Abstract
Paenibacilli are gram-variable, endospore-forming bacteria that occupy various ecologic niches. These microorganisms have been known to infect humans occasionally at various anatomic sites. However, in humans, as well as in other vertebrate animals, the relationship between disease and isolation of Paenibacillus spp. remains poorly understood. We report here a case of infection in an adult Poodle dog. The animal had nodules in the lungs and multifocal osteolytic expansile bone lesions. From bone, Paenibacillus amylolyticus was recovered by culture and identified by MALDI-TOF mass spectroscopy and 16S rDNA sequencing; pyogranulomatous inflammation was observed in lung and bone specimens. The microorganism was resistant to clindamycin and imipenem. Four-month treatment with amoxicillin–clavulanate resulted in clinical resolution of disease in this dog. Nevertheless, therapy for more prolonged periods should be considered because recurrent infections can occur as a result of the transition of Paenibacillus spores to vegetative cells. Disease caused by a Paenibacillus species has not been reported previously in dogs, to our knowledge.
Genus Paenibacillus comprises a wide variety of known or emerging soil-related bacteria that are pathogenic for animals and humans. 15 Bacteria belonging to this genus are rod-shaped, gram-positive or gram-variable, endospore-forming, aerobic or facultatively anaerobic. 3 Originally classified as a Bacillus group 2 and reassigned to the novel genus Paenibacillus in 1993, 3 the number of novel species being identified as Paenibacillus has been increasing, and taxonomic subdivision continues to change. The genus includes beneficial bacteria isolated from the plant endosphere and known as plant growth–promoters.25,29 Some species produce antimicrobial agents, particularly polymyxins,9,30 enzymes, and exopolysaccharides of industrial interest used for biomass and chemical degradation and waste management.1,36,38 However, a large number of paenibacilli have been found to be responsible for occasional local and systemic infections in humans, especially those who are immunocompromised.27,28
In honeybee larvae, Paenibacillus larvae causes lethal intestinal infection of considerable relevance, known as American foulbrood. 11 Paenibacillus infections have not been reported in non-human vertebrates. To our knowledge, Paenibacillus spp. have not been reported in canine infectious diseases. Here, we present a case of canine infection caused by Paenibacillus amylolyticus. As well, we reviewed the available literature on Paenibacillus infections by searching the electronic databases PubMed, Google Scholar, and Scopus for original research articles and reviews using the terms Paenibacillus, Paenibacillus infection, Paenibacillus amylolyticus, Paenibacillus bacteremia, Paenibacillus sepsis, canine Paenibacillus, Paenibacillus and dog, and veterinary Paenibacillus.
A 4-y-old male Poodle dog was presented to a veterinary clinic in Tuscany (Central Italy) because of weight loss, lethargy, and back pain. The past medical history only included removal of dental plaque 6 mo before the onset of the presenting signs. Physical examination confirmed thoracolumbar spine pain, which was initially assumed to be associated with a recent episode of traumatic pressure mentioned by the dog owner. Spinal column radiography revealed no morphologic abnormalities of the thoracolumbar spine and surrounding soft tissues. CBC indicated a mild reduction of RBCs (5.2 × 1012/L; RI: 5.8–9.0 × 1012/L) and neutrophilia (13 × 109/L; RI: 2–12 × 109/L). A NSAID was prescribed.
Approximately 3 wk later, the dog showed no response to the treatment, but the presenting clinical signs persisted along with fever. Moreover, lameness and tumefaction had developed in the right hindlimb. Capillary electrophoresis of serum proteins was performed. A low albumin:globulin ratio was found (0.20; RI: 0.95–1.41) because of marked hypoalbuminemia (10.5 g/L; RI: 31–39 g/L) and values of α-, β-, and γ-globulins of 21.3 g/L (RI: 11–18 g/L), 16.3 g/L (RI: 8–18 g/L), and 14.4 g/L (RI: 3–11 g/L), respectively. CBC indicated severe anemia (RBCs 3.9 × 1012/L, RI: 5.8–9.0 × 1012/L; Hct 0.30 L/L, RI: 0.37–0.54 L/L; hemoglobin 111 g/L, RI: 112–184 g/L; MCV 78 fL, RI: 56–72 fL), marked neutrophilia (17 × 109/L; RI: 2–12 × 109/L), and a high-normal concentration of platelets. One week later, the platelet count had increased to 778 × 109/L (RI: 175–500 × 109/L).
