Abstract
The zebrafish (Danio rerio) provides a powerful model for analyzing genetic contributors to cancer. Multiple zebrafish lines with cancer-associated genetic mutations develop soft tissue sarcomas that are histologically consistent with malignant peripheral nerve sheath tumor (MPNST). The goal of this study was to determine the phenotype of soft tissue sarcomas in a brca2-mutant/tp53-mutant zebrafish line using immunohistochemical markers that are commonly expressed in mammalian MPNST. We classified 70 soft tissue sarcomas from a brca2-mutant/tp53-mutant zebrafish cohort as MPNST, undifferentiated sarcoma, or other tumor based on histologic features. The expression of S100, CD57, and glial fibrillary acidic protein (GFAP) was analyzed in nonneoplastic neural tissues and tumor specimens by immunohistochemistry. Each marker was expressed in nonneoplastic neural tissues. In MPNST, S100 and CD57 were widely expressed in neoplastic cells, with greater consistency observed for CD57 expression. In undifferentiated sarcomas, results were variable and correlated to anatomic location. Coelomic undifferentiated sarcomas often exhibited widespread CD57 expression but limited S100 expression. In comparison, ocular undifferentiated sarcomas exhibited limited expression of both CD57 and S100. Overall, CD57 and S100 expression was significantly higher in MPNST than in undifferentiated sarcomas. GFAP was not expressed in any of the tumors. This study identified commercially available antibodies that are useful for analyzing S100, CD57, and GFAP expression in zebrafish. This study further shows a correlation between degree of histologic differentiation and expression of these markers in soft tissue sarcomas from brca2-mutant/tp53-mutant zebrafish and suggests that these cancers are derived from the neural crest with differentiation toward myelinating Schwann cells.
The zebrafish (Danio rerio) is a widely used vertebrate animal model in cancer research. 27 Zebrafish models for a range of cancer types have been developed and characterized, and cancers from several of these models were shown to express markers that are also expressed in the analogous human cancer. 1,2,7,13,14,16,17,22,24 A notable exception is soft tissue sarcoma. Among genetically engineered zebrafish cancer models, multiple lines develop soft tissue sarcomas that are histologically consistent with malignant peripheral nerve sheath tumor (MPNST). 1,2,7,22,24,28 Despite the frequency of MPNST development in these models, immunohistochemical characterization has been limited. This may reflect the relative paucity of zebrafish-specific antibodies that have been tested and validated.
We have previously reported that zebrafish from a brca2-mutant/tp53-mutant line frequently develop soft tissue sarcomas with histologic features of either MPNST or undifferentiated sarcoma. 28,29 Herein, we use commercially available antibodies for S100, CD57 (HNK-1, Leu-7), and glial fibrillary acidic protein (GFAP), 3 markers used for the diagnosis of MPNST in other species, for immunohistochemical analysis of soft tissue sarcomas in this zebrafish line. The objectives of this study were to (1) characterize S100, CD57, and GFAP expression by immunohistochemistry in nonneoplastic zebrafish tissues; (2) apply these markers to determine the phenotype of soft tissue sarcomas arising in brca2-mutant/tp53-mutant zebrafish; and (3) establish the relatedness of MPNST and undifferentiated sarcoma based on patterns of marker expression.
Materials and Methods
Animals and Genotyping
All animal studies were approved by the Intramural Animal Care and Use Committee, National Cancer Institute, National Institutes of Health, Bethesda, Maryland. The zebrafish cohort was derived from an incross of zebrafish with heterozygous brca2Q658X mutation and homozygous tp53 M214K mutation. 2,28 The brca2Q658X -mutant zebrafish line was generated at the National Institutes of Health using a previously established ENU-mutagenized library 30 in the EK zebrafish genetic background and outcrossed to wild-type AB zebrafish. The tp53M214K -mutant zebrafish line was acquired from the Zebrafish International Resource Center (ZIRC). The tp53M214K -mutant zebrafish line was generated from an ENU-mutagenized library in the Tübingen zebrafish genetic background. 2 Mutant alleles are designated hereafter as “m.”
Zebrafish used in this study were reared in a multirack recirculating containment system. From 5 to 9 days of age, zebrafish larvae received live cultured Brachionus plicatilis (L-type rotifers), and from 10 to 30 days of age, zebrafish fry received commercially available, appropriately sized powder diets (Zeigler Larval AP100 Diet [Pentair Aquatic Ecosystems, Apopka, FL]; Hatchfry Encapsulon [Argent Laboratories, Redmond, WA]) supplemented with live cultured Artemia sp (brine shrimp). Juvenile and adult zebrafish received commercially available dry zebrafish diets (Zeigler Larval AP100 Diet and Zebrafish Adult Diet; Pentair Aquatic Ecosystems) supplemented with live cultured Artemia sp. Zebrafish were monitored for clinical and gross evidence of tumor development, such as ocular enlargement or coelomic distension. Zebrafish were observed for tumor development up to 10 months of age, and the median age at tumor onset during this observational period was 8 months of age. Tumor-bearing zebrafish were euthanized with tricaine methanesulfonate (0.625 g/l) in system water buffered with sodium bicarbonate (0.7 g/l).
