Abstract
Social housing is recommended where possible for laboratory mice. In order to achieve this, mice must be individually identifiable. Although, various methods are available, permanent identification is often required, such as ear notching. This method is likely to be painful and to date there is limited literature on pain assessment and alleviation for this routine husbandry practice. Here we aimed to determine if the mouse grimace scale (MGS) could be used to assess pain in C57BL/6 mice following routine ear notching. Langford et al. found that very acute noxious stimuli (i.e. < 10 min in duration) did not produce a change in MGS score in comparison to baseline. Here, no significant difference was found between MGS scores at baseline and immediately post ear notching, potentially indicating that the pain associated with ear notching is either too acute to assess using the MGS tool or the practice is not painful. Studies in other species indicate that ear notching is painful, therefore, unless we can confidently conclude that the process of ear notching is not painful, we should err on the side of caution and assume it is painful due to the large number of mice ear-notched and potential welfare consequences. Alternative methods of assessing pain following this routine practice should be used in order to assess both the potential pain in mice, and the effectiveness of analgesics or local anaesthetics to relieve any associated pain.
Over three million mice were used in scientific procedures in the UK in 2012 with 64% of these being genetically-modified (GM) or harmful mutants. 1 As a social species, it is recommended that mice are group-housed wherever possible to promote welfare,2–4 thus some method of identification of individuals within a group is essential to allow for independent tracking throughout studies. Permanent methods of identification, such as ear notching, ear tattooing, ear tagging or implantation of a subcutaneous transponder, are often used rather than semi-permanent methods such as tail marking or hair clipping, especially in long-term studies to limit errors.
Pain and stress resulting from ear notching has been studied in some species. Ear notching in piglets induces pain-related behaviours (e.g. head shaking, shivering and grunting), non-specific behaviours and an increase circulating lactate levels.5,6 Ear notching has also been shown to increase heart rate and blood pressure in rats, predominantly in the first hour post procedure. 7
Ear notching is a technique that has been in regular use for over 75 years in laboratories. 8 It is considered a routine husbandry practice for identification of mice in the laboratory but is also commonly used to collect samples in order to genotype mice. 9 Ear notching is likely to be painful in mice,10,11 however to date, very limited data have been published on pain assessment and alleviation in this species. Due to the large numbers of mice requiring permanent identification and/or to be genotyped, this is an important issue to address as pain not only compromises welfare, but also potentially the validity of the data collected from these animals.
The mouse grimace scale (MGS) developed by Langford et al. 12 is a mouse pain assessment tool found to be useful in a range of laboratory procedures.13,14 It has been suggested to be most effective at detecting pain that is present for more than 10 min, but less than 4 h. 12 The process of ear notching involves application of very acute noxious stimulus, however pain associated with the process may be present for a period following the procedure as indicated by changes in heart rate and blood pressure in the rat. 7
Eutectic mixture of local anaesthetics (EMLA) cream can easily be applied to provide topical anaesthesia. EMLA cream has been used in a range of species to reduce or prevent pain associated with a range of procedures including vaccination, vessel canulation and minor surgical procedures.15–17 It is low cost and is available without prescription in the UK and North America. Its effectiveness has also been demonstrated during the process of ear tattooing in New Zealand white rabbits. Application resulted in less struggling and vocalization, lower peak heart rate and blood pressure and reduced rabbit grimace scale score in comparison to animals that were tattooed without any pain relief. 18
The aim of this study was to determine if the MGS is an appropriate technique for assessing pain in mice following ear notching. If so, a secondary aim was to determine the effectiveness of EMLA application to alleviate pain associated with ear notching.
Animals
Thirty male C57BL/6 mice (Charles River Laboratories, Kent, UK) aged eight weeks were used in this study. The mice were housed in groups of three or four in individually ventilated cages (Type 2; Arrowmight, Hereford, UK) (30 cm × 18 cm × 13 cm) with sawdust bedding and nesting material (sizzle nest, Datesand Ltd, Manchester, UK). Food (CRM (P); SDS Ltd, Essex, UK) and tap water were provided ad libitum. Chew blocks and cardboard tubes provided environmental enrichment. A seven-day acclimatization period was given prior to the start of the study. The animal room was maintained at 23 ± 1℃, 48% humidity and on a 12/12 h light dark cycle (lights on at 07:00 h).
