Abstract
Vasectomized mice are needed in the production of genetically-modified animals. The BVAAWF/FRAME/RSPCA/UFAW Joint Working Group on Refinement recommended that vasectomy should be performed via an incision in the scrotal sac, rather than via laparotomy, arguing that the former could be less painful due to minimal tissue trauma. This study was undertaken to assess the validity of this recommendation. Mice underwent vasectomy via either abdominal or scrotal approach surgery. Mice were filmed for 15 min presurgery and at one, 24 and 48 h postsurgery. Data were obtained using automated behaviour recognition software (HomeCageScan). Meloxicam was administered either alone or combined with acetaminophen prior to surgery. A third group received only saline subcutaneously. Postsurgery behaviour changes were compared between groups at each time point. Exploratory behaviours such as rearing, walking and sniffing were most greatly reduced at one hour following surgery whereas the duration of grooming increased. By 48 h these changes had largely subsided. Results indicated mice undergoing scrotal approach surgery fared better at one hour postsurgery, but the magnitude of this was relatively insignificant compared with the overall effects of surgery. If the observed behaviour changes resulted from pain, results suggested there was no significant advantage of scrotal versus abdominal approach vasectomy. These and other recently obtained data on the effects of non-steroidal anti-inflammatory drugs (NSAIDs) in mice suggest considerably larger doses of these or more potent analgesics, more precise monitoring of surgical outcomes, or a combination of these factors are needed to determine the extent of pain experienced by mice undergoing vasectomy.
Of the total number of animals used in regulated procedures in the UK, genetically-modified animals has increased from 8% (1995) to 54% in 2010. 1 Almost all of these animals were mice, of which a significant proportion being males undergoing vasectomy for inducing pseudopregnancy in females for transgenic animal production. It is presently unknown whether these mice, and those currently involved in these types of studies, experience pain. Despite virtually no supporting evidence the BVAAWF/FRAME/RSPCA/UFAW Joint Working Group on Refinement 2 recommended vasectomy should be performed via scrotal approach surgery, where the vas deferens could be accessed via a relatively minor incision. By comparison with laparotomy, avoiding incising the abdominal muscles bearing the weight of the viscera (ca. 30% of body mass) 2 was considered likely to be less painful. However, scrotal approach vasectomy also involves dissection and tearing of tissue to access the vas deferens, so significant postoperative pain remains likely.
Behavioural analysis is a key method of identifying pain postsurgery in rats and mice. In addition to suppression of normal behaviours including walking and rearing, a range of discrete identifiable acts have been characterized following vasectomy including; belly pressing, twitching, writhing and hopping behaviours. 3–5 These studies attest to the excessive time needed to conduct the necessary behavioural analyses as a possible reason for the lack of evidence concerning the effects of vasectomy. However, a validated and effective software-based method of automating the collection of behaviour data in mice has emerged relatively recently.
HomeCageScan (HCS) enables rapid analysis of behaviour and has been shown to be effective for detecting differences in behaviour from before to after surgery in mice. 5,6 Use of automated systems not only reduces time constraints but also minimizes observer bias. The validity of this system is based on having been shown to be as effective as manual scoring of mouse behaviour for a range of specific activities. 7 In accordance with the welfare principles underpinning the 3Rs, 8 we sought to determine the need for refining the vasectomy procedure by establishing the relative impact of scrotal versus abdominal approach surgery, and whether mice benefit from analgesic treatment.
Materials and methods
All mice were required for use in the university's transgenic mouse production programme so no animals were used specifically for this study (so conforming to the reduction principle of the 3Rs). The work was also conducted in accordance with the ethical principles of the International Association for the Study of Pain (IASP) and in accordance with the Animals (Scientific Procedures) Act 1986 and was approved by a local ethical review body.
A pilot study was necessary to determine if behavioural differences could be detected between mice that had undergone vasectomy via the abdominal approach compared with the scrotal approach. Following this a larger study was undertaken to assess the effect of analgesic treatments.
Pilot study
Animals
Twenty male CD1 mice (Charles River Laboratories Inc, Kent, UK) weighing 35–39 g at the start of the study were used. All mice were singly housed upon arrival in individually ventilated cages (IVC) (Arrowmight, Hereford, UK) for a seven-day acclimatization period. The animal room was maintained at 23 ± 1°C, 35% humidity and on a 12/12 h light/dark cycle (lights on at 07:00 h). Food (CRM (P), SDS Ltd, Essex UK) and tap water were provided ad libitum. Sawdust bedding (Aspen, BS and S Ltd, Edinburgh, UK) was provided along with nesting material (Shredded paper, DBM, Broxburn, UK).