For genotyping, fin clips were collected from tumor-bearing zebrafish at the time of death or euthanasia, and DNA extraction was performed as described previously. 28 Zebrafish were genotyped for the brca2Q658X mutation by polymerase chain reaction (PCR) amplification using primers designed to create a Tsp45I restriction site based on the presence of the brca2Q658X point mutation. PCR products were incubated with Tsp45I digestion buffer (0.125 U/μl Tsp45I in CutSmart buffer; New England Biolabs, Ipswich, MA) at 65°C for 2 hours. In some instances, genotyping for the brca2Q658X mutation was achieved by sequencing as described previously. 29 Zebrafish were genotyped for the tp53M214K mutation by sequencing as described previously. 29
Histologic Analysis
Tumor-bearing zebrafish were fixed and decalcified as described previously 29 and were routinely processed to prepare 5-μm hematoxylin and eosin–stained sections (Histoserv, Gaithersburg, MD). Zebrafish with coelomic tumors were prepared by sectioning sagitally through the midline. Zebrafish with ocular tumors were prepared by sectioning the head transversely across the midline and sectioning the body sagitally through the midline. Two sections from each zebrafish were independently evaluated by 2 investigators (L.A.W. and H.R.S.). Specimens were evaluated without knowledge of genotype. Phenotypic sex was determined at the time of histologic analysis.
Immunohistochemistry and Scoring
Immunohistochemistry was performed with a mouse monoclonal anti-CD57 antibody (MA5-11605; Thermo Scientific, Rockford, IL) without antigen retrieval, a rabbit polyclonal anti-S100 antibody (Z031129; Dako, Carpinteria, CA) without antigen retrieval, and a mouse monoclonal anti-GFAP antibody (36705; Cell Signaling Technology, Danvers, MA) with citrate-buffered heat-induced epitope retrieval methods. Antibodies targeting conserved regions of each protein were selected based on protein-protein alignments of human, mouse, and zebrafish orthologues.
Unstained 5-μm serial sections from tumor-bearing zebrafish were routinely prepared from paraffin-embedded tissues and processed for immunohistochemistry (Dako Autostainer Plus). Serial sections exposed to secondary antibody alone (either anti-mouse or anti-rabbit antibody) were used as negative controls, and multiple negative controls were included for each experiment. Sections were independently evaluated and scored (Suppl. Table S1) by 2 investigators (L.A.W. and H.R.S.). Specimens were evaluated without knowledge of genotype. Following statistical analysis for interobserver agreement (see below), consensus scores were determined by 2 investigators (L.A.W. and H.R.S.) for use in subsequent analyses.
S100, CD57, and GFAP expression was assessed in individual zebrafish specimens (Suppl. Tables S2, S3). As an internal control for variation in fixation time between individual specimens, S100, CD57, and GFAP expression was assessed in nonneoplastic nervous tissues in the same specimen for each tumor-bearing zebrafish.
Statistical Analysis
To determine interobserver agreement, a weighted κ statistic was applied to individual scores as determined by 2 observers (L.A.W. and H.R.S.), and values above 0.7 were considered to indicate observer agreement. To compare the distribution of tumor types across brca2 genotypes (Suppl. Table S2), Fisher’s exact test was applied, and P < .05 was accepted to indicate statistical significance. To compare the level of expression of S100 and CD57 between MPNST and undifferentiated sarcoma, tumors from coelomic and ocular locations were combined, and scores for approximate percentage of cells exhibiting expression and scores for intensity of expression were compared (Suppl. Table S3). An ordinal logistic regression analysis was applied with internal control tissue intensity as a covariate, and P < .05 was accepted to indicate statistical significance.
