Abstract
Mycoplasma hyopneumoniae has a primary role in the porcine respiratory disease complex (PRDC). The objective of this study was to determine whether fumonisin mycotoxins influence the character and/or the severity of pathological processes induced in the lungs of pigs by Mycoplasma hyopneumoniae. Four groups of pigs (n = 7/group) were used, one fed 20 ppm fumonisin B1 (FB1) from 16 days of age (group F), one only infected with M. hyopneumoniae on study day 30 (group M), and a group fed FB1 and infected with M. hyopneumoniae (group MF), along with an untreated control group (group C). Computed tomography (CT) scans of infected pigs (M and MF) on study day 44 demonstrated lesions extending to the cranial and middle or in the cranial third of the caudal lobe of the lungs. The CT images obtained on study day 58 showed similar but milder lesions in 5 animals from group M, whereas lungs from 2 pigs in group MF appeared progressively worse. The evolution of average pulmonary density calculated from combined pixel frequency values, as measured by quantitative CT, was significantly influenced by the treatment and the age of the animals. The most characteristic histopathologic lesion in FB1-treated pigs was pulmonary edema, whereas the pathomorphological changes in Mycoplasma-infected pigs were consistent with catarrhal bronchointerstitial pneumonia. FB1 aggravated the progression of infection, as demonstrated by severe illness requiring euthanasia observed in 1 pig and evidence of progressive pathology in 2 pigs (group MF) between study days 44 and 58.
The porcine respiratory disease complex (PRDC) mainly affects growing and finishing pigs and reduces animal growth and profitability. 3,29,37 It is caused by the combined effects of multiple pathogens and predisposing environmental factors. Mycoplasma hyopneumoniae was first isolated in the 1960s from pigs suffering from respiratory disease and later became known as enzootic pneumonia of pigs. 13,19 Subsequently, M. hyopneumoniae was identified as one of the major causative factors in PRDC.
Among the predisposing environmental factors reported for PRDC, mycotoxins present in the diet may exert a deleterious effect on the health of pigs already infected with M. hyopneumoniae. Fumonisins, such as fumonisin B1 (FB1), fumonisin B2 (FB2), fumonisin B3, (FB3), and so on, are secondary metabolites of the mold species, Fusarium verticillioides (Sacc.) Nirenberg (previously called Fusarium moniliforme Sheldon), 12 which may occur in considerable amounts in various grains (particularly maize) all over the world. 7 The mycotoxin FB1 is considered responsible for inducing porcine pulmonary edema 15 and pulmonary fibrosis that develops in cases of chronic exposure. 41 It may cause immunosuppression 20,33 and has been associated with secondary infections, including intestinal colonization by pathogenic Escherichia coli, 22 or respiratory pathogenic agents including Pasteurella multocida 14 and porcine reproductive and respiratory syndrome (PRRS) virus. 25
Few studies have addressed the combined effect of FB1 and respiratory pathogens in pigs. In one experiment, 25 more severe lung alterations were observed in animals consuming FB1 and subsequently inoculated with PRRS virus. Increased lesion severity was thought to be due to the immunosuppressive effect of the toxin, which allowed the virus to progress. The immunosuppressive effect of the toxin may be related to the accumulation of free sphingoid bases, which inhibits the proliferation of lymphocytes. 34 Others 22 demonstrated that dietary exposure to FB1 decreased resistance to E. coli, resulting in increased bacterial colonization of the small intestine. The effect of FB1 on P. multocida infection was theorized to be related to the suppressed immune response and the reduced phagocytic ability of pulmonary macrophages. 14 Increased susceptibility of pulmonary capillaries to FB1 may also render the lungs more prone to infection. 14 Several studies have also demonstrated that FB1 mycotoxin has a proinflammatory effect. 34
Previous studies have shown that even low contamination levels of FB1 (10 ppm) can decrease the immune response in pigs as expressed by the significant decrease in antibody titers against Morbus Aujesky at days 21 and 35 postvaccination. 33 Another study found that feeding a diet containing 10 mg FB1 per kilogram of feed, in combination with Bordetella bronchiseptica and toxigenic Pasteurella multocida serotype D infection, increased the incidence, extension, and severity of pulmonary lesions in pigs. 23
This study reviews the interaction of M. hyopneumoniae, recognized as one of the most important porcine respiratory pathogens, with fumonisin toxins in the lungs of pigs by both computed tomography (CT) and histopathologic examination. The objective was to determine whether FB1 given at feed levels encountered in a production environment can influence the character and/or the severity of the pathological processes induced by M. hyopneumoniae in the lungs of pigs. The level of FB1 contamination used in the present study (20 mg/kg feed) corresponded to the higher levels observed in naturally contaminated feed for pigs. 31,32
Materials and Methods
Experimental Animals and Their Management
Piglets were obtained from a Seghers hybrid herd that was free from brucellosis, leptospirosis, Aujeszky disease, and PRRS and in which the incidence of respiratory diseases was low. The sows delivering the piglets selected for the experiment (n = 10) were serologically negative for M. hyopneumoniae.
