Abstract
Immunosuppressive (IS) medication is needed to avoid graft rejection in porcine transplantation models. An ideal IS therapy should have no side-effects, but increased susceptibility to infections, disturbed intestinal microflora and toxic effects on organs and tissues are commonly reported. The aim of the present study was to design an IS protocol with tacrolimus and mycophenolic acid to be used for maintenance therapy in the post-transplant period. An eligible whole blood trough value for tacrolimus was 5–15 μg/L. Conventional specific pathogen-free pigs were fitted with an indwelling catheter under general anaesthesia, and after the acclimatization period three groups were formed: group A (n= 4) received 0.15 mg/kg body weight (BW) twice daily tacrolimus and 500 mg twice daily mycophenolic acid; group B (n= 4) received 0.3 mg/kg BW twice daily tacrolimus and 500 mg twice daily mycophenolic acid; group C (n= 2) did not receive any medication. Daily clinical examinations and analyses of blood concentrations of tacrolimus and glucose were performed. Total and differential white blood cell counts, enzyme activities, bilirubin and electrolyte concentrations were measured every fourth day. At the end of the experiment, the pigs were killed with an overdose of pentobarbital intravenously and a necropsy was performed immediately. All animals seemed to tolerate the IS treatment well. No alterations in their clinical state of health were observed throughout the study and daily weight gain was similar for the three groups. The necropsy did not reveal any pathological findings related to medication. The study showed that 0.25 mg/kg BW twice daily tacrolimus and 500 mg twice daily mycophenolic acid would be an appropriate maintenance dosage for conventional pigs.
Immunosuppressive (IS) medication is compulsory for allo- and xenotransplantation procedures to avoid graft rejection, regardless of whether it is a human patient or a research animal. Often, an induction therapy is given for a short period of time in conjunction with the transplantation followed by maintenance therapy given throughout the lifespan of the graft. Side-effects associated with IS treatment have to be considered, and in general the recommended trough levels (i.e. lowest drug level that is needed to reach therapeutic range) are lower today than those previously used. The IS protocol has to be adjusted to the species involved, and the age and general health status of the recipient. Different protocols and combinations of drugs have been utilized but during the last decade, tacrolimus, a second-generation calcineurin inhibitor that suppresses interleukin-2-dependent T-cell activation, and mycophenolic acid, which suppresses leukocyte proliferation, have been widely used as maintenance therapy. 1,2 In addition to general adverse effects of IS therapy, such as increased susceptibility to infections and an increase in malignancies, tacrolimus and mycophenolic acid can be nephrotoxic and disturb the glucose metabolism, respectively. 1–6
Human type 1 diabetes mellitus (T1DM) patients with multiple hypoglycaemic episodes or uncontrolled diabetes despite compliance with an insulin regimen may benefit from transplantation of islets of Langerhans
7,8
(ITR Giessen, see
The pig is a valuable large animal model in diabetes research, and DM can be successfully induced by streptozotocin in both minipigs 11 and conventional Landrace pigs. 12 In addition to its similarities to humans with respect to organ size and physiology, the diabetic pig's clinical signs of disease as well as metabolic alterations are similar to that of humans. 13 Conventional pigs have also been used in a non-survival transplantation study, undertaken to quantify early islet engraftment in the liver with a positron emission tomography/computed tomography scanner. 14 In both pigs and humans the islets stay in the portal vein lumen or wall and are not incorporated in the liver parenchyma as seen in rodents. 15 To improve the outcome of clinical islet transplantation a preclinical porcine model using surviving animals can be of great value. Such an animal model has to include appropriate IS medication. Therefore, the aim of the present study was to design an IS protocol for pigs, including tacrolimus and mycophenolic acid with an eligible whole blood trough value for tacrolimus of 5–15 ng/mL. This value is the same as that chosen for recipient pigs in small bowel transplantation. 16 Mycophenolic acid was given at a fixed dose since no consensus on drug monitoring exists. 17
Materials and methods
Animals and experimental design
One litter consisting of 10 high health certified specific pathogen-free (SPF) pigs (Yorkshire × Landrace, Serogrisen, Ransta 18 ) weighing 22–30 kg on arrival were used. The animals were habituated for two weeks and housed in individual pens measuring ∼3 m2 in a separate stable. Straw was used as bedding and the animals were kept within sight and sound of one another. A 12:12 h light/dark schedule was used and the room temperature was 20 ± 2°C. The pigs were fed on actual weight twice daily with commercial finisher diet without growth promoters (Singel Flex, Odal, Sweden) and had free access to water.