Results
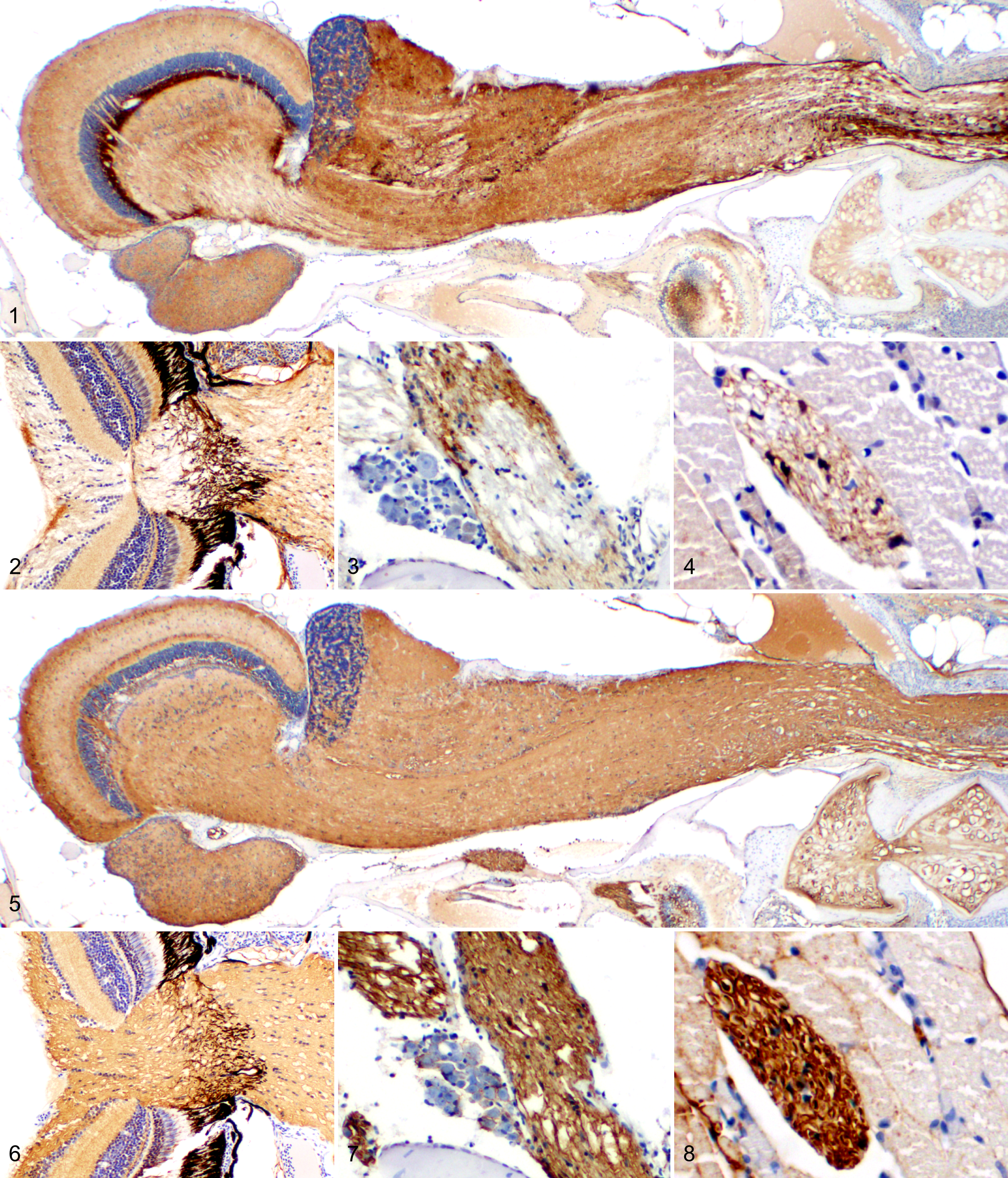
A total of 68 tumor-bearing adult zebrafish siblings from a brca2-mutant/tp53 mutant zebrafish line 28 were examined histologically and by immunohistochemistry. Nonneoplastic neural tissues from zebrafish within the study population were analyzed for S100, CD57, and GFAP expression. In the central nervous system (CNS), S100 was widely and strongly expressed in the brain and spinal cord (Fig. 1). S100 expression was variable in the optic nerve and retina (Fig. 2), vagus nerve (Fig. 3), and peripheral nerves (Fig. 4). CD57 was widely and strongly expressed in the brain and spinal cord (Fig. 5), optic nerve and retina (Fig. 6), vagus nerve (Fig. 7), and peripheral nerves (Fig. 8). Cross-sectioned nerves displayed well-demarcated, intense CD57 expression circumferentially around axons (Fig. 8), consistent with membrane labeling of myelinating cells. In contrast, GFAP was expressed in defined zones in the brain and spinal cord (Suppl. Fig. S1) and was minimally expressed or absent in the optic nerve and retina (Suppl. Fig. S2), vagus nerve (Suppl. Fig. S3), and peripheral nerves (Suppl. Fig. S4). Nonspecific labeling was minimal to absent for all antibodies (Figs. 1–8, Suppl. Figs. S1–S4), although moderately intense, specific CD57 labeling was regularly observed in chondrocytes and renal glomeruli. No background labeling was observed in negative control slides exposed to secondary antibody alone.