Piglets were randomly divided into 4 groups using the principle of equality, so that the average body weight of the groups was 2.35 ± 0.20 kg on day 3 of life. The 4 groups were split into 2 separate rooms on the day of arrival, which corresponded with day 0 of the study. The 2 uninfected groups, control animals (group C, n = 7) and pigs fed FB1 toxin (group F, n = 7), were kept in one of the rooms, with the groups segregated in two separate elevated-level battery cages of identical size. The pigs infected with M. hyopneumoniae (group M, n = 7) and those infected with M. hyopneumoniae and fed FB1 toxin (group MF, n = 7) were housed in the second room and were similarly segregated in 2 battery cages. Both rooms were kept at air temperature (27°C) with the required air exchange ensured by exhaust fans. The battery cages, the drinking systems, and the rooms were cleaned twice a day, and the piglet-rearing installations were dismantled and washed every second day. All operators were required to wear protective clothing and perform foot and hand disinfection with an aqueous solution of Virkon S (KRKA d. d.; Antec International Ltd, Novo Mesto, Croatia) upon entry and exit from the rooms.
Feeding of the Experimental Animals
The piglets received a milk replacer consisting of skimmed milk powder, vegetable fats, and whey powder, containing 23% crude protein, 23% crude fat, and 1.6% lysine (Salvana Ferkel Ammen Milch; Salvana Tiernahrung GmbH, Klein-Offenseth Sparrieshoop, Germany), from an automatic feeder (Sloten B.V., Deventer, the Netherlands) until day 16 of the study. Beginning on day 7, piglets also received a dry meal ad libitum, containing 16 MJ/kg energy, 18.5% crude protein, 9% crude fat, and 1.65% lysine (Salvana Pre-meal; Salvana Tiernahrung GmbH). From day 16 until the end of the experiment (day 58), milk replacer was eliminated and only the dry meal made available.
Drinking water was available from nipple drinkers throughout the study. Between days 0 and 7, water was also provided from plastic drinking bowls.
Mycotoxin Treatment
F. verticillioides fungal culture containing 3691 mg/kg FB1, 650 mg/kg FB2, and 350 mg/kg FB3 was produced by previously described methods. 9 Beginning on study day 16, a defined quantity of the fungal culture was dried, ground, intimately homogenized, and step by step diluted and homogenized with the dry meal to give the required concentration of 20 mg/kg feed FB1 (3.5 mg/kg FB2 and 1.9 mg/kg FB3). The toxin-containing diet was fed to groups F and MF until the end of the study (day 58), for a period totaling 42 days.
The mycotoxin contents of the fungal culture alone, the normal dry meal diet, and the artificially mycotoxin-contaminated dry diet were checked prior to feeding using a liquid chromatography–mass spectrometry (LC-MS) system (LC-MS 2020 Single Quadrupole Mass Spectrometer, LC-20 AD pumps with DGU-20A degasser, SIL-20ACHT autosampler, CTO-20-AC Column Owen, and CBM-20A Interface; SHIMADZU, Kyoto, Japan). The detection limits for FB1, T-2, zearalenone, deoxynivalenol, ochratoxin A, and aflatoxin B1 were 10, 3, 5, 3, 10, and 10 μg/kg, respectively. No other mycotoxins (including T-2, zearalenone, deoxynivalenol, ochratoxin A, aflatoxin B1) were present in detectable quantities in the fumonisin fungal cultures or contaminated diets fed to groups F and MF. The basal diet was also free of fumonisin contamination.
Experimental Infection and Microbiological Investigations
On study day 30, pigs from 2 groups (M and MF) were infected with a virulent strain of M. hyopneumoniae obtained from a herd experiencing clinical disease. The pigs were inoculated intratracheally with a lung homogenate suspended in Friis medium 11 containing 3 × 10 5 color-changing units of M. hyopneumoniae in a volume of 3 ml. Control animals (group C) were treated similarly but with sterile Friis medium. Preliminary investigations revealed no pathological response in the lungs of pigs inoculated with the sterile medium. Microbiological investigations revealed no respiratory pathogens in the inoculums, in the lungs of the control and infected pigs at the end of the study, or in the single-group MF pig that died during the experiment, except the M. hyopneumoniae pathogen. M. hyopneumoniae was detected only in the experimentally infected pigs by polymerase chain reaction (PCR) 2 on bronchoalveolar lavage fluid collected at necropsy.
Samplings and Test Parameters
Clinical signs and body temperatures were recorded daily. Piglets were weighed and blood samples were taken on study days 30, 44, and 58.
Serology
Blood samples from infected and control pigs were tested for M. hyopneumoniae antibodies with the DAKO Mh ELISA (DAKO, Glostrup, Denmark).
