Abstract
The hearts of 27 Bull Terriers and 6 control dogs were evaluated. Heart murmurs were auscultated in 14 (52%) Bull Terriers. At necropsy, 25 Bull Terriers (93%) had myxomatous degeneration of the mitral valve or abnormalities of the left ventricular outflow tract. Small vessel arteriosclerosis in the myocardium and fibrosis of cardiac conduction tissue were common histologic findings in Bull Terriers with clinical cardiac disease. These lesions were also detected in dogs without clinical evidence of cardiac disease and only mild murmurs or structural valvular disease.
Keywords
Familial, early-onset myxomatous degeneration of the mitral valve (MVD) with regurgitation is common in Bull Terriers.8, 23 Congestive heart failure from MVD is associated with coronary arteriosclerosis and myocardial interstitial fibrosis.5, 10 Death results from sudden arrhythmia, hypoxemia, pulmonary embolism, or multisystemic organ failure.15
Subvalvular left ventricular outflow tract obstruction (LVOTO), or subaortic stenosis, is also common in Bull Terriers;18, 19, 23 aortic valvular stenosis has also been reported.28 These types of LVOTO may be inherited in Bull Terriers just as subaortic stenosis is in other breeds.26 Sudden death from LVOTO is attributed to myocardial ischemia and arrhythmia and is associated with left ventricular hypertrophy, systolic left ventricular hypertension, and coronary arteriosclerosis.11
The purpose of this study was to evaluate the hearts of Bull Terriers with and without clinical cardiac disease to identify cardiac defects and to investigate lesions that might explain sudden death.
Materials and Methods
Dogs
Bull Terriers were obtained from breeders and owners. The dogs or postmortem tissues were received after sudden death or euthanasia for various reasons. Dogs were divided into 4 groups by clinical cardiologic findings: 1) Bull Terriers without clinical heart disease, 2) Bull Terriers with heart murmurs, 3) Bull Terriers with congestive heart failure or “sudden death,” and 4) 6 unclaimed shelter dogs that were free of clinical heart disease and did not resemble Bull Terriers phenotypically. Groups 1–3 were all English Bull Terriers except dog 17, which was a Miniature Bull Terrier. Pedigrees were scrutinized for most Bull Terriers and reflected a range of breeding lines typical of those in Australia.
Specimen collection and evaluation
Complete necropsy examination was performed on 2 dogs (Nos. 24 and 27). In 20 dogs, postmortem examinations were of thoracic and abdominal organs only. Formalin-fixed tissues were examined in 11 dogs (6, 8, 10, 13, 16, 20–23, 25, 26). Hearts were opened by the inflow-outflow method.21 Cardiac valvular lesions were graded by established criteria.30 In grade 1 aortic and pulmonic valve myxomatous degeneration, valvular cusps were excessively long and voluminous, with mild transverse fibrous ridges, and/or 1–2 mid-cusp nodules. Valves with grade 2 degeneration had marked transverse fibrous ridges and coalescing fibrous nodules resulting in narrowing of the orifice. Valves with grade 3 degeneration had diffusely thickened, nodular, and distorted cusps, with orifice stenosis. Fixed, subvalvular LVOTO was graded according to established criteria.27 Concentric left ventricular hypertrophy was diagnosed if the interventricular septum, left ventricular free wall, and papillary muscles were thickened without increase in chamber diameter.22
For histologic examination, samples were taken from the long axis of the heart through the mural mitral valve leaflet and left ventricular free wall, through the mural tricuspid valve and right ventricular free wall, and from the left ventricular outflow tract through the junction of the right and dorsal aortic valve cusps. Samples were also taken from the dorsal surface of the right and left auricles; from the upper and lower interventricular septum perpendicular to the long axis of the heart for evaluation of myofiber arrangement;21 from the crest of the septum, just below the interatrial septum, for evaluation of the atrioventricular node or nearby conduction tissue;25 and from the junction of the cranial vena cava and right auricle for evaluation of the sinoatrial node or nearby conduction tissue.25
Tissue samples were fixed in 10% neutral buffered formalin and processed routinely. Paraffin sections (4–5-μm thick) were routinely stained with hematoxylin and eosin, Masson's trichrome, periodic acid–Schiff, Alcian blue (pH 2.5), Miller's elastin stain, and Perl's Prussian blue.
Results
Control dogs
Control dogs did not have symptomatic cardiac disease or gross cardiac lesions postmortem.