A total of 70 tumors from 68 zebrafish, consisting of 25 ocular tumors (Fig. 9) and 45 coelomic tumors (Fig. 10), were analyzed histologically (Suppl. Table S2). Tumors were categorized by assessment of histologic features, with MPNST and undifferentiated sarcoma identified as the most common tumor types in both ocular and coelomic locations (Suppl. Table S2). Nine zebrafish within the study population exhibited 2 tumors in distinct anatomic locations (ocular region and coelom). Three of these 9 zebrafish developed tumors of 2 different histologic types. In 7 of these 9 zebrafish, serial sectioning precluded immunohistochemical evaluation of both tumors, as they were not both present in serial sections. Tumors that could not be assessed for this reason were not included in the final histologic analysis (Suppl. Table S2). No correlation between genotype and tumor type was observed (P = .9672, Fisher’s exact test).
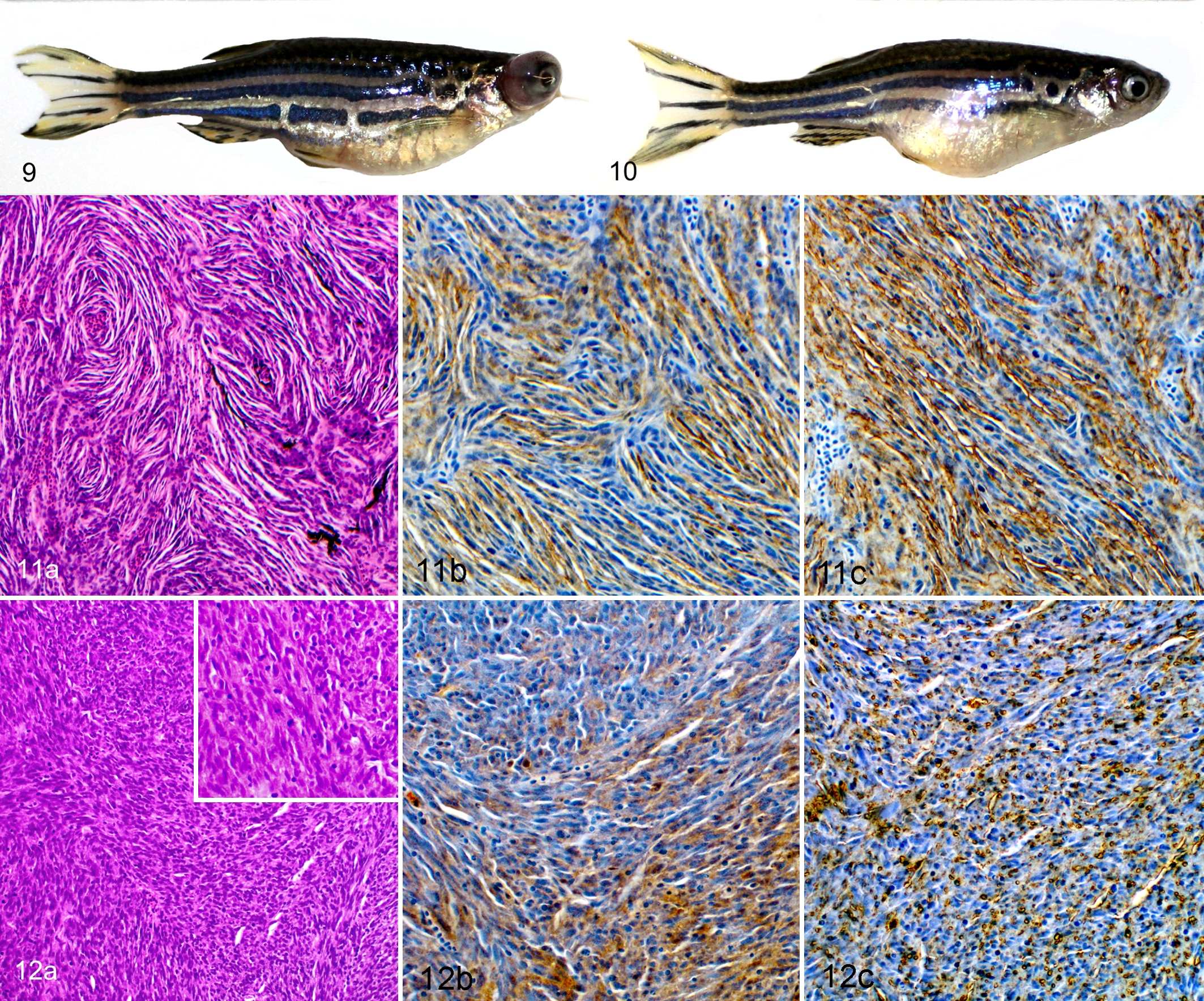
Tumors classified as MPNST exhibited classic features that include spindloid neoplastic cells with oval or tapered nuclei organized in fascicles, whorls, and bundles (Fig. 11a). Tumors classified as undifferentiated sarcoma exhibited variably pleomorphic spindle cells containing round to oval nuclei that frequently formed solid bundles without distinct organization (Fig. 12a). Other ocular tumors included a primitive neuroectodermal tumor, medulloepithelioma, and retinoblastoma. Other coelomic tumors included a hemangiosarcoma, thyroid carcinoma, lymphosarcoma, presumed yolk sac carcinoma, and gonadal stromal tumor.