Sphingolipid Profile Test
On study day 58, the free sphinganine to sphingosine ratio, known to be the most sensitive biomarker of fumonisin toxicosis, 21 was determined using a previously described method. 17
Computed Tomography Examination
CT examination to detect lesions in the lung was performed on study days 30, 44, and 58. Combinations of the following active ingredients were used for narcosis and premedication: azaperone (Stresnil [Janssen, Beerse, Belgium]; 4 mg/kg body weight [BW], intramuscular [IM]), ketamine (CP-Ketamin 10% [CP-Pharma, Burgdorf, Germany]; 10 mg/kg BW IM), xylazine (CP-Xylazine 2% [CP Pharma]; 1 mg/kg BW IM), and atropine (Atropinum sulphuricum 0.1% [EGIS, Budapest, Hungary]; 0.04 mg/kg BW IM). After premedication, a balloon-type endotracheal tube was inserted, and anesthesia was maintained with isoflurane inhalation (Forane; Abbott Laboratories, Abbott Park, IL) in a mixture with 2% (v/v) oxygen. Anesthetized pigs were placed into the CT machine in supine position on a special supporting structure. Artificial breath holding was applied during CT scanning of the lungs by occluding the expiratory valve. For the duration of the thoracic scans, intrapulmonary pressure was kept on a standard value (5 kPa).
CT scans of the entire lung region were taken with a SIEMENS Somatom Emotion 6 multislice CT scanner (Siemens, Erlangen, Germany) using the following parameters: tube voltage, 130 kV; dose, 100 mAs; and FoV [field of view], 200 mm. From the collected data, 2- and 5-mm-thick cross-sectional images were reconstructed, with full overlap. Scans of 2-mm slice thickness were used for diagnostic (ie, qualitative) evaluation, whereas 5-mm slice thickness scans collected quantitative data using the Medical Image Processing software (version 1.0, Ferenc Závoda, Kaposvár, Hungary). On cross-sectional images, the regions of interest (ROIs) of the imaged lung were marked out with the help of a Wacom Bamboo MTE-450 digital table (Wacom Europe GmbH, Krefeld, Germany). The x-ray absorption values (Hounsfield units, HU) of pixels from these ROIs were added, and their calculated average values (average density) formed the basis of quantitative analysis.
Gross Pathology and Histopathologic Examination
At the end of the study period (day 58), the pigs were humanely euthanized and then subjected to postmortem examination. Lung samples were taken for histologic examination and fixed in 10% neutral buffered formalin. Fixed tissues were embedded in paraffin wax, sectioned at 4 μm, and stained with hematoxylin-eosin (HE). Periodic acid–Schiff (PAS) stain was also used for highlighting lipoprotein, glycoprotein, or mucoprotein substances in various tissues and cell components. Some materials were stained with Weigert iron hematoxylin for evaluation of the presence or absence of fibrin in edematous areas.
Statistical Analysis
The statistical model included treatment, age, and their effect on clinical, CT, and histopathologic signs, which were studied by using multiway analysis of variance (ANOVA). The significance of differences was tested by Tukey and least significant difference (LSD) post hoc tests. Because a significant age × treatment interaction occurred (P < .05), data were further subjected to 2 types of statistical analyses: within the same age (among the 4 treatments) and within the same treatment (between ages). Data were analyzed by using the GLM procedure of SAS (SAS Institute, Cary, NC).
Animal Welfare Permission
The experimental infection and the CT examinations applied in this study were authorized by the Food Chain Safety and Animal Health Directorate of the Somogy County Agricultural Office, under permission number 23.1/02322/008/2008.
Results
Growth Rate and Clinical Signs
No significant differences in the body weights of the pigs between groups were observed (data not shown).
During the study, 1 pig in group MF was euthanized on day 55 (39 days after the start of toxin feeding and 25 days after M. hyopneumoniae infection), due to clinical signs of progressive dyspnea and hypoxia (open-mouth breathing, cyanotic discoloration of the mucous membranes, and protruding body parts).
No clinical signs were observed in groups C and F during the study. Starting from day 7 after M. hyopneumoniae infection (study day 37), all infected animals (groups M and MF) exhibited mild and progressively increasing coughing or hoarseness, with some dyspnea. From day 4 postinfection (study day 34), infected animals (groups M and MF) had temporary febrile periods, whereas control and toxin-fed pigs (groups C and F) maintained normal physiological body temperatures (between 38.5°C and 39.5°C).
Serology
All pigs tested negative for M. hyopneumoniae antibodies in the DAKO Mh ELISA at the start of the study. All pigs infected with M. hyopneumoniae had seroconversion by 28 days postinfection.
Blood Sphingolipid Profile
On study day 58, the free sphinganine (SA) to sphingosine (SO) ratio (SA/SO), known to be a biomarker of FB1 toxin, was statistically significantly elevated (P < .05) in groups F and MF (1.43 and 1.29, respectively) as compared with groups C and M not fed toxin (0.41 and 0.31, respectively), indicating that there was an effect exerted by the toxin at a cellular level.
CT Image Evaluation of Lung Lesions
The CT scans from study day 30 did not show appreciable lesions in the lungs of pigs in any of the groups. On study day 44, the CT scans of infected pigs (M and MF) showed lesions involving 1 or multiple neighboring lobules in the cranial and middle lung lobes, as well as in the cranial third of the caudal lung lobe (Fig. 1). Lesions consisted of patchy ground glass opacification, whereas more severe cases also had ventral consolidation.
