Abstract
Abstract
The severity of osteogenesis imperfecta (OI), the associated reduced quality and quantity of collagen type I, the degree of bone fragility, ligamentous laxity, vertebral fractures and multilevel vertebral deformities all impair the mechanical integrity of the whole spinal architecture and relate to the high prevalence of progressive kyphoscoliotic deformities during growth. Bisphosphonate therapy may at best slow down curve progression but does not seem to lower the prevalence of deformities or the incidence of surgery. Brace treatment is problematic due to pre-existing chest wall deformities, stiffness of the curve and the brittleness of the ribs which limit transfer of corrective forces from the brace shell to the spine. Progressive curves entail loss of balance, chest deformities, pain and compromise of pulmonary function and eventually require surgical stabilization, usually around puberty. Severe vertebral deformities including deformed, small pedicles, highly brittle bones and chest deformities, short deformed trunks and associated issues like C-spine and cranial base abnormalities (basilar impressions, cervical kyphosis) as well as deformed lower and upper extremities are posing multiple peri- and intraoperative challenges. Hence, an early multidisciplinary approach (anaesthetist, pulmonologist, paediatric orthopaedic spine surgeon) is mandatory.
This paper was written under the guidance of the Spine Study Group of the European Paediatric Orthopaedic Society. It highlights the most pertinent information given in the current literature and various practical aspects on surgical care of spine deformities in young OI patients based on the personal experience of the contributing authors.
Introduction
Young patients with severe osteogenesis imperfecta (OI) bear a high risk of developing progressive scoliotic spine deformities. The prevalence is 25% at the age of five years, reaching up to 80% in adolescence.1–3 Risk factors are the type and severity of OI, the associated degree of bone fragility, late achievement of motor milestones, vertebral fractures and multilevel vertebral deformities (biconcave vertebrae with typical codfish appearance in the lateral view), ligamentous laxity and muscular weakness.3–5 A vicious cycle of increasing curvature and subsequent secondary inhibitory vertebral growth disturbances in the concavity of the curve fuels progressive vertebral and spinal deformity. Multiple compression fractures and loss of vertebral height entail global sagittal trunk imbalance due to thoracic hyperkyphosis and subsequent compensatory lumbar hyperlordosis primarily diminished lumbar lordosis. 6
Bisphosphonate (BP) therapy does not alter scoliosis prevalence and has at best a limited effect on curve progression.7–9 Brace treatment is controversial any may even negatively effect chest wall morphology and pulmonary function. 3
Hence, progressive curves require surgical stabilization, usually around puberty. The literature on surgical treatment of spinal deformities in young OI patients is scarce and limited to case reports and case series of eight to 27 patients, mostly including different OI severities and age groups.10–15 Some studies date back more than 30 years with the use of Harrington instrumentation.15,16
This paper aims at shedding light on the most crucial aspects of the treatment of progressive spine deformities in young patients with severe OI by congregating practical experience from various paediatric orthopaedic spine centres. It was written under the guidance of the Spine Study Group of the European Paediatric Orthopaedic Society (EPOS) and is based on the most pertinent information given in the current literature and the presentations given at the 37th annual meeting of the EPOS in Oslo/Norway (13 April 2018) in a focus session on genetics, metabolism and spine deformities in OI by the involved authors who all practice in high volume paediatric orthopaedic spine units.
Natural history and specific perioperative aspects in OI
Natural history
The natural history of spine deformities in OI patients is not well defined. However, it seems that the incidence and risk of progression is linked to the severity of the disease and the ambulatory status of the patients, similarly to patients with cerebral palsy (Fig. 1).