Valvular and outflow tract lesions
Cardiac lesions are summarized by group in Table 1. No gross or histologic abnormalities were detected in the cardiac valves of control dogs. Mitral valvular degeneration was evident in 25 of 27 Bull Terriers: 9 of 11 in group 1, 11 of 11 in group 2, and 5 of 5 in group 3 (Table 1). Grossly, affected valves had short, opaque, glistening, white leaflets with multiple firm nodules. The septal cusp was often lengthened and ballooned. Papillary muscles were thickened and originated more basally than in control dogs. Chordae tendinae were short, thick, and fused. In group 3 (congestive heart failure or sudden death), 4 of 5 dogs had MVD of moderate severity. Two grade-3 dogs also had mitral endocarditis. Another had mitral stenosis, with thickened, nodular, stiff leaflets and fused commissures.
Signalment and cardiac lesions in 27 Bull Terriers. ∗
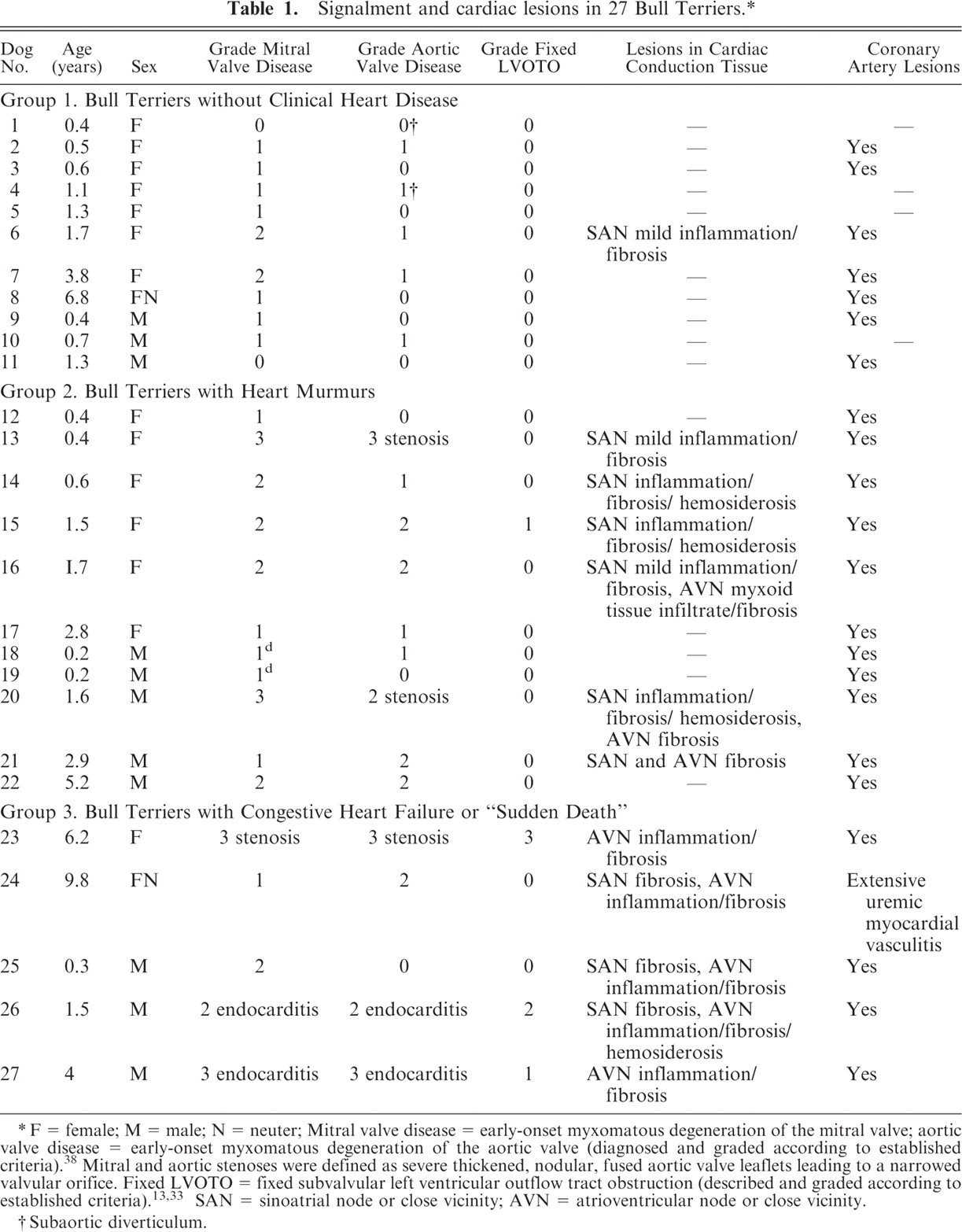
F = female; M = male; N = neuter; Mitral valve disease = early-onset myxomatous degeneration of the mitral valve; aortic valve disease = early-onset myxomatous degeneration of the aortic valve (diagnosed and graded according to established criteria).8 Mitral and aortic stenoses were defined as severe thickened, nodular, fused aortic valve leaflets leading to a narrowed valvular orifice. Fixed LVOTO = fixed subvalvular left ventricular outflow tract obstruction (described and graded according to established criteria).13, 33 SAN = sinoatrial node or close vicinity; AVN = atrioventricular node or close vicinity.
