant mitochondria, and rough endoplasmic reticulum. In some cases, subgroups of tumor cells were completely or partially surrounded by discontinuous lamellar aggregates of amorphous electron-dense extracellular material. Necrotic foci had abundant extracellular fibrin and lipid (Fig. 16).
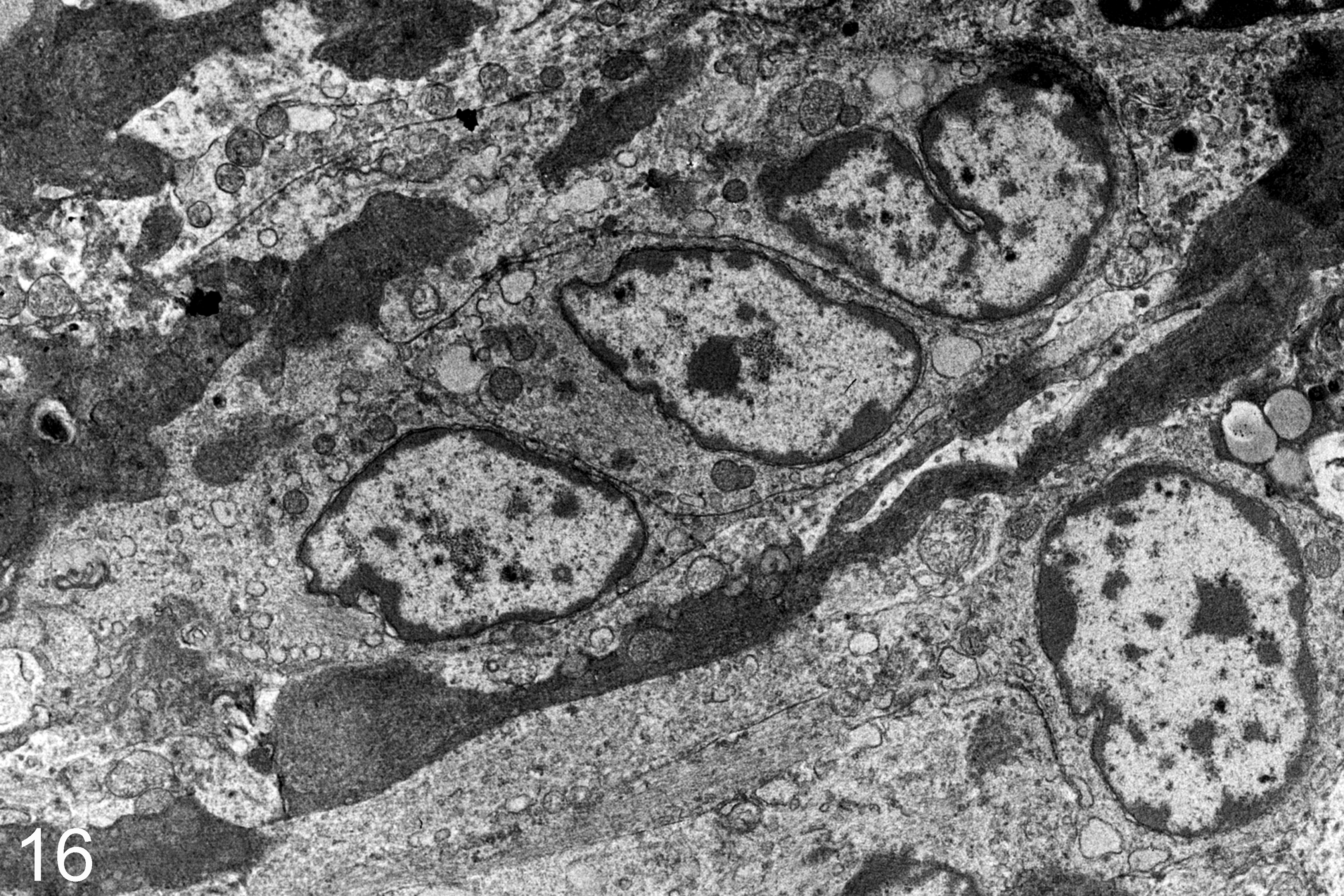
Olfactory tumor; raccoon, case No. 3. Variable pleomorphic spindle-shaped cells with moderate amounts of cytoplasm. Nuclei are pleomorphic oval to reniform with occasional indentations of the nuclear membrane and distinct nucleoli. Extracellular matrix is electron dense and separates groups and clusters of cells.