The protocol ran for a total of six weeks and all procedures were approved by the Ethical Committee for Animal Experimentation, Uppsala, Sweden. During the acclimatization period the pigs were handled frequently and trained to step onto an electronic scale. In order to make the oral administration of IS medication easier the pigs were trained to be hand-fed with meatballs which were later prepared with IS drug capsules. The pigs were weighed three times per week and daily weight gain was calculated, clinical examinations were performed daily by a veterinarian. Blood for haematology and clinical chemistry was collected by venepuncture from the jugular into EDTA tubes during the acclimatization period before onset of IS medication, and then every fourth day during the medication period. Also, whole blood concentrations of tacrolimus were analysed daily during this part of the experiment. In order to facilitate frequent blood sampling the animals were fitted with an indwelling silicon catheter (SIL-C70 with rounded distal tip, Instech Solomon, Plymouth Meeting, PA, USA) into the jugular vein after the acclimatization period. Surgery was performed under general anaesthesia. In brief, after an overnight fast atropine sulphate, 0.05 mg/kg body weight (BW) (Atropin®, Merck NM, Sweden), was given intramuscularly and anaesthesia was induced with a combination of medetomidine (Domitor® vet 1 mg/mL, Orion Pharma Animal Health, Sollentuna, Sweden), tiletamine and zolazepam (Zoletil® 250 mg of each compound/mL, Reading, Carros, France) at a dose of 0.03 mL/kg BW. Buprenorphine (Temgesic® 0.3 mg/mL, Schering-Plough, Brussels, Belgium) was provided for additional analgesia and administered intramuscularly preoperatively at a dosage of 0.01 mg/kg BW. After intubatation, anaesthesia was maintained with isoflurane vaporized in oxygen, with 25% nitrous oxygen added initially (5–10 min). The concentration of isoflurane (1–2.5%) was continuously adjusted to achieve an adequate depth of anaesthesia. The silicon catheter was brought through a subcutaneous tunnel and exteriorized on the back between the scapulae, and finally covered with a piece of canvas. Flushing was carried out twice daily with 2% heparinized saline (5000 IU/mL, Heparin, Leo, Apoteksbolaget, Sweden). The anaesthetic and surgical protocols have been described in detail previously. 13,19 After the surgical procedure, penicillin (Penovet® 300 mg/mL, 0.7 mL/10 kg; Boehringer-Ingelheim, Sweden) was administered intramuscularly once daily for three consecutive days. At the end of the experiment the pigs were killed with an overdose of pentobarbital sodium (Avlivningsvätska®, Apoteksbolaget, Sweden) intravenously and a necropsy was performed immediately.
IS medication
One week after the surgery, three treatment groups were formed and the IS medication was administered orally twice daily for a two-week period: group A (n= 4) received 0.15 mg/kg BW twice daily tacrolimus (Prograf®, Astellas Pharma, Malmö, Sweden) and 500 mg twice daily mycophenolic acid (CellCept®, Roche, Stockholm, Sweden); group B (n= 4) received 0.3 mg/kg BW twice daily tacrolimus and 500 mg twice daily mycophenolic acid; group C (n= 2) did not receive any immunosuppression. All blood samples for tacrolimus trough level determination were collected daily at 07:00 h, immediately prior to IS treatment, with the exception for the second day of medication when samples were collected at 19:00 h. During the IS period the commercial diet was supplemented with sour milk and the animals were fed one hour after the IS administration.
Blood analyses
EDTA-preserved blood was analysed for total and differential white blood cell counts with an electronic cell counter validated for porcine blood (Cell-Dyn 3500, Abbott, Wiesbaden, Germany). Serum samples were analysed for activities of γ-glutamyltransferase (γ-GT, μkat/L) and concentrations of magnesium, sodium, potassium, creatinine and total bilirubin using an automated equipment (Konelab 30, Espoo, Finland).
Tacrolimus whole blood levels were determined at Uppsala University Hospital by an immunoassay (CEDIA® Tacrolimus Assay, Microgenics Corporation, Fremont, CA, USA; Hitachi 912, Roche).
Blood glucose concentrations were measured using test strips (Accu-Chek, Roche Diagnostics, Basel, Switzerland; this test was validated for porcine samples at the Department of Clinical Chemistry, SLU).
Statistical methods
Data are presented as mean ± SEM or given as range. Differences between groups were evaluated by the Mann-Whitney U-test.
Results
All animals seemed to tolerate the IS treatment well, they had a good appetite and no gastrointestinal disturbances were observed throughout the study. Neither were any alterations in their clinical state of health noted. At the end of the experiment the animals weighed between 34 and 58 kg, their daily weight gain was ∼650 g, which was similar for the three groups.