Subaortic diverticulum.
Left ventricular outflow tract obstruction occurred in 1 of 11 Bull Terriers in group 2 and 3 of 5 in group 3. Three affected dogs had white, fibrous subaortic nodules; 1 had an asymmetric subaortic fibromuscular shelf.
Aortic valve myxomatous degeneration occurred in all dogs with fixed LVOTO (aortic or subaortic stenosis). Aortic valve disease occurred in 5 of 11 dogs in group 1, 9 of 11 dogs in group 2, and 4 of 5 dogs in group 3. Valvular aortic stenosis, manifested as severe thickening, nodularity, and fusion of leaflets with a narrowed aortic orifice, was detected in 2 dogs in group 2 and 1 dog in group 3. Aortic valvular endocarditis occurred in 2 dogs in group 3; these dogs also had fixed, subvalvular LVOTO.
Grade-1 pulmonic valve myxomatous degeneration was found in 2 dogs (Nos. 13 and 20) in group 2 and in 2 dogs (Nos. 24 and 26) in group 3. Grade-3 myxomatous degeneration of the pulmonic valve, with stenosis, was the major lesion in 1 dog (No. 22) in group 2. Myxoid degeneration of the septal cusp of the tricuspid valve occurred in 1 dog (No. 2), 3 dogs (Nos. 15, 20, and 22), and 1 dog (No. 27) in groups 1, 2, and 3, respectively.
Twenty-five of the 27 Bull Terriers had at least 1 valve affected by myxomatous degeneration. Histologically, affected valves had irregularly thickened spongiosa with bundles and whorls of spindloid cells in myxomatous stroma, fragmented and