Virology
Specific PCR primers designed to amplify the gene encoding the T antigen of JCV and degenerate primers previously reported to amplify human and multiple animal cytomegaloviruses did not amplify any product from raccoon tumor tissue (LCM). No expression of JCV T antigen was found in any of the tumors by immunohistochemistry. CDV (cases No. 1–4) and WNV (case No. 4) antigens were not detected in sections of either cerebellum/medulla oblongata or heart and kidney, respectively.
Toxicology
None of the organophosphorus or carbamate insecticides were detected in the liver from case No. 4. In case No. 5, the liver contained AR 0.36 mg/kg of brodifacoum and trace amounts of bromadiolone (below 0.05 mg/kg) and diphacinone (below 0.25 mg/kg). The liver of case No. 10 contained 1.3 mg/kg of brodifacoum. In case No. 5, lead was detected in toxic concentrations in the fresh liver (29 mg/kg) and formalin-fixed liver (26 mg/kg), kidney (8.1 mg/kg), and brain (3.7 mg/kg). All other tissues contained metals in concentrations not considered toxicologically significant.
Ancillary Test Results
Bacterial aerobic and fungal cultures yielded either negative or unremarkable results, and Salmonella sp. was not detected in feces by PCR. All brains tested for rabies were negative by FAT.
Discussion
This article describes the unique occurrence of 10 spontaneous cases of either intranasal and intracranial olfactory MPNST and/or undifferentiated sarcomas in free-ranging raccoons. Nine of the 10 raccoons in this series were either captured (n = 8) or found dead (n = 1) in a small geographic region (44.13-km radius) in Northern California within a 26-month period from 2010 to 2012. The temporal and geographic clustering of a tumor type not previously described in raccoons strongly suggests either a common cause or a precipitating factor leading to induction or potentiation of neuro-oncogenesis. Potential factors that may contribute to such neuro-oncogenic mechanisms include exposure to environmental toxicants or ionizing radiation, a small genetic pool with genetic inheritance, or infection with oncogenic and/or immunosuppressive viruses, many of which were explored in the diagnostic investigations of these cases. The consistent anatomic location in the olfactory system suggests that exposure or transmission was via the nasal/upper respiratory tract or olfactory mucosa.
Viruses that have been associated with human brain tumors include the polyomavirus JCV and HCMV (herpesvirus). 5,9–11,30,33,49,54,59 Glioblastoma and other astrocytic, glial-derived primary brain tumors have been reproduced experimentally in laboratory rodents (Sprague Dawley rats) and nonhuman primates (new world owl and squirrel monkeys) after intracranial inoculation of JCV. In these studies, large T antigen was demonstrated in neoplastic cells, and the JCV genome was integrated into the cellular DNA, indicating a clonal origin for the tumors. 36,37,43,46,64 MPNSTs developed after direct inoculation of JCV in hamsters 73 and in the brain of transgenic mice expressing the T-antigen coding region of JCV under the regulation of its own promoter. 59 On the basis of these reports, Dela Cruz et al investigated whether polyomaviruses were present in the tumor tissue, and a newly discovered member of the genus Orthopolyomavirus named RacPyV was detected in tumor tissue of all affected animals. 13 Sequencing of full (n = 6) or partial (n = 4) RacPyV genomes in all 10 of the raccoons revealed a genomic structure typical of the members of the Orthopolyomavirus genus (Fig. 17). Members of this genus are mammalian polyomaviruses, and many are oncogenic, including the type species SV40. 28 By sequence prediction alone, T-antigen proteins of RacPyV encode putative binding sites for p53 and the retinoblastoma protein, which have been shown in experimental models of polyomavirus-induced transformation as necessary for oncogenic activity. Further studies would be necessary to assess RacPyV T-antigen binding affinity to these proteins. RacPyV DNA was not present in nontumorous brain tissue or other tissues of affected animals.
