Abstract
The pig is commonly used in renal transplantation studies since the porcine kidney resembles the human kidney. To meet the requirements of intense caretaking and examination without stress, a 2-week socialisation and training programme was developed. Conventional cross-breed pigs (n = 36) with high health status were trained for 15 min/day in a four-step training programme before kidney transplantation. The systematic training resulted in calm animals, which allowed for ultrasound examination, blood sampling and urine sampling without restraint. When a 2-methacryloyloxyethyl phosphorylcholine polymer-coated jugular catheter introduced via the auricular vein was used for post-operative blood sampling, clotting was avoided. To assess renal function, urinary output was observed and creatinine and cystatin C were measured; the latter was not found to be useful in recently transplanted pigs. The results presented contribute to the 3Rs (refine, reduce, replace).
Introduction
Renal transplantation is a life-saving procedure for patients with end-stage renal disease, 1 a disabling condition with a high mortality rate. 2 Despite improved short- and long-term survival of renal grafts over recent last decades, 50% of transplanted grafts from living donor organs are still lost within 12 years 3 and the life-saving benefit of renal transplantation lasts only as long as the transplant. 4 Thus, more research is needed to improve the survival of renal grafts. The pig is now the major large animal model used in translational research and has replaced dogs and non-human primates in many research areas.5,6 The anatomical and physiological features of the species make the pig an appropriate model for many human diseases, and they are commonly used in renal transplantation studies since the porcine kidney closely resembles the structure and function of the human kidney. 7
Pigs have to be carefully prepared to meet stressful experimental situations. Thus, optimal socialisation, training and nursing during the peri-operative period facilitate procedures, and benefit animal welfare and scientific outcomes. Some general guidelines for the handling of pigs in research have been published,5,8,9 but information about socialisation and pre-operative training for specific procedures is very limited.
A potentially stressful situation in many porcine studies is blood sampling due to the need for restraint. 10 Indwelling venous catheters are therefore preferred for repeated blood sampling, 11 but must be meticulously managed to avoid occlusion caused by mechanical obstruction, fibrin sheet formation, thrombosis or infection, of all which have been described in humans.12,13 Monitoring of urine production is central for assessing the function of renal grafts.14,15 Urine production can be measured but requires catheterisation of the urinary tract, which is technically difficult in pigs and may introduce microbes.16,17 In humans, ultrasound examination of the bladder is recommended to measure urine production. 18 Automatic portable devices have been developed; BladderScan® is part of routine care for urine measurement in humans, but has not been evaluated in pigs. Further, estimation of glomerular filtration rate (GFR) by measuring plasma cystatin C concentrations can be used to assess renal function.19,20
The aim of the present study was: (a) subject pigs to systematic socialisation and training during the pre-operative acclimatisation period to enable post-operative blood sampling, urine sampling, and ultrasound examination of the bladder and transplanted kidney without restraint; (b) evaluate the use of the Seldinger technique for minimal invasive fitting of venous catheters (conventional and 2-methacryloyloxyethyl phosphorylcholine (MPC)-based polymer-coated) through the auricular vein and into the jugular vein; and (c) investigate whether plasma concentrations of cystatin C can be used for estimation of GFR in recently transplanted pigs.
Materials and methods
All procedures were approved by the Ethics Committee for Animal Experimentation, Uppsala, Sweden (C123/14).
Animals and housing
Thirty-six pigs (Yorkshire x Hampshire, both sexes, 7–8 weeks upon arrival) obtained from the university farm, Lövsta, Swedish University of Agricultural Sciences (SLU), were included in a training programme during a 2-week acclimatisation period. Twenty-five of these 36 animals were used as recipients in renal transplant studies (referred to as studies I–IV) and the remaining 11 pigs were used in other studies. In each transplant study, the pigs came from the same litter. In addition, 18 pigs used as organ donors were similarly acclimatised, but not included in the training programme. The animals arrived 14 days before the surgical procedure and were housed at the Department of Clinical Sciences in individual pens measuring 3 m2, within sight and sound of one another. Prior to surgery, straw and wood shavings were provided as bedding, whereas synthetic fleece was used post-operatively. The temperature in the housing area was 18 ± 2℃, a 10:14-hour light:dark schedule was used, and an infrared lamp was provided in a corner of each pen. The pigs were fed commercial finisher diet (Solo 330 P SK, Lantmännen, Sweden) every day at 7 a.m. and 4 p.m., the amount depending on body weight (BW) and according to the SLU regimen for growing pigs. Water was provided ad libitum. The general condition of the pigs was examined once a day during the acclimatisation period.
