Abstract
Background: Historically, blood loss and the requirement for subsequent transfusion have been a significant, while expected, complication of total knee arthroplasty (TKA). While many developments with regard to pharmacologic management and prevention of blood loss have rendered this complication rarer in recent years, orthopaedic surgeons should continue to explore any mechanical techniques with the potential to complement their efforts.
Methods: We evaluated an intraoperative technique during total knee arthroplasty to mitigate blood loss in a randomized controlled trial. Ninety-eight patients undergoing TKA at one institution (three surgeons) were randomized into two groups: Group I received 6 minutes of knee hyperflexion intraoperatively and Group II did not. We compared drain output, hematocrit, blood transfusions, and complications.
Results: The percent reduction in hematocrit on postoperative day 3 was significantly less in the hyperflexion group, and 24 hour drain outputs were higher in the control group, a difference which approached but did not reach significance. No significant differences in blood transfusions or complications were reported.
Conclusions: Based on hematocrit and drain output, holding the knee flexion intraoperatively may play a role in mitigation of postoperative blood loss after TKA. This technique has promise and may be useful as an adjunct to other blood conserving therapies.
As the average age and activity level of our geriatric population continues to rise, so too does the need for joint replacement surgeries, among the most common of which is total knee arthroplasty (TKA). According to the Agency for Healthcare Research and Quality, more than 600,000 knee replacements are performed each year in the USA. 1 The issue of blood loss after TKA is a serious one, having been reported to be as high as 1500 mL per operation, which corresponds to a hemoglobin drop of more than 3 g/ dL. 2 Several investigators have also reminded readers that total blood loss must take into account the concept of “hidden hemorrhage” (i.e., bleeding into local tissues or residual blood in the joint), and therefore true total blood loss may be even higher than is conventionally reported. 3 , 4
Significant bleeding may lead to increased pain and interference with rehabilitation therapy following surgery. 5 Furthermore, bleeding increases the rate of blood transfusion, which can portend its own associated complications, including, but not limited, to infection rates, allergic reactions, and significant costs to the hospital. These consequences translate into increased morbidity and mortality for patients. 6 Given the amount of TKAs being performed and the suggestion that this number will continue to rise, surgeons must learn techniques both old and new to forestall this substantial problem.
Certain known associated factors have been investigated that may contribute to postoperative blood loss, only some of which can be modified. For example, timing of tourniquet release, low molecular weight heparin use, total operative time, and female gender have been associated with increased blood loss and transfusion rates. 7 In a study that also found an increased risk of blood loss in female patients, use of drains has been shown to have a moderate effect on transfusion rate, 8 while four-hour clamping of drains reduced blood loss and need for transfusion in another study. 9 The use of drains in general remains controversial, with some surgeons choosing to use them for 24 hours, others not at all, and some deciding based on intraoperative findings.
Conventional means of mitigating blood loss include maintaining low mean arterial blood pressures with spinal anesthesia, tourniquet use when possible, and persistent hemostasis. Despite these efforts, reports suggest that the major blood loss incurred as a result of TKA in fact occurs postoperatively. 10 - 12 Systemic and topical agents have been pursued using various agents, including tranexamic acid (TXA), epinephrine, fibrinogen, fibrin, and thrombin-based sealants to decrease blood loss. Local injection of the wound with bupivicaine and lidocaine has been shown to reduce blood loss, 13 and norepinephrine administered similarly reduced blood loss and need for transfusion. 14 Lidocaine with epinephrine is also a well-accepted local injection solution to mitigate blood loss, 15 - 17 while epinephrine alone was shown to offer no effect in one study. 18 Tranexamic acid has been studied extensively and has been accepted as a method to reduce postoperative blood loss in several recent metaanalyses of randomized controlled trials. 19 - 22
The aforementioned pharmacological techniques offer compelling promise, and in recent years, the need for transfusion following TKA has evolved from relatively common to relatively rare. With the use of an effective blood loss protocol that integrates these methods, TKA now confers no higher expected blood loss than the patellofemoral arthroplasty, a shorter, less-invasive procedure. 23 Much of this progress is attributable to the profound effect that TXA has had on minimizing blood loss in joint arthroplasty surgery. Still, we should continue to explore any mechanical techniques with the potential to complement their efforts, especially in light of the fact that there are certain populations for which TXA is contraindicated. In theory, an intraoperative technique that tempers postoperative blood loss could reduce the quantity of pharmacological agents necessary for each procedure or simply work in concert with them to achieve the same goal. Mechanical techniques influencing blood loss and range of motion, such as knee positioning and bandage compression, have been studied, though less extensively. 5 , 24 - 28 These studies have focused mainly on patient positioning during recovery rather than intraoperatively; one, for example, showed a 25% reduction in total blood loss when the knee was left elevated and extended postoperatively. 25 To our knowledge, only one study currently exists in the literature that has compared positioning of the knee intraoperatively using flexion and compression methods to influence postoperative blood loss. 29 The outcome measures of the study were blood loss volumes and changes in hemoglobin, hematocrit, and red blood cells on the first and the second postoperative day. The study results revealed no differences between the groups in any of the observed parameters.
