Abstract
Gas bubbles were found in 15 of 23 gillnet-drowned bycaught harp (Pagophilus groenlandicus), harbor (Phoca vitulina) and gray (Halichoerus grypus) seals, common (Delphinus delphis) and white-sided (Lagenorhyncus acutus) dolphins, and harbor porpoises (Phocaena phocaena) but in only 1 of 41 stranded marine mammals. Cases with minimal scavenging and bloating were chilled as practical and necropsied within 24 to 72 hours of collection. Bubbles were commonly visible grossly and histologically in bycaught cases. Affected tissues included lung, liver, heart, brain, skeletal muscle, gonad, lymph nodes, blood, intestine, pancreas, spleen, and eye. Computed tomography performed on 4 animals also identified gas bubbles in various tissues. Mean ± SD net lead line depths (m) were 92 ± 44 and ascent rates (ms-1) 0.3 ± 0.2 for affected animals and 76 ± 33 and 0.2 ± 0.1, respectively, for unaffected animals. The relatively good carcass condition of these cases, comparable to 2 stranded cases that showed no gas formation on computed tomography (even after 3 days of refrigeration in one case), along with the histologic absence of bacteria and autolytic changes, indicate that peri- or postmortem phase change of supersaturated blood and tissues is most likely. Studies have suggested that under some circumstances, diving mammals are routinely supersaturated and that these mammals presumably manage gas exchange and decompression anatomically and behaviorally. This study provides a unique illustration of such supersaturated tissues. We suggest that greater attention be paid to the radiology and pathology of bycatch mortality as a possible model to better understand gas bubble disease in marine mammals.
Introduction
Air-breathing, diving mammals are cyclically exposed to increased ambient pressure. The increase in environmental pressure directly increases the solubility of inspired gas in blood and tissue. Depending on the amount of gas exposure and depth and duration of a dive, inert gas accumulates in tissues until reaching saturation. Upon returning to the surface, the situation reverses and absorbed gas is released from blood and tissue. If decompression is rapid, bubbles may grow, disrupting tissue and circulation and causing the pain and damage of “the bends.” It has been a longstanding paradox that aquatic mammals appear to tolerate and suffer no ill effects from tissue gas tensions and behavior that would result in bubble formation and injury in terrestrial animals. 22, 34 Gas bubbles have become a greater concern since the publication of a brief report in 2003 and then fuller descriptive papers of atypical mass strandings of beaked whale and other single strandings of small odontocetes. 13, 18, 19 These papers established the pathobiologic basis for a hypothesis that beaked whales are acutely 13 and other odontocetes are perhaps repeatedly 19 subject to the risk of gas bubble formation similar to those experienced by humans exposed to marked changes in barometric pressure: caisson workers, pilots, and divers. 28 The clinical significance of these bubbles in humans depends on the severity of the case, with “silent” nonpathologic emboli being common. Postmortem bubbles can also outgas from supersaturated tissues regardless of barotrauma and are not of themselves generally considered diagnostic of the bends in human fatalities. 6 The case for clinical significance in the animals studied by Fernandez and Jepson was made on the basis of the association of gas bubbles with gross and histologic lesions. 13, 19
These observations have in turn led to an intense nongovernmental organizational scrutiny of the complex relationship between ocean noise, bubble injury, and marine mammal strandings (http://www.awionline.org/oceans/Noise/IONC/index.htm). There has also been significant scientific interest in the topic, with a workshop held in Baltimore, MD, USA, in April 2004. 9 That workshop concluded that “gas-bubble disease, induced in supersaturated tissue by a behavioral response to acoustic exposure, is a plausible mechanism for the morbidity and mortality seen in cetaceans associated with sonar exposure.” The need was stated for further investigation of beaked whale pathobiology and diving behavior and the onset of lung collapse and its impact on nitrogen gas kinetics and for conducting additional postmortem experiments. Knowledge of beaked whale behavior was recently advanced when it was concluded that “the natural behavior of beaked whales is inconsistent with known problems of acute nitrogen super-saturation and embolism, but that possible decompression problems are more likely to result from an abnormal behavioral response to sonar.” 38 Furthermore, a recent model suggested that the depth at which behavioral change would most likely result in decompression sickness was just above the depth of lung collapse (70–80 m). 41 This is supported by earlier work demonstrating that bottle-nosed dolphins are not protected from gas accumulation in tissues at this depth range. However, despite having a diving regimen that would produce symptoms in humans, dolphins appear to avoid decompression sickness. 34
It is important therefore to understand the origin of bubbles observed at necropsy and determine their source. The bubbles may form pre- or postmortem and result from supersaturated tissue, embolized tissue, bacterial decomposition, or dissection artifact. To better differentiate between these bubble sources, an experimental model of marine mammal gas bubble formation is required.
Bycatch are species caught in a fishery targeting another species. There have been previous studies of the pathology of bycaught marine mammals, 11, 21, 23 where net trauma and lesions specific to drowning have been reported. The study reported here is part of a broad survey of stranded and bycaught marine mammals for zoonotic infectious agents and the presence or absence of associated gross and histologic lesions with the goal of understanding the clinical significance of such organisms. 4 In this paper, we describe gross histologic and radiographic observations related to the presence of gas bubbles in bycaught seals and dolphins within that survey. Although gas bubbles are a common finding in this study group, our examination of the literature suggests that this is the first report of acute gas bubble events in drowned marine mammal bycatch.
Methods
NOAA Fisheries Observers recorded date, latitude, longitude, depth, dimensions of fishing gear, depth of net, number of nets in the set, haul duration, target species, and surface water temperature when a net was set and when hauled. Rate of individual net ascent was calculated as the depth of set, divided by the total time spent retrieving the net, divided by the number of nets in the set, with the assumption that each net rises completely before the next net starts to ascend. Deep muscle temperature of all marine mammal carcasses was recorded using a Taylor 9841 Digital Instant-Read Pocket Thermometer (Taylor Precision Products, Oak Brook, IL, USA) once the animals were on deck to estimate the elapsed time since death. Surface water temperature was measured using a 30-cm Spirit-Filled Lab Thermometer (Forestry Supplier, Inc., Jackson, MS, USA). Dolphin and seal mortalities deemed to be relatively fresh on the basis of minimal scavenging, orifice discharges, and bloating were placed on ice as practical, landed when the vessel returned to port, and transported by road to Woods Hole, MA, USA. They were then held at 4°C until necropsied, <12 hours after arrival.
Four animals (case Nos. 17, 18, 20, 23) were randomly selected for computed tomography (CT) scanning prior to necropsy. A beach-stranded harp seal (case No. 24: CCSN 07-116Pg) was scanned as a control. Additionally, a beach-stranded white-sided dolphin (case No. 25: CCSN 08-004La) was scanned on arrival, refrigerated for 3 days, and then scanned again to examine the possibility of decomposition gas increasing with time. A Siemens Somatom Volume Zoom 4-detector row multi slice scanner (Siemens Medical Solutions USA, Inc., Malvern, PA, USA) was used for all image acquisitions. All scans were performed in helical mode using a technique of 120 kV and 300 mAs with the animals placed in sternal recumbency and centered within the gantry. Acquisitions were reconstructed using a low-frequency image reconstruction algorithm for optimizing soft-tissue detail and a high-frequency image reconstruction algorithm for optimizing bone detail. Whole body scans and head scans (nasal planum to C2 vertebral body) were performed for each animal. For the whole body scans, 5- to 8-mm slice thickness was selected according to patient size, with matching 5- to 8-mm intervals, and a pitch (table movement during a 360° gantry rotation divided by slice thickness) of 10. For the head, images were acquired using 3-mm slice thickness with 3-mm interval and a pitch of 5. After image acquisition, analysis was performed using a dedicated workstation and efilm software (Merge Healthcare, Milwaukee, WI, USA). Soft-tissue acquisitions were viewed with a soft-tissue window and level with a window width (WW) of 350 and a window level (WL) of 90. The thorax was evaluated using the soft-tissue reconstruction algorithm acquisitions with a lung window and level (WW 1,200 and WL 600) to optimize the evaluation of lung tissue. Skeletal features were evaluated from the bone reconstruction algorithm acquisitions with a bone window and level (WW 2,500 and WL 480). All images were initially subjectively evaluated. Suspected gas bubbles and other hypoattenuating regions were then objectively evaluated by placing a region of interest within the boundaries of the area in question and measuring the Hounsfield units (HU).
Routine gross external and internal observations were made following a standard protocol, 33 including documentation of net trauma, scavenging, and other abnormalities. Particular attention was given to the presence and absence of intravascular and interstitial gas bubbles. Grossly normal and abnormal tissue samples were fixed in formalin for histopathologic examination. Fixed tissue samples were trimmed, processed routinely, sectioned at 5 µm, and stained with HE. Bubbles were scored as present or absent. Additional tissues were archived in formalin.