A total of 67 tumors from 65 zebrafish were analyzed by immunohistochemistry for expression of S100, CD57, and GFAP (Suppl. Table S3), including scoring of the approximate percentage of neoplastic cells expressing each marker and assessment of the intensity of expression (Suppl. Table S1). Comparison of observer scoring for approximate percentage of cells and intensity in tumors and internal control intensity for S100 and CD57 indicated interobserver agreement across each category (weighted κ statistic >0.7).
Within the major category of MPNST, immunohistochemical findings were similar in tumors from ocular and coelomic locations. Moderate to strong expression of S100 was present in a majority of cells in 7 of 12 (58%) ocular MPNSTs and 12 of 27 (44%) coelomic MPNSTs (Fig. 11b and Table 1). In comparison, moderate to strong CD57 expression was present in a majority of cells in 9 of 12 (75%) ocular MPNSTs and 27 of 27 (100%) coelomic MPNSTs (Fig. 11c and Table 1). CD57-expressing neoplastic cells exhibited intense membrane labeling (Fig. 11c). Comparison of scores indicated that CD57 expression was more consistent than S100 expression in MPNSTs from both ocular and coelomic locations (Fig. 13).
Extent and Intensity of S100 and CD57 Expression in 67 Ocular and Coelomic Sarcomas in Zebrafish.a
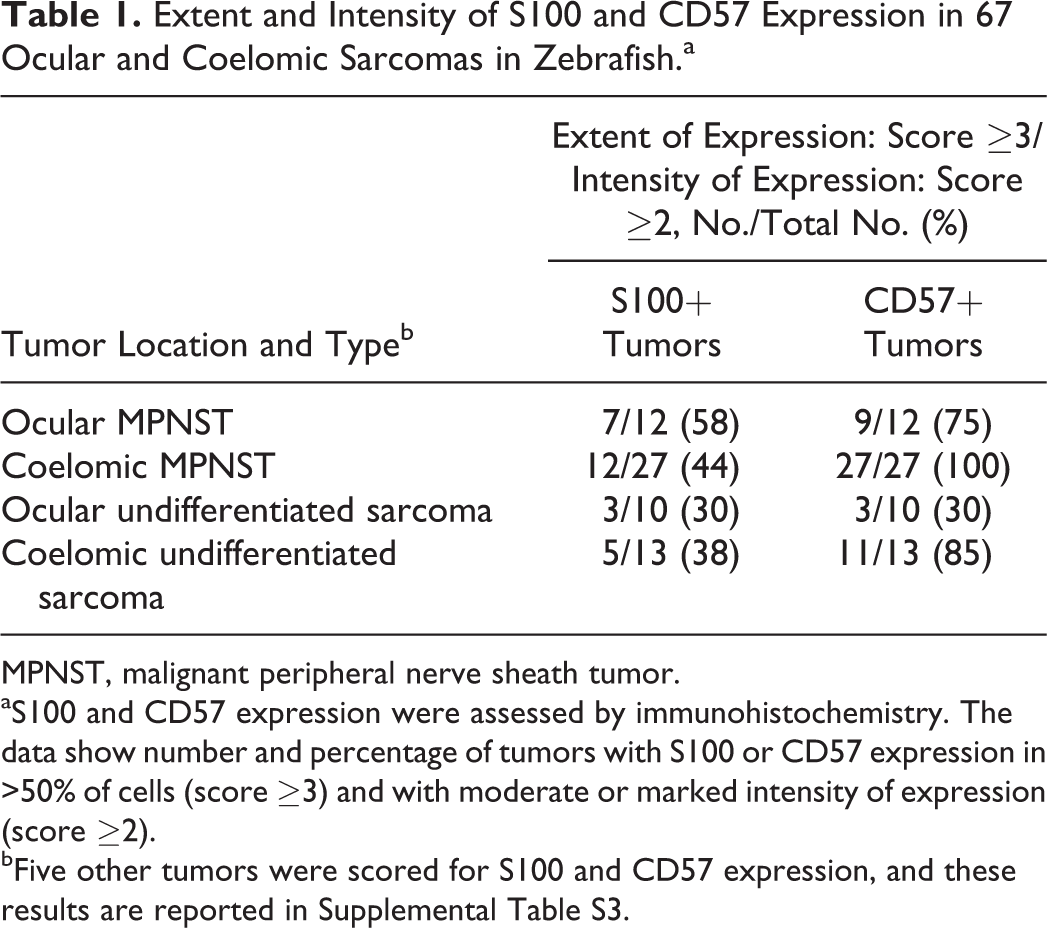
MPNST, malignant peripheral nerve sheath tumor.
aS100 and CD57 expression were assessed by immunohistochemistry. The data show number and percentage of tumors with S100 or CD57 expression in >50% of cells (score ≥3) and with moderate or marked intensity of expression (score ≥2).
bFive other tumors were scored for S100 and CD57 expression, and these results are reported in Supplemental Table S3.