Socialisation and training protocol
Each pig was trained in a four-step training programme, which is summarised in Table 1. Five persons, with experience of training pigs, took equal part in the social training programme. In step 1, pigs were allowed to adapt to the new environment for three days. During this time, no specific training or socialisation took place. The staff only entered the housing area for feeding and cleaning of the pens (Figure 1). In step 2, the trainer sat in the pen for 15 min per day, allowing the pig to get accustomed to the person. Once the pig was close enough, the trainer started to gently touch and brush the animal and offered it pieces of fruit from the hand. The pigs were also trained to accept touching and palpation of the ears as preparation for blood sampling from the auricular vein. In step 3, the training from step 2 continued and further included touching with an ultrasound transducer dummy over the abdomen to tolerate ultrasound examination of the urinary bladder and transplanted kidney post-operatively. Pigs were accustomed to urine collecting by a trainer who held a paper kidney dish for free-flow sampling. In step 4, the training from step 2 and 3 continued, and the pigs were also trained to undergo a clinical examination including auscultation of the heart and lungs. For each individual pig, a new step was introduced once the pig had been completely accustomed to the procedures in the previous step. In addition, from start of step 2, the pigs were trained to step onto a spring scale (Ecco 101, Farmer Tronic Industries A/S, Vamdrup, Denmark) for repeated recording of BW throughout the study.
Socialisation and training of pigs in renal transplantation studies. Step 1: left to settle, step 2: interaction with trainer and brushing, step 3: urine sampling and step 4: clinical examination. Socialisation and training of pigs in four steps during a two week acclimatisation period.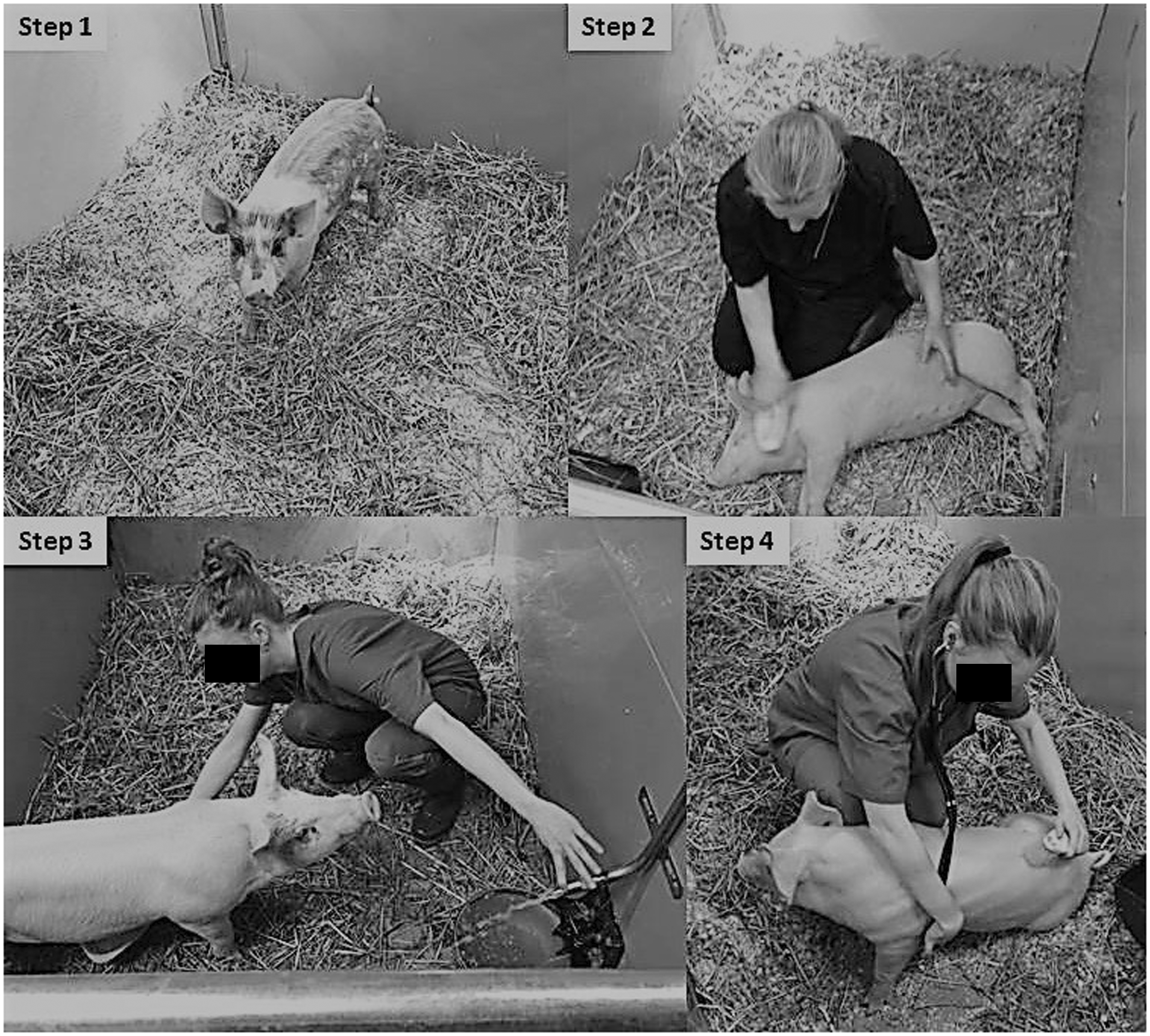

Anaesthesia and catheterisation