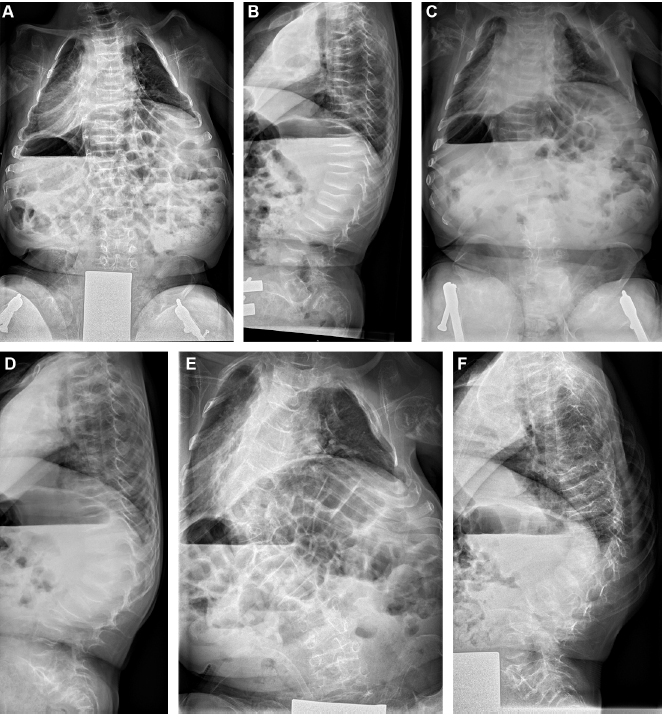
Non-ambulating boy with osteogenesis imperfecta type III. He underwent bilateral femur and tibia osteotomies with subsequent telescopic rodding at the age of four years and had three to four monthly IV. Biphosphonate therapy since the age of three years. The natural spine history shows a rapid delopment of a left thoracic scoliosis and thoracolumbar kyphosis: within two years the deformity progressed from initially 10° scoliosis and 33° kyphosis (
Spinal deformities are known to occur in anywhere between 39% to 78% of all patients with OI, depending on the severity of the disease as classified according to the Sillence type. 17 Patients with Sillence type I were shown to have the lowest scoliosis prevalence and rate of progression (1° per year), patients with type III progress at an average of 6° per year.1,18–20 Most often a progressive scoliosis occurs, but kyphosis due to vertebral collapse may also pose a problem in the growing skeleton. Spondylolisthesis, as a stress fracture of an elongated interarticular pars, has been suggested to occur more frequently than in the general population. 20 Although others have reported similar percentages as in the general population. 21 Untreated scoliosis in OI will normally progress over time and may become very severe in adulthood, associated with vertebral malformation, deformities of ribs, pectus carinatum and compromised pulmonary function. 18 OI, unlike its name suggests, does not only impair the mechanical qualities of the bone, it is a hereditary disorder that affects the quality and quantity of all collagen type I, thus spinal ligaments and discs are involved as well. So the mechanical integrity of the whole spinal architecture is impaired, and as vertebrae collapse, the growth centre in the ring apophysis is damaged, and the already poor soft tissues become more and more mechanically insufficient, creating a vicious cycle. Ishikawa et al 5 showed that the occurrence of biconcave vertebrae is related to curve development; in patients that have more than six biconcave vertebrae, the prevalence of severe scoliosis (> 50°) was 93%, whereas less than six biconcave vertebrae was not found to be a risk factor. Engelbert et al 19 found that regardless of the severity of the underlying disease, an earlier age of achieving motor milestones was related to a later development of spinal deformities. Spinal collapse into severe scoliosis can lead to impairment of pulmonary function, and restrictive lung disease is one of the most common causes of death in young OI patients, accounting for 68% of deaths, largely related to chest wall and spinal deformities.22–24 Whereas in idiopathic scoliosis a decrease in pulmonary function and overall lung capacity only occurs in very large curves (> 90°) or early onset scoliosis, in OI, restrictive lung function can occur in curves as small as 60°, also due to concomitant rib and chest wall deformities. 24 Because of this deleterious effect of scoliosis on pulmonary function, an attempt at prevention of curve progression at an early stage is of paramount importance for maintaining function and quality of life. 25
Specific perioperative aspects
Interdisciplinary preoperative assessment (paediatric orthopaedic spine surgeon, pulmonologist, intensivist, anaesthesiologist and geneticist) is mandatory. Routine standard anteroposterior and lateral radiographs of the cervical spine are needed since the C-spine is commonly involved in skeletal dysplasias (see further below). All specialists involved must be aware of the OI specific features and risks which do not only impact the core surgical procedure but also challenge all aspects of perioperative management, such as intubation, positioning of the patient on the operative table and the intraoperative management 26 (Table 1).