malaligned collagen fibers, and loosely arranged elastin fibers (Fig. 1). The myxomatous matrix was Alcian blue and periodic acid–Schiff positive. Similar myxomatous degeneration accounted for the subvalvular LVOTO. Valvular endothelium was focally hypertrophic and hyperplastic. One dog (No. 20) also had hemosiderosis and amyloidosis of the aortic and mitral valves.
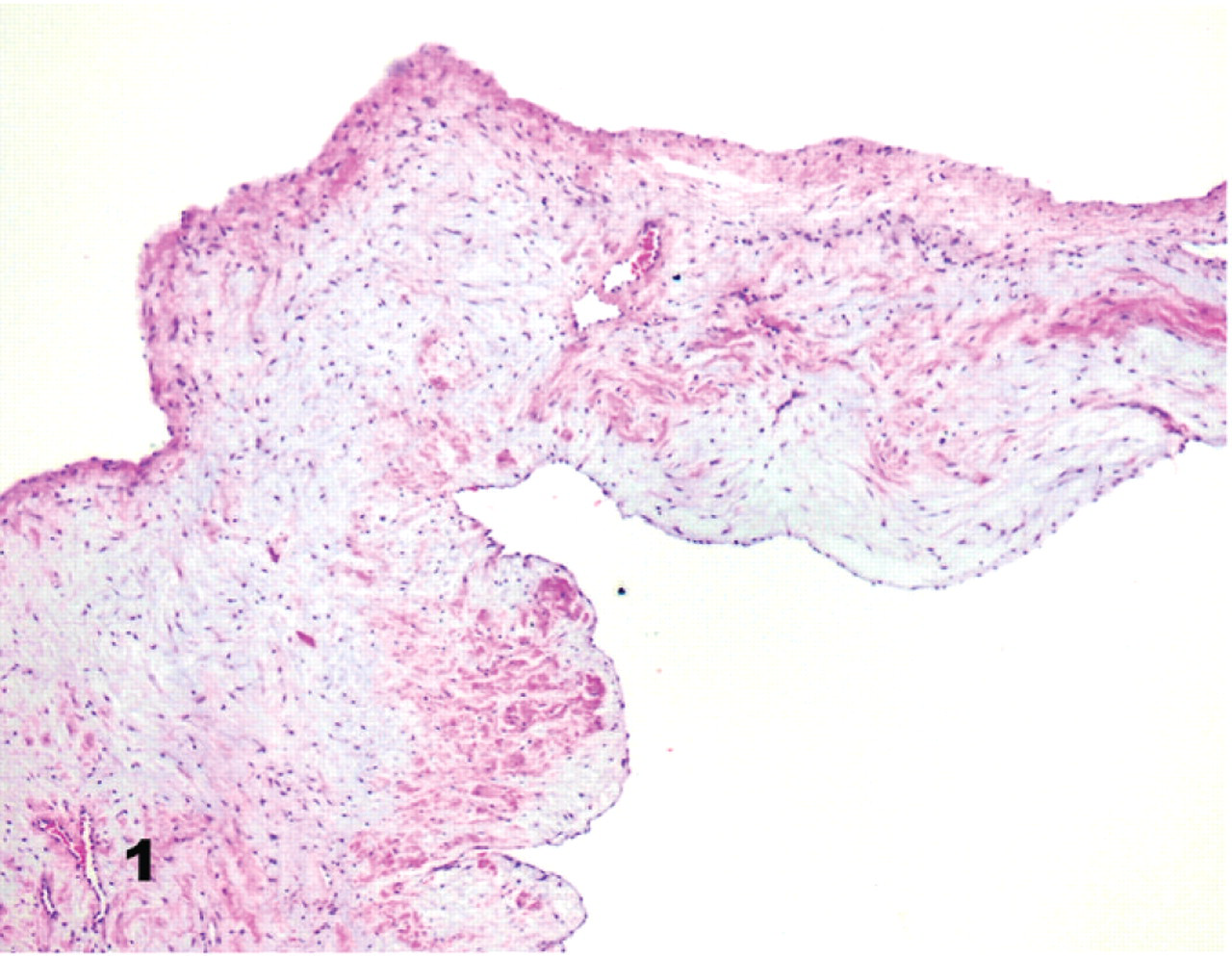
Mitral valve, heart; dog No. 6, Bull Terrier without clinical cardiac disease. Expansion of the valvular leaflet by spindloid to stellate cells in abundant myxomatous matrix. HE.
Coronary arterial and arteriolar lesions
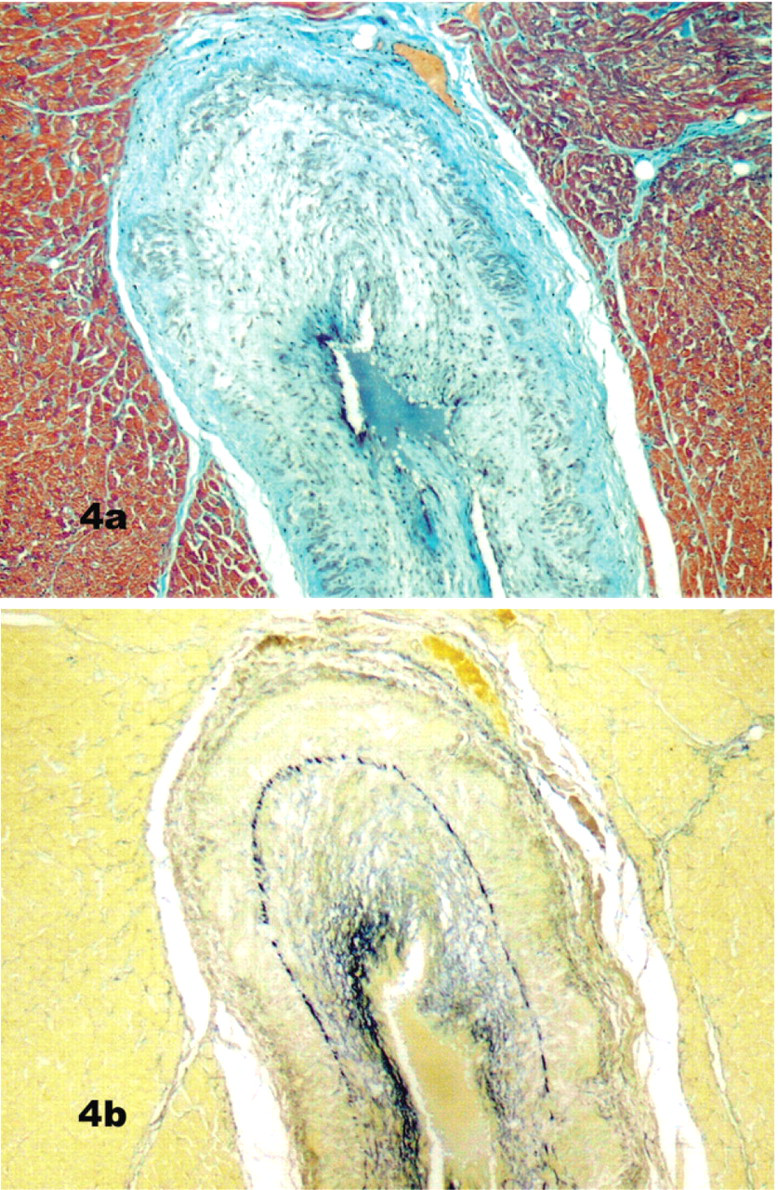
Mild subintimal and medial fibromuscular hyperplasia in a few intramural coronary vessels was found in 2 control dogs (Group 4; Fig. 2). In the Bull Terriers, however, segmental or continuous, mild to severe, intramural (and occasionally extramural) lesions were present in 5 of 11 dogs in group 1, 9 of 11 in group 2, and 4 of 5 in group 3. The lesions were common in, but not exclusive to, coronary arterial branches in the interventricular septum, left ventricular free wall, and papillary muscle. These lesions consisted of a roughly concentric fibrous and myxomatous intimal proliferation (Figs. 3, 4a, b) and were detected in 7 of 11 Bull Terriers with no murmurs (group 1) and in all Bull Terriers with symptomatic heart disease (Groups 2 and 3).