Anaesthesia was induced in the pigs’ home pens with tiletamine-zolazepam mixed with medetomidine intramuscularly (IM). 21 Pre-operatively, buprenorphine was given at a dose of 0.01 mg/kg BW IM and bensylpenicillinprokain (Penovet® vet. 300 mg/mL, Beohringer Ingelheim, Ingelheim, Germany) at 20 mg/kg BW IM. In each case, the animal was covered with a blanket and transported to the preparation room, where oxygen saturation and pulse rate measurements were started. To prevent heat loss, socks were put on the claws and distal legs. An intravenous (IV) catheter (BD Venflon™ 20 G × 32 mm, BD Medical, Franklin Lakes, US) was placed in an auricular vein, and blood was sampled for the analysis of complete blood cell count (CBC) and enzyme activities. Endotracheal intubation was facilitated by use of a laryngoscope. Oxygen (4 L/min) was delivered by a face mask until the animal was connected to the anaesthesia circle. Anaesthesia was maintained with isoflurane (IsoFlo® vet. Orion Pharma Animal Health, Sweden) in an oxygen/air mixture (FIO2 0.3) from a rebreathing circuit and all pigs were mechanically ventilated (FLOW-i® Anaesthesia Delivery System, MAQUET Medical Systems, US). Epidural morphine (Morfin Epidural Meda 2 mg/mL, Meda AB, Sweden) 0.1–0.12 mg/kg was administered according to the protocol of Malavasi et al. 22 The pigs received IV infusion of lactated Ringer solution (Ringer-acetate, Fresenius Kabi AB, Sweden) 10–15 mL/kg/hour and succinylated gelatine (Gelofusine®, B. 40 mg/mL, Braun Melsungen AG, Germany) as a bolus of 3 mL/kg during 10 min, followed by 3 mL/kg/hour. To achieve adequate arterial blood pressure (≥60 mmHg), dobutamine (DOBUTAMIN Carino® 250 mg/50 ml, Carinopharm, Germany) was given as an infusion (5–15 µg/kg/min). During anaesthesia, circulatory and respiratory parameters were continuously monitored (AS/3 Anesthesia Monitor, Datex-Ohmeda, Finland). Arterial blood pressure was intermittently measured oscillometrically using an inflatable cuff placed around a forelimb and connected to the monitor.