Red flags in perioperative management of patients with severe osteogenesis imperfecta

A thorough preoperative baseline clinical neurological examination by a paediatric neurologist and a MRI of the whole neuraxis is recommended. The surgeon should be present to position the patient preoperatively to minimize the risk of fractures of the extremities, to gain optimal access to the small patient for the core surgical procedure and for surveillance of the cord signals during transfer to the prone position. Temporary paraplegia/quadriplegia has been reported for syndromic patients by subtle change of head flexion/extension in prone position. 27 Hence, intraoperative multimodal (somatosensory evoked potentials (SSEPs), transcranial motor evoked potentials (tcMEPs) of the upper and lower extremities) spinal cord monitoring is to be commenced immediately after anaesthetic induction, immediately after prone positioning and until supine positioning after the end of the procedure. Intraoperative spinal cord monitoring (IOM) may also be considered for non-spine surgery in case of previous neurological events, hyperkyphosis, interventions which presumably last more than 45 minutes and if significant blood loss is to be expected. 26
Deformity treatment in OI
Medical treatment
BPs were deemed to prevent, slow down or even stop the vicious cycle of fragile vertebrae, secondary vertebral deformities (dystrophic ‘codfish’ vertebrae) and gross spinal deformity but hitherto the evidence is limited that they do so.
Pamidronate therapy has been shown to be effective to enhance the bone quality in OI. The main structural effect of pamidronate is to increase both cortical and cancellous bone volume, especially in the first two to four years after initiation of treatment. Pamidronate also increases the number of bone trabeculae while reducing the cancellous bone turnover. 28 The effect of medical treatment with cyclical intravenous BPs on curve progression rates is not clear.7–9 BPs interfere with bone resorption, thus leading to more bone volume and a decrease in fracture risk. Anissipour et al 1 showed that specifically in type III according to Sillence, if BP treatment is started before the age of six years, the rate of curve progression is decreased from 6° to 3.8° per year, which was considered a significant difference. In other types of OI or if administered at an older age, BPs were not found to be beneficial. This was explained by the fact that vertebral height and bone density is preserved better if given at a young age, whereas after the age of six bigger curves and more vertebral deformation has already occurred. Nevertheless, the administration of BPs at an older age may be beneficial for other purposes such as prevention of long bone fractures, but also better preservation of vertebral morphology. BPs disturb the balance between osteoblast and osteoclast activity, which is necessary for maturation of a fusion mass. It has been shown to delay healing after long bone osteotomies in children with OI. 29 Thus it is advised to interrupt BP treatment six months before planned spinal fusion and not resume it until six months after the procedure. 25
Brace treatment
The role of nonoperative treatment of established structural deformities be exercises or custom made braces is uncertain. No change in natural history has been noticed which is not astonishing. In view of the fragile ribs which are supposed to transfer corrective forces to the spine, it seems logical that bracing is problematic or may even be harmful. Pre-existing chest cage deformities may render satisfactory brace fitting difficult, result in relatively excessive correcting forces, further deformation of the pre-existing altered thoracic geometry and even compromise respirator function and soft tissues. Benson et al 3 reported on the unsuccessful conservative treatment of scoliosis in nine OI patients. In a multicentre survey by Yong-Hing and MacEwen 16 on the experience of 51 orthopaedic surgeons in 14 countries, details on 73 patients treated in different types of braces were collected. Bracing was started at an average age of ten years nine months with an average curve magnitude of 43°. The average curve magnitude at the last follow up was 65.9° and 22 complications were related to the brace treatment itself, such as rib deformities or pressure sores. Braces failed to stop progression in 82% of the braced patients. Although it has been suggested that, with the advent of medical (BP) treatment that as become ‘state of the art’ since these reports, brace treatment may have become a more viable option, this was not substantiated in the recent study by O'Donnell et al, 25 who does not advise bracing for scoliosis in OI.
Operative treatment
In cases of progressive curve progression beyond 40° to 50° Cobb angle instrumented surgical stabilization needs to be discussed since further progression and severe functional and respiratory compromise is to be expected. Further care should happen in dedicated specialized centres with vast experience in the treatment of OI patients.