All mice in this study were being ear-notched as a standard husbandry procedure for identification for use in other non-related research projects. The animals were not ear-notched for the purposes of data collection in this study and no regulated procedures were carried out.
Materials and methods
Baseline image collection
One week prior to notching, the mice were transferred to a quiet room and placed individually in custom-made photography cubes (80 × 80 × 80 mm) that consisted of two clear acrylic sides and two matt white sides. Close-up photographs of the face of the mice were taken across a 10 min period using a high definition camera (Casio EX-ZR100; Casio Computer Co, Ltd, Tokyo, Japan). The mice were photographed on every occasion when they directly faced the camera, apart from when grooming.
Ear notching
Random allocation of mice to each treatment group.

EMLA: eutectic mixture of local anaesthetics.
At the time of notching, the mice were fully restrained and one ear was notched using a standard ear punch (Vet-tech Solutions Ltd, Cheshire, UK) by a trained and experienced member of the technical staff, who was blind to pre-treatment. Those mice that did not receive an ear notch were restrained in the same manner and the ear punch was clicked in the immediate vicinity of the mouse ear without making any contact.
Post notch image collection
Immediately post notching, the mice were placed into the custom-made photography cubes and the process of image collection was repeated as described above. Images were taken across a maximum period of 5 min.
Image selection and scoring
For each mouse at each time point, three photographs were randomly selected, using a random number generator, from all the clear photographs for future scoring. Each selected photograph was cropped and only the head of the mouse remained to prevent any bias in scoring the images due to the posture of the mouse. The selected cropped photographs of each strain were added to pre-designed excel MGS scoring files in a random order, and were assessed by two MGS-trained coders blind to the project aims and methods. For each picture, four facial action units (FAUs), orbit tightening, cheek bulge, nose bulge and ear position were scored based on the MGS method developed by Langford et al., 12 each facial unit was scored on a three-point scale separately (0 = not present, 1 = moderate, 2 = severe), and the sum of all four FAUs were analysed. Whisker position was excluded from the analysis due to the high number of pictures where scorers were unable to assign a score.
Statistical analysis
Data were analysed using SPSS software (version 21; SPSS Inc, Chicago, IL, USA). As data collected were not on the continuous scale, they were analysed non-parametrically. MGS scores between the treatment groups (see Table 1) at each time point were compared using a Kruskall–Wallis test. Pre and post ear notching MGS scores were compared within treatment groups using a Wilcoxon test. Results were considered statistically significant when P < 0.05.