The main aim of this preliminary investigation was to assess differences in behaviour between the two surgical approaches. As abdominal vasectomy is known to result in changes in behaviour of CD1 mice, 4 to minimize animal use untreated vasectomized, anaesthesia only and anaesthetized drug treated control groups were not included in the pilot study. In a previous study, it was shown that isoflurane anaesthesia alone did not result in changes in spontaneous behaviour at the time points studied here. 4 The interest in this study was between the procedures, not the dosages of analgesia. Therefore, if a high dose was used, it is possible that all mice would receive more analgesic than necessary; hence the chances of detecting group differences would be lessened. A low but potentially useful dose (1 mg/kg) of carprofen was therefore administered.
Surgery
All mice were weighed on the morning of surgery. Ten mice were randomly assigned to each of the two surgery groups; abdominal versus scrotal approach. Surgery began at 09:00 h, with the same surgeon operating on all mice. Anaesthesia was induced with isoflurane in oxygen (induction 5%, 2 L/min) following which mice were placed on bedding (Vetbed, Kennel Needs and Feeds, Morpeth, UK) where a heating blanket (Harvard Apparatus, Edenbridge, Kent, UK) maintained body temperature between 36 and 38°C. Anaesthesia was maintained using a facemask at 2.5% isoflurane at 1 L/min. All mice then received 1 mg/kg carprofen subcutaneously. The lower abdomen was shaved and sprayed with chlorhexidine (Hydrex Derma spray, Adams Healthcare, Leeds, UK) when loss of the pedal reflex was confirmed, vasectomy was then carried out. Abdominal surgery involved a 1 cm transverse incision made through the skin and abdominal wall. The testes were exteriorized, vasa deferentia were located and a small section ablated using electrocautery, the testes were then returned to the abdomen. The incision in the abdominal wall was closed with Vicryl 5.0 (Johnson & Johnson, New Brunswick, NJ, USA). Tissue glue (Nexaband, Abbott Laboratories, Chicago, IL, USA) and sutures (Vicryl 5.0) were used to close the skin. Scrotal surgery involved making a 0.5 cm incision in the scrotal sac, identifying the vas deferens and again removing a small section of these using cautery. The skin incision was closed using tissue glue only (Nexaband, Abbott Laboratories). No intraoperative complications were reported and all mice recovered from anaesthesia uneventfully.
Postsurgery data collection
Behaviours used in the assessment of post vasectomy pain during the pilot study
Statistical analysis
Behaviours were compared between the two surgery groups by independent Student's t-tests.
Main study
Animals
Thirty-six male CD1 mice (Charles River Laboratories Inc) weighing 29–45 g at the start of the study were used. All mice were singly housed upon arrival in IVCs, for a seven-day acclimatization period, under the same conditions described above.
Treatment groups
Eighteen mice were randomly assigned to each surgery group, subdivided equally (n = 6) across analgesic treatments: A, saline s/c (at an equivalent volume to group B meloxicam); B, meloxicam (5 mg/kg subcutaneously; Boehringer-Ingelheim, Labiana Life Sciences SA, Terrassa, Spain); C, meloxicam (5 mg/kg subcutaneously) and paracetamol (50 mg/kg intraperitoneally) (Perfalgan, TM Bristol–Myers Squibb Pharmaceutical Ltd, Uxbridge, UK). Injections were given 30 min prior to surgery. Surgery was exactly as described in the pilot study with the same surgeon operating on all mice. As before, all mice recovered uneventfully.
Filming
The day prior to vasectomy, mice were placed individually in the clear plastic cages and filmed for 15 min using a video camera (Sony DCR-HC96, Sony) positioned 60 cm from the cage for analysis by HCS (Version 3.0, Clever Sys Inc, Reston, VA, USA). This automated method of data collection was used in order to practically and effectively carry out a longer period of analysis. This method of analysis has been deemed equally acceptable to manual scoring of particular behaviours. 7 Following filming mice were returned to their home cages. This was repeated at one, 24 and 48 h following vasectomy. As before, no animals provided specific cause for concern and no intervention analgesic treatments were given.
Data collection
Descriptions of the HomeCageScan behaviours analysed
Statistical analysis
Data were analysed using SPSS software (version 12.0 for Windows, SPSS Inc, Chicago, IL, USA). All data were tested for normality and homogeneity of variance. As data were not normally distributed and could not be transformed, they were analysed using appropriate non-parametric tests (Friedman and Wilcoxon) with post hoc (Bonferroni) corrections of probability. Interactions (surgery type by analgesic treatment) were analysed using a Kruskall–Wallis test.