During this same period, an additional 41 beach-stranded seals and cetaceans, either found freshly dead or euthanatized using intravenous pentobarbital, were also examined using the same gross and histologic protocols. 14
Students t-tests were run to compare bubbled and unbubbled bycaught case groups for differences in time from collection to necropsy, mean leadline depth, surface water and core temperature, and haul rate.
Results
Figure 1 shows the locations of the bycaught cases from the Gulf of Maine. Table 1 lists the cases and their associated life history and environmental data. Case Nos. 1 to 8 exhibited no bubbles, whereas case Nos. 9 to 23 all had bubbles present to varying degrees. Table 2 summarizes the differences between the unbubbled and bubbled animals. In general, the animals with bubbles (Fig. 2) had a longer time from collection to necropsy, a faster ascent, and a warmer core temperature and were recovered from greater depths than the animals without bubbles. However, there were no statistically significant differences between the 2 groups.
Seals, dolphins, and porpoises examined after drowning in gillnets: environmental data and bubble status.
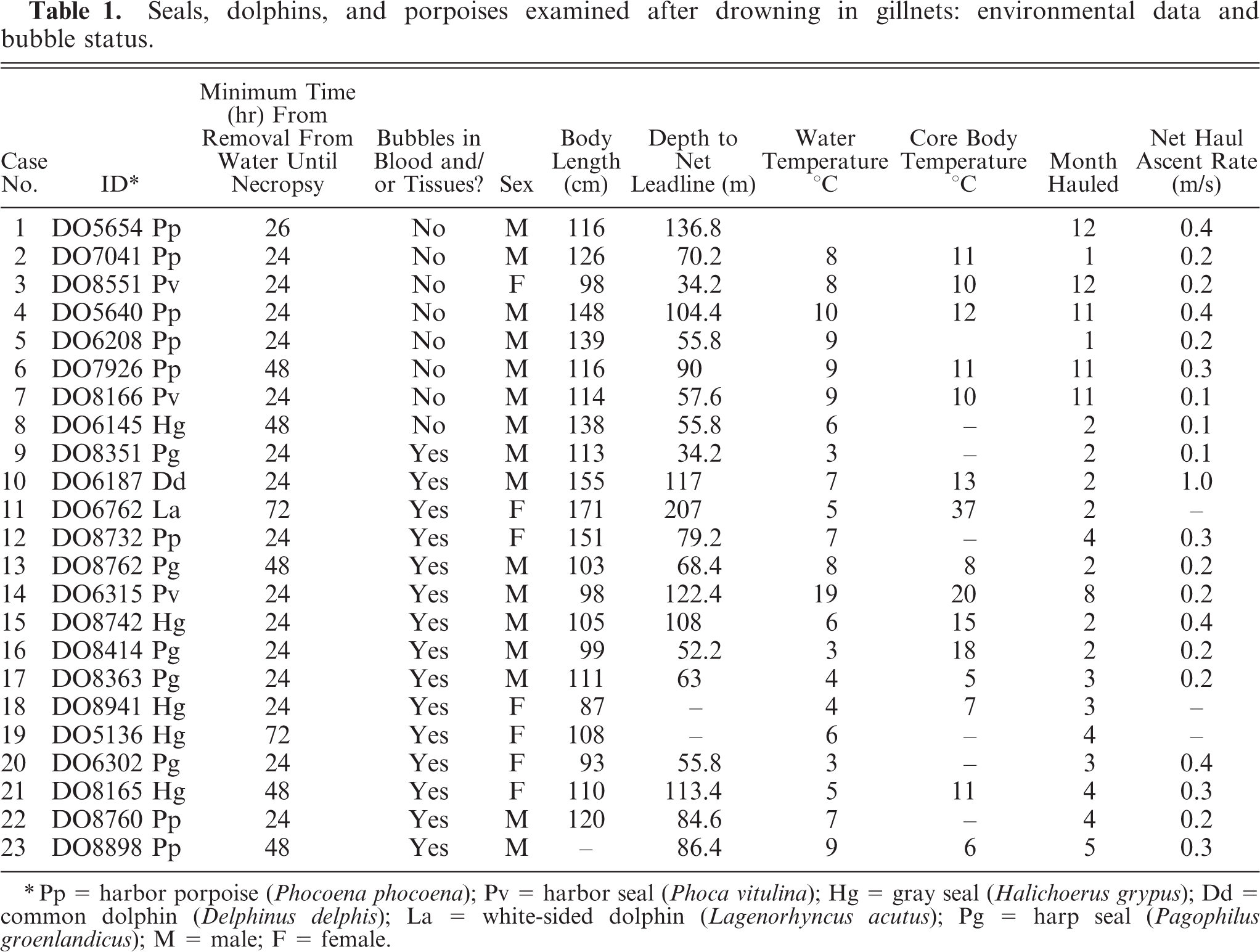
∗ Pp = harbor porpoise (Phocoena phocoena); Pv = harbor seal (Phoca vitulina); Hg = gray seal (Halichoerus grypus); Dd = common dolphin (Delphinus delphis); La = white-sided dolphin (Lagenorhyncus acutus); Pg = harp seal (Pagophilus groenlandicus); M = male; F = female.
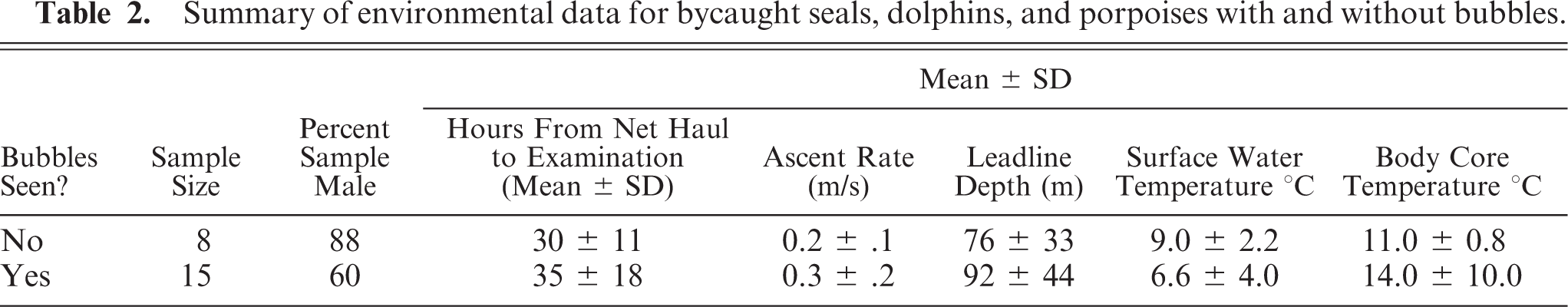
Summary of environmental data for bycaught seals, dolphins, and porpoises with and without bubbles.
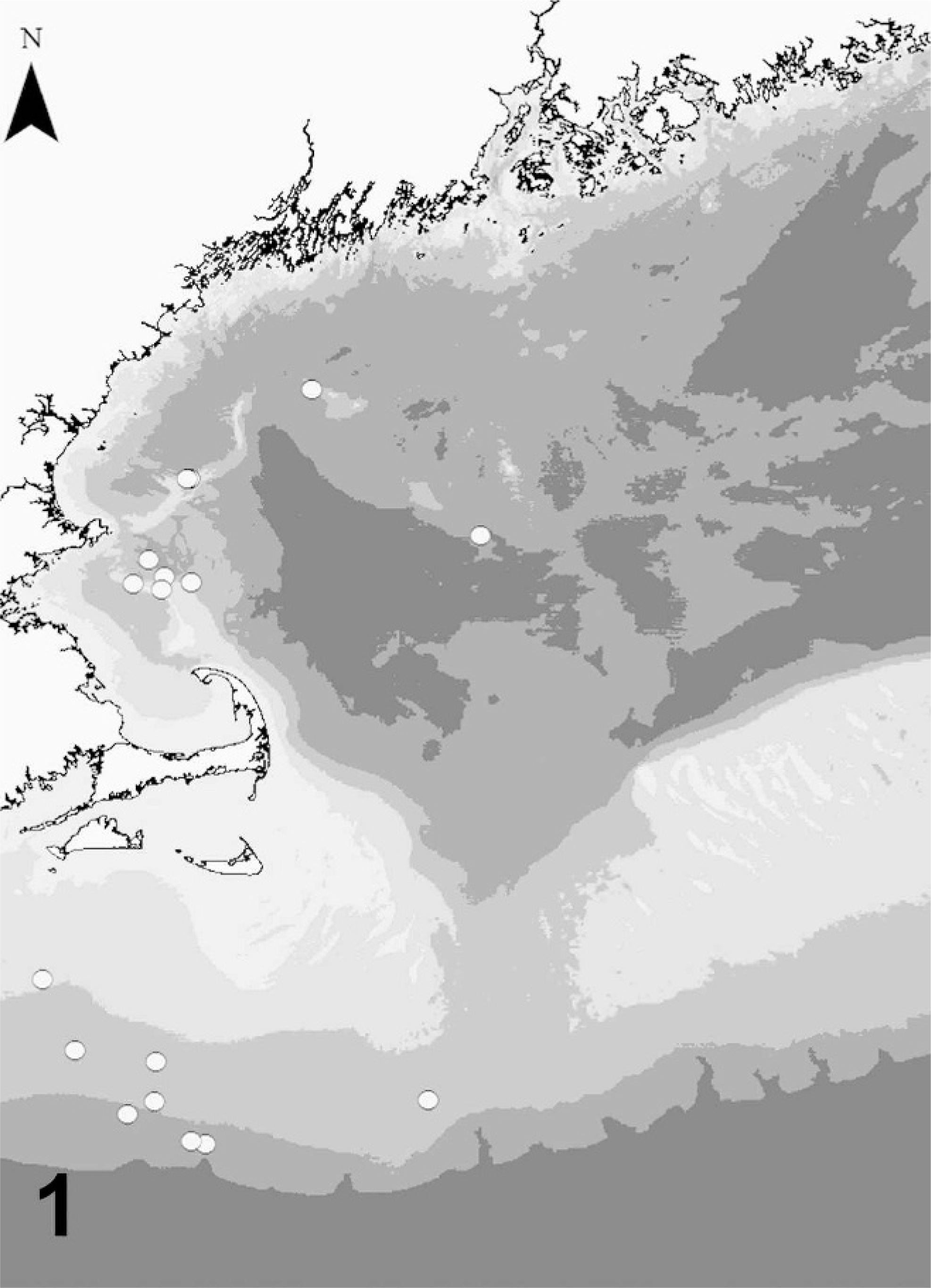
Locations of gillnetting operations in the Gulf of Maine, NW Atlantic, USA, where seals, dolphins, and porpoises were caught.