Operative treatment for scoliosis in OI probably started with King and Bobechko, 20 who described spinal fusion in two patients. Yong-Hing and MacEwen 16 presented a multicentre survey on 121 patients with OI, 60 of whom had undergone some sort of spinal fusion, most had received Harrington rod instrumentation and some were only fused in situ without instrumentation. They note that the ligamentous laxity, present in these patients, may contribute to a better correction than in idiopathic scoliosis. They also advise having a bone bank available, since the iliac crest may not contain much bone. Janus et al 13 described 20 patients with OI, all treated according to the same protocol with HALO-gravity traction (HGT) before posterior spine fusion. They advise use of ten pins as the use of six pins was shown to cause migration. Patients were subsequently mobilized in an upright HALO frame with increasing weight up to half of the patient's body weight. The operation was performed under HALO traction when no further improvement of the curve could be observed, which took a mean of 90 days, using Cotrel-Dubousset (CD) or Harrington rods, without attempts at obtaining more correction. Postoperatively, patients continued the HGT for a number of weeks, after which they wore a body jacket for up to 1.5 years. They describe a correction of the kyphosis after an average of almost five years of 32% regarding the scoliosis and 24% for the kyphosis. One of the important advantages they describe is that the patients could remain ambulatory during the whole course of the pre- and postoperative period, which is beneficial for the general condition, muscle strength and bone quality and compares favourably with older series that prescribed bed rest after the operation. HGT may still have a place in the treatment of scoliosis in OI, to improve alignment of the spine in all planes, exercise the patient and improve pulmonary and general conditions. O'Donnell et al 25 advise the use of as many pins as possible, usually eight to 12 and use of low insertional torque (22.6 N-cm to 45.2 N-cm).
Modern day segmental pedicle screw fixation allows for rigid fixation, three-column control of the spine and distribution of forces along the construct and has become the state of the art in OI as well. Piantoni et al, 23 Yilmaz et al 10 and O'Donnell et al 25 advise performing spinal fusion in patients with OI who have a curve of 50° or more, and have reached the age of eight years. Goals are to halt curve progression and to prevent progression of restrictive pulmonary disease. Fusion should include all the curves and typically extends from T2 to L3 or L5; the inferior end plate of the lowest instrumented vertebra should be horizontal. In cases of significant pelvic obliquity (> 10°), fusion to the sacrum-pelvis should be considered. Piantoni et al 23 mention the use of Smith Peterson osteotomies in 12 patients to increase flexibility of the spine, which may protect implants from pull out although the pre-existing ligamentous laxity and a modest attempt at correction may be sufficient to avoid that complication. They report one dural tear but do not state whether that complication occurred during an osteotomy. Their mean correction rate was 58.5%. Cement augmentation of spine anchors has been described in the past and was used in pedicle screws recently by Yilmaz et al. 10 They present ten consecutive patients, all who had Ponte osteotomies, some had rib osteotomies, and seven received polymethylmethacrylate (PMMA) cement augmentation of the pedicle screws at the proximal and distal end of the construct. They advise use of high viscosity cement that has a long curing time and to prepare the pedicles and vertebral body meticulously, only injecting cement into the vertebral body. They reported no cement leakage or neurological injury.
In summary, modern-day segmental pedicle instrumentation has improved the ability to correct spinal curves, also in the OI patients, hopefully preventing disturbed sitting balance, pulmonary compromise and general poor quality of life. Medical (BP) treatment may improve implant stability and cement augmentation can be used if necessary. In selected cases, there seems to be a role for HALO-gravity treatment, to improve general medical condition, nutrition and pulmonary function.
Surgical difficulties and challenges in weak bone diseases
Disease-specific alterations of the morphology
Because the morphological alterations of bone develop with time, early intervention is critical to have optimal results with low complication risk. OI is associated with poor bone quality. In OI spine, soft and brittle vertebral bodies are prone to microfractures in the vertebral endplates. Multiple compression fractures and disturbed symmetrical spinal growth leads to the development of progressive scoliosis.3,5,18 Another issue is elongation of the pedicles caused by repetitive stress fractures. It may complicate pedicle screw insertion and posterior fusion may fail to prevent the pedicle elongation. 30 In patients with OI, progressive deformation of the pelvic bone named as ‘champagne glass appearance’ also may hinder the insertion of iliac screws in a regular way.
Surgical considerations
The first step of the surgery is subperiosteal exposure of the posterior elements between proximal and distal levels to be fused. Spine deformity in OI is usually rigid whereas bone quality is poor and any forceful manoeuvre to correct the deformity generally results in implant failure. Hence, facetectomy of each level to be fused combined with multiple Ponte osteotomies at the apex of deformity seems to be a reliable option to improve the curve flexibility. In addition, multiple rib osteotomies to the concave side may contribute to deformity correction. This approach may avoid the surgeon having to apply excessive loads to weak bone in any attempt of deformity correction.