The necropsy did not reveal any pathological findings except in two pigs, which displayed minor abscesses (∼1 mm2) between the scapulae at the site of the orifice for the catheter, and in one pig an ulcer (1 × 1 cm) in the cutaneous portion of the stomach was noted.
Blood analyses
In all pigs, the total WBC counts (8.1–20.0 × 109/L), the differential counts and the haemoglobin concentrations were within the reference values throughout the experiment. No morphological changes of the erythrocytes were observed at any sampling occasion. The creatinine and bilirubin concentrations, and activity of the enzyme γ-GT did not differ between treatment groups and sampling occasions. Also, no significant changes in the serum concentrations of magnesium, sodium and potassium were observed throughout the experiment. The blood glucose levels were within reference values throughout the experiment and ranged from 3.9 to 5.3 mmol/L.
Tacrolimus concentrations
One day after the onset of IS medication (day 1 in Figure 1) the whole blood trough values of tacrolimus in both treatment groups reached the eligible level of 5–15 μg/L. Because the trough levels were above 15 μg/L the dosage of tacrolimus in group B was lowered to 0.25 mg/kg BW twice daily from day 9 onwards. Accordingly, the tacrolimus dosage in group A was increased to 0.25 mg/kg BW twice daily from day 10 onwards since trough levels were below 5 μg/L. Both modifications resulted in trough levels that were within 5–15 μg/L.
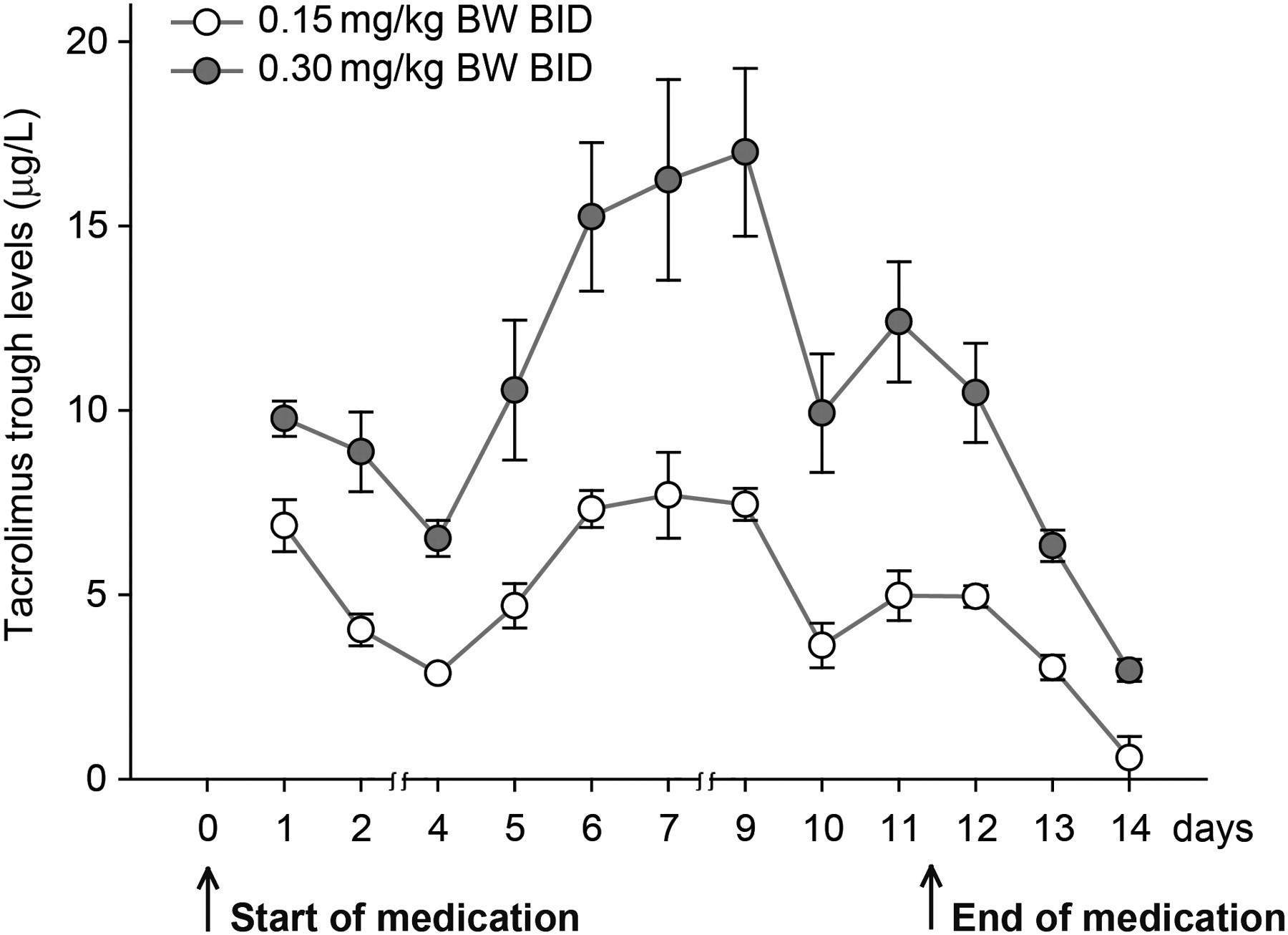
Tacrolimus trough levels during a two-week period. Group A (white circles, n= 4) and group B (black circles, n= 4) received 0.15 and 0.3 mg/kg BW tacrolimus (twice daily), respectively. From days 9 (group B) and 10 (group A) of the medication period the administration of tacrolimus was changed to 0.25 mg/kg BW. Data are presented as mean ± SEM. BW: body weight
Discussion
Transplantation procedures in mice and rats are performed on inbred strains and therefore IS medication is not necessary. However, transplantation studies on outbred species such as dogs, monkeys and pigs require sufficient IS to avoid graft rejection in the post-transplant period. Obviously, IS treatment has side-effects such as increased susceptibility to infections but also toxic effects on organs and tissues, and therefore a balance is sought between efficacy and toxicity. In humans, long-term IS medication result in a four-fold higher incidence of tumours. 20 Corticosteroids have diabetogenic effects and cause a need for insulin. 20 To avoid this a steroid-free IS regimen was introduced by the Edmonton group. 21 For maintenance therapy sirolimus and tacrolimus were used in that study. However, sirolimus frequently gives rise to side-effects such as mouth ulcers and joint pain and is commonly replaced by mycophenolic acid. 22,23