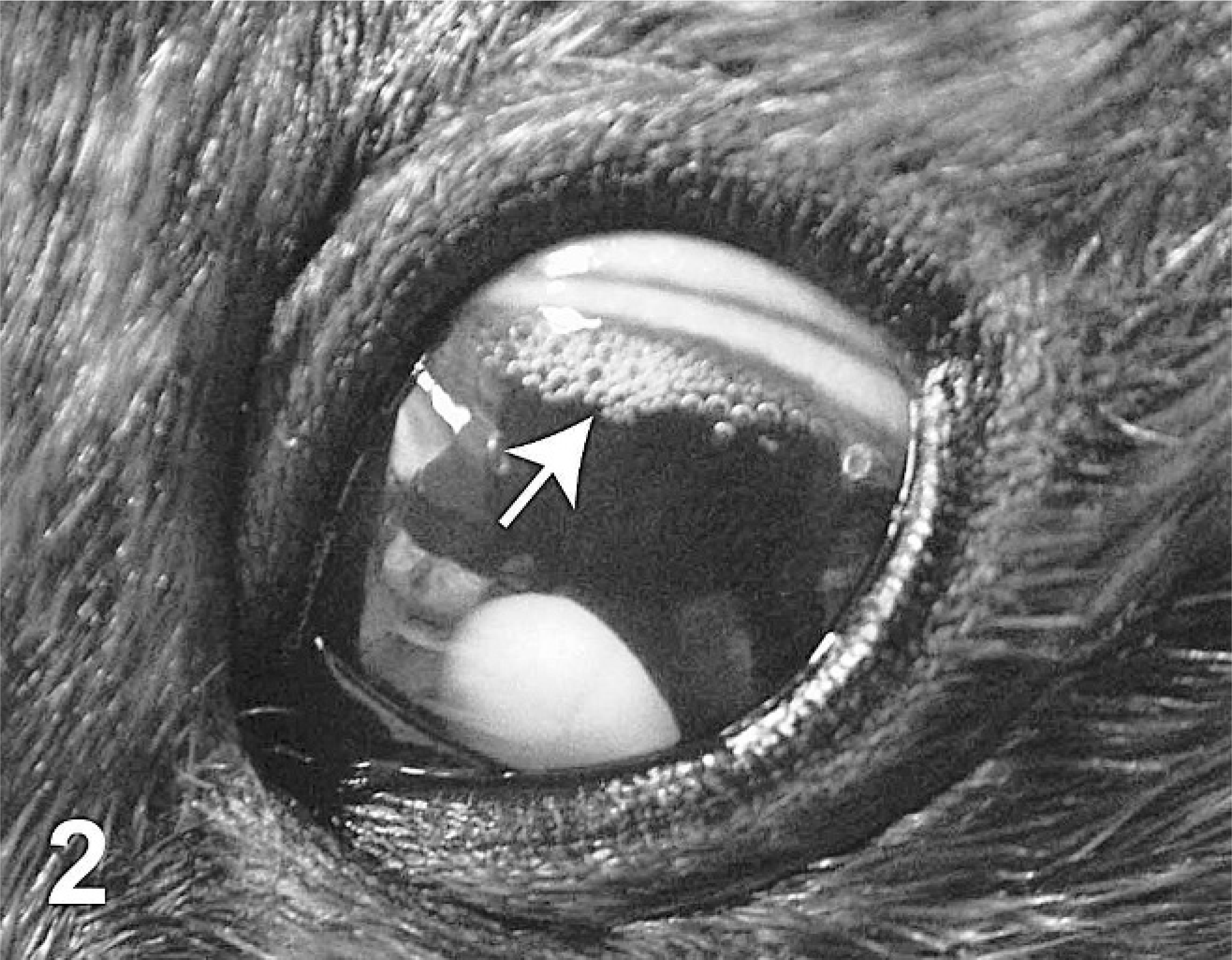
Left eye, harp seal; case No. 18. Bubbles in the aqueous humor.
Intraparenchymal and intravascular, spherical, hypoattenuating regions identified on CT images exhibited HU measurements of −900 to −1,000, levels consistent with gas (Figs. 3–4 5 6 7). Hounsfield unit levels associated with fat in the individuals studied here were confirmed by placing regions of interest over the subcutaneous blubber, which produced a mean result of −117 HU (SD 3.6) after 30 random measurements. Computed tomography of 2 reference-stranded animals (case Nos. 24 and 25) showed no bubbles in the stranded harp seal (Case No. 24) and in the stranded white-sided dolphin (Case No. 25), despite 3 days of refrigerated storage. In contrast, the 4 bycaught cases showed extensive systemic bubbles in both vascular and tissue spaces (Case Nos. 17, 18, 20, and 23), and this correlated with gross and histopathologic findings. Interestingly, in 2 cases, CT permitted identification of free gas within the abdominal cavity. Of the 41 beach-stranded animals examined by necropsy within 3 to 36 hours of discovery or euthanasia, only one case, a Cuvier's beaked whale (Ziphius cavirostris), had intravascular bubbles. Table 3 summarizes the observations of bubbles, both grossly and histologically, in the bycaught case series. Case Nos. 6, 7, and 17 to 21 were not examined histologically. Bubbles were observed in the following tissues, in decreasing order of occurrence (with number of cases showing bubbles in parentheses): blood and heart (11) (Fig. 8), lymph node (10), kidney (9), skeletal muscle (7), brain and lung (5), intestine and spleen (3), eye and pancreas (2), and liver (1). The nature of the observation in terms of gross, histologic, or both is detailed in Table 3. In all tissues, 23 cases were examined except brain (21). Gross findings included the presence of single or multiple, variably sized, clear, spherical spaces that were oval to round microscopically. Inflammation and hemorrhage were not associated with affected areas. In the skeletal muscle, myofibers were mildly compressed by a well-circumscribed, but unencapsulated, clear space. In the heart, there were multiple, variably sized bubbles within a coronary vessel and endocardium (Fig. 8).