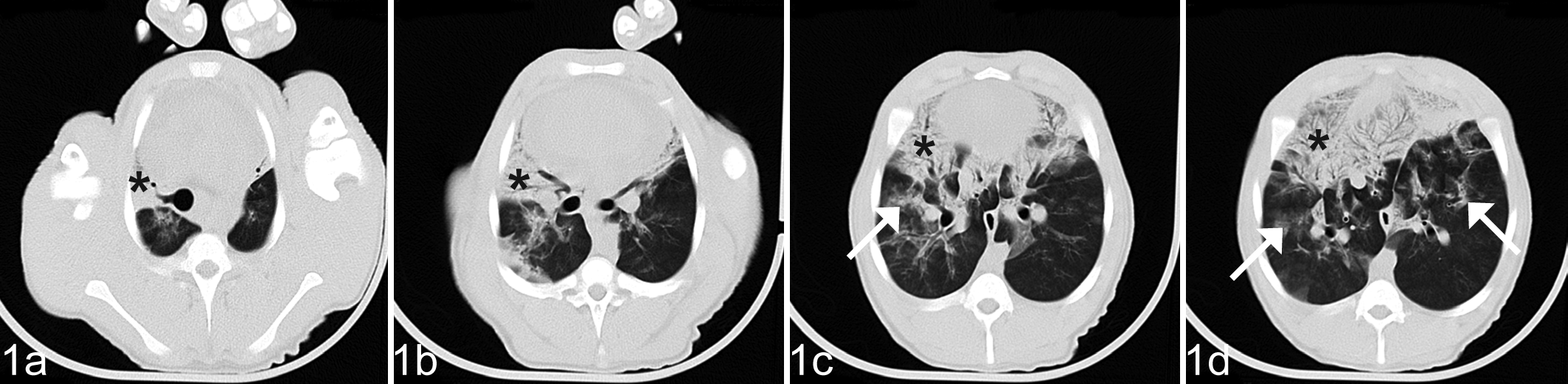
Lung; pig, group MF. Computed tomograpy images in different sectioning planes on study day 44 (day 14 after infection). Patchy ground glass opacification (arrows) and ventral consolidation (*).
CT images at the end of the study (day 58) showed similar lung lesions that were milder in 5 group M animals (Fig. 2) and 2 pigs in group MF but progressively aggravated in 2 other pigs in group MF (Fig. 3). Lesions indicative of pulmonary edema (groups F and MF) could not be identified on the CT images.
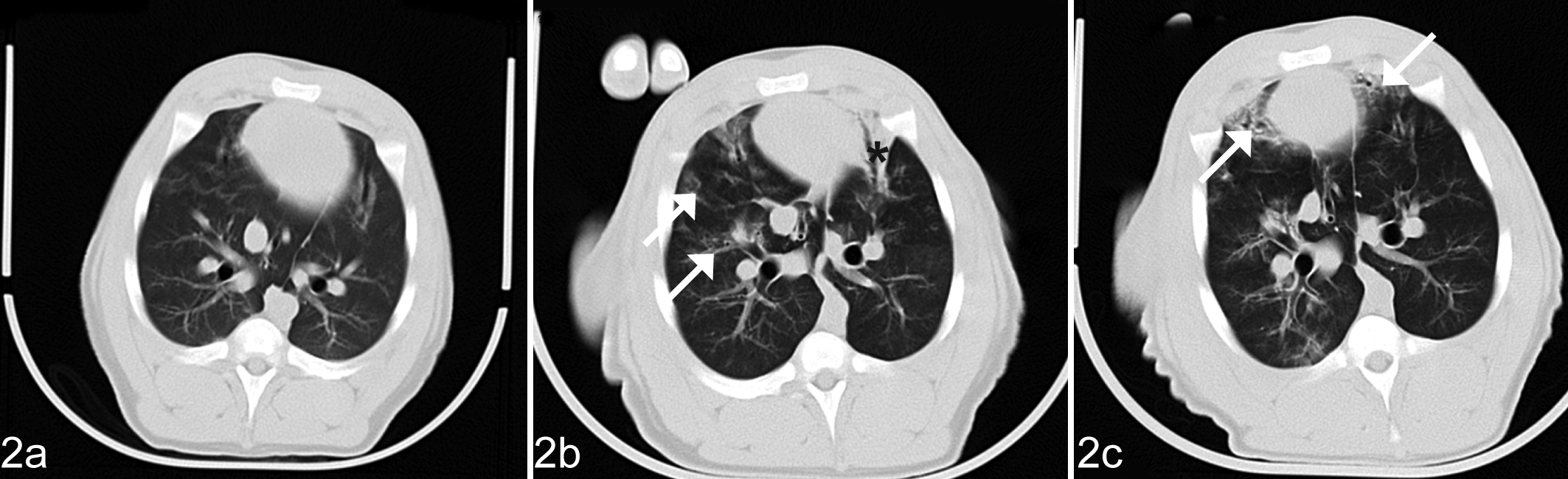
Lung; pig, group M. Computed tomography images showing time course of the development of lung lesions in a pig infected with Mycoplasma hyopneumoniae on study day 30 (a). Pigs treated only with M. hyopneumoniae showed gradual healing in the lungs observed by comparing the images taken on study day 44 (b) and study day 58 (c). Patchy ground glass opacification (arrows) and ventral consolidation (*).