Selection of implants
Another consideration in spine deformity surgery for weak bone diseases is regarding the selection of implants. Preoperative HGT is a useful option in severe and rigid deformities with poor bone quality. Previous studies report successful outcomes using preoperative HGT to reduce the deformity followed by in situ fusion with or without instrumentation, and was recommended in such cases.13,23 In the past two decades, pedicle screw fixation became the benchmark in spine deformity surgery. In OI spine, because of poor bone quality, purchase of pedicle screw at the cancellous bone of vertebral body is diminished. There are several strategies to decrease the risk of failure for pedicle screw fixation. First of all, reduction screws should be the choice of implant. Secondly, the largest possible diameter screws should be selected and applied under-tapped. Very gentle and gradual correction should be applied by using titanium rods rather than stainless steel or cobalt rods, without any effort to overcorrect. In addition, cross-links may be used to augment the segmental spinal instrumentation. Alternatively, hook fixation – especially claw configuration – or sublaminar wire are two useful supplemental fixation options in the osteoporotic spine. 31 The rationale behind this is the relative sparing of lamina (cortical bone) from osteoporosis as compared with vertebral body (cancellous bone) (Fig. 2).
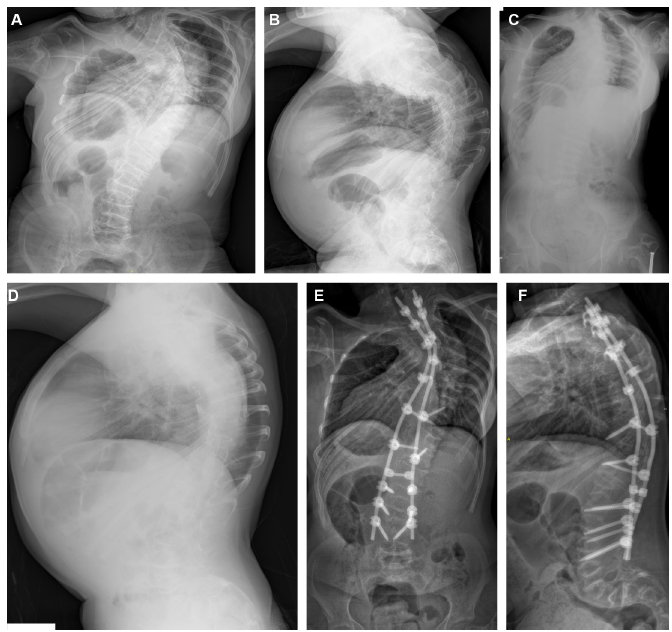
A 14-year-old girl with osteogenesis imperfecta type III previously treated with bisphosphonates and with a thoracolumbosacral orthosis (TLSO) brace: (
Augmentation techniques
Augmentation technique with PMMA, hydroxyapatite (HA) or calcium phosphate may be used to improve the axial pull-out force of the pedicle screws. PMMA can be applied through fenestrated pedicle screws either in all instrumented levels of the most cephalad and caudad levels. Biomechanical studies reported a 110% to 190% increase in pull-out strength of pedicle screws with PMMA augmentation. 32 However, cement leakage to the spinal canal and embolus are two major concerns for the application of bone cement to vertebral body. Reported incidence of pulmonary thromboembolism varies between 2.1% to 26% which are mostly asymptomatic. 33 The prevalence of intracardiac embolus following vertebroplasty is 3.9% which is also mostly asymptomatic. 34 There is limited data regarding the safety of PMMA augmentation technique in paediatric population. Maximum safe dose per level in the paediatric population is still unknown. In their retrospective series, Yilmaz et al 10 reported that there was no fixation failure following PMMA augmented pedicle screw instrumentation in OI spine.
Another way to enhance pedicle screw fixation is HA coating. Biomechanical studies reported that HA coating improves the pull-out resistance and reduce the risk of loosening. 35 However, at least 12 weeks is needed for a significant bone reaction around the HA-coated screws. Again, calcium phosphate cement (CPC) augmentation can increase the maximum pull-out strength up to 77%. 36 On the other hand, CPC converts to HA to have osteoconductivity which takes several weeks after implementation. Because poor bone diseases like OI require a good immediate pull-out resistance, HA or CPC augmentation techniques do not seem to be suitable options for such a condition.