Presence or absence of bubbles in drowned bycaught seal, dolphin, and porpoise tissues. ∗
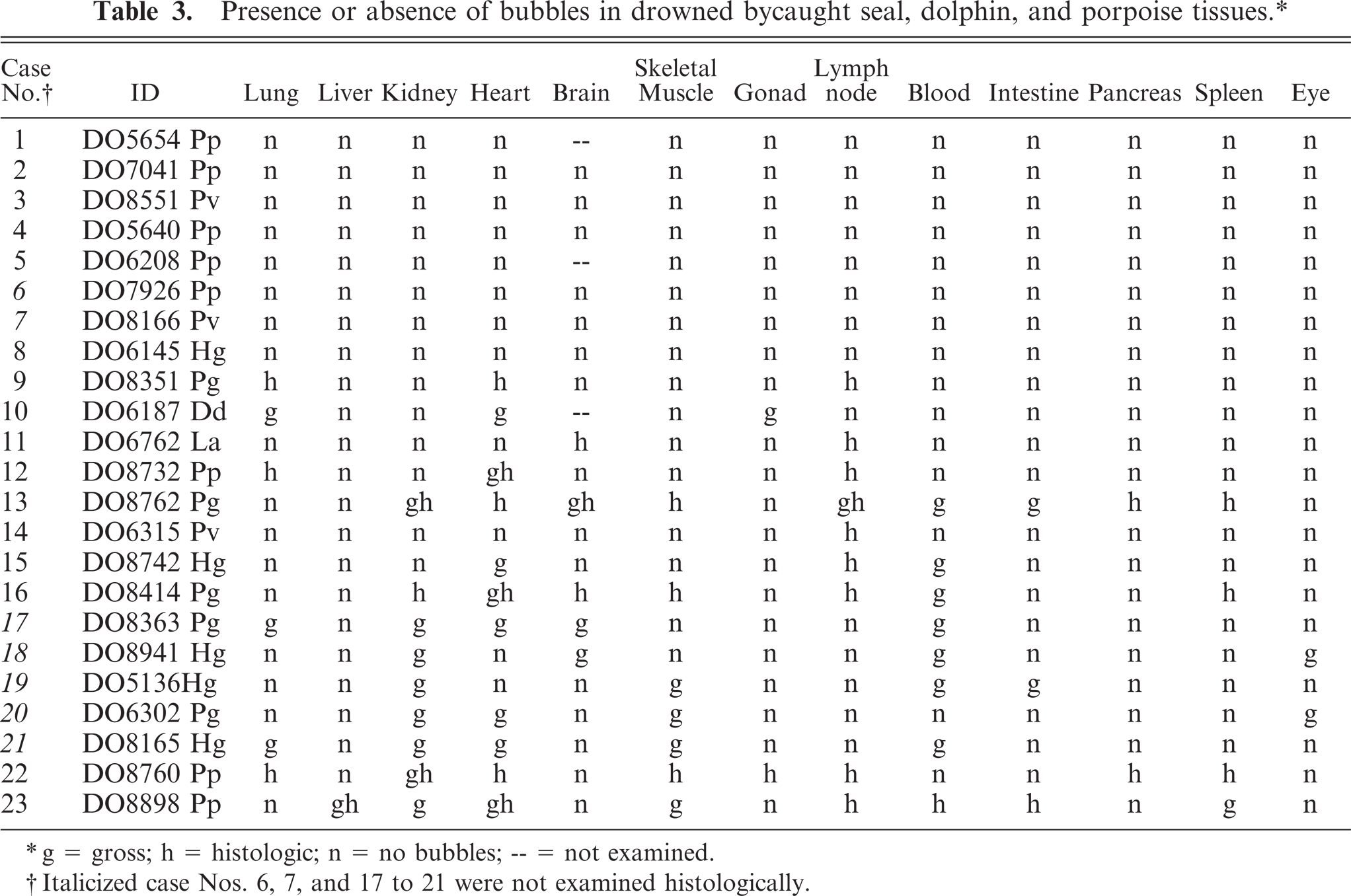
∗ g = gross; h = histologic; n = no bubbles; – = not examined.
† Italicized case Nos. 6, 7, and 17 to 21 were not examined histologically.
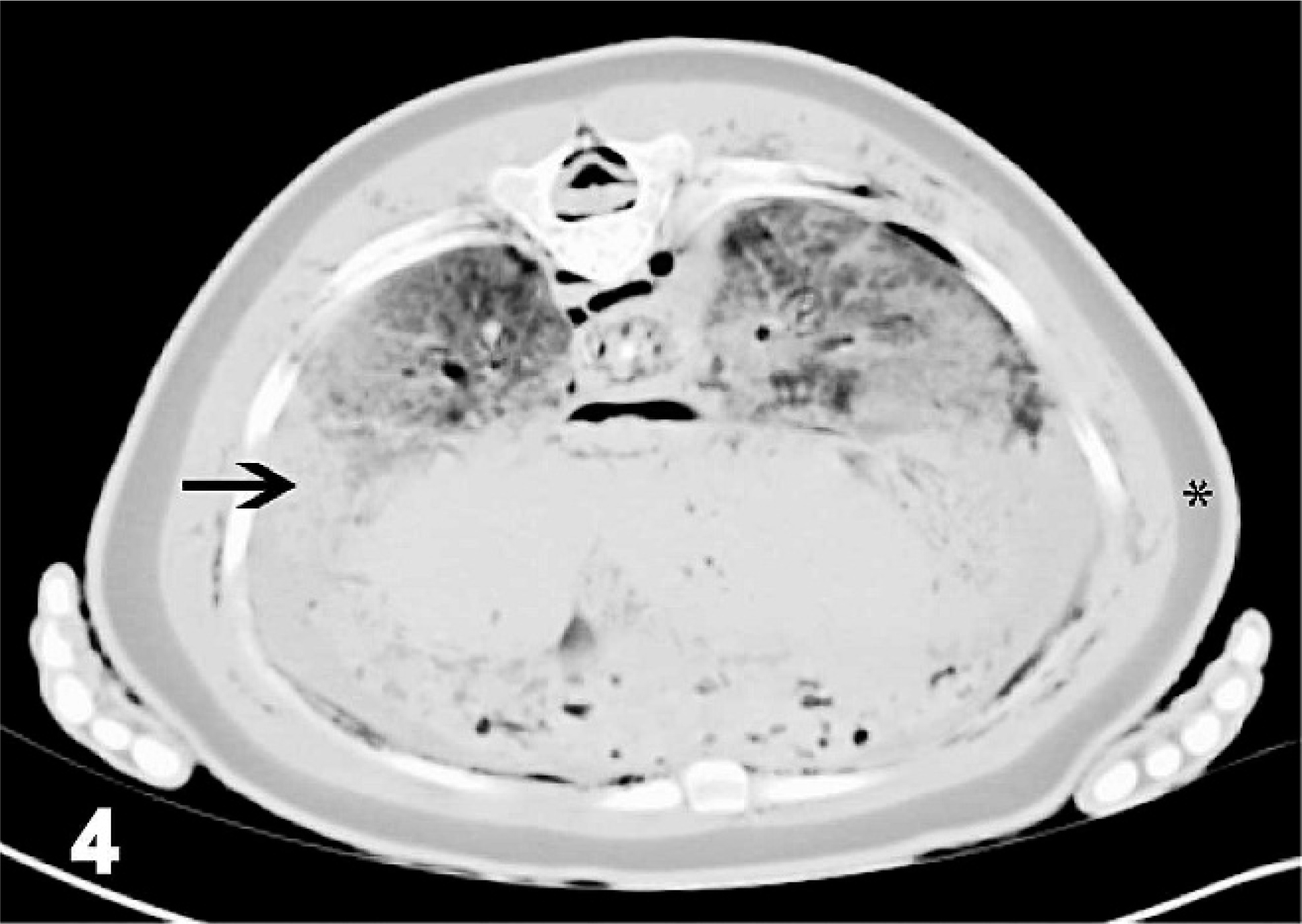
Transverse computed tomographic image of cranial abdomen, harp seal; case No. 17. Image acquired using soft-tissue algorithm, 8-mm slice thickness, and viewed with window width 1,470 and window level 684. Right is to the left of the image. Rectangle outlines vertebral canal containing gas (seen as hypoattenuating region [black]), open arrow points to gas within the large venous sinus, medium white arrow points to gas within stomach (normal), thin white arrows point to gas believed to be within the mesenteric vasculature (abnormal). Gas is also present within the small intestines (normal) and within the extrathoracic musculature (abnormal). Fish have recently been ingested and are the cause of the hyperattenuating (white) material within the stomach and caudalesophagus.