Radiography revealed an osteolytic expansile lesion in the proximal end of the right tibia (Fig. 1A), which was confirmed by total body computed tomography (CT) performed without contrast and after administration of an iodine-based contrast medium. The CT scan revealed multifocal osteolytic expansile osteopathy, including lysis of cancellous bone and rarefaction of cortical bone of the T12 vertebral body (Fig. 1B), and severe bilateral osteolysis of the distal femoral epiphysis and proximal tibial epiphysis. The right proximal tibia had an irregular periosteal reaction. This was observed also in the ischium and, to a lesser extent, in the humerus and scapula. Numerous nodules of similar size were found in the lungs (Fig. 1C). Vertebral sarcoma with bone and pulmonary metastasis; multicentric neoplasia such as multiple myeloma, lymphoma, or histiocytic sarcoma; pulmonary carcinoma with lung and bone metastasis; and hematogenous osteomyelitis also involving the lungs, were considered in the differential diagnosis.
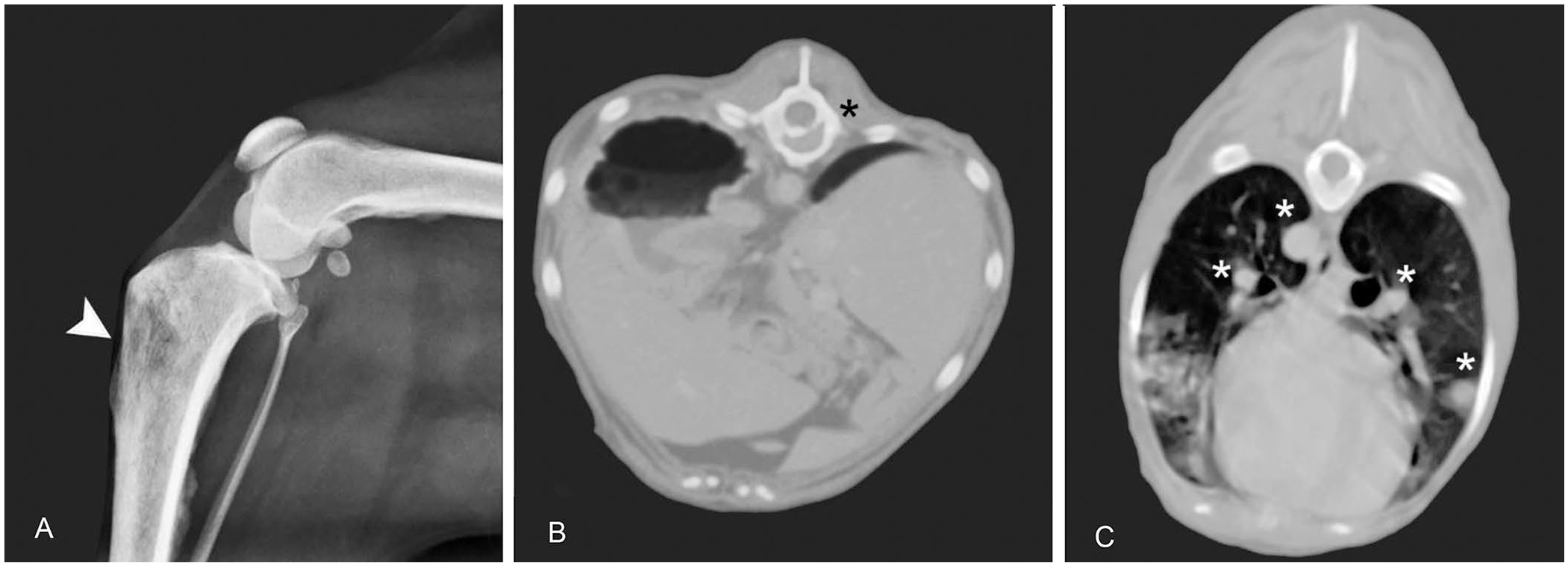
Radiography and computed tomography (CT) of Paenibacillus lesions in a dog.
Lung and bone samples were collected for laboratory investigation. Lung nodules were sampled by ultrasound-guided, fine-needle aspiration for cytologic examination using Romanowsky stain; bone samples were obtained from the osteolytic lesion in the proximal end of the right tibia using a Jamshidi needle and processed for bacterial culture and histopathology. Bone biopsies for histopathology were fixed in 10% neutral-buffered formalin; 4-μm thick sections were stained with H&E. PAS and Gram stains (Bio Optica) were subsequently performed to identify fungal or bacterial agents.