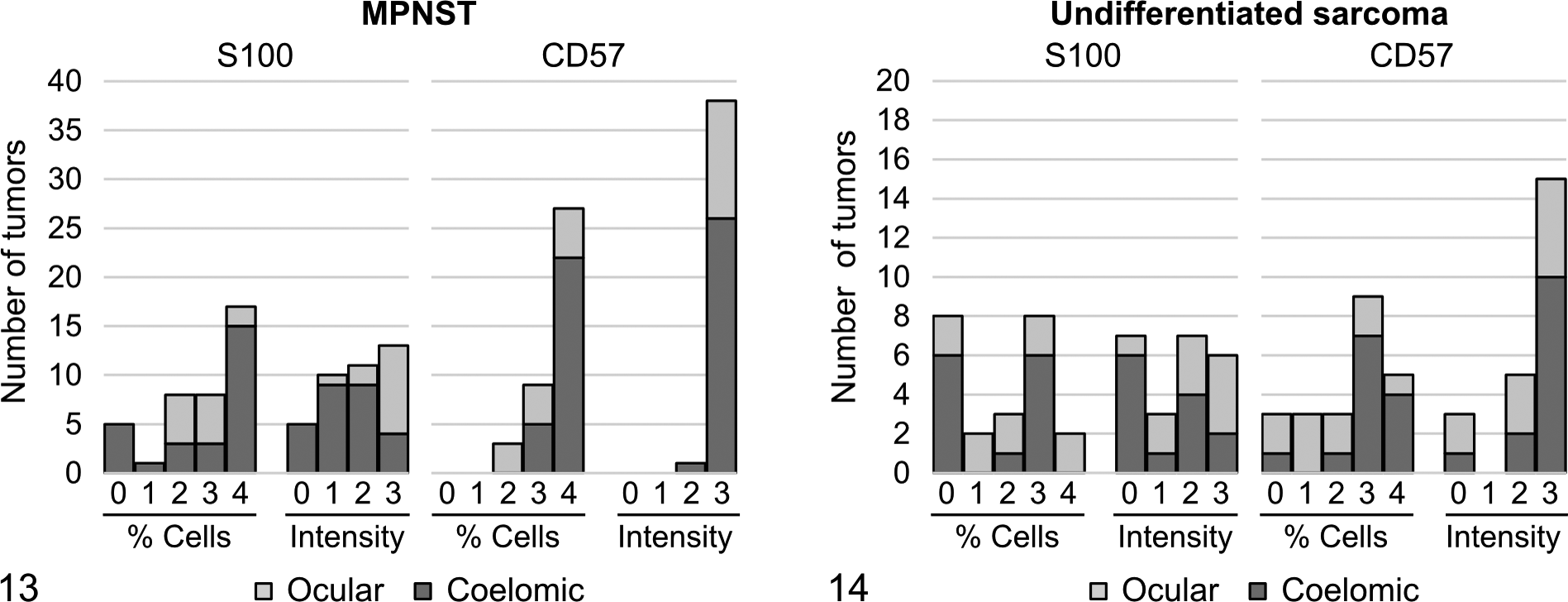
Within the major category of undifferentiated sarcoma, immunohistochemical findings were varied in tumors from ocular and coelomic locations. Only 3 of 10 (30%) ocular undifferentiated sarcomas and 5 of 13 (38%) coelomic undifferentiated sarcomas exhibited moderate to strong expression of S100 in a majority of cells (Fig. 12b and Table 1). In comparison, 3 of 10 (30%) ocular undifferentiated sarcomas exhibited moderate to strong expression of CD57 in a majority of cells. However, 11 of 13 (85%) coelomic undifferentiated sarcomas exhibited moderate to strong expression of CD57 in a majority of cells (Fig. 12c and Table 1). CD57-expressing neoplastic cells exhibited intense membrane labeling (Fig. 12c). Comparison of scores indicated greater variability in S100 and CD57 expression in undifferentiated sarcomas (Fig. 14) compared with MPNSTs (Fig. 13) from both ocular and coelomic locations.