To allow repeated blood sampling, a polyurethane catheter (BD Careflow™ 3 Fr 200 mm, BD Medical, US) was introduced via the auricular vein into the jugular vein using the aseptic Seldinger technique. In study IV, the same type of catheter additionally coated with MPC was used. The catheters were sutured onto the ear with monofil-coated polyamide (Supramid 2-0, B Braun Medical, Sweden) and covered with a bandage (Snøgg AS, Norway).
Surgical procedure
The recipient pigs underwent allogeneic single kidney transplantation. Through a 15–20-cm abdominal midline incision, the iliac vessels in the right iliac fossa were identified and gently mobilised. The distal segment of the vena cava as well as the iliac artery (from its emergence from the aorta) were mobilised, sealing the surrounding lymphoid tissue. The renal graft was thereafter placed in the right iliac fossa in the proximity of the iliac vessels. The renal vein and artery were trimmed, and subsequently anastomosed in an end-to-side fashion to the recipient’s right iliac artery and distal vena cava using a polypropylene 7/0 running suture (PROLENE®, Ethicon, USA). The ureters were implanted by extravesical ureteroneocystostomy to the top of the bladder by 6-0 polydioxanone suture (PDS®, Ethicon, US). Thereafter (in studies I, II and IV), a bilateral nephrectomy of the native kidneys was performed. The midline incision was closed by running a 2/0 polyglactin (VICRYL® Ethicon, US) fascia suture and skin clips. The mean duration of the surgery was 3 hours (2.5–5.5 hours).
Post-operative care
Towards the end of surgery, isoflurane administration was discontinued, the fraction of inspired oxygen was increased (FIO2 1.0) and the pigs were weaned off the mechanical ventilator. The animals were placed in a cage where they could easily be monitored continuously by the staff. Oxygen supplementation was delivered by a face mask. Heart rate, respiratory rate, oxygen saturation and body temperature were monitored until the pigs regained consciousness. At the end of anaesthesia and during the remaining post-operative period, additional buprenorphine (0.03 mg/kg) was given IV after assessment of the general condition and behaviour of each pig. 22 If needed, the pigs were hand-fed fruit to stimulate appetite, supported to drink water, and assisted to stand and walk post-operatively.
Blood sampling and analyses
In studies I, II and IV, after induction of anaesthesia, blood was collected and EDTA-preserved for analysis of CBC, including leukocyte differential counts, with an electronic cell counter validated for porcine blood (Advia 2120, Siemens, Germany). Serum samples were analysed for enzyme activities of aspartate amino transferase, alanine amino transferase, γ-glutamyltransferase and glutamate dehydrogenase by automated equipment (Architect C4000, Abbott Diagnostics, Australia).
Blood samples were then collected during surgery and daily up to 4 days post-operatively in all studies. In studies I, II and IV, creatinine was analysed via an enzymatic method (8L24-01, Abbott Laboratories, USA). In study I, cystatin C was analysed with a particle-enhanced immunoassay (reagent: 1014; Gentian, Moss, Norway) on an Architect Ci8200 analyser (Abbott Laboratories). After blood sampling in studies I–III, the catheters were flushed with saline and filled with heparinised saline (100 IU/mL). In study IV, the MPC-coated catheters were flushed and filled with saline without heparin. During handling of catheters, all personnel used gloves and followed aseptic routines. The caps and bandages were changed when appropriate.
Ultrasound examinations and urine sampling
Ultrasound examinations were performed pre-operatively in non-trained, anaesthetised (IM tiletamine-zolazepam-medetomidine) donor pigs to screen for renal