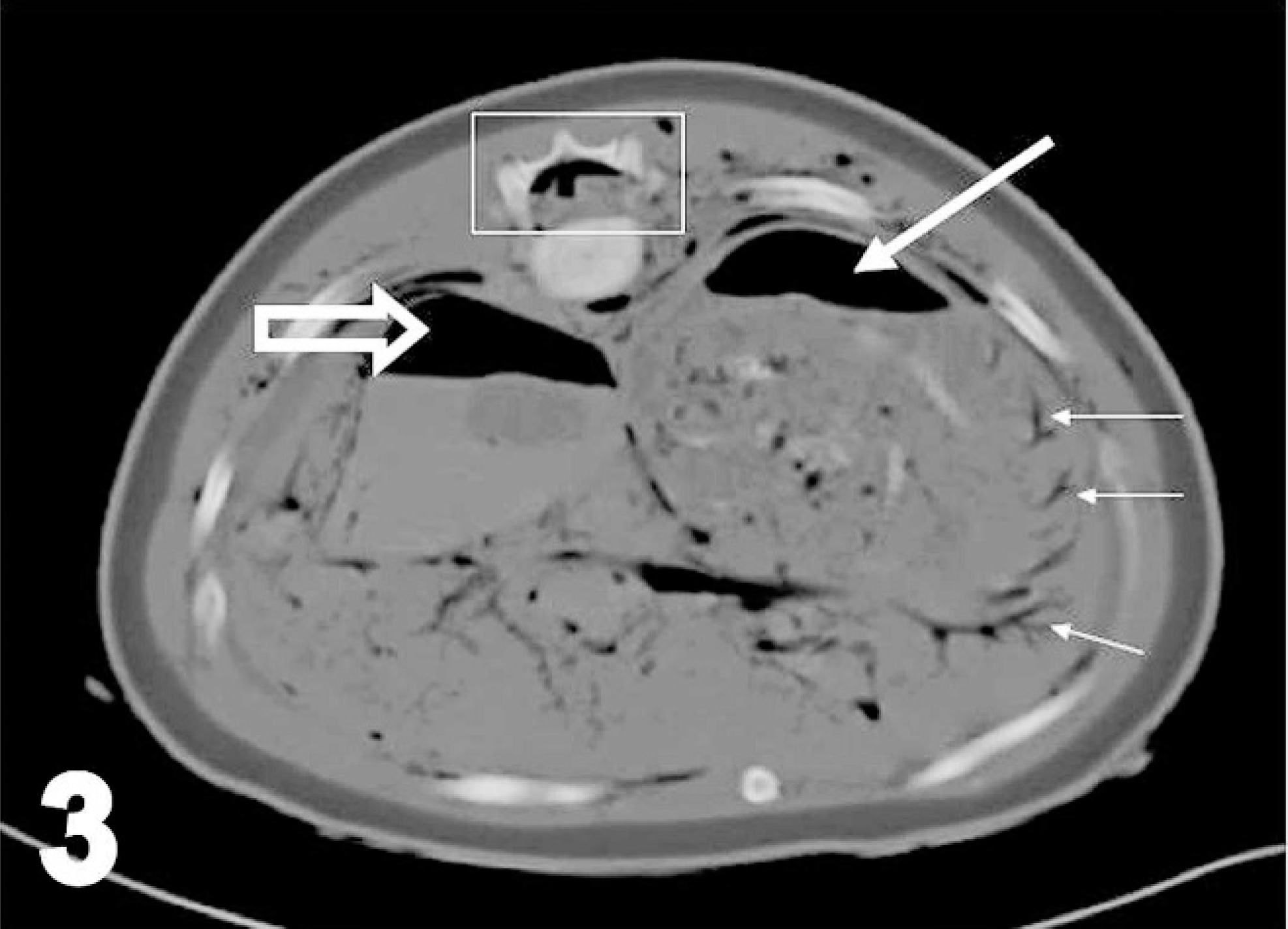
Transverse image of caudal lung fields, harp seal; case No. 17. Image acquired using soft-tissue algorithm, 8-mm slice width, and viewed with window width 463 and window level −151. Right is to the left of the image. Thick black arrow points to hyperattenuating (whiter than normal) lung tissue (differentials would include consolidation, atelectasis, and hemorrhage). ∗ indicates the subcutaneous fat layer, which had an average attenuation value of −117 Hounsfield units. Normal gas accumulations (hypoattenuating or black) are present within bronchi, and additional abnormal hypoattenuating bubbles, some confirmed and some suspected as gas, are within the vertebral canal, the mediastinum, and the extrathoracic musculature. Suspected bubbles of gas were those too small for the measurement of Hounsfield units by the placement of regions of interest.
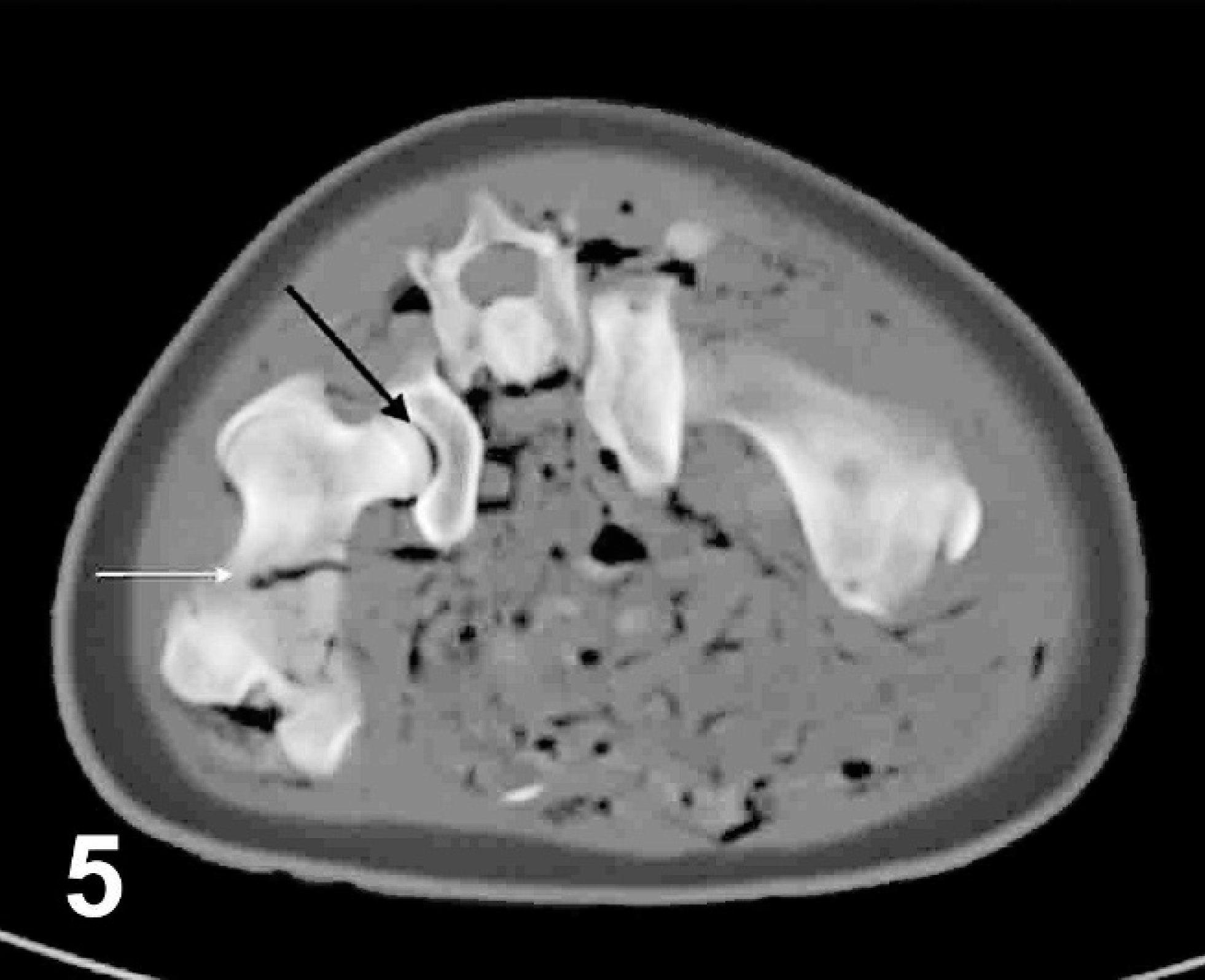
Transverse computed tomographic image of pelvis, harp seal; case No. 17. Image acquired using bone algorithm, 8-mm slice width, and viewed with window width 653 and window level 462. Right is to the left of the image. Black arrow points to gas within the right coxofemoral joint, white arrow points to gas within the femoral nutrient artery. Additional gas accumulations and discrete bubbles can be identified in the musculature (abnormal) and intestinal (normal) lumen.