One form of convincing data that polyomaviruses are causative rather than bystanders in tumor formation is genomic integration. MCPyV, for example, is clonally integrated in cases of Merkel cell carcinoma. In the raccoon tumors, RacPyV exists in episomal form, 13 so either the virus is nonintegrated, or integrated copies are in low copy number and difficult to detect. If RacPyV is contributing to cell transformation and/or unchecked cell replication, a mechanism other than integration should be considered. Polyomaviruses, including members of the Avipolyomavirus genus, can have a canonical life cycle of replication, assembly, and release (Fig. 18a). In this case, the multifunctional large T-antigen plays a key role in regulating the viral life cycle by binding to the viral origin of DNA replication to promote late gene (VP) transcription and DNA synthesis. An alternate life cycle for these viruses, however, is thought to occur when large T antigen is present in the absence of virion assembly and cytolysis. 7 This could occur in a number of ways, including mutations of the late genes or their promoter, by integration with or without disruption of capsid genes or by epigenetic control of gene expression (Fig. 18b).
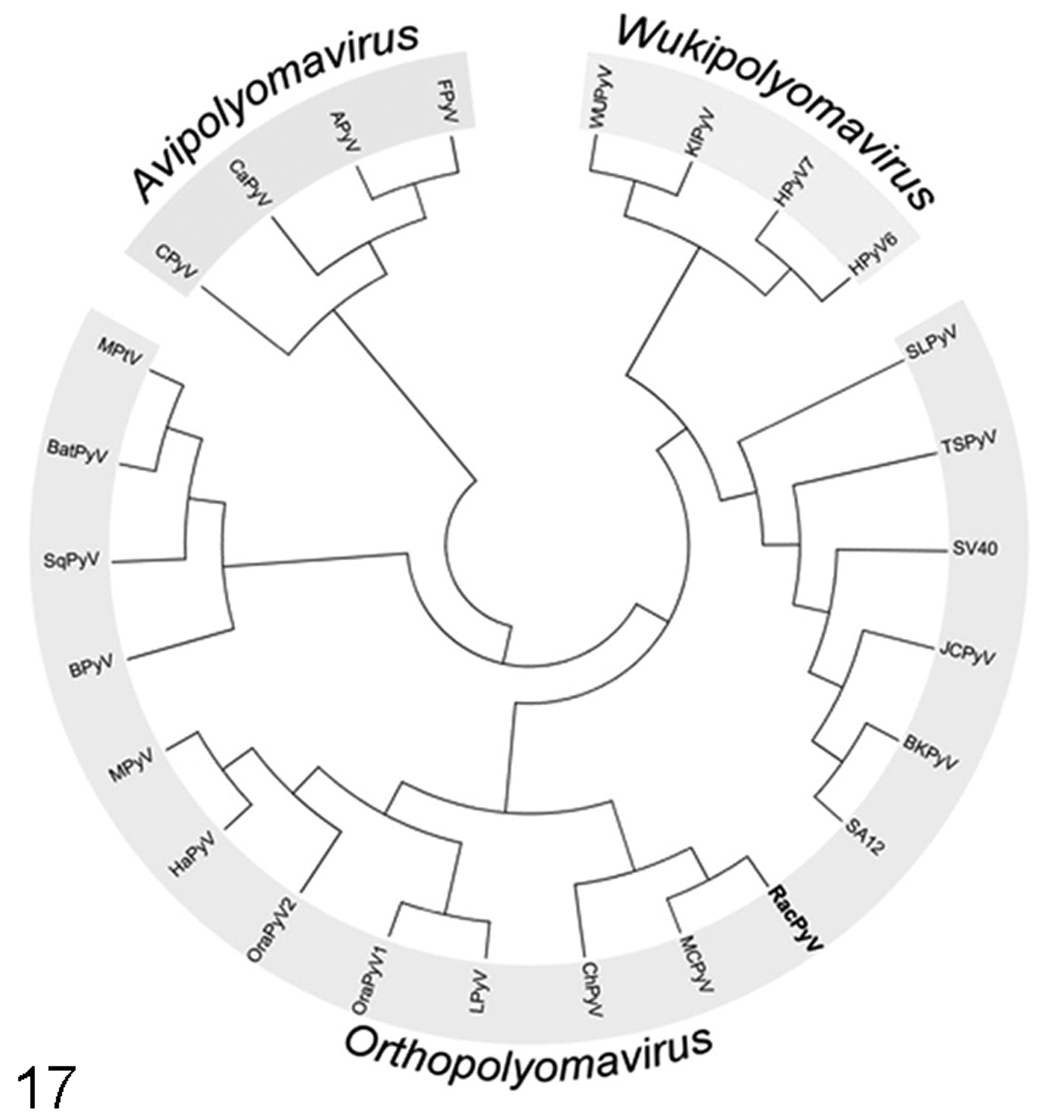
Phylogenetic relationships are based on whole genomic nucleotide sequences, using ClustalW alignments of 26 polyomaviruses. Viruses are assigned to 1 of 3 newly created genera as described by the Polyomaviridae Study Group of the International Committee on Taxonomy of Viruses. 27 RacPyV falls into the genus Orthopolyomavirus and is phylogenetically most closely related to MCPyV.