Inflammatory cell infiltrates consisting primarily of neutrophils and macrophages or epithelioid cells were observed on lung cytology (Fig. 2A); the same features were seen in bone histologic samples, characterized by neutrophilic and macrophage infiltrates with a small number of lymphocytes and plasma cells intermingled with small areas of hemorrhage and necrosis (Fig. 2B). Based on the cytologic and histologic observations, chronic pyogranulomatous osteomyelitis and pneumonia were diagnosed.
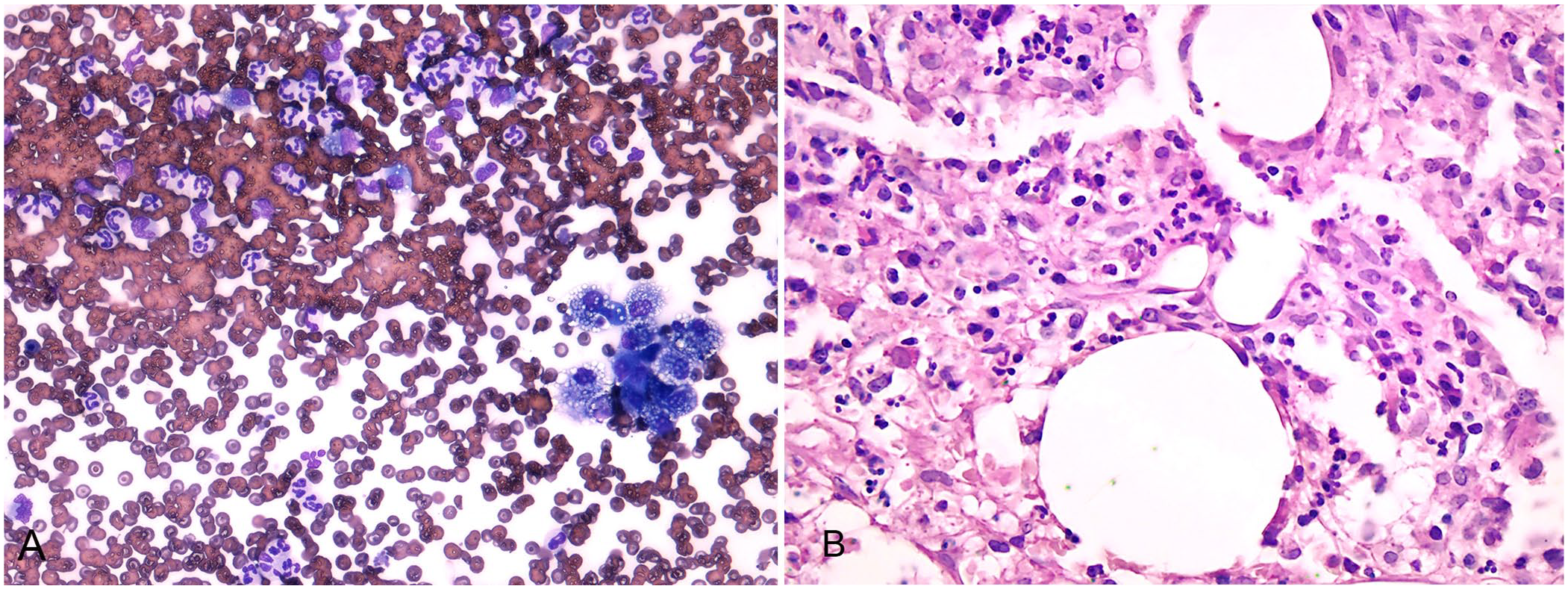
Cytologic sample of lung and biopsy of tibial bone.
For microbiologic analysis, the bone biopsy was homogenized in sterile saline. The suspension and its dilutions were seeded onto 5% defibrinated sheep blood agar, mannitol salt agar, and McConkey agar (Liofilchem). The plates were incubated aerobically at 37°C for up to 5 d. For the anaerobic culture, the plates were incubated anaerobically at 37°C for 5 d; no anaerobes were isolated. Aerobic bone cultures grew pure, non-hemolytic, gray-white, translucent, smooth, and flat colonies on 5% sheep blood agar. The colonies were visible 48 h after the start of incubation. The bacterial burden did not exceed 5 × 102 cfu/g. Microscopically, long, gram-variable rods were observed (Fig. 3A). The Schaeffer–Fulton method differentiated the green endospores from red vegetative cells (Fig. 3B). The bacterium was identified as Paenibacillus amylolyticus by matrix-assisted laser desorption/ionization time-of-flight mass spectroscopy (MALDI-TOF MS Biotyper, v.4.1.100; Bruker). The Biotyper was able to identify the isolate to the species level based on standard manufacturer interpretive criteria (triplicate score values of ≥2.00).