Authors’ preferred treatment algorithm
Because of the limited data about the surgical management of spine deformities in patients with OI, we developed a treatment algorithm based on our clinical experience. In severe and complex deformities (> 70°), we use preoperative HALO-gravity traction to gain a partial correction. The key point to achieving a reasonable deformity correction and preserve it until fusion is early surgical intervention when the deformity magnitude is still moderate (40° to 60°). In such deformities, there is no requirement for HALO-gravity traction. Our strategy is to improve curve flexibility to perform a less aggressive correction manoeuvre. For this purpose, we perform bilateral facetectomy at each instrumented level, sequential posterior column osteotomies at the deformity apex and multiple concave rib osteotomies. We use high-density pedicle screw constructs with reduction screws and titanium rods. We also liberally use screw-hook-sublaminar wire hybrid constructs as alternative fixation methods when morphological alterations hinder pedicle screw insertion.
In summary, it is possible to achieve a good deformity correction and to maintain this with the current technology in OI patients with spine deformity. Early intervention is critical for an optimal outcome while avoiding complications.
Essentials of the cervical spine
Cervical spine disorders in children with OI can be divided into three main pathologies: basilar invagination, atlantoaxial instability and fractures of the cervical spine.
Basilar invagination
The soft bone of the skull in OI may result in the settling of the base of the skull over the cervical spine resulting in secondary basilar invagination. 37 The suggested mechanism for basilar invagination has been infolding of the foramen magnum and the base of skull, which results in the medialization of the occipital condyles, secondary stenosis of the foramen magnum and typical form of skull (‘Darth Wader skull’). 38 This mechanism requires upright posture and therefore Sillence has recommended avoiding upright posture before 18 months of age. The definition of basilar invagination is based on the relationship of the foramen magnum and the tip of dens (Fig. 3).
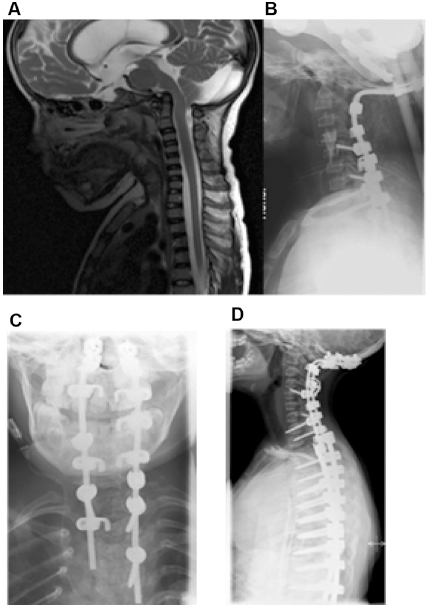
(
If the tip of dens is above the McRae's line drawn from the anterior rim of foramen magnum (Basion) to the posterior rim of foramen magnum (Opisthion), the patient has basilar invagination. The normal position of the tip of dens is 5 mm below this line. 39 In a cross-sectional study of 76 patients with OI, 13% presented basilar invagination. 40 Of the 13 patients with type III OI, five (39%) showed basilar invagination, which was higher than in patients with type I (2%) or type IV patients (25%). Early BP treatment may delay the development of basilar invagination but it is unclear whether this treatment can prevent its development.41,42 It has been reported that a height Z-score below -3 is a risk factor for basilar invagination. 42 It is also unclear how much detected basilar invagination will progress after skeletal maturity. 40 Basilar invagination may interfere with cerebrospinal fluid pathways resulting in: ventricular dilatation and secondary hydrocephalus; pressure on the cerebellum producing bilateral cerebellar disturbance; compression of the brainstem causing lower cranial nerve deficits; disturbance of respiratory centre; compression of the spinal cord; and sudden death.37,43 It has been the author's practice and also recommendation in the literature to obtain a single lateral cervical spine radiograph at the age of six years for surveillance of craniocervical junction abnormalities.37,43,44 In cases where there is a suspicion of basilar invagination remains, an MR image of the head and cervical spine is indicated (Fig. 4).