Results
Pilot study
The majority of activities thought to be closely tied to pain occurred significantly more frequently at one hour following abdominal approach surgery compared with scrotal approach (P = 0.024, <0.001, 0.016, 0.018, for twitch, writhe, flinch and hop respectively) Phenotypic descriptions of these can be obtained elsewhere.
9
However, belly-pressing, another activity previously linked to post vasectomy pain occurred significantly more often following the supposedly lesser painful scrotal approach (P < 0.001) (Figure 1). Time spent wound licking was not different between the two groups, neither was the time spent grooming the face and forepaws.
Mean frequency of occurrence (±1SD) of behaviours, thought to be related to postoperative pain, in mice during 6 min of analysis at one hour post vasectomy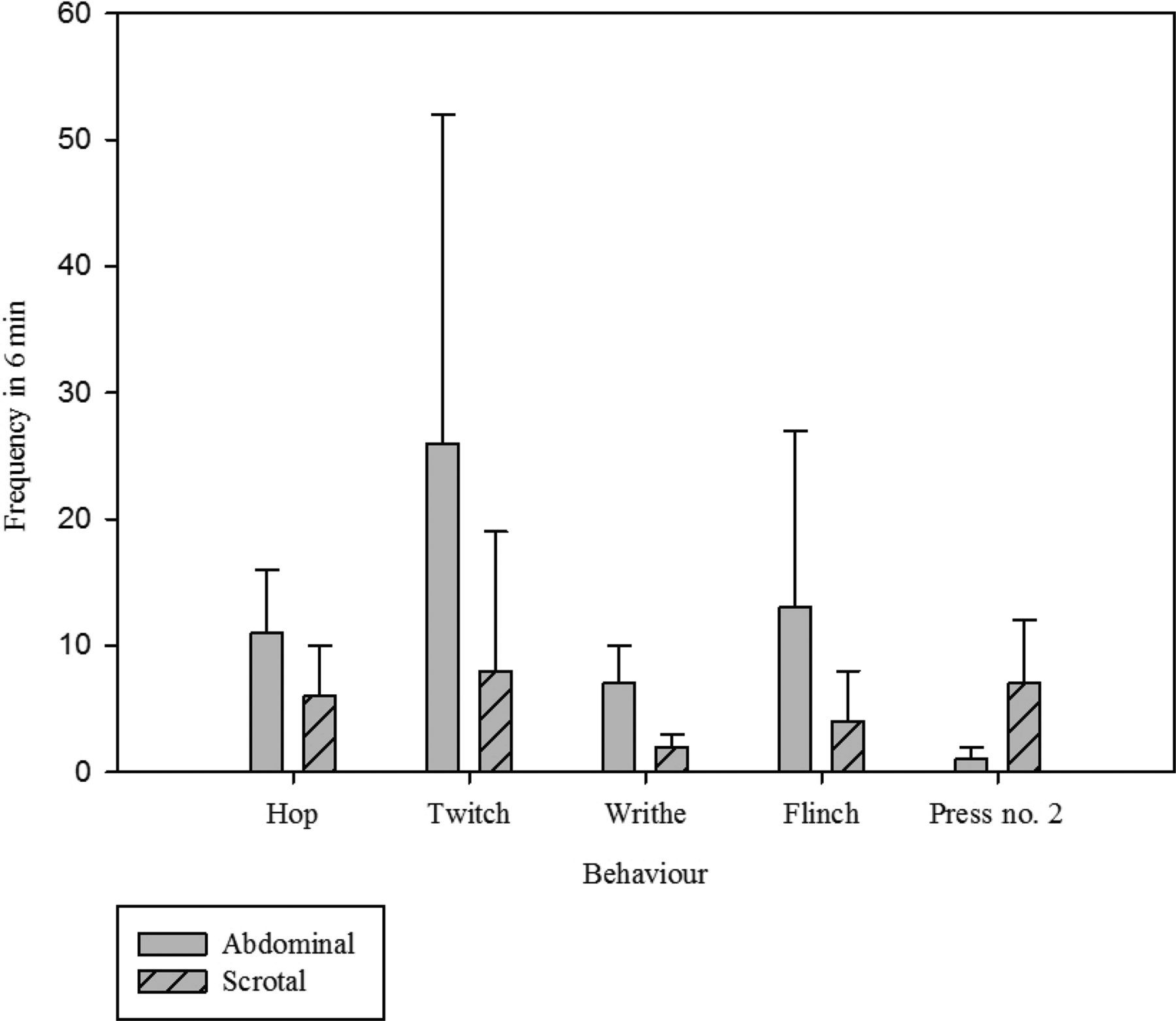
Main study
Effects of surgery
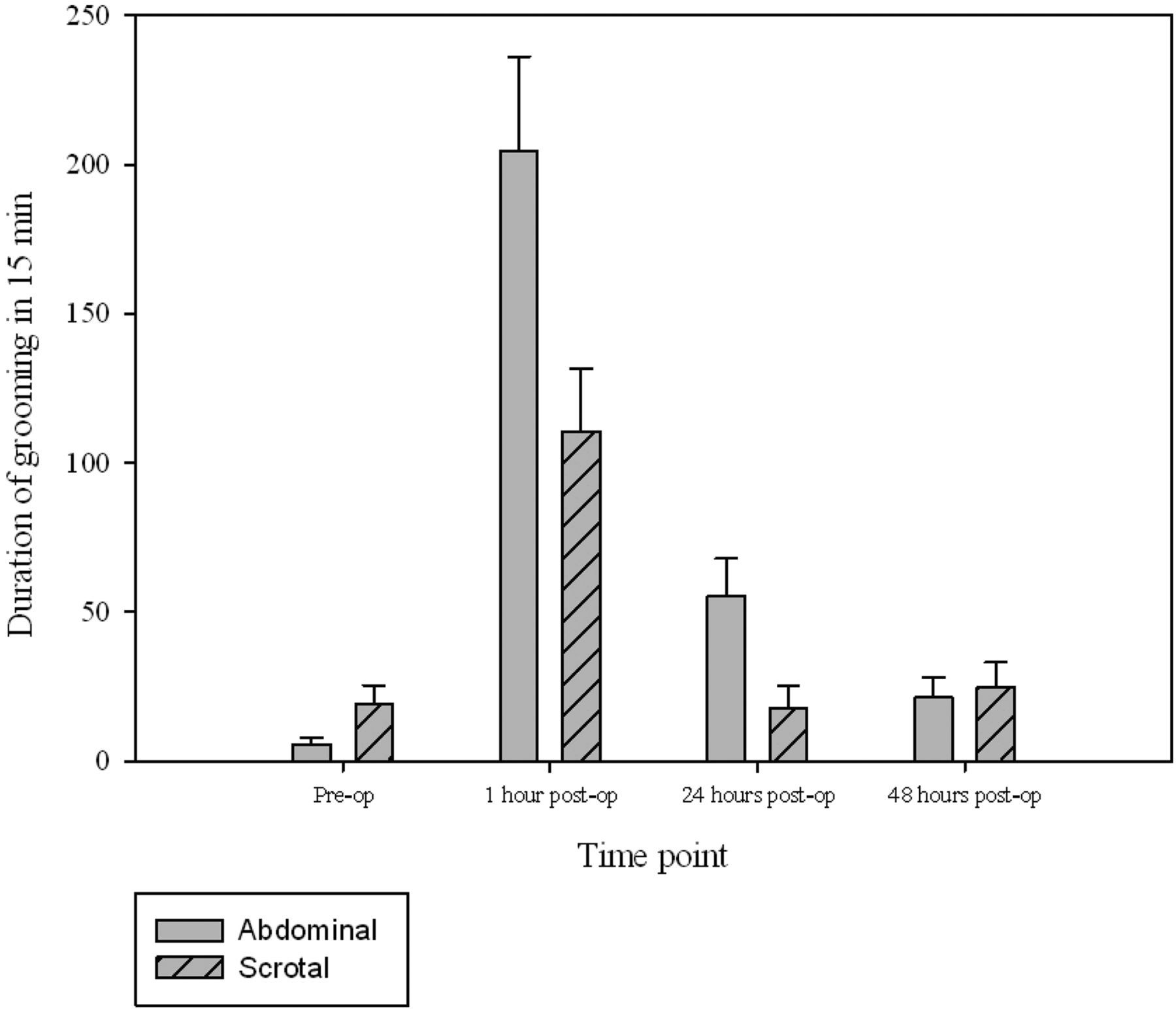
The surgery groups were compared at each time point, with the majority significant differences occurring one hour following vasectomy (Table 3). Rearing was significantly more frequent following scrotal approach vasectomy at both one and 24 h following surgery (P= 0.001 in both cases; Figure 2). The time spent in a stretched position was found to be greater in the scrotal group than the abdominal approach group at 24 h postsurgery (P = 0.035). Time spent walking was significantly greater in the scrotal approach group compared with the abdominal approach group at one hour postsurgery (P = 0.042). This was not significantly changed from walking preoperatively, unlike in the abdominal approach group where walking duration was significantly less at one hour following surgery compared with baseline (presurgery) (P = 0.02). Sniffing significantly decreased in frequency in both the abdominal and scrotal approach groups at one hour postsurgery (P = 0.001 in both cases). Compared with baseline, time spent grooming increased following both surgical methods (P = 0.001 in both cases) and remained so at 24 h in the abdominal surgery group (P = 0.002). Grooming was significantly longer in the abdominal group at both the one and 24 h (P = 0.025 and 0.013 respectively; Figure 3). Table 3 gives details of the remaining group differences.