The initial theory that intraoperative knee flexion may mitigate blood loss after TKA came from anecdotal observations in our institution that the patients of a surgeon whose TKA technique involves the knee positioned in flexion seemed to experience less postoperative bleeding than other patients. Due to the lack of available data and our assumption that flexion and compression techniques intraoperatively may influence postoperative bleeding, we believed further investigation to be warranted.
Sample size calculations were done using Sample Size Tables for Clinical Studies. 30 We used an α of 0.05 and a power of 0.80 to determine the total sample size needed to experience a difference in blood loss through drain output. Assuming that the true population mean blood loss for ordinary standard of care with tranexamic acid is 373.8 ± 75.1 mL based on meta-analyses of randomized controlled trials of TKAs, if the blood loss by our intervention is half as effective as this method in decreasing postoperative blood loss, then we would expect a mean blood loss of 308.9 ± 60.8 mL. 31 If those were the population values, then with 38 subjects per group, we would have 80% power to find a statistically significant difference in our intervention.
Material and Methods
The protocol used was approved by our institutional review board. All patients or their legal guardians provided informed consent to participate in the study and were informed at the time consent was obtained that case-related data would be submitted for publication.
Patients
Patients were recruited as new or existing patients from the outpatient orthopaedic surgery offices affiliated with our inpatient Joint Replacement Center. With each patient, all options were discussed, including conservative medical management versus surgical intervention, and informed consent was obtained. Following a separate informed consent procedure, patients were asked by their care provider whether they were interested in the present study, and a member of the study team explained the risks and benefits of participation. Inclusion criteria consisted of adult patients receiving TKA for either knee who were willing to provide written, informed consent. Exclusion criteria included pregnancy, revision arthroplasty, cancer, and a history of a bleeding disorder or coagulopathy. Patients with prior knee surgery or hardware were not specifically excluded. Randomization took place in the operating room in which one of an equal number of unmarked envelopes containing a note indicating either experimental or control group was selected by a member of the nursing staff.
Procedure
At the inpatient Joint Replacement Center, 98 patients underwent identical total knee arthroplasty (TKA) surgery under the care of three joint replacement surgeons. In the operating room prior to the procedure, the surgeon was handed an unmarked envelope containing the patient’s assignment to either the control group or the treatment group; envelopes were prepared by staff members uninvolved in the procedure to ensure blinding. All patients underwent identical preoperative protocols and operative technique, (the arthrotomy was done through a midline skin incision and a median parapatellar capsular incision), and the same type of prosthesis was implanted (NexGen® Complete Knee Solution Legacy® Posterior Stabilized Knee; Zimmer, Warsaw, IN); all patients received spinal anesthesia. Before the procedure, a tourniquet was placed on all patients’ operative leg and inflated to 300 mmHg; the patient also received a preoperative dose of 10 mg/kg tranexamic acid. After the primary TKA was completed, patients in the treatment group had their knee hyperflexed as indicated in Figure 1. The tourniquet was then released. After release, the wound was compressed for 6 minutes intraoperatively before hemostasis with electrocautery and surgical closure. This number was chosen based on the known expected range for clotting time (the time required for a sample of blood to coagulate in vitro under standard conditions) of 2 to 10 minutes. Patients in the control group had their knee incision closed in the usual extended leg fashion after tourniquet release and hemostasis with electrocautery. This step was deliberately executed for 6 minutes to eliminate any potential confounding effect that differences in operative time would have on the final data. Both groups had a Hemovac drain inserted deep to the fascial layer before wound closure, which was removed 24 hours after the surgery at which point drain output volumes were recorded. At the time of tourniquet release, patients in both groups received a second dose of 10 mg/kg tranexamic acid.