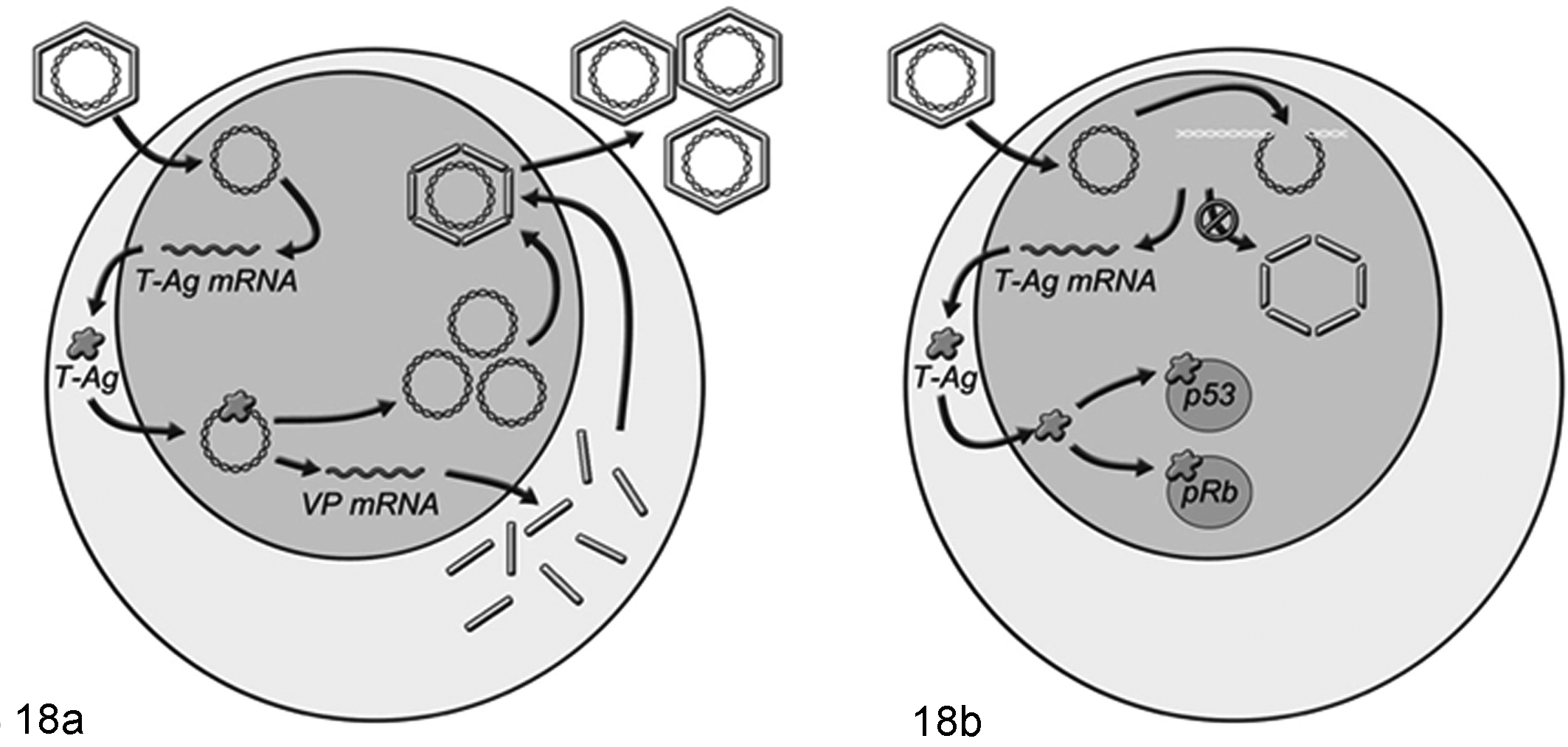
Models of the life cycles of Polyomaviridae. (a) In permissive hosts, polyomaviruses can have a “lytic” life cycle, or one that would result in cellular shedding. Polyomaviruses uncoat and replicate in the nucleus of the host. In the first of 2 distinct replication phases, the nonstructural T antigens are expressed. T antigens in turn regulate the second phase of structural protein transcription and are required for viral DNA replication. Viral capsid proteins form pentamers within the cytoplasm and then are transported to the nucleus where new virus particles are assembled. (b) Alternate viral life cycles occur in either nonpermissive hosts or are cell type specific. Among proposed life cycles that would potentially contribute to oncogenesis or nonlytic persistence, there is in common a block or attenuation of structural protein production with preservation of T-antigen expression. Possibilities include integration, mutation, or epigenetic control of translation.
In both JCV- and MCPyV-associated tumors as well as the tumors described here, large T antigen, which is both necessary and sufficient to cause tumor formation in experimental models, is expressed in the nuclei of neoplastic cells. Given the close phylogenetic proximity of RacPyV with MCPyV and the importance of polyomavirus T antigens in cell transformation and oncogenesis, Dela Cruz et al 13 performed immunohistochemistry using MCPyV T-antigen-specific antibody to demonstrate expression of T antigen in the nuclei of neoplastic cells, which has been demonstrated in 6 of the 10 tumors so far. Interestingly, the pattern of expression parallels that observed for JCV T antigen in brain tumors in which several but not all tumors cells express T antigen. 11,12,49 Also, similar to human tumors, p53 was found in the nuclei of neoplastic cells in the same tumors. A subset of cells in the raccoon tumors described here was also shown to coexpress large T antigen and p53 by double labeling imunofluorescence, 13 suggesting that T antigen is sequestering and inactivating p53. Interestingly, expression of JCV T antigen was not demonstrated in any of the tumors in our study. Large T antigen is a multifunctional protein expressed by many oncogenic polyomaviruses and is involved in viral replication and oncogenesis, the latter in part by inactivation of the tumor suppressor proteins retinoblastoma protein and p53. 17,38,50,59 In people, expression of T antigen has been documented in subsets of neoplastic cells in a variety of brain tumors in adult and pediatric patients, 9 –12,33,46,49,54 and in MPNSTs in a transgenic mice model, 25% of tumor cells expressed large T antigen to varying degrees, and there was concurrent expression of p53. 59