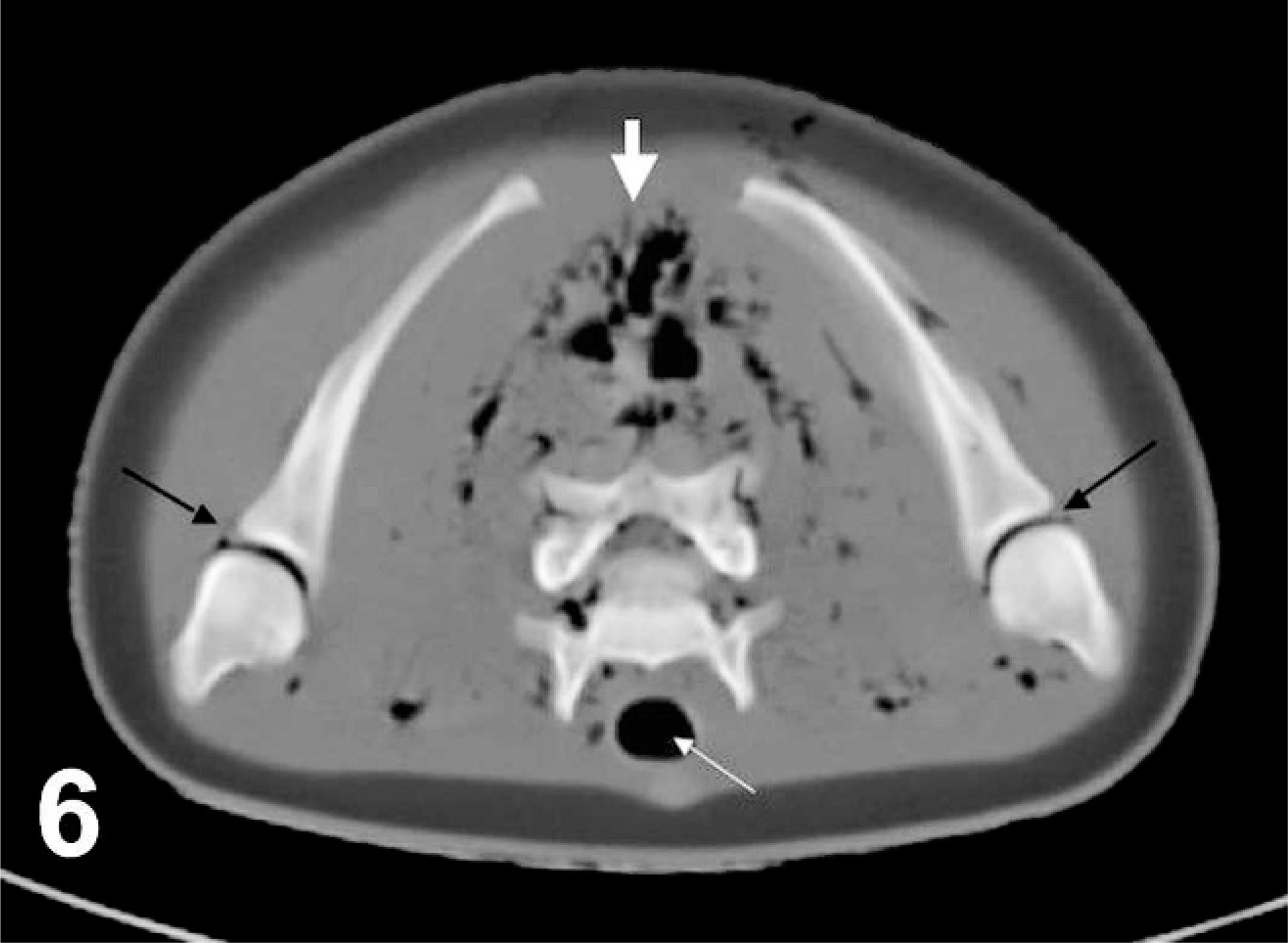
Transverse computed tomographic image of shoulders, harp seal; case No. 17. Image acquired using bone algorithm, 8-mm slice width, and viewed with window width 544 and window level 342. Right is to the left of the image. Black arrows point to abnormal gas bubbles within glenohumeral joints, thin white arrow points to normal gas accumulation within the trachea, thick white arrow points to abnormal gas bubble accumulations within the dorsal musculature. Additional abnormal gas bubbles can be identified within the vertebral canal.
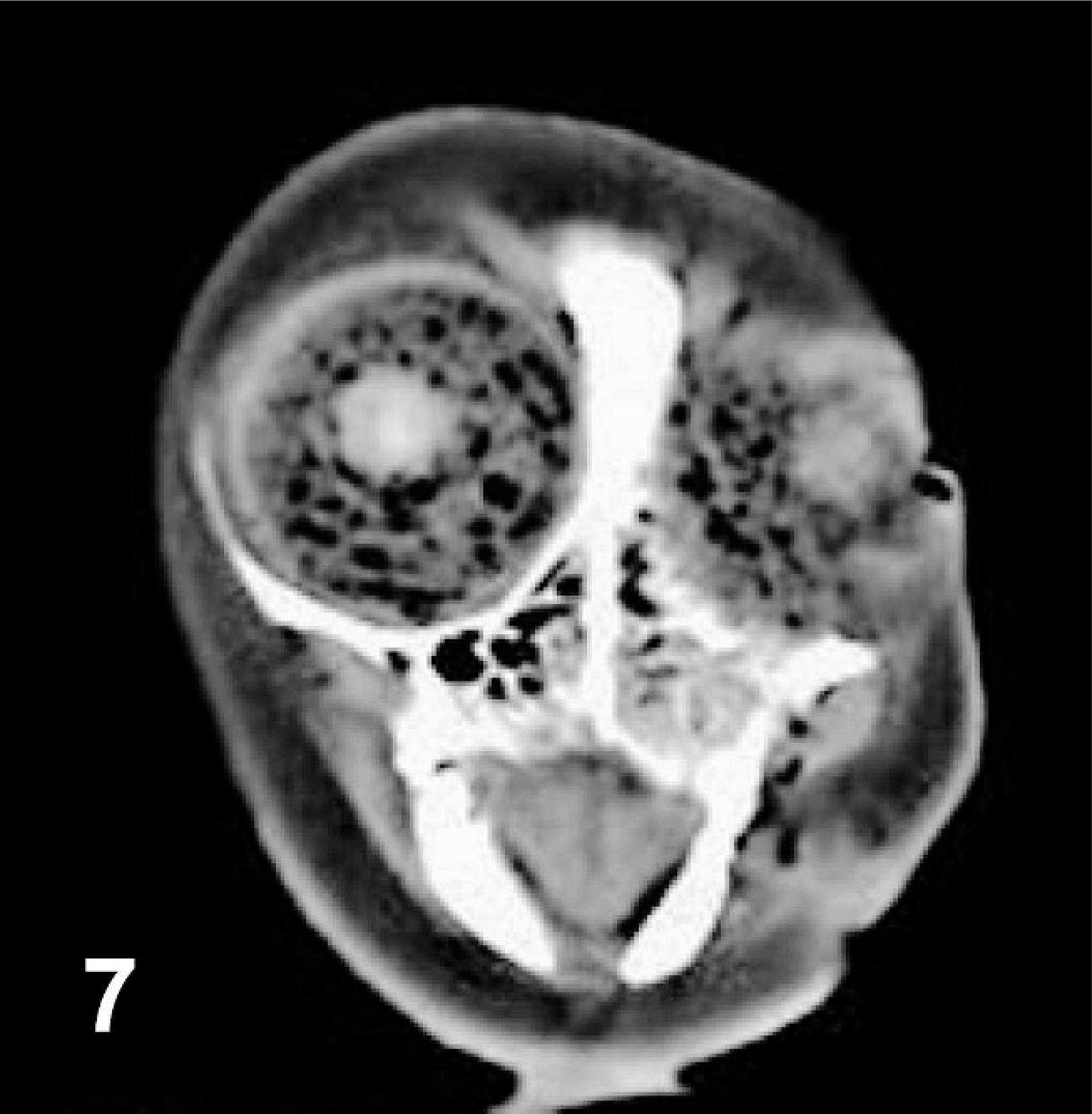
Transverse computed tomographic image of head at level of eyes, harp seal; case No. 20. Image is slightly oblique due to difficulties straightening out the cadaver for scanning. Image acquired using soft-tissue algorithm, 3-mm slice width, and viewed with window width 300 and window level 40. Right is to the left of the image. Gas bubbles are evident as round hypoattenuating structures within both eyes. Normal gas accumulation can also be identified within the nasal cavities.