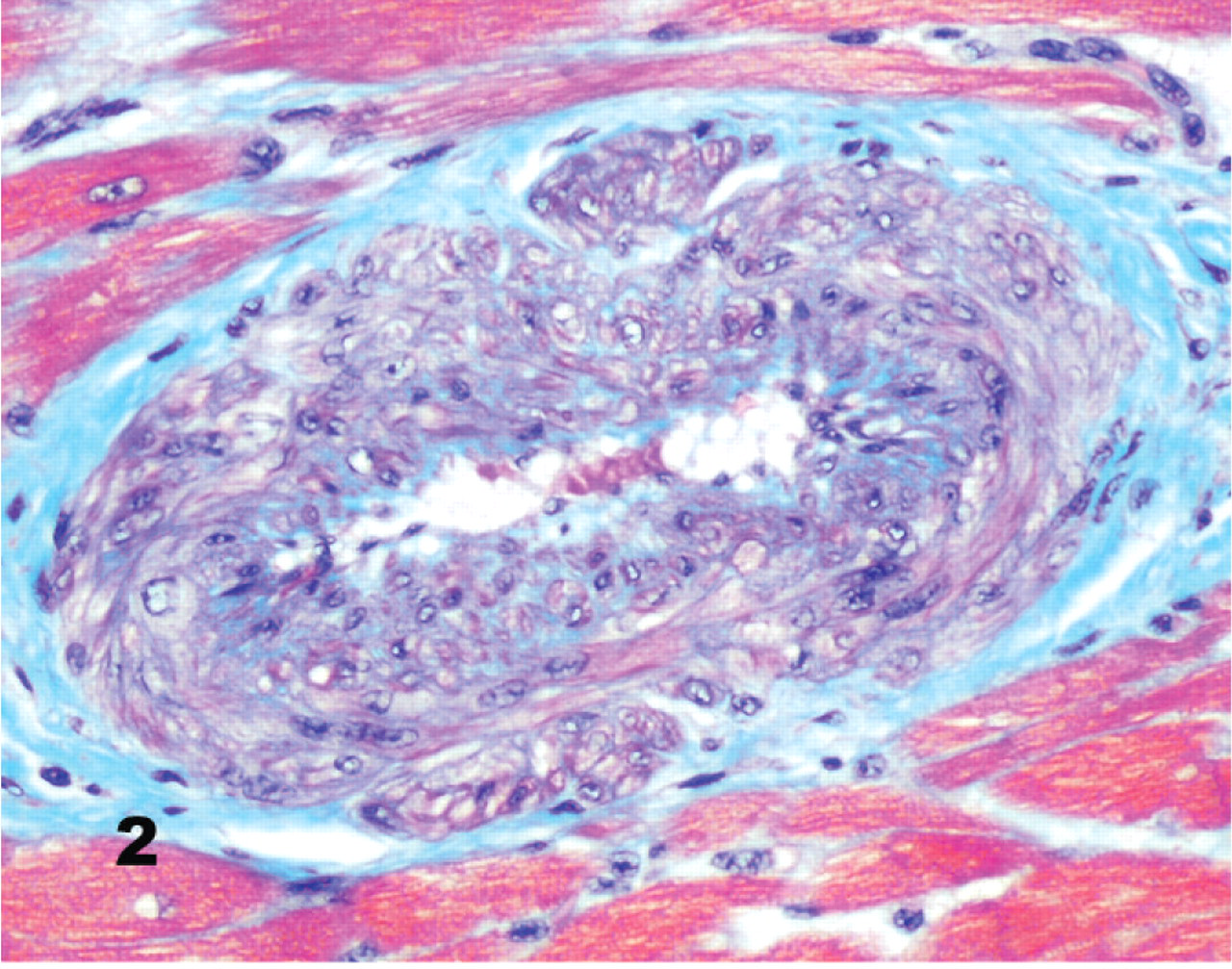
Myocardium; control dog No. 29. Intramural coronary artery with mild intimal proliferation and mild dysplasia of the tunica media with myocyte depletion and infiltration by collagen fibers. Masson's trichrome stain.