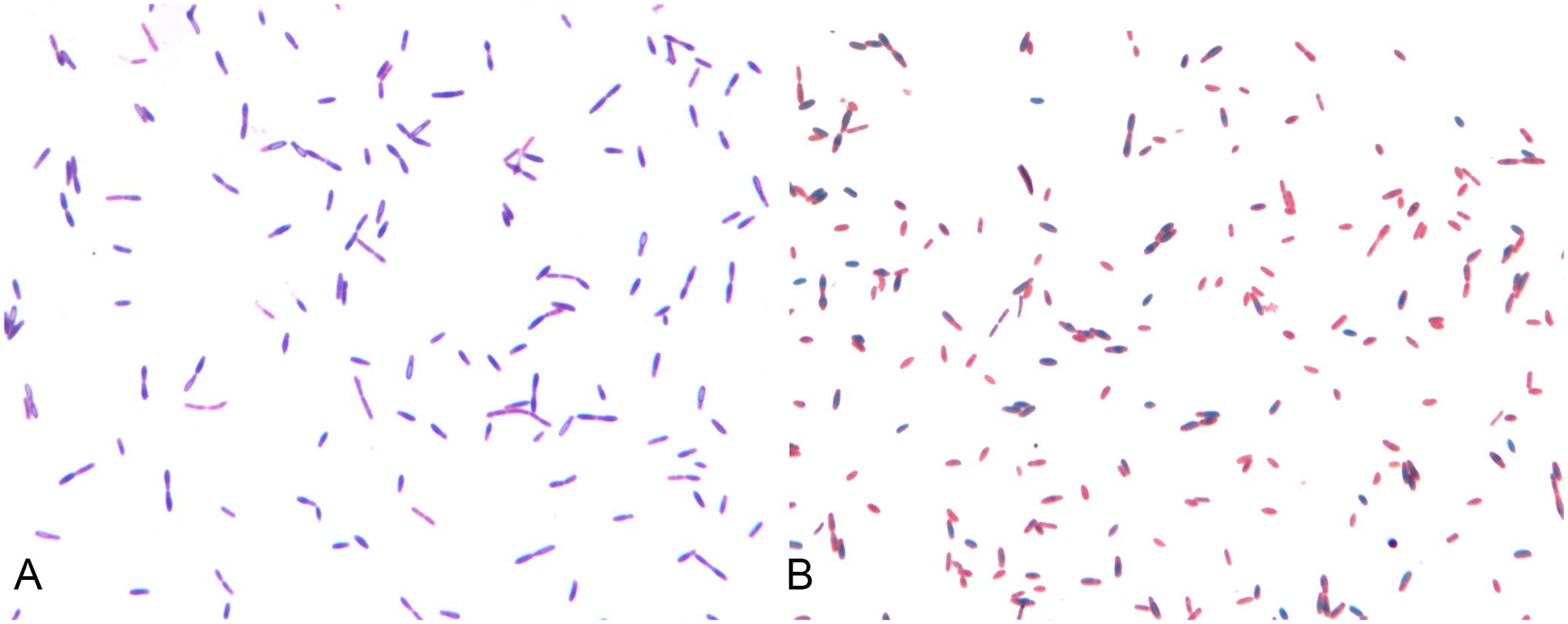
Microscopic images of Paenibacillus amylolyticus from culture plate.
Moreover, total DNA was extracted (Quick-DNA miniprep kit; Zymo Research), and amplification of bacterial 16S rDNA was performed using universal primers at 0.4 µM (Bio-Fab Research). 20 The PCR product of 466 bp was purified and submitted for sequencing. Analysis of the consensus sequence using BLAST (http://blast.ncbi.nlm.nih.gov/Blast.cgi) found 99.78% identity to P. amylolyticus. Antimicrobial susceptibility testing was performed by disk diffusion on cation-adjusted Mueller–Hinton agar with 5% sheep blood (Liofilchem) for fastidious growth. 6 Inhibition zone diameters were interpreted based on CLSI guidelines for Bacillus spp. 7 The bacterium was susceptible to most antibiotic classes, but it was interpreted as resistant to clindamycin and imipenem (Table 1).