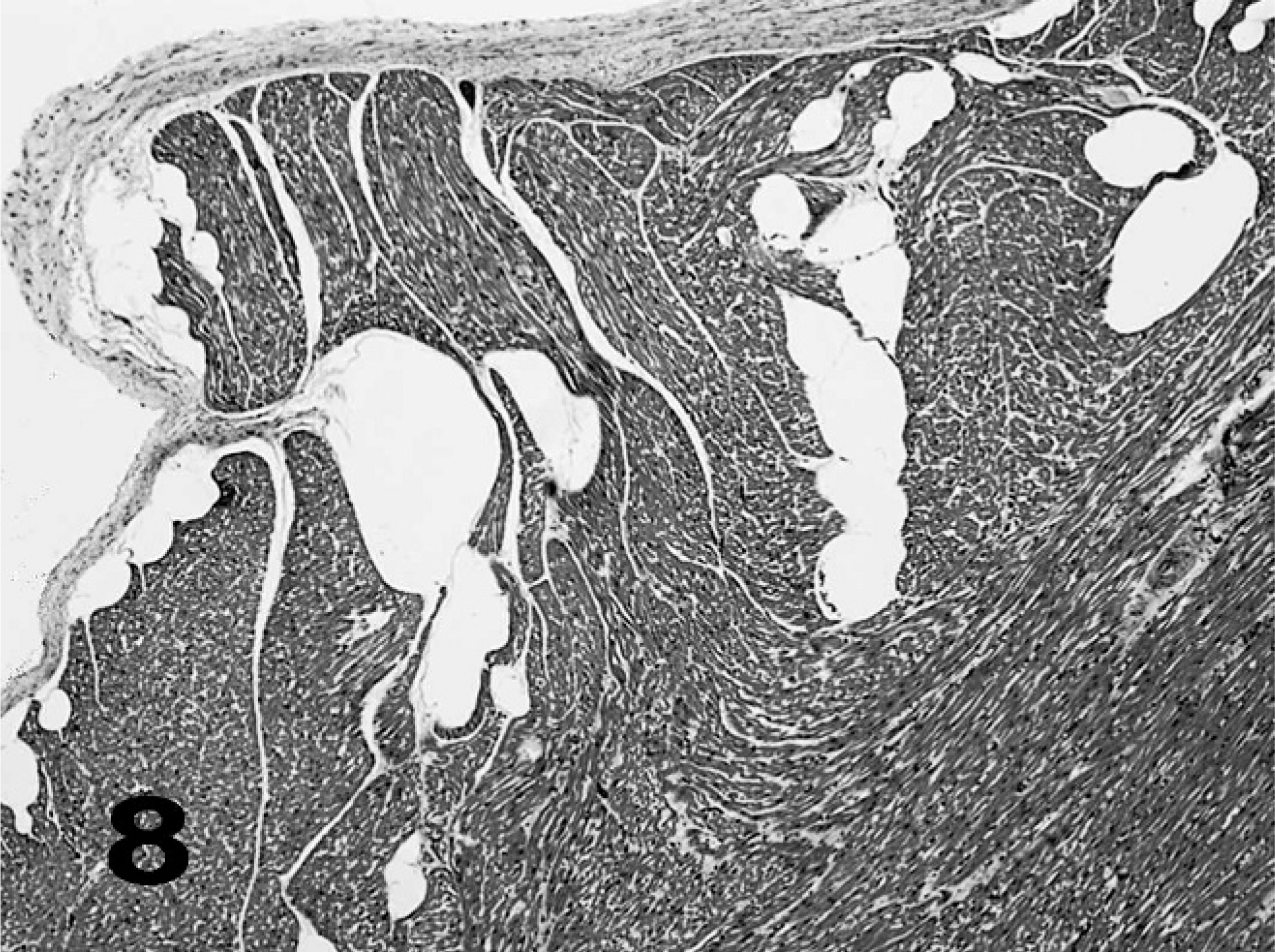
Heart, common dolphin; case No. 10. Gas bubbles (clear spaces) in endocardium and myocardium; 5-µm section, HE.
In summary, the majority (15/23) of the 23 seals and dolphins examined after drowning at depth in bottom-set gillnets had extensive bubble formation in multiple tissues and blood. Histopathologic findings associated with drowning and other sublethal chronic lesions were observed and will be reported in a subsequent publication.
Discussion
This study found a high prevalence of vascular and interstitial bubbles in seals and dolphins drowned in gillnets set at a depth of approximately 80 m. In contrast, 1 of 41 beach-stranded animals in this study and 10 of 2,376 beach-stranded marine mammals (1,970 cetaceans and 406 pinnipeds) in the UK 19 showed a very low prevalence of gas bubble lesions (−0.4%). This would seem to indicate a strong predisposition for gas bubble development in animals bycaught and recovered from deep-set ground fish gillnets.
Negative and positive findings of gas during CT imaging correlated with gross and histopathologic findings in animals that underwent all studies. Differentiation between gas and fat is important for accurate image interpretation. Thus, the HU associated with fat in these species was determined by measurement of the subcutaneous fat. The gas bubbles measured between −900 and −1,000 HU. The slight variation may have been due to the spherical nature of the bubbles permitting a small volume of other tissue to be present within the voxel being measured. Despite being represented as a 2-dimensional image, the data visualized and subsequently measured in the range of interest is in fact a volume. The addition of any other tissue into that volume will artifactually raise the HU measurement even if the pixel on the 2-dimensional image appears black. However, to have such a negative number, a majority of the voxel must be gas filled; thus, these bubbles were confirmed as gas. Another possibility for the measurement fluctuation is a small amount of image noise not discernible to the naked eye but sufficient to subtly affect the measurement of data.
Possible sources of gas bubbles include embolized inspired air forced into the circulation, gas released from supersaturated tissue, gas-producing bacteria, and the dissection procedure. Release of gas from supersaturated tissue can occur in a live or dead animal. It is also possible that some of these clear spaces included fat emboli, although the sheer extent and size of the clear spaces, absence of cellular structures (nuclei or cell membranes) in the histologic sections of the bubbles, and the HU measurements of the bubbles in the region of −900 to −1,000 HU in the CT scan images suggest that the majority of the voids, seen as hypoattenuating regions on CT, were gas bubbles.
Postmortem gas-producing bacteria are also unlikely, given the absence of bacteria in the lesions and absence of similar changes in beach-stranded animals of very comparable postmortem history, with the exception that they died at surface pressure rather than at depth. The only exception to this was one beaked whale. It is interesting that beaked whales have been shown elsewhere to be prone to bubble formation after sonar exposure. 13
The well-preserved nature of the tissues and the absence of bacteria and autolytic changes in histologic section are further evidence that the gas bubbles in these animals were not a product of decomposition. In a controlled experiment using a sheep as a model, it was reported that small gas pockets could be imaged on CT within 24 hours postmortem with no dysbaric exposure. 7 The amount of gas, however, was minimal and would likely be overlooked in dissection. The failure of case No. 25 to develop bubbles after 3 days postmortem further dilutes the concern that these bubbles could be decompositional.
Embolic barotrauma is also a highly unlikely source of the bubbles. This is mostly a problem for human divers breathing compressed gases when failing to exhale to compensate for expanding lung gas during ascent results in lung rupture and introduction of gas into pulmonary vessels.
A comprehensive set of conditions have been described that would result in air escaping from the alveolus into circulation and/or interstitial tissue. 26 The only condition that might be applicable in cases of animals drowned at depth involves damage to alveolus due to overpressurization of the thorax due to struggling and straining. Although there are instances of embolism and emphysema from rupture of alveoli documented in human cases, this is unlikely to be the cause of gas bubbles in this instance for 2 likely reasons. First, in human cases the gas released is generally minimal and is only forced through lung tissue while straining occurs. 26 Once the straining stops, so too does the escape of gas. In this instance, one can assume that violent struggling would be rather brief and the amount of damage that might be done to lung tissue would be minimal and not account for the amount of gas seen at necropsy or on CT. In dairy cattle, straining has been associated with the development of pneumomediastinum and pneumothorax. Here the alveolar rupture is associated with preexisting alveolar disease, and the volume of air entering the mediastinum and traveling along fascial planes can be substantial. Importantly, there is always communication between all affected regions rather than the diffuse distribution of gas identified in these study animals. 37 Second, animals were recovered from or below depths (approximately 70 m for dolphins and 25–50 m for seals) at which their lungs would have been fully collapsed. 12, 34 So even if struggling increased thoracic pressure, it would be unlikely to compress air trapped in alveoli.
Disease-damaged alveoli may also leak respiratory gas into the circulation and surrounding tissues. Mediastinal and interstitial emphysema have been described in marine mammals as sequelae to necrotizing pneumonias due to influenza 15 and morbillivirus. 1 This is highly unlikely in this case, because there was no evidence of viral disease or necrotizing pneumonia detected in the bycaught animals, gas bubbles were found in several species from different locations, the distribution of gas in diseased animals tends to be localized to respiratory and mediastinal tissue, and animals with severe respiratory compromise are not likely to be actively foraging at depth as were the bycaught specimens.
For the widespread systemic bubble distribution as described in this study to have occurred as a result of barotrauma, these animals would have to have been alive at the time of their ascent. In the case series, their essentially ambient core body temperature (Table 1) showed that they had (except case No. 11) died before the net was hauled. Although there is no reference data for body cooling rates for cetaceans, human data suggest that a body in water cools about twice as fast as in air (roughly 2.5°C per hour for humans) and reaches ambient temperature within 12 hours. 32 Differences in body and surface water temperatures may be explained due to death just prior to collection (such as in case No. 11); insulation of the carcasses with blubber with some postmortem decompositional heat production; warmer water temperatures at depth, which is the case for December through April; or differences in thermometers used to measure water and body temperature. Table 1 shows that the core temperatures that were significantly higher than surface water were all in the winter months. Thus, this is considered the primary factor leading to the temperature differences. When swimming, grey seals are known to ascend at 0.6 to 0.8 ms−1, 2 harbor seals at 0.6 to 3.3 ms−1, 24 and harbor porpoises at 0.5 to 1.5 ms−1. 30 The deeper they went, the faster the ascent rate. In contrast, these nets were hauled at 0.1 to 1.0 ms−1 (Table 1). Thus, the dead animals in this study were hauled at or below a normal ascent rate for a live animal.