The mean frequency ( ± SEM) of rearing during 15 min preoperatively and at one, 24 and 48 h postabdominal or scrotal approach vasectomy The mean duration ( ± SEM) of grooming during 15 min preoperatively and at one, 24 and 48 h postabdominal or scrotal approach vasectomy Significant differences in behaviour between abdominal and scrotal approach vasectomy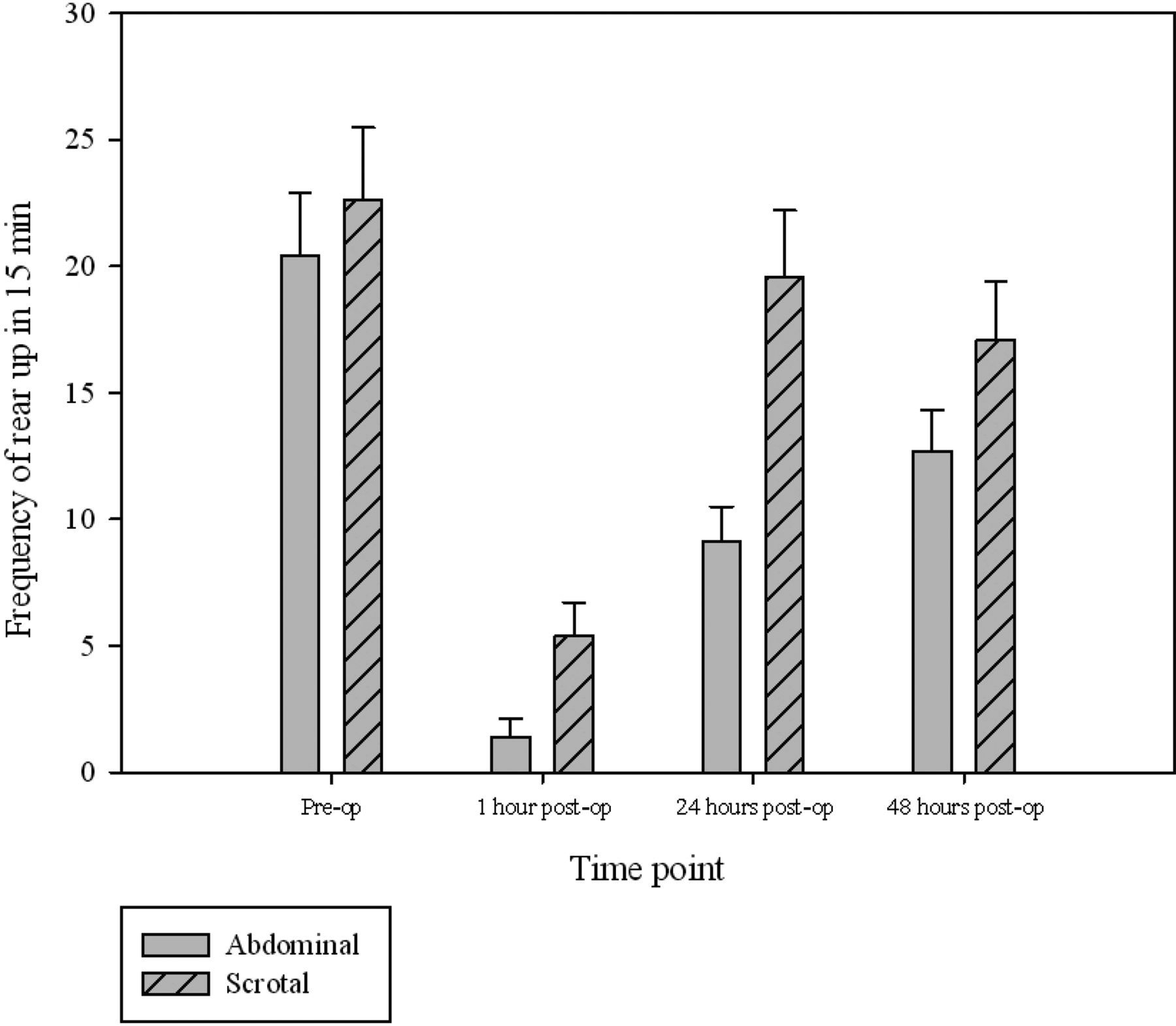
Effects of analgesic treatment
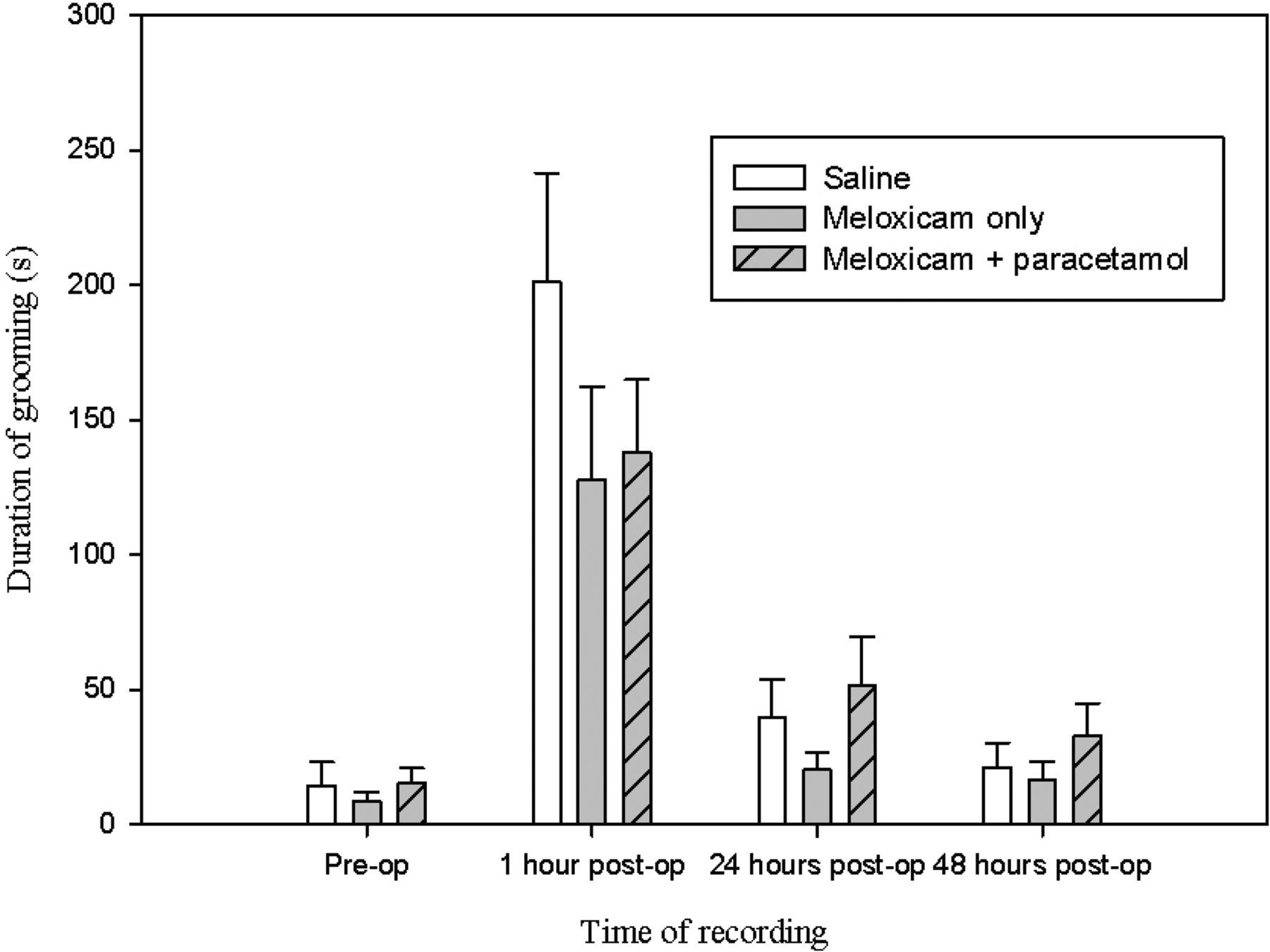
The behaviour of mice following administration of meloxicam alone (M) or the combination of meloxicam and paracetamol (MP) were compared with mice given only saline (S); however, neither treatment had any significant effect. This was because they failed to abate the changes in behaviour observed following surgery, for example rearing, which remained significantly depressed at one hour following surgery in all groups (Figure 4; P = 0.003, 0.008 , 0.005 for M, MP and S groups respectively). Similar effects were observed for grooming duration (Figure 5; P < 0.01 for each group).