Myocardium; dog No. 16, Bull Terrier with cardiac murmur. Intramural coronary artery with marked arteriosclerosis and narrowed lumen. The tunica intima is thickened by proliferative smooth muscle cells in myxomatous matrix. Degeneration is mild and focal in the tunica media. Masson's trichrome stain.
Myocardium; dog No. 16. Advanced arteriosclerosis of an intramural coronary artery with fibromuscular hyperplasia, loss of smooth muscle, and fibrosis of the tunica media; fibromuscular or myxomatous intimal hyperplasia, and narrowed lumen. Masson's trichrome stain.
The intimal thickening consisted of individual or clusters of smooth muscle cells and loosely arranged collagen fibers in myxomatous matrix (Fig. 3), which increased total wall thickness and narrowed the lumen. In addition to the intimal proliferation, there was multifocal to coalescing loss of the medial smooth muscle with collagen replacement (Fig. 4a). The internal elastic lamina was still evident, but multiple irregular strands of elastin were also distributed through the thickened tunica intima (Fig. 4b). Mild to moderate medial muscular hypertrophy of intramural coronary vessels was observed focally in some Bull Terriers in Group 1 (those without clinical cardiac disease).
Dog 18 (Group 2) had severe uremic cardiac vasculitis with hyaline and fibrin deposits, medial necrosis and calcification, and adventitial proliferation. There were periarteriolar infiltrations of polymorphonuclear and mononuclear leukocytes. This dog also had mineralizing myocarditis.
Conduction tissue
Four Bull Terriers had solitary, irregularly shaped, transmural auricular plaques. These were mottled red and white and up to 3 cm in diameter and affected 1 or both auricles. In group 2, this lesion was present in the right auricle in 2 dogs (Nos. 14 and 20) and in both auricles in 1 dog (No. 15). In group 3, 1 dog (No. 23) had this lesion in the left auricle.
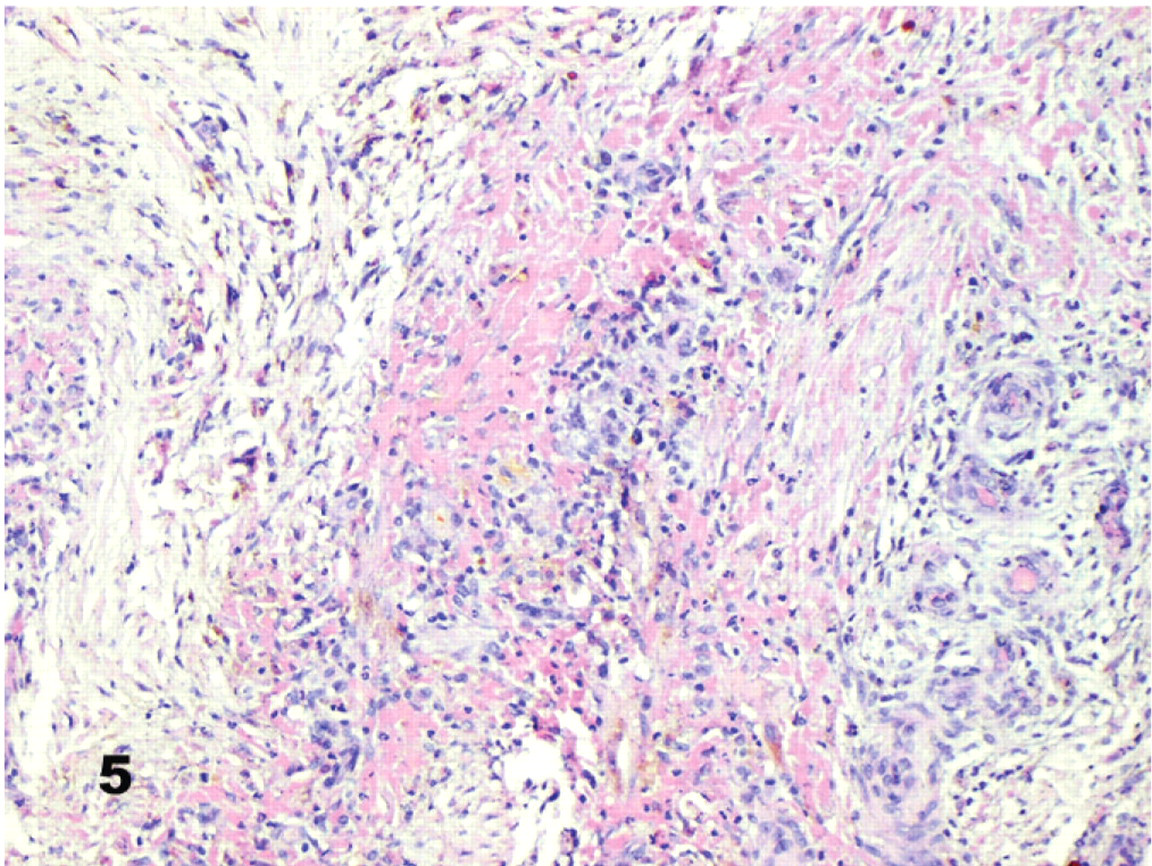
The sinoatrial node was difficult to locate in 3 Group 2 dogs (Nos. 14, 15, and 20) because of severe multifocal to diffuse right auricular epicarditis and endocarditis with dissecting myocardial fibrosis (Fig. 5). Throughout these lesions were aggregates of macrophages (many containing hemosiderin), plasma cells, and lymphocytes. Neutrophil clusters were also present, especially near foci of myocardial necrosis and in adjacent endocardium.