Exposure to some toxic agents or immunosuppression can be contributory factors to cellular transformation and tumor formation; therefore, both were investigated as part of a complete diagnostic workup. Studies have shown increased risk of brain cancer in humans with use of select pesticides, 8,65 insecticides, 45 and fungicides. 39,52 Odds ratios of gliomas in humans exposed to pesticides bufencarb, chlorphyrifos, and coumaphos were significantly higher than in nonexposed people. 53 N-nitroso alkyl ureas present in some fungicides and other N-nitroso compounds are potent nervous system carcinogens in animals and have been found in many pesticides in common use. 31 Exposure to any of these agents, with or without RacPyV, may be a contributory factor to rapid cellular transformation and tumor formation in raccoons. In one of the raccoons of this study (case No. 4), an extended carbamate and organophosphorus insecticide screen designed to detect 55 compounds yielded negative results, so association with exposure to these compounds is not likely, but more animals should be tested in future studies. N-nitroso alkyl ureas are metabolized quickly so could not be evaluated in these raccoons. However, DNA alkylation occurs with N-nitroso alkyl ureas; therefore, formation of DNA adducts can be monitored in future studies. At least 2 of the 10 raccoons were exposed to one or more anticoagulant rodenticide compound. This finding is not considered of toxicologic significance, as lesions did not support evidence of a coagulopathy; however, it demonstrates that these raccoons were exposed to environmental toxicants. There is also no knowledge of anticoagulant rodenticides having tumor-promoting properties. One raccoon had toxic concentrations of lead in multiple organs, consistent with lead poisoning. While lead has not been directly associated with carcinogenesis, it certainly could have contributed to neurologic signs in this individual. Further studies are needed to determine if a toxin is contributing to the rapid tumor progression of polyomavirus-infected raccoons.