In the present porcine preclinical model we evaluated an IS maintenance therapy with tacrolimus and mycophenolic acid. A tacrolimus dosage of 0.25 mg/kg BW twice daily resulted in the eligible whole blood trough level 5–15 μg/L. It is known that children require approximately a 1.5–2 times higher dose of tacrolimus than adults (Prograf monograph, Fujisawa/Astellas Pharma, 2003) because of a higher clearance rate. Accordingly, the young pigs used in the present study may have the same requirements as children and the dosage might need to be adjusted if older pigs are utilized. Nevertheless, daily measurements of the trough levels are needed at the beginning of the therapy and then once weekly. The interindividual variations in the concentration of tacrolimus among the pigs were less than those observed in humans. One explanation for this finding is that animals from a high health herd were used but patients subjected to transplantation are for obvious reasons in a poor health condition, which affects the metabolism. Further, even if conventional crossbreed pigs are outbred, the genetic variation is less than among humans, and in the present study all animals used were littermates. The pigs were fed twice daily on a strict conventional diet supplemented with vitamins and minerals, thus the diet was well nourishing and the energy intake was balanced to the daily need. Human patients cannot be expected to stick to such a restricted diet, and the drug uptake may be related to, e.g. variation of fat and carbohydrate intake.
Tacrolimus and mycophenolic acid medication often results in alterations of haematopoiesis, metabolic disturbances, e.g. hyperglycaemia, renal dysfunction, nausea and diarrhoea. To prevent disturbances in the gut microflora the animals included in the present study were given sour milk daily. To facilitate oral medication and to minimize the stress of handling, the drug capsules were hidden in meatballs. The taste is greatly appreciated by the animals and meatballs were also used as rewards in connection with blood sampling and weighing. Throughout the experiment none of the pigs showed any signs of disease and the daily weight gain was similar in principals and control animals. Neither was any alteration noted in the haematopoiesis, and the clinical chemical data were within the reference values. Finally, necropsy confirmed that the pigs had been healthy. In long-term preclinical animal studies, when IS treatment is needed for months, adverse effects may develop, but if careful clinical examinations and frequent blood samples are taken for both drug concentrations and complete blood count, the IS medication can be balanced.
Taken together, a porcine preclinical diabetic model can help to improve islet transplantation procedures. An optimal IS regimen is needed in the post-transplant period and in the present study it was shown that 0.25 mg/kg BW twice daily tacrolimus and 500 mg twice daily mycophenolic acid would be an appropriate initial dosage for conventional pigs.
Footnotes
ACKNOWLEDGEMENT
The financial support from the Swedish Research Council for Environment, Agricultural Sciences and Spatial Planning (FORMAS) is gratefully acknowledged.