Right auricle; dog No. 16. Focal degeneration with loss of cardiomyocytes and partial replacement by loose fibrous tissue infiltrated by macrophages and lymphocytes. HE.
Focal osseous metaplasia occurred in some lesions. Similar, but milder, changes were also present in 1 dog in group 1 (No. 6), 3 additional dogs in group 2 (Nos. 13, 16, and 21), and 3 dogs in group 3 (Nos. 24–26). The left auricle was similarly affected in 1 dog in group 2 (No. 15) and 1 dog in group 3 (No. 23). Severe subintimal fibromuscular hyperplasia of vessels near the sinotrial node was present in 2 dogs in group 1, 1 dog in group 2, and 3 dogs in group 3.
In 1 dog (No. 16) in group 2, myxomatous degeneration extended from the tricuspid, mitral, and aortic valves into the atrioventricular node and bundle branches. In all dogs (Nos. 23–27) in group 3, dissecting fibrous tissue and mononuclear inflammatory cells infiltrated the atrioventricular node, its approaches, or the bundle of His. In 1 of these dogs (No. 20), there was also hemosiderosis in this area. Excessive interstitial fibrous tissue in the region of the node or conduction fibers was also noted in 3 Group 2 dogs (Nos. 16, 20, and 21) and 1 Group 3 dog (No. 23). One dog (No. 16) in group 2 and 1 (No. 24) in group 3 had fibromuscular hyperplasia of intramural coronary arteries and arterioles adjacent to interventricular septal conduction tissue.
Additional cardiac abnormalities
A subaortic diverticulum consisting of a triangular depression, 3 mm in diameter, was detected in 2 sibling dogs (Nos. 1 and 4) in group 1. The diverticulum was just ventral to the insertion of the right and dorsal valvular cusps, in the area of the left fibrous trigone, and extended into the membranous part of the interventricular septum. A white, mucinous nodule, approximately 0.5 cm in diameter, spanned the membranous interventricular septum between the insertion of the tricuspid septal leaflet and the origin of the aorta in 3 dogs (Nos. 7, 16, and 23).
Subjectively, 2 Bull Terriers in Group 3 (Nos. 24 and 25) had left ventricular hypertrophy, together with aortic valvular degeneration and MVD. One of these dogs had fixed, subvalvular LVOTO, whereas the other had chronic renal failure.
Systemic manifestations of cardiac disease
All dogs in group 3 had some or all of the following histologic changes consistent with congestive heart failure: pulmonary alveolar edema, hemorrhage, and siderophages and periacinar hepatic congestion and necrosis. Two dogs (Nos. 25 and 26) had pleural and pericardial effusion and pulmonary edema with excess stable foam in airways. One of these dogs (No. 26) also had pulmonary thromboembolism and hemorrhage.
Discussion
Cardiac valvular disease occurred in a high proportion (25 of 27) of the Bull Terriers in this study. Twenty-one of the dogs were less than 3 years old, which is consistent with a familial condition. The valvular dysplasia probably resulted from early-onset and progressive myxomatous degeneration and may be related to the histologically similar coronary vascular lesions. A correlation between the 2 conditions has been suggested.9, 10, 13 In humans, an association has also been found between “myxoid heart disease” or mitral valve prolapse and microvascular disease.7, 17 The lesions in the human cases appear to be identical to those of the Bull Terriers in this study.