To statistically assess correlations between histologic type and marker expression, scores for the 2 parameters assessed in this study (Suppl. Table S1) were compared between all MPNSTs and all undifferentiated sarcomas by ordinal logistic regression analysis. Staining intensity of internal control tissue (nonneoplastic nervous tissue) in each specimen was used as a covariate to address potential differences in efficacy of immunohistochemical labeling in individual samples.
For S100, MPNSTs had a significantly higher likelihood of having higher percentages of expression than undifferentiated sarcomas (P = .0065). This outcome was independent of the intensity of control tissue expression. However, MPNSTs did not have a significantly higher likelihood of exhibiting high intensity of S100 expression in tumor cells than undifferentiated sarcomas (P = .1474). This outcome was affected by the intensity of control tissue expression (P = .0006); specimens with a higher control tissue intensity score were more likely to have a higher tumor intensity score.
For CD57, MPNSTs had a significantly higher likelihood of having higher percentages of expression than undifferentiated sarcomas (P = .0001). MPNSTs also had a significantly higher likelihood of exhibiting high intensity of CD57 expression in tumor cells compared with undifferentiated sarcomas (P = .0061). The intensity of control tissue expression for CD57 was the same for all specimens and thus did not affect either outcome.
GFAP was not expressed in any of the ocular or coelomic MPNSTs or undifferentiated sarcomas. However, GFAP was expressed in the optic nerve pathways that were disrupted by tumor infiltration, particularly in nonneoplastic cells lining the interface between optic nerve tissue and invading neoplastic cells (Suppl. Figs. S5–S7).
S100 and CD57 expression was also analyzed in 5 other tumors (Suppl. Table S3). The 2 coelomic mesenchymal tumors of nonneural origin (hemangiosarcoma and gonadal stromal tumor) that occurred in the study population did not express either marker (Suppl. Figs. S8, S9). The 3 ocular tumors of neuroectodermal origin (medulloepithelioma, retinoblastoma, and primitive neuroectodermal tumor) exhibited variable S100 and CD57 expression (Suppl. Table S3).
Discussion
Zebrafish models are widely used in comparative cancer research: they can be analyzed in large numbers, are highly amenable to genetic manipulation, and share many conserved cancer-associated genes with humans and other species. Broad use of zebrafish cancer models requires careful and complete tumor phenotyping, including the application of ancillary tests such as immunohistochemistry, to define neoplastic cell type and pattern of differentiation. These analyses can provide the basis for determining genetic effects on neoplastic development.
Soft tissue sarcomas arising in several cancer-prone zebrafish models are frequently reported as MPNSTs 1,2,7,22,24,28 but have been poorly characterized by immunohistochemistry. In mammalian species, soft tissues sarcomas can exhibit common histologic features across tumor types. 4,5,8,23,31 Differential diagnoses for MPNSTs include mesenchymal tumor types such as poorly differentiated fibrosarcoma, hemangiosarcoma, perivascular wall tumor, rhabdomyosarcoma, liposarcoma, leiomyosarcoma, and synovial sarcoma. Melanocytic (or chromatocytic) tumors derived from the neuroectoderm may also be considered. In this study, we analyzed soft tissue sarcomas in zebrafish from a brca2-mutant/tp53-mutant line by histology and immunohistochemistry to (1) characterize the reactivity of antibodies against S100, CD57, and GFAP in nonneoplastic zebrafish tissues; (2) apply these markers to assess the phenotype of MPNST and undifferentiated sarcoma; and (3) determine the relationship between well-differentiated MPNST and undifferentiated sarcoma.
Multiple markers are used to assist in the diagnosis of MPNST in human and animal species, including S100, CD57, GFAP, nestin, vimentin, nerve growth factor receptor (NGFR), CD34, neurofilament protein, and others, 4,5,8,23,26,31 although no marker is specific for MPNST in any species. We identified 3 antibodies that can be used to analyze expression of S100, CD57, and GFAP in zebrafish neural tissues by immunohistochemistry. These markers were selected for study based on regions of protein homology between human, mouse, and zebrafish orthologues. For each antibody, specificity was supported by minimal to absent nonspecific labeling and lack of background labeling in negative controls.
We observed robust, widespread S100 expression and moderate, discrete GFAP expression in the zebrafish central and peripheral nervous systems, findings that are generally similar to prior studies. 9,12 To our knowledge, CD57 expression has not been analyzed previously in the developed nervous system in adult zebrafish. Our data indicate robust, widespread CD57 expression in the central and peripheral nervous systems, with intense membrane labeling of presumptive myelinating cells. We also observed less intense labeling of chondrocytes and renal glomeruli. In comparison, human oligodendrocytes and Schwann cells express CD57, 10 and the CD57 antibody used for these studies labels human myelin-associated glycoprotein. 19 Interestingly, CD57 is also reportedly expressed in human chondrosarcoma and some types of renal cancer. 6,10,32 Although CD57 expression is not limited to the nervous system in zebrafish, the consistently robust CD57 labeling, particularly in presumptive myelinating cells, supports a conserved and specific pattern of expression in zebrafish neural tissue.