Antimicrobial susceptibility testing by disk diffusion for Paenibacillus amylolyticus.
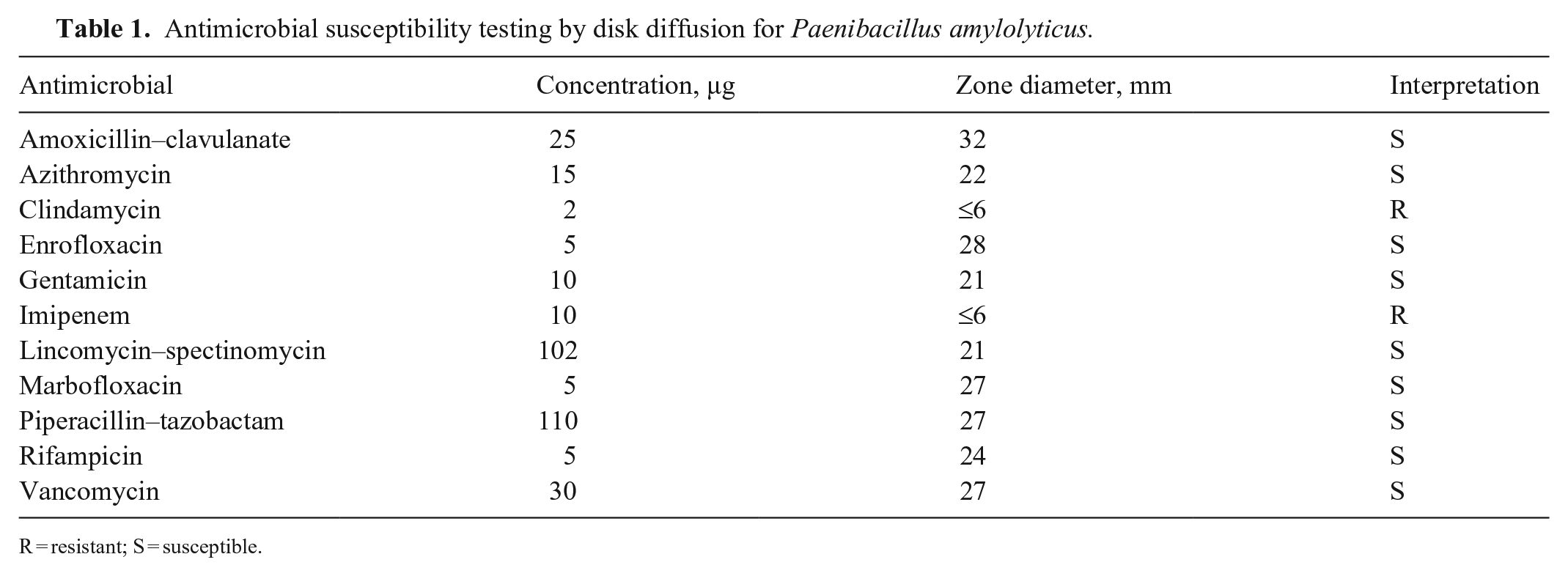
R = resistant; S = susceptible.
Once antimicrobial susceptibility results became available, amoxicillin–clavulanate therapy was administrated at a dosage of 20 mg/kg q12h PO for 4 mo in combination with a proton pump inhibitor for gastric protection. After 3 mo of treatment, clinical improvement of the dog was evident on physical examination; body condition was ideal, and mentation and body temperature were normal. Pulmonary lesions were reduced by 90% on follow-up CT, which also suggested remission of the osteomyelitis with signs of bone remodeling and absence of pain. At the end of treatment, a CBC showed that RBCs and neutrophils fell within their RIs, and the platelet count had decreased to 562 × 109/L (RI: 175–500 × 109/L), indicating resolution of the infection.
Paenibacillus spp. have been isolated from various ecologic niches, being ubiquitous in the environment. P. larvae, the etiologic agent of American foulbrood, is the most pathogenic Paenibacillus in honeybees. Some species are also known to infect humans as opportunistic pathogens, with Paenibacillus alvei proposed as the most common. 34 Diseases caused by Paenibacillus spp. have not been reported previously in dogs and other companion animals, to our knowledge. In humans, as well as in other vertebrates, the relationship between disease and isolation of Paenibacillus thus remains poorly understood.