Immunosuppression has been associated with an increased risk of viral-induced oncogenesis. In humans, HIV is a major determinant that predisposes affected humans to cancers yet does not directly cause cellular transformation. Among common causes for immunosuppression in raccoons, CDV—a paramyxovirus that is commonly diagnosed in raccoons in California and Oregon—targets the lymphoid tissue resulting in lymphopenia and immunosuppression. 1,66 CDV-induced immunosuppression in young raccoons may facilitate RacPyV infection at an early age and thus be an indirect contributing factor to neoplastic transformation in raccoons. Affected raccoons had no histologically or immunohistochemically detectable concurrent CDV infection, but this does not rule out previous infection with CDV and its immunosuppressive effects in the absence of neurologic invasion. CDV serology would have been a better diagnostic technique to demonstrate previous infection with CDV; however, antemortem serum samples were not available.
A familial aggregation for gliomas (astrocytomas) has been proposed in people, based on epidemiologic studies, 18,40 and genetically inherited disorders associated with PNSTs, such as neurofibromatosis, are well characterized in people 21 and recognized in cattle. 58 To estimate probability of any familial relatedness for raccoons with brain tumors and other raccoons from the same population, it would be necessary to create a database of genotype profiles by genotyping a minimum of 30 unrelated individuals to determine allele frequencies within the population from which these raccoons originated. Affected raccoons may be related, and this will be examined in future studies.
The histogenic classification of several types of tumors suggest that the presumed polyomavirus-driven neuro-oncogenesis has a number of cell targets. Diagnosis of a MPNST was made most confidently in the cases with microscopic involvement of branches of the olfactory nerve with an intraneural tumor cell population expressing a basal lamina defined by the strong uniform pericellular network of reticulin fibers and confirmed with immunoreactivity for laminin. The histogenesis of this tumor would thus be most likely be a Schwann cell or an olfactory ensheathing cell (OEC) originating in the olfactory nerve. In some species, the Schwann cell can be differentiated from an OEC by the exclusive immunoreactivity of Schwann cells to CD57 and low affinity nerve growth factor receptor (p75), despite both being immunoreactive with GFAP, but in vitro, these cells can assume either phenotype. Whether this is true also for raccoons remains conjectural. In some of the olfactory nerve tumors with presumed secondary intracranial spread through the cribriform plate, the intracranial and intranasal components had the same MPNST phenotype.
Another feature expected of an MPNST was that, indeed, the brain–tumor interface was characterized by an expansive mode of growth. Normal myelinated peripheral nerves in the raccoon (including olfactory, trigeminal, and sciatic nerves) are strongly and uniformly immunoreactive with GFAP; thus, this antigen is not a specific astrocytic marker in this species (Giannitti, personal communication, 2012). Further support for such a histogenesis was the demonstration of strong immunoreactivity of GFAP and S-100 in most of the tumors. Two intracranial tumors, examined before the nasal tumors were detected, had a spindle cell population immunoreactive with GFAP and with serpentine necrosis, which had a differential diagnosis, including a glioblastoma and gliosarcoma. However, the uniform individual pericellular reticulin fibers, confirmed as a basal lamina by laminin reactivity, excluded both these diagnoses.