Arteriosclerosis occurs in less than 10% of dogs less than 4 years of age but affects 50% of dogs more than 13 years of age.9, 13 Arteriosclerosis as an incidental age-related change cannot be distinguished from that due to other pathologic processes;14 however, the fact that most affected Bull Terriers in this study were young suggests an underlying pathologic process. Vascular injury due to any insult produces a stereotypical response that results in intimal thickening. This response includes the migration of smooth muscle cells or their precursors into the intima, where they proliferate and produce extracellular matrix.17 Bull Terriers in this study not only had coronary arteriosclerosis, but it was remarkably severe in young animals. This could reflect an exaggerated response to some mild vascular injury, such as that from the turbulence that occurs in all vessels, especially at bifurcations.9 Indeed, the most severe effects appeared at or near arterial bifurcations in our cases. Bull Terriers in this study also had marked degeneration of the tunica media, a lesion that has not been described previously with mitral valve prolapse. A similar intimal lesion was illustrated, but not well described, in ischemic heart disease that was not associated with valvular lesions.9
In a study of human patients, intimal musculoelastic hyperplasia in coronary arterioles transformed with age into a fibroelastic layer.31 We found coronary arteries in a few of the subclinical Bull Terriers (Group 1) that had more cellular and compact intimal proliferations, suggestive of a maturation process.
We speculate that the “sudden deaths” in dogs in this study might have resulted either from myocardial infarction or dysrhythmias associated with arteriosclerosis. Coronary arteriosclerosis increases the risk of local ischemia, infarcts (that may be difficult to detect), focal fibrosis, and eventual heart failure.9, 10 Dysrhythmias may result from electrical inactivity or instability in cardiac conduction tissue.7, 14
In this study, LVOTO was common and either subvalvular or valvular. Subvalvular LVOTO may originate with morphologic abnormalities of the left ventricular outflow tract, including a steep aortoseptal angle and small aortic root, the latter having been demonstrated echocardiographically in the Bull Terrier.24 Structural abnormalities lead to elevated septal shear stress, subaortic cellular proliferation, and development of a fixed lesion.12
The auricular lesions in 6 of 27 Bull Terriers may have been due to elevated atrial pressure; atrial cell loss, degeneration, and interstitial fibrosis have all been reported in dogs with congestive heart failure.20 However, chronic atrial dilation, even without heart failure, leads to interstitial fibrosis and chronic inflammation.29 Right atrial fibrinous epicarditis is reported in dogs with right heart failure due to tricuspid valve dysplasia.4 Similarly, fibromuscular hyperplasia in epicardial vessels in cases of idiopathic hemorrhagic pericardial effusion has been attributed to local hypertension.2 Alternatively, the auricular lesions may have been caused by infarction, because fibrinohemorrhagic epicardial thickening may overlie areas of myocardial necrosis.14 Other possible causes include bleeding from Thebesian vessels, turbulent blood flow, and rupture of pectinate muscles.
Myocardial interstitial inflammation and fibrosis were present in all dogs that died suddenly and in 1 dog with congestive heart failure. Additionally, 6 dogs had coronary arteriosclerosis near conduction tissue. Similar changes have been reported in and around cardiac conduction tissue in dogs, horses, and humans that died suddenly.1, 3, 6, 7, 16
In summary, a variety of cardiac lesions were identified in these Bull Terriers, including myxomatous degeneration of mitral and aortic valves, LVOTO, left ventricular hypertrophy, interstitial myocarditis and fibrosis associated with cardiac conduction tissue, and an unusual form of coronary arterial degeneration. Because severe histologic lesions were present in some young dogs without gross lesions, careful histologic evaluation of coronary arteries and the cardiac conduction system is warranted in cases of sudden death in Bull Terriers, even when no macroscopic lesions are apparent.
Footnotes
Acknowledgements
We thank the following individuals for help with this study: Professor Clive Huxtable, Ken Wyatt, Jenny Richardson and the radiology staff (Murdoch University), Professor Keith Hughes, Bruce Mackay and Isobel Johnstone (The University of Queensland), and Richard Malik (University of Sydney). Staff from the University of Queensland Veterinary Clinical Pathology Laboratory processed blood and urine samples; Veterinary Pathology and Anatomy staff collected some tissues; and the Histopathology Laboratory staff processed some tissues. Staff from the Veterinary Histology Laboratory (Murdoch University) also performed tissue processing. Financial support was provided by the Queensland Canine Control Council Breeders Research Fund and the Kibble Bequest. We also thank the Bull Terrier breeders and owners involved in this research, particularly those in The Bull Terrier Club of Queensland, Inc.