The mean frequency ( ± SEM) of rearing during 15 min preoperatively and at one, 24 and 48 h postoperatively The mean duration ( ± SEM) of grooming during 15 min preoperatively and at one, 24 and 48 h postoperatively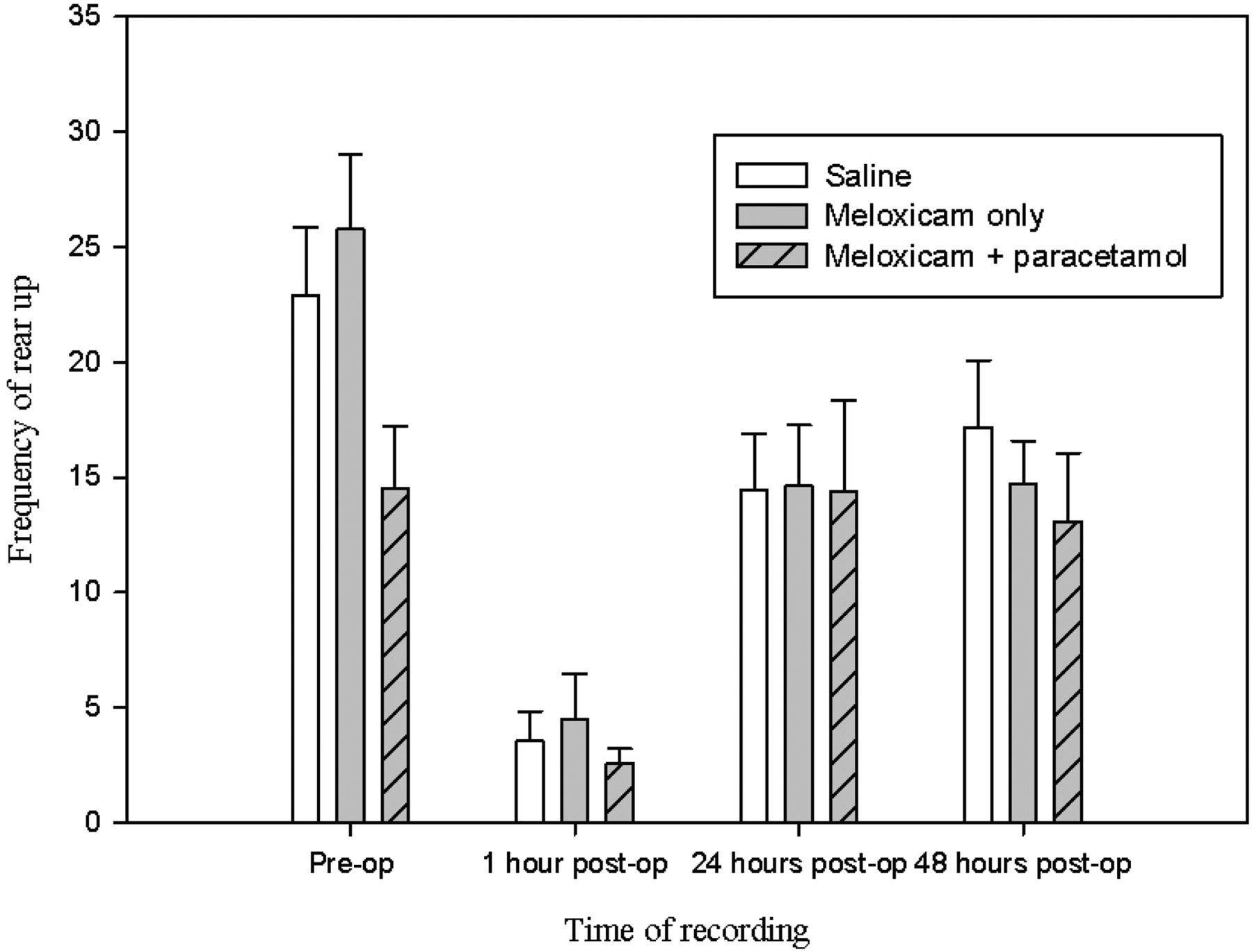
The combined effect of the type of surgery and the analgesic treatment were analysed. The largest change in behaviour occurred between presurgery and one hour postsurgery; therefore, analysis was focused on this time point. The difference in frequency and duration behaviour scores (ethogram shown in Table 3) were calculated (presurgery−one hour postsurgery) and the six groups (abdominal S, abdominal M, abdominal MP, scrotal S, scrotal M and scrotal MP) of mice were compared. From this extensive list of behaviours no significant differences were found between the groups.
Discussion
The pilot study results indicated greater concern for mice undergoing abdominal vasectomy. This was because, with the exception of belly-pressing, behaviours previously linked to pain following vasectomy (e.g. twitching, flinching, hopping and writhing 10 ) occurred more frequently than in the scrotal model. The data therefore warranted a more detailed investigation into the responses of mice to two analgesic treatments and an extension of the period of assessment to 48 h. Obtaining meaningful data necessitated including an untreated (saline injected) group. Evaluating the responses of mice to drug treatment provides another important method of evaluating the effects of surgery, since prospectively at least, procedures that have greater impact would require more aggressive therapy. This was the reason a combined (MP) group was included in the main study.