Results
The four treatment groups were compared at each time point. There was no significant difference in MGS score between the groups pre or post ear notch (Figure 1). The baseline and post notch MGS scores were compared. There was no significant difference between the two time points.
Mean mouse grimace scale (MGS) score ( ± SEM) at each time point. Maximum obtainable MGS score was 8. EMLA: eutectic mixture of local anaesthetics.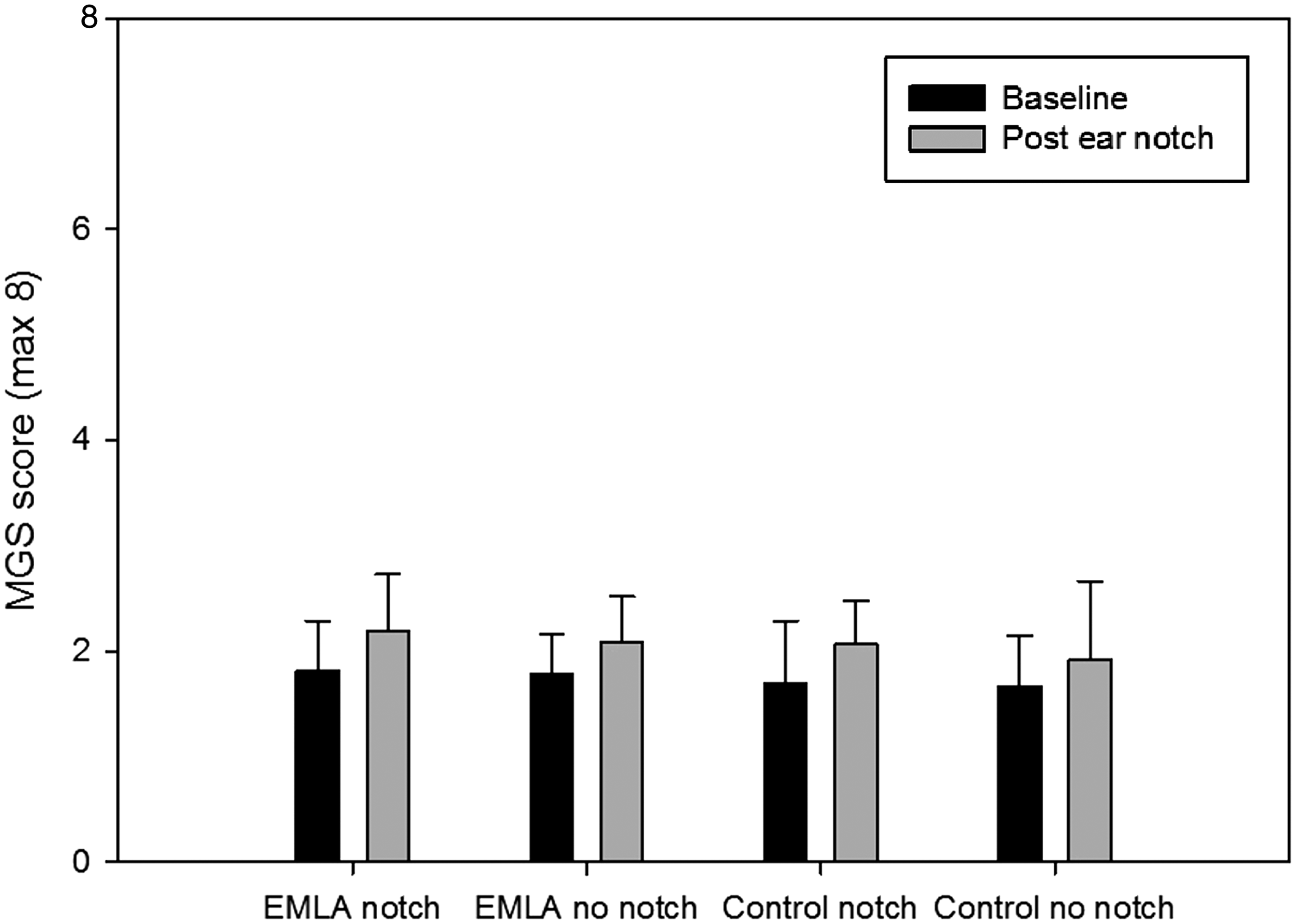
Discussion
Ear notching is a routine husbandry practice used in laboratories to identify individual mice or for genotyping. Based on studies in other species including pigs and rats, the assumption is made that this practice is likely to be painful in mice.5–7 The MGS has been demonstrated to be an effective research tool in pain assessment following a range of procedures where pain is present for between 10 min and 4 h. 12 Here, we did not see any change in MGS score immediately following ear notching in male C57BL/6 mice thus indicating that the MGS is not a suitable pain assessment technique for this practice. The data could be interpreted in an alternative way, arguing that no change in MGS score demonstrates that ear notching is not painful in male C57BL/6 mice. However, based on data collected in other species, e.g. rats and pigs5–7 and the suggested limitation with the MGS that it is not effective for painful stimuli with a duration of less than 10 min, 12 the assumption can be made that this procedure is likely to be painful unless further evidence can be provided to demonstrate otherwise. Although this finding appears to contrast with change in grimace score observed in rabbits following ear tattooing, 18 this procedure is likely to be more traumatic and so to potentially cause pain over a longer period.
Due to the potential lack of sensitivity of the MGS in this study, potential benefits of applying EMLA prior to notching could not be assessed. However, it is noted that application of EMLA in itself did not result in any changes in the MGS score of the control group, male C57BL/6 mice. Alternative methods of pain assessment should be trialled, for example, focusing on behaviour of the mice immediately following the notching process.
Footnotes
Acknowledgements
The authors would like to thank the animal care staff of the Comparative Biology Centre, Newcastle University for technical assistance and Katherine Tyson for assisting with the data collection.
Funding
This work was funded by the National Centre for the Replacement, Refinement and Reduction of Animals in Research (NC3Rs; Grant/Award No. G1100563).