Intraoperative photograph demonstrating the high flexion maneuver. After the implants have been cemented into place, the knee is place in hyperflexion, and the tourniquet is deflated. The knee is held in this position for 6 minutes.
An identical inpatient postoperative protocol was implemented for all patients. Patients were never informed as to whether they were in treatment or control groups to ensure double blinding. Patients began participating in physical therapy with the rehabilitation staff on postoperative day 1. Drains were removed 24 hours postoperatively. Low molecular weight heparin was given daily at 10:00 a.m. beginning postoperative day 1, cefazolin was given for 24 hours following surgery unless the patient was penicillin-allergic, in which case the patient received vancomycin, and identical protocols were used for analgesics.
Data Collection and Analysis
Several data items were collected from all patients prior to commencement of the study to ensure proper randomization and to avoid any effect on results from outside factors. These included age, gender, presence or absence of cardiac, pulmonary renal, or metabolic disease, and surgeon performing the procedure. As per our established protocol, specific data points of interest were collected at four specific time periods. Hemoglobin, hematocrit, and red blood cell count were recorded preoperatively, then at 24 hours, 48 hours, and 72 hours postoperatively. At the 24-hour time point, the additional data point of drain output was also collected and recorded. Finally, each patient’s tourniquet time, hospital length of stay, and any complications were recorded and compared between groups to ensure no significant differences existed that could skew our data of interest. All the aforementioned data points from the control and experimental groups were compiled and analyzed: categorical variables were summarized as percent of the study group and compared with Fisher’s exact test and two-sample T-tests from continuous variables. A two-tailed alpha of 0.05 was used to denote statistical significance.
Results
Table 1 shows patient and surgery characteristics for the two study groups. Patients were mostly female (82%). There were no significant differences in patient characteristics with respect to age, preoperative hematocrit, or comorbid conditions, suggesting no differences between groups in bleeding risk. There were no significant differences in tourniquet time (average: 45.63 minutes in the experimental group and 49.27 minutes in the control group) or length of stay (average: 2.8 days in the experimental group and 2.7 days in the control group). The great majority of patients experienced no complications at all; there was one reported case each of delirium, postoperative hypertension, and cellulitis; complications were seen in both groups with no significant differences.
Distribution of Patient and Surgery Characteristics for the Two Study Groups and All Patients Total

Blood loss as measured by percent decrease in hematocrit showed no differences between groups on postoperative days 0, 1, and 2. Table 2 shows postoperative measurements of blood loss across all patients. On postoperative day 3, the experimental group demonstrated an average of 20% decrease in hematocrit (SD 6%) as compared to the control group that demonstrated an average of 24% decrease (SD 7%), which was statistically significant (p = 0.023). This is highlighted in Figure 2. Put another way, the experimental group experienced essentially no drop in hematocrit between days 2 and 3 (an average of 31.29 to an average of 31.04), while the control group experienced a continued steady decline at this time interval (an average of 31.15 to an average of 28.67).