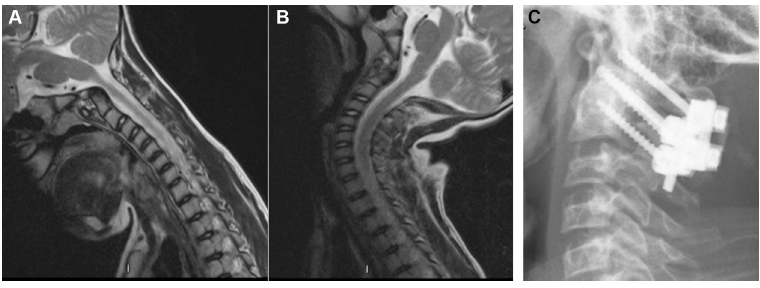
Nine-year-old boy with atlantoaxial instability (AAD interval 7 mm) underwent C1-C2 instrumented fusion using the Harms’ technique.
A rigid cervical collar has been reported to alleviate the neurological symptoms of basilar invagination. 45 In the authors’ practice this treatment modality has been used to postpone surgery in the hope of bigger and stronger bone and fixation points in the cervical spine. The surgical indications for basilar invagination during childhood remain poorly described. 44 In the authors’ practice signs or findings of neural element compression due to basilar invagination are an indication for occipitocervical spinal fusion. Existing surgical strategies include posterior decompression (foramen magnum and C1 laminectomy) and modern rigid cervical instrumentation without preoperative halo traction44,46–48 or combined approach with anterior odontoidectomy and posterior occipitocervical spinal fusion, when there is a severe neurologic deficit preoperatively.45,49 With the current rigid cervical spine instrumentation an all posterior occipitocervical spinal fusion and decompression with preoperative HALO traction and/or ventriculoperitoneal shunting has resulted in resolution of neurological symptoms.44,47,50,51 To reduce risk of nonunion it is the authors’ practice to halt BP treatment for four months postoperatively and use a structural rib grafting to span the instrumentation.
Cervical instability and fractures
The literature on OI associated cervical instability is very limited. According to the authors’ experience atlantoaxial instability in OI may be related to os odontoideum, dysmorphic dens or ligamentous laxity (Fig. 4). In general C1/C2 instability is defined as an atlantoaxial distance > 4 mm or space available for spinal cord < 13 mm.52,53 Neck pain, signs of myelopathy or space available for spinal cord less than 13 mm has been regarded as indications for C1-C2 instrumented spinal fusion and rigid fixation has improved fusion rates as compared with traditional wiring techniques. 54
Cervical spine fractures are rare even in OI. Dens, 55 C2 pars or Hangman 56 and subaxial compression fractures 57 have been reported during growth periods in the literature. Most of these fractures in children have healed well with external support (individual rigid cervical collar) or HALO body jacket 57 and a malunited dens fracture has even shown signs of remodellation during continued growth. 55
Conclusions
Progressive spinal deformities in OI patients are frequent, particularly in severe types of OI. Although BP therapy may slow down curve progression and deformation of vertebrae, it does not seem to lower scoliosis prevalence at maturity or the incidence of surgery.7–9 Brace treatment is controversial; chest wall deformities, rib fragility and stiffness of the curve are limiting factors. 3 Rib deformity and fragility limits force transfer to the spine and bracing carries the risk of further deforming the thoracic cage. Surgical stabilization for progressive curve is challenging for multiple reasons: curve severity and stiffness, associated sagittal plane deformities and loss of balance, vertebral deformity including deformed, small pedicles, highly brittle bones and chest deformities, difficulties to attain stable anchorage of the implant, short deformed trunks and associated issues like C-spine and cranial base abnormalities (basilar impressions, cervical kyphosis) as well as deformed lower and upper extremities.
A multidisciplinary setting and approach in a highly specialized and experienced paediatric orthopaedic environment is key for success in this highly challenging cohort of patients.
Footnotes
C. Hasler: Manuscript basic concept and structure, Abstract, Introduction, Natural history and specific aspects, Table 1, ![]() , References, Layout, Submission.
, References, Layout, Submission.
I. Helenius: Essentials of the cervical spine, Figures 3 and ![]() .
.
M. Yazici: Surgical difficulties and challenges in weak bone diseases.