The automated analysis system HCS, in its default format, is primarily a phenotyping tool, and as such records ‘normal’ rather than abnormal activities which were the main focus of the pilot study. By necessity, the main focus shifted from increased abnormal activity to reduced normal behaviour. Vasectomy reduced the frequency and duration of a range of these normal (mainly exploratory) behaviours including walking, rearing up (Figure 2) and sniffing whereas grooming duration significantly increased (Figure 3). This latter effect could indicate either pain or increased anxiety. Stress and anxiety that occur following surgery are known to result in an increase in short bouts of grooming activity which are often incomplete, i.e. the mouse begins the grooming process at the paws and face but then does not continue to the rest of the body. 10,11 Due to the nature of HCS specific sequences of grooming behaviour were not recorded. By 48 h following surgery normal activity resumed in the majority of cases, but there were exceptions to this with some animals showing continued suppression of exploration at 48 h. Although results indicated scrotal approach vasectomy impacted less on normal behaviour, the findings must be interpreted cautiously, since the actual magnitude of the observed differences was relatively small. In addition, while the frequency or duration of some exploratory behaviours (e.g. rearing and walking) were greater following scrotal vasectomy, this was still a marked reduction from preoperative exploration observed in these mice. As a result, we cannot state conclusively that either method is preferred. By 24 h there were only minor differences between groups. This indicates that the most important time for assessing these differences is during the early postoperative period and further study should focus on the time period between one and 24 h.
The Joint Working Group recommendations indicated a preference for scrotal approach versus abdominal surgery, due to the comparatively smaller incision needed to access the vas deferens. 2 However, this still requires tissue tearing that could result in additional pain, and could explain the relatively similar findings with respect to changes in normal behaviour. It is possible, indeed likely, that surgeon skill rather than technique is more significant in predicting surgical outcomes.
The relative lack of positive effect of drug treatment was disappointing, but perhaps not surprising. Previous studies using meloxicam have shown that relatively high doses are needed to illustrate positive effects following abdominal surgery in mice: up to 20 mg/kg. 9 By contrast, in mice undergoing partial hepatectomy 2 mg/kg meloxicam has been reported to be effective and to be superior to 2.5 mg/kg flunixin. 12 The conclusions of this study may have been misled by lack of a placebo-treated group. Paracetamol used alone, at doses shown to be effective in antinociceptive tests 13 had no significant positive effect after vasectomy. 5 However, synergistic effects have been demonstrated by its combination with various non-steroidal anti-inflammatory drugs (NSAIDs) including ibuprofen, meloxicam, diclofenac and ketoprofen. 13 We had hoped to demonstrate similar effects here (as large doses of NSAIDs can have other detrimental effects), but in terms of the clinical responses of the mice as opposed to effects in antinociceptive testing. However, neither the meloxicam treatment alone, or combined with paracetamol had any demonstrable beneficial effects on behaviour. As such we are forced to conclude, either; the dose rates used were ineffective, or the behavioural scoring was not sufficiently sensitive to detect any small beneficial effects of the treatment. Such consideration, importantly, serve to emphasize the necessity for including an untreated surgery group, since without these data the conclusion that both analgesic regimens were equally effective would be as likely as one claiming they were equally ineffective.
Further to this study other methods of pain assessment should be considered in order to further understand differences in pain resulting from alternative methods of surgery. This will require additional methods of evaluating pain. Evaluations that rely exclusively on behavioural changes may merely be assessing the secondary consequences (or ‘affect’) of pain, whereas methods that probe the subjective impact of pain may be more predictive. One possibility is use of facial expressions, which are known to change during periods of acute pain. 14 A recently published paper has shown that following scrotal approach vasectomy, assessment of mice using the mouse grimace scale (MGS) showed mice that received analgesic treatment (meloxicam [20 mg/kg, a larger dose than used here] or bupivacaine [5 mg/kg]) to have a significantly lower MGS score than those treated with saline. 15 This indicates a more sensitive method of pain assessment for this surgical model. Alternatively, studies designed to assess affective state 16,17 may be more appropriate in determining whether scrotal approach vasectomy is less painful than abdominal approach and to assess different analgesic regimens accordingly.
Footnotes
ACKNOWLEDGEMENTS
The authors thank Charles River UK for the supply of animals and the staff of the Comparative Biology Centre for technical support. This project was funded by the BBSRC and Pfizer Global Research and Development.