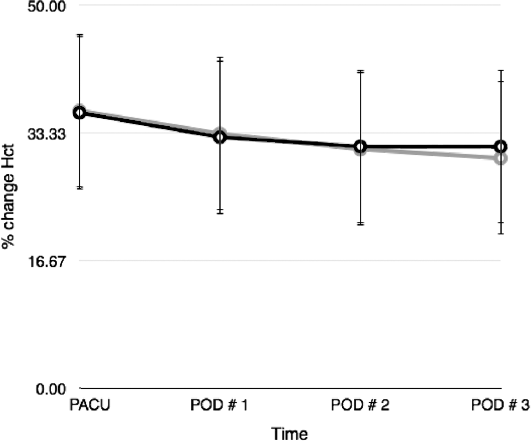
On postoperative day 3, the experimental group demonstrated an average of 20% decrease in hematocrit as compared to the control group which demonstrated an average of 24% decrease, which was statistically significant (p = 0.023).
Postoperative Measurements of Blood Loss, All Patients

Across all surgeons, the difference in average 24-hour drain output was 239.8 (SD: 147.4) for the experimental group versus 288.8 (SD: 142.9), which approached but did not reach statistical significance (p = 0.100).
Discussion
The results of this randomized, controlled, blinded prospective study show that the simple technique of intraoperative knee hyperflexion reduced certain measures of blood loss after TKA, namely percent reduction in hematocrit on postoperative day 3 and potentially drain output at 24 hours. Indeed, it has been suggested that this maneuver can aide in immediate reduction in blood loss intraoperatively and is a practice taught to residents at many institutions to control bleeding vessels when the tourniquet is released prior to wound closure. As mentioned earlier, anecdotal evidence had suggested that knee flexion may result in a reduction in blood loss not only in the immediate setting but in the postoperative setting as well. We believe this is a novel finding as the investigation of mechanical interventions intraoperatively has been explored only minimally thus far in the literature, and prior studies have been unable to demonstrate significant effects.
These results are promising for a number of reasons. First, the goal of reducing blood loss postoperatively is one shared by all surgeons across and beyond the field of orthopaedics. As mentioned earlier, only recently did the value of reducing the blood loss that continues postoperatively become as prominent in the discussion as reducing the value of blood loss reported at the end of each procedure. Advances in TKA itself, as well as associated protocols, have made commendable progress in the efficiency with which patients can begin rehabilitation therapy and ultimately recover their ambulatory status more quickly. Postoperative blood loss can potentially hinder this progress whether it be limited by active wound drainage or symptomatic anemia. Further, this anemia garners its own associated risks and complications. Second, aside from the consequences of 6 extra minutes of operative time—a factor that is largely neutralized based on the reduced time needed for coagulating bleeding vessels with electrocautery—intraoperative patient positioning maneuvers are safe and effectively cost-free. The maneuver could also potentially be performed at the end of the case while the patient is awakening from anesthesia in order to ensure that no additional operative room time is added by this modification. By demonstrating that such a simple adjustment does indeed have an effect on a consequence as potentially serious as postoperative blood loss, we believe this finding could open the door to the study of new and different mechanical maneuvers or encourage surgeons in the future to try this in addition to their standard protocol.
As mentioned earlier, a review of the literature reveals exciting progress in the potential for intraoperative pharmacological interventions to reduce postoperative blood loss, 13 , 19 - 22 but fewer studies have investigated mechanical manipulation of the knee and its effect on postoperative blood loss, and fewer still have found significance. Speck et al. 28 showed that knee flexion at 70° for 6 hours following TKA reduced wound drainage at 48 hours by 33%. Ong and Taylor 25 expanded beyond wound drainage and demonstrated that a group of patients positioned with their knee flexed and elevated for 6 hours following TKA demonstrated a 25% reduction in hemoglobin loss. Ma et al. 27 compared patients who were splinted in flexion for 24 hours postoperatively to patients splinted in full extension and found no differences in any of their recorded measures, which included postoperative day 1 drain volume and changes in hematocrit. Madarevic et al. 29 compared different combinations of knee positioning after tourniquet release, electrocoagulation, and wound closure with and without compressive dressing. They also found no differences in any of their recorded measures, which included intraoperative blood loss and hematocrit on postoperative days 1 and 2. 29
Some previous investigators have mentioned that prior reports of blood saving during TKA via flexion of the operative knee may have been misleading because their reporting methods (i.e., wound drainage) did not take into account total or hidden blood loss. 25 , 29 Our study sought to better delineate the reduction in blood loss by using both wound drainage output and 3-day hematocrit trends as measuring tools. One possibility for why our study more nearly approached significant differences in blood loss between experimental and control groups is that the difference became evident in our population on postoperative day 3, a time point at which some other studies had already concluded their data collection. At the same time, whether differences in hematocrit at this time point has clinical relevance is not clear, especially given the fact that the difference did not correspond to any differences in the need for transfusion. Because the postoperative protocols were standardized among all participants, it is unlikely (yet still possible) that this difference is reflective of a third variable between groups such as disparate fluid management or nutritional parameters. Alternatively, the decision by our surgeons to place the knee in hyperflexion prior to cauterization and wound closure may have offered a more significant reduction in blood loss than those who performed this maneuver after closure.