As both CD57 and S100 can be expressed in mammalian tumors of neural and nonneural origin, immunohistochemical analysis must be coupled with careful histologic assessment. MPNSTs in ocular and coelomic locations displayed classic histologic features observed in tumors arising from the neural sheath. Most ocular MPNSTs and all coelomic MPNSTs exhibited moderate to strong, widespread CD57 expression. CD57-expressing neoplastic cells exhibited intense membrane labeling, similar to that observed in presumptive myelinating cells in peripheral nerves. S100 expression of similar intensity and extent occurred in a smaller proportion of ocular and coelomic MPNSTs. The utility of these markers in zebrafish for the diagnosis of MPNST is supported by the lack of S100 and CD57 expression in mesenchymal tumors that were clearly of nonneural origin based on histologic features. The combined histologic and immunohistochemical results reported herein indicate that zebrafish MPNSTs are of neural crest origin, with the pattern of CD57 expression suggesting differentiation toward myelinating Schwann cells.
In comparison to MPNST, expression of both CD57 and S100 in undifferentiated sarcomas was more variable, less intense, and less widespread. Despite this difference, CD57-expressing neoplastic cells exhibited similar strong membrane labeling as observed in MPNSTs. CD57 expression was more intense and widespread in coelomic undifferentiated sarcomas in comparison to ocular undifferentiated sarcomas. In comparison, S100 expression and distribution in undifferentiated sarcomas were relatively low in both ocular and coelomic locations. This finding suggests that sarcomas arising in coelomic vs ocular locations may originate from different cell populations or may undergo different patterns of differentiation. Further molecular and immunohistochemical analyses of tumors from these locations will be required to distinguish these possibilities.
Statistical analyses of marker expression using control tissue expression as a covariate suggested that the intensity of S100 expression in tumors, but not the distribution of S100 expression, may be affected by external variables (eg, fixation time). This is in contrast to the robust and uniform expression of CD57 in control tissues, which did not affect tumor expression outcomes. These findings highlight the importance of evaluating control tissue expression in individual samples for immunohistochemical studies, particularly when validating untested antibodies in model organisms.
GFAP was not expressed in any zebrafish tumor specimens, although the findings in normal tissues indicate reactivity of the antibody in zebrafish tissues. The lack of GFAP expression in these neoplasms is not inconsistent with analyses of GFAP expression in other species, as GFAP expression is reportedly variable in tumors of neural sheath origin and may occur more frequently in those that are benign or low grade. 4,11,20,25,26
Interestingly, nonneoplastic GFAP-expressing cells were observed lining the interface between neoplastic cells and optic nerve tissue in zebrafish with ocular tumors. These findings may indicate the presence of reactive resident astrocytes in the infiltrated optic nerve in response to tumor invasion. An alternative source of GFAP-expressing cells could be migration of astrocytes along the optic nerve pathway. Multiple prior studies have indicated that GFAP-expressing cells are not normally present in fish optic nerve, 3,15,18 which is consistent with our findings in nonneoplastic ocular tissue. In addition, optic nerve astrocytes in fish remain GFAP negative following optic nerve injury. 15,21 Although our current analyses do not allow determination of the source of GFAP-positive cells, given the lack of reactivity in injury models, we favor migration of astrocytes along the optic nerve.
In conclusion, histologic and immunohistochemical analyses of soft tissue sarcomas in zebrafish from a brca2-mutant/tp53-mutant line strongly suggest that the majority of these sarcomas are of neural crest origin with differentiation toward myelinating Schwann cells. Although less certain, the partial immunoreactivity to S100 and CD57 in many of the undifferentiated sarcomas is supportive of a common progenitor cell for these 2 neoplasms with varying degrees of differentiation between MNPST and undifferentiated sarcoma. Given the apparent frequency of MPNST in zebrafish cancer models, S100 and CD57 may be useful markers for diagnosis of these tumors in other zebrafish lines. It is critical to incorporate detailed histologic and immunohistochemical analyses in the assessment of zebrafish neoplasms to achieve accuracy in tumor identification. Definitive diagnosis of zebrafish cancers by such methods will strengthen the use of this model in comparative cancer research and will provide important phenotypic data for toxicologic or toxicogenomic studies in zebrafish.
Footnotes
Acknowledgements
We thank Sandra Horton and Laura Shewmon for their assistance in performing immunohistochemical analyses and Dr Emily Griffith for her assistance with statistical analyses.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Intramural Research Program of the US National Institutes of Health, National Cancer Institute, Center for Cancer Research, and the North Carolina State University, College of Veterinary Medicine.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