It has been hypothesized that Paenibacillus can occupy suitable anatomic niches opportunistically. 15 Such niches includes the urinary tract,23,26 cornea, 16 nervous system, 26 joints, 37 skin and soft tissues,8,10,13 and lungs and bone. 22 Paenibacillus spp. were also the dominant bacterial pathogen in Africa in hydrocephalic infants, with frequent viral coinfection. 18 In our case, the isolation of P. amylolyticus from bone, neutrophil detection, and observation of a lymphocyte-macrophage-plasma cell infiltrate met the diagnostic criteria for bacterial osteomyelitis,32,35 even though bacteria were not detected in histopathology sections. As a rule, bone infections are characterized by a low pathogen count in samples, limiting the direct observation of pathogens in bone tissues. 32 The lung specimen was too small to be cultured. Therefore, the diagnosis of P. amylolyticus pneumonia was only presumptive, based on the presence of pyogranulomatous nodules in the lungs. From bone, P. amylolyticus was correctly identified by MALDI-TOF MS at scores indicating species-level accuracy and confirmed by 16S rDNA sequencing. Previous versions of the MALDI-TOF MS system failed to identify Paenibacillus strains or were unable to identify P. amylolyticus at the species level, likely because of the lack of uncommon bacterial species in previous databases. 5 As a result, the clinical relevance of certain Paenibacillus species from human and animal clinical samples might have been underestimated and the antimicrobial treatment misled.
Paenibacillus spp. are characterized as poor biofilm formers. 17 Nevertheless, they have been implicated in human infections related to medical devices,12,19 which are usually associated with microbial biofilms. 4 An intravenous injection has also been reported as a source of Paenibacillus infection. 34 Our case occurred without a history of any implantation or intravascular catheter insertion. Therefore, the origin of P. amylolyticus penetration and infection in the Poodle remains unknown. However, considering the multifocal nature of P. amylolyticus infection, we suspect that this might have been secondary to bacteremia, hypothetically from poor dentition, despite the long time that had elapsed between removal of dental plaque and the occurrence of clinical signs. A human case report proposed that an undrained dental abscess could have been a source of persistent bacteremia by P. amylolyticus and Lysinibacillus. 34 Paenibacilli were also isolated from the subgingival dental plaque of a human gingivitis lesion 24 and the oral cavity of a healthy dog, 14 further supporting our hypothesis. Paenibacillus spp. tend to infect immunocompromised people and elderly patients. 15 In our case, P. amylolyticus infected an adult dog in the absence of previous or concomitant immunosuppressive therapies and known immunodeficiency disorders.
Innate and acquired antimicrobial resistance in Paenibacillus spp. has been addressed briefly in the literature. Paenibacilli isolated from humans have demonstrated a variety of strain-dependent antimicrobial resistance profiles. P. amylolyticus in our case was susceptible to most antimicrobial classes tested, but it was resistant to clindamycin and imipenem. Resistance or low susceptibility of Paenibacillus spp. to clindamycin22,31,33 and lincomycin 21 has been reported from various hosts and sources, suggesting that intrinsic resistance to lincosamides may exist, although the underlying mechanisms remain to be explored.
An important question to address for future studies is also the presence of virulence properties in Paenibacillus spp. In the literature, there are a few examples of the pathogenic potential of Paenibacillus spp., including a human P. amylolyticus isolate that secreted proteases and harbored genes nheA/C encoding for non-hemolytic enterotoxins. 5 Revealing the production of toxins and other virulence proteins by Paenibacillus spp. may provide additional support for the opportunistic pathogenic behavior of these bacteria.
Long-term treatment of our case with amoxicillin–clavulanate resulted in clinical resolution of disease. Nevertheless, therapy for more prolonged periods should be considered in the event of Paenibacillus involvement because infections can recur as a result of the transition of Paenibacillus spores to vegetative cells. 31
Footnotes
Acknowledgements
We thank Drs. Chiara Francesca Magistrali, Francesca Blasi (Istituto Zooprofilattico Sperimentale Umbria e Marche “Togo Rosati”), Giovanni Angeli (Department of Veterinary Medicine, University of Perugia), and Amato Violini (independent veterinary practitioner).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