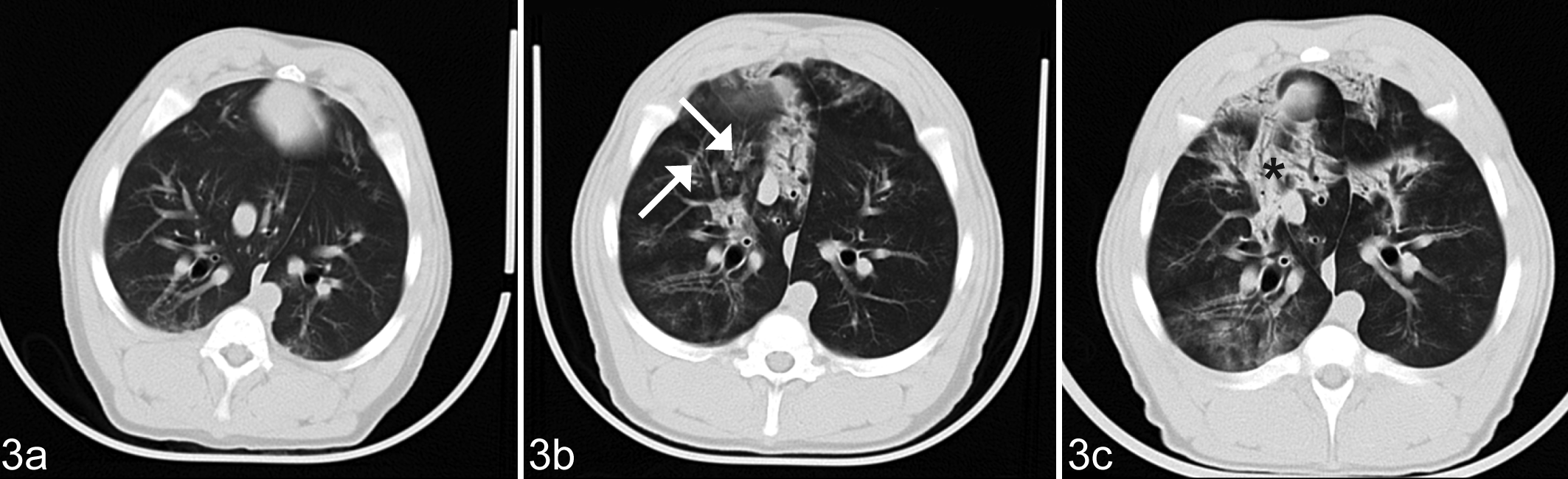
Lung; pig, group MF. Computed tomography images showing time course of the development of lung lesions in a pig infected with Mycoplasma hyopneumoniae and exposed to fumonisins on study day 30 (a). Pigs treated with both the bacteria and toxin showed progressive pulmonary disease by comparing the images taken on study days 44 (b) and 58 (c). Patchy ground glass opacification (arrows) and ventral consolidation (*).
The evolution of average density calculated from combined pixel frequency values obtained for designated areas of the lungs (Table 1) was significantly influenced by the treatment and the age of animals at the time of the examination (P = .024). The average pulmonary density decreased between study days 30 and 44 (P < .01) but did not change between study days 44 and 58. The average density of the lungs of infected groups (M and MF) was 5% to 10% higher when compared with those of uninfected animals (groups C and F) (P < .001). The average pulmonary density values found in infected pigs (groups M and MF) on days 44 and 58 did not differ significantly from the values obtained on day 30. Treatment with FB1 (groups F and MF) did not cause a significant difference in average pixel density in the case of both the uninfected (C and F) and the infected (M and MF) groups.
Result of the statistical analysis of density in the regions of interest of pulmonary parenchymal areas of the lungs on the Hounsfield scale (HU means ± SD).