Data from the Central Brain Tumor Registry of the United States indicates that 7% of all primary nervous system tumors occur in the cranial nerves. 6 Of these tumors, 63% occur as benign acoustic neuromas (as PNSTs), while less than 1% are classified as olfactory nasal tumors. Intracranial PNST in the subfrontal region in people, classified as subfrontal schwannomas or olfactory groove schwannomas, are rare, with only 34 cases reported until 2010. 69 In recent years, the OEC—glial cells that ensheathe the axons of the olfactory nerve—has been proposed as the presumed cell of origin for these olfactory nerve-associated PNSTs. 69,70 To date, only 1 human subfrontal malignant PNST with presumed origin in olfactory nerve has been reported. 57 The possible status of a polyomavirus infection has not yet been determined. In domestic animals, PNSTs of the cranial nerves are very rare with the trigeminal nerve most commonly involved, 32 and to our knowledge, none have been reported in the olfactory nerves of any species, including the raccoon. However, some of the intracranial tumors (eg, case No. 5) remain classified as undifferentiated or pleomorphic sarcomas of unknown histogenesis despite immunocytochemical attempts to characterize most of the neoplastic cell types involved. Last, there were some very pleomorphic intranasal tumors with yet another different phenotype, with different cytologic and immunocytochemical marker expression from the other tumors, most likely malignant pleomorphic sarcomas. Finally, this histologic study was somewhat limited by the absence of any published data on similar tumors in the raccoon and the major lack of validated cell-specific antigenic markers of nervous system tumors in raccoons (eg, for CD57 and p75 for identification of possible OEC).
One raccoon was collected in Oregon adjacent to Highway 101 (which also runs through the region of clustering in California). This raccoon may have “hitched a ride” north: a scenario that is well documented, 61 as no other raccoons with brain tumors were found in a data search of 144 raccoons necropsied at the OSUVDL. Yet, the geographically outlying case from Oregon may prove very valuable epidemiologically to further investigate contributing factors.
The presence of a recently discovered polyomavirus associated with olfactory/brain tumors in 10 temporally clustered free-ranging raccoons, 9 of which were also geographically clustered, suggests that RacPyV is highly associated with neuro-oncogenesis, particularly since polyomavirus was detected in tumorous tissue in all 10 raccoons but not in unaffected brain or other tissues from affected raccoons. 13 A causative relationship between the viral infection and oncogenesis cannot be made based solely on the findings of the present study; however, the nuclear coexpression of the oncogenic protein large T antigen of MCPyV and its binding protein p53 strongly supports a role for polyomavirus in the pathogenesis of these tumors. 13
The diagnostic investigations thus far have not identified other potential factors that could have contributed to neuro-oncogenesis. The life expectancy of captive raccoons is typically 10 years; however, free-ranging raccoons usually do not survive beyond 2 to 3 years in an urban or suburban environment. 29,72 The short life expectancy of wild raccoons suggests that oncogenesis in these raccoons with olfactory/brain tumors started early in life and progressed rapidly. Long latent periods are required for tumor formation even with experimental intracranial inoculation of JCV in nonhuman primates (16–36 months). 36,37 Moreover, serologic studies have demonstrated that 80% of the adult human population worldwide has antibodies against JCV, 2,47,63 yet the incidence of brain tumors is comparatively low. 34 Additionally, tumors occur decades after initial infection with oncogenic virus. Therefore, there may be one or more cofactors contributing to neoplastic transformation in the polyomavirus-infected raccoons. There are numerous examples of neoplasias in wildlife that are highly associated with concurrent exposure to environmental toxins and infection with oncogenic viruses. The incidence of fibropapillomatosis in sea turtles infected with papillomavirus or genital cancer in sea lions infected with herpesvirus is significantly higher in individuals exposed to polluted waters 20,26 or to polychlorinated biphenyls, 71 respectively. This diagnostic workup examined possible risk factors that may contribute to development of tumor transformation. Further investigation of the epidemiology and pathogenesis of this viral infection and its possible interactions with other genetic and environmental risk factors in this population of raccoons is needed to understand the possible causal relationship between RacPyV and neuro-oncogenesis.
Footnotes
Acknowledgements
We thank Diane Naydan, Terrence Wildman, Karen Sverlow, Robert Nordhausen, Robert Munn, Scott Fish, and Alyssa Miller from the California Animal Health and Food Safety Laboratory and Lora Konde from the California Department of Fish and Wildlife for their technical assistance.
Declaration of Conflicting Interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Funding provided by the California Department of Fish and Wildlife and WildCare.