Bowel gas is an unlikely source of the bubbles observed in these cases. Jepson, Deaville, Patterson, Pocknell, Ross, Baker, Howie, Reid, Colloff, and Cunningham review the sources of portal gas in humans in detail, and their conclusion, that an enteric source of the bubbles they described in marine mammals is highly unlikely, applies here also. 19 It is thus not a significant participant in the blood and tissue gas saturation dynamics in a diving mammal. Similarly, once the pressure has increased enough to compress the lungs such that the alveoli are devoid of gas, the lung also is no longer a source of tissue gas.
Therefore, the most likely explanation for these bubbles is peri- or postmortem off gassing of supersaturated tissues and blood, during and after the animals were hauled to the surface. This is supported by previous controlled studies in which guinea pigs drowned under pressure would bubble when returned to the surface, while those that were drowned at the surface or immediately upon reaching depth did not. 6 Although the subjects had to be exposed to increased pressure for a period of time (at least 30 minutes in this study) before death for bubbling to occur, the depth was relatively shallow and need not exceed 18 m. The rate of ascent did not seem to affect the formation of bubbles to any great degree. In a second study, sheep were euthanatized after a period of simulated diving in a pressurized air; the cadavers were then examined by sequential postmortem CT at surface pressure. 7 All the sheep developed significant quantities of gas in the vascular system within 24 hours, as demonstrated by CT and necropsy, while the control animals did not. Those authors concluded that the presence of gas in the vascular system of human cadavers after diving-associated fatalities was to be expected and is not necessarily connected with gas embolism after pulmonary barotrauma, as had previously been claimed. These experimental studies appear to accurately model what has been described in the present study and strongly suggest that the seal and cetacean species studied here were “gas loaded” from repetitive breath-hold dives to depth and therefore supersaturated at the time they were entangled in the nets and then drowned. It is also significant that breath-holding human divers can show signs of decompression sickness. 31 It is unlikely that extensive gas exchange occurred after death in the cases described here. The specific mechanisms of drowning in marine mammals are not well known, although they likely differ from man and other animals given that there is unlikely to be an involuntary inspiration of water after unconsciousness. It is unclear nonetheless how much air would be retained in the respiratory tree after death and relaxation of upper respiratory muscles. Furthermore, the depth at which many of the specimens showing the most bubbling were recovered is below depths known to fully collapse the lungs. 12, 34 Therefore, in live animals, gas exchange stops at these depths, and it is unlikely that inspired air would contribute significantly to the gas loading in dead animal tissue.
There have been inferences from blood gas measurements and modeling studies that seals and dolphins may be gas loaded to some degree or supersaturated during dives and are not free from bubble formation in tissue and blood. 12, 17, 34, 35 In one case, researchers noted that bubbles formed in blood drawn at depth when left at the surface. 12 The current study serves to support such suggestions. Given that supersaturation of tissues may be a routine occurrence in marine mammals, it is reasonable to surmise that gas emboli are also a common if not ubiquitous feature of diving mammal physiology. Such bubbles will likely risk embolic infarcts in end arteriolar, poorly collateralized tissues, such as joint bone surfaces. Thus, our findings also lend some credence to the controversial hypothesis that sperm whales at least may exhibit dysbaric osteonecrosis induced by the chronic sublethal effects of prolonged circulation of gas bubbles. 27, 36
We have described both intravascular and interstitial bubbles in extracellular locations. Such bubbles have been predicted as more likely than intracellular bubbles. 29 The bubble formation described in this study may have utility in the ongoing debate as to origin of bubbles in decompression sickness in humans. It is worth considering undertaking a series of CT scans of individual bycatch mortality cases through time to observe where the bubbles first develop and how they enlarge and are distributed. Bubbles are most likely to form in tissue where the most gas is dissolved and in areas where bubbles are likely to nucleate. The lipid content of a tissue may have a great effect, because nitrogen is >5 times more soluble in lipid than in water at 1 absolute atmosphere and 37°C. 3, 7 Factors that influence bubble nucleation are less clearly understood. Although sound impacts have been hypothesized to have the potential to cause bubble nucleation in humans and marine mammals, 10 less is known about the mechanisms inhibiting bubble formation. A single bout of strenuous activity suppresses bubble formation in humans. 40 Results from a study in rats suggest that bubble suppression may be due to increased release of nitric oxide reducing platelet aggregation and adhesion and inhibiting leucocytes. 39 This in turn reduces the overall hydrophobicity of the lining of the blood vessels and reduces bubble nuclei formation. 16 The cessation of inhibition at death undoubtedly promotes bubble nucleation and growth unchecked.
The precise events that accompany entanglement and drowning of a marine mammal in a gillnet are unknown. Presumably they struggle and swim in vain disentanglement attempts, developing an oxygen debt and lactic acid and carbon dioxide loading well beyond what they would experience in a normal dive. In fact, early experimental studies of diving physiology on marine mammals under conditions of forced submersion produced the most profound manifestations of the diving response, including extreme reduction of peripheral circulation and greatly increased lactic acid levels. 8 Further study of the dynamics of these events can shed light on the physiology of marine mammals under extreme conditions as may be encountered responding to acoustic stressors. It is interesting that these bubble lesions have not been described in bycaught marine mammals previously. 21, 23 It is likely that we were sensitized to look for these mostly subtle changes in the light of recent publications on gas bubbles in marine mammals. The bubbles were clearly evident on CT images, a technique not routinely used prior to necropsies of marine mammals. Negative and positive findings of gas during CT imaging correlated with gross and histopathologic findings in animals that underwent all studies and provided information prior to the necropsy regarding distribution of the gas bubbles. However, limitations do exist. Although CT has a high spatial resolution, the capability to differentiate between truly intraparenchymal gas bubbles and gas bubbles within the small blood vessels of the tissues is not possible. Thus, correlation with histopathology remains extremely important.
As early as 1908, 5 caution was advised in interpreting bubbles found at postmortem, stating the “the presence of bubbles in vivo must be inferred from their discovery post mortem with considerable caution. The super saturation of the body may be such that the separation of the gas bubbles may take place after death.” This caution may also be extended to the investigations of death by drowning, decompression injury, or anthropogenic sound, because there are no postmortem findings that are pathognomonic for any of these conditions and reported pathology may overlap. For example, in humans, middle ear and mastoid sinus hemorrhage has been reported in bodies recovered from water and was considered suggestive, but not proof of, barotrauma and/or drowning. 20, 25, 32 Hemorrhaging near and around ears of stranded beaked whales has likewise been reported as possibly related to exposure to sonar. 13
The present study highlights that under certain conditions considerable gas can be released from the tissue of a diving mammal. In the normal event of repeat diving and surfacing, gas supersaturation is presumably managed by offgassing at the lung at each surfacing. Where they die at depth, this cannot happen. Thus, they offgas directly to the tissues when brought to the surface postmortem. What we show here, then, is an end-stage lethal scenario and a unique visualization of the narrow physical and physiologic line that likely exists for these animals. There is not a large body of evidence of pathology due to gas loading in marine mammals, and it therefore seems prudent not to confuse such pathology with simple evidence of physiologically normal supersaturation. There is therefore a need for closer examination of animals that have died under known conditions to better understand pathophysiology of drowning and gas release in these breath-hold divers. It is also important that when gas bubbles are encountered in beached animals, serious consideration should be given to these findings and any resultant pathology, because it seems that animals that have been able to surface normally blow off supersaturated gases, and for bubbles to persist, this may represent a pathologic condition perhaps reflecting stressors that have precluded behaviors that normally manage gas tensions to keep bubble growth to a minimum.
Footnotes
Acknowledgements
We thank the many fishermen and NOAA Fisheries Observers and Observer Program staff who went to great trouble to bring these animals ashore and the Cape Cod Stranding Network for the beach-stranded cases. We also thank our colleagues of the WHOI CT scanner facility, Darlene Ketten, Julie Arruda, and Scott Cramer. We thank the Histopathology Department at the University of Tennessee, College of Veterinary Medicine Histopathology Laboratory, in particular Dee Stephenson, and the North Carolina State College of Veterinary Medicine University Histopathology Laboratory. We also thank Judy St Leger and anonymous reviewers for valuable comments. This study was funded by NOAA Grant No. NA05NOS4781247, Office of Naval Research Award No. N00014-08-1-1220, Woods Hole Oceanographic Institution Ocean Life Institute, and the MS Worthington Foundation. Samples from bycatch were collected under NOAA Permit 939-1680-00 and from stranded animals by authorization of the NOAA North East Region in Gloucester, MA, USA.