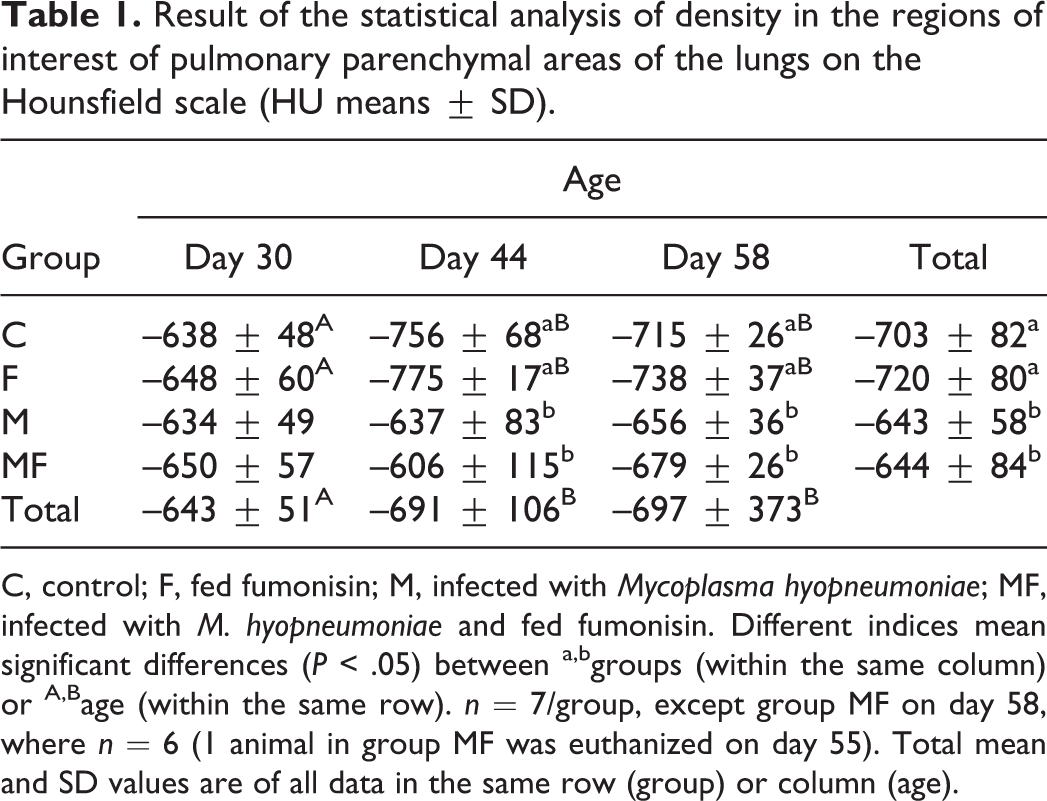
C, control; F, fed fumonisin; M, infected with Mycoplasma hyopneumoniae; MF, infected with M. hyopneumoniae and fed fumonisin. Different indices mean significant differences (P < .05) between a,bgroups (within the same column) or A,Bage (within the same row). n = 7/group, except group MF on day 58, where n = 6 (1 animal in group MF was euthanized on day 55). Total mean and SD values are of all data in the same row (group) or column (age).
Evaluation of the Lung Lesions on the Basis of the Gross Pathological Findings
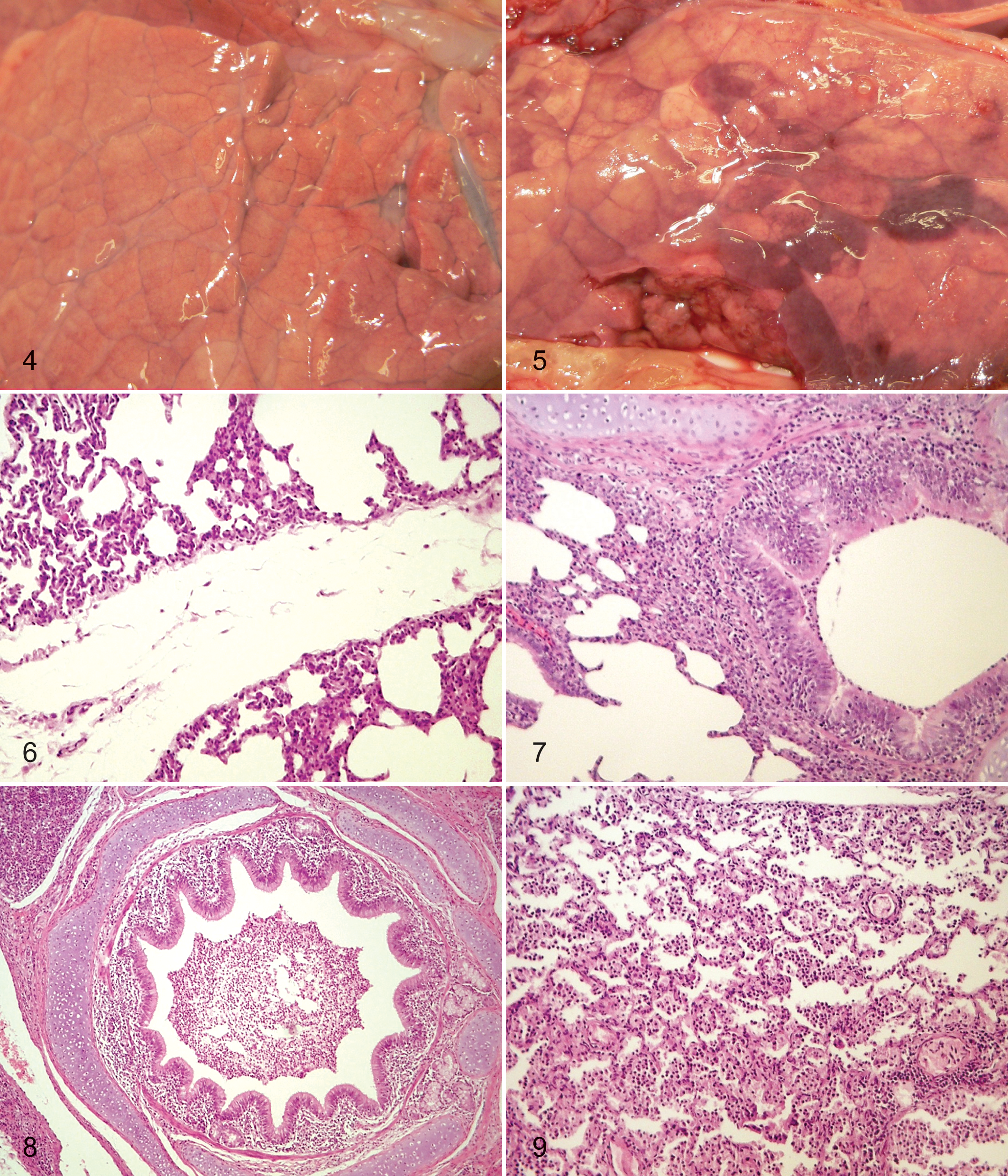
In group C, macroscopic lesions were not seen in the lungs of any of the pigs. In group F, there was mild interstitial edema in the lungs of all pigs (Fig. 4). All animals in groups M and MF had developed subacute or acute catarrhal bronchointerstitial pneumonia. The lesions were located mainly in the cranial and middle lung lobes and in the cranial third of the caudal lung lobe and varied in severity by extending to a few lobules or involving the entire lobe. In group MF animals, mild interstitial edema involving the entire lung was seen in all animals in addition to the pneumonic changes (Fig. 5), and 2 pigs had also developed serofibrinous pleuritis and pericarditis. The pig that was euthanized on study day 55 (group MF) had acute to subacute catarrhal bronchointerstitial pneumonia and severe interstitial edema, along with serofibrinous pleuritis, pericarditis, and peritonitis.