This study is not without limitation, the most notable of which being the fact that the differences in blood loss noted do not yet correlate with any significant clinical outcomes. As mentioned earlier, recent blood loss protocols involving tranexamic acid, spinal anesthesia, drains, and other methods have transformed blood transfusion from a relatively common to a relatively rare complication following TKA. Given this fact, future studies would need to evaluate a much larger sample size in hopes of delineating a difference in transfusion requirements between groups that do and do not receive this intervention. The theoretical increase in operative time may bring along with it a slight increase in risk of deep vein thrombosis (DVT); therefore, future studies should investigate whether the incidence of DVT is increased in patients who receive this intervention.
The second limitation of our study is that while we noted differences in average 24-hour drain output between groups, this difference did not achieve significance. This suggests that difference may indeed exist in blood loss as measured by drain output, but that either our study design or, certainly, our sample size, was not appropriate to properly elucidate these differences in a statistically significant manner.
Though we did control for differences in operative time by performing 6 minutes of electrocautery in extension prior to closure in the group that did not receive the knee flexion maneuver, the fact remains that the implementation of a required 6 extra minutes of hyperflexion would likely ultimately add minutes to a surgeon’s total operative time. In light of this fact, it is important to consider the potentially increased deep vein thrombosis (DVT) as a theoretical limitation of this practice. As with the need for transfusion, the orthopaedic community has made great strides in recent years with mechanical and chemical methods of DVT prevention; as such, the incidence of this complication has decreased across the board. Since this complication was not seen in either our experimental or our control group, clearly a much larger sample size would have been required to delineate potential differences in DVT risk.
In light of these limitations, there is certainly more to be explored regarding the promise of this easy and cost-free maneuver for controlling blood loss. One could examine differences in knee flexion angle or track the amount of time that the knee is held in flexion and whether it affects the magnitude of differences noted postoperatively. Future studies could also seek to evaluate incidence of any symptoms of anemia beyond transfusion requirements, such as fatigue, headache, or shortness of breath, particularly with physical therapy. Furthermore, alternative mechanical or procedural measures could be studied and compared with the experimental group from the current study, including a group in which the tourniquet was released gradually over 6 to 7 minutes or variations in postoperative positioning. In order to provide insight into the mechanism of postoperative blood loss, one could even design a study in which radiolabeled red blood cells are injected preoperatively in order to assess postoperative volume and location of blood loss, as well as potentially accounting for the possibility of third-variable problems such as hemodilution. We believe the findings from the current study show significant promise and that further research is warranted to explore its use as an adjunct to other blood conserving therapies.
Conclusion
A simple technique of intraoperative knee hyperflexion reduced measures of blood loss after TKA, including percent reduction in hematocrit on postoperative day 3 and 24-hour drain output. This technique has promise and may be useful as an adjunct to other blood conserving therapies.
Conflict of Interest Statement
None of the authors have a financial or proprietary interest in the subject matter or materials discussed, including, but not limited to, employment, consultancies, stock ownership, honoraria, and paid expert testimony.