Evaluation of the Lung Lesions on the Basis of the Histopathologic Findings
In the lungs of all examined pigs exposed to fumonisin (group F), there was thickening of interalveolar septa due to epithelial hyperplasia and accumulation of serofibrinous exudate or fibrin. There was also edema and accumulation of serous or serofibrinous exudates in the interlobular tissue of all group F pigs (Fig. 6), whereas pronounced perivascular edema and hyperemia of vessels were seen in 1 group F pig.
In the lungs of all pigs infected with M. hyopneumoniae (group M), there was thickening of interalveolar septa adjacent to bronchioles due to hyperplasia of type II alveolar epithelial cells and the presence of macrophages or lymphoid cells. Proliferation and hyperplasia of bronchial or bronchiolar epithelium, sometimes associated with cloudy swelling and eosinophila, were often observed (Fig. 7). Accumulation of fibrin (fibrinous exudates) with prominence of leukocytes, macrophages, and desquamated epithelial cells was seen in the septa, alveolar spaces, and lumina of bronchi or bronchioles (Fig. 8) in all examined pigs.
Pathomorphological changes in lungs of infected and FB1-fed pigs (group MF) corresponded to the morphologic pattern of a catarrhal bronchointerstitial pneumonia, with development of prominent peribronchial and peribronchiolar lymphocyte infiltration and a strong interstitial edema. The alveolar architecture was often obscured by thickened alveolar septa and atelectatic or exudate-filled lumina. There was thickening of interalveolar septa adjacent to bronchioles by hyperplasia of type II alveolar epithelial cells and the presence of macrophages or lymphoid cells. Proliferation and hyperplasia, along with cloudy swelling in some cells, of bronchial or bronchiolar epithelium were observed along with hyperplasia of the alveolar epithelium. Accumulation of fibrin with or without accumulation of leukocytes, macrophages, and desquamated epithelial cells in the interalveolar or interlobular septa, alveolar spaces, and bronchi or bronchioles was present in all group MF pigs (Fig. 9). Small pulmonary parenchymal hemorrhages were seen in some areas. Slight to moderate edema (serous exudates) (Fig. 9) or accumulation of fibrinous exudates was seen in the pleura and in the interlobular tissue in almost all group MF pigs.
No visible pathomorphological changes were seen in the lungs of the control pigs (group C), except slight hyperemia of some pulmonary vessels.
Discussion
Data on the possible predisposing effects of mycotoxins in the manifestation of different infectious diseases are very limited. In production practices, FB1 is usually combined with small quantities of other metabolites, such as FB2 and FB3, which are produced by the same fungus, F. verticillioides. The quantities of FB2 and FB3 metabolites are usually much lower, less toxic, and not deserving of the same attention relating to disease alteration as the main feed concentration of FB1. 9,23,33 The effect exerted by the fumonisin mycotoxins on the course and characteristics of the disease produced by M. hyopneumoniae and the interaction between the toxin and the bacterium have not been studied; however, in pigs, the lungs are the primary site of action of both components. Both agents occur frequently in pig production, and their presence may lead to severe economic losses, partly attributable to poorer production parameters. As an important causative agent of PRDC, M. hyopneumoniae may play a role in reducing the growth performance and body weight gain of pigs, 29 although the results of some studies, including this one, indicate that M. hyopneumoniae infection does not always cause this effect. 5,30
According to the literature, some Fusarium toxins may affect the performance of pigs in very low amounts; for example, T-2 toxin induces feed rejection, resulting in reduced body weight gain in a concentration as low as 0.5 mg/kg of feed. 24 On the other hand, FB1 did not affect feed intake and body weight gain even when fed in much higher concentrations (eg, 40 mg/kg of feed), despite the fact that the pigs ingesting it had developed rather severe pulmonary edema but no obvious clinical signs of pulmonary disease. 38 Another similar experiment conducted with growing pigs showed that dietary exposure from 1 to 10 ppm of FB1 caused uneven body weight gain in the first 4 weeks of life, but growth stabilized between weeks 4 and 8. Dietary exposure to 10 ppm FB1 reduced the body weight gain of boars by 10%. 27
The combined effect of FB1 toxin and P. multocida serotype A was studied in experimentally infected piglets, 14 and a significant decrease in the body weight gain was found only in piglets fed the toxin. In a previous study, simultaneous infection with P. multocida and B. bronchiseptica did not cause a significant reduction in feed intake or body weight gain, even when combined with dietary exposure to FB1, despite the fact that FB1 exposure resulted in a more severe bacterial infection. 23
The appearance of clinical signs characteristic of respiratory disease has been previously reported to occur on days 7 to 8 after experimental infection with M. hyopneumoniae, 5,37 which was similar to findings in this study. The increase in body temperature noticed postinfection was probably due to the toxic metabolites produced by multiplication of the pathogen in the respiratory tract, the cytokines formed by the immune system, and damage to the epithelial cells. 10,18 According to the results of previous studies, acute fumonisin toxicosis typically causes pulmonary edema, whereas chronic exposure (>6–8 weeks) at low doses (1–10 mg/kg of feed) results in pulmonary fibrosis. Generally, as in this study, these cases occur without recognizable clinical signs. 41 Much higher doses of FB1 (100–300 mg/kg of feed, or above 15 mg/kg of body weight) were required for expression of clinical signs. 15,16
M. hyopneumoniae infection on its own can produce lung lesions in growing pigs. CT images obtained 14 days postinfection (study day 44) showed appreciable changes in all infected animals. Dietary exposure to FB1 aggravated the course of infection: the CT images obtained on study day 58 (day 28 postinfection) indicated progressive pulmonary disease in 2 of the 7 pigs infected with M. hyopneumoniae and fed FB1 toxin (group MF).
The edematous changes and increased permeability of vessels provoked by FB1 could facilitate the distribution of M. hyopneumoniae infection in the lung, resulting in further pathogen dissemination and aggravation of pneumonic damage, which could also change the typical macroscopic findings characteristic for M. hyopneumoniae infection. This process could be facilitated by the potent immunosuppressive effect of FB1 on the humoral immune response in pigs, which was observed in recent studies at even lower (10 ppm) contamination levels of FB1. 33 The interaction between M. hyopneumoniae and other respiratory pathogens has been previously studied, and many demonstrated that pneumonia caused by mixed respiratory infections has a more severe clinical course. 1,4,6,28,35,36,39
By numerically expressing the CT lung lesions as average density values, a significant difference was clearly demonstrated between the M. hyopneumoniae–infected and uninfected pigs, but not between FB1-exposed and unexposed pigs. The absence of a significant increase in the average lung density value in the pigs exposed to both FB1 and M. hyopneumoniae infection could be due to the low numbers of experimental animals or, in part, because 1 MF group pig with the most severe lung lesions was euthanized before the end of study and its average lung density value was not included in the statistical analysis. Between 30 and 44 days of age, there was a significant change in the average lung density values in the 2 uninfected groups (C and F), which could presumably be due to the increased growth in lung size during this time. In the 2 infected groups (M and MF), however, no age-related difference was observed; in these groups, the HU values of x-ray absorption in the lungs did not change with age most likely due to the inflammatory processes produced.
Dietary exposure to 20 ppm of FB1 resulted only in pulmonary edema, which was not demonstrated on the CT images and, therefore, caused no significant change in the average density values. However, the toxin did exert its effect at the cellular level, as indicated by the significantly elevated SA/SO value. At necropsy, pulmonary edema typical of the toxin could be seen in all of the animals exposed to fumonisins (groups F and MF).
Histopathologic findings in the lungs of M. hyopneumoniae–infected animals (group M) were very similar to those described in experimentally infected pigs, 26 with a progressive bronchointerstitial pneumonia from days 7 to 28, which became less severe at day 35. As in this study, previous authors 26 observed that infiltrating neutrophils and mononuclear cells were frequently seen interspersed between epithelial cells; lymphocytes, plasma cells, and neutrophils also were often observed within alveolar spaces, and the alveolar epithelium showed hypertrophy and hyperplasia of type II pneumocytes. From days 14 to 28, the expansion of alveolar septa by infiltrating plasma cells, lymphocytes, and macrophages was also observed.
Pathomorphological damages in the lung of pigs in this study fed FB1 (group F) were typical for previously described fumonisin toxicosis in pigs, including interlobular edema of noninflammatory origin extending to the peribronchial, peribronchiolar, and perivascular regions of the lungs. 8,16,40 Damages in the lung of pigs fed a diet containing FB1 are probably due to the increased permeability of vessels, which is subsequently responsible for the perivascular and especially pericapillary edema observed.
In infected and toxin-fed animals (group MF), the histopathologic alterations typical to both M. hyopneumoniae infection and FB1 effect had developed, which resulted in aggravation of the pneumonic process in 2 animals.
In summary, dietary exposure to FB1 may complicate or facilitate the course of M. hyopneumoniae infection, as demonstrated by the progressive pulmonary disease observed in 2 pigs and the severe clinical signs resulting in early euthanasia of another pig. Additional exploration of the effect exerted by the fumonisin toxin in the lungs and the apparent synergism between FB1 and M. hyopneumoniae requires further studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Hungarian Scientific Research Fund (OTKA, project No. K 81690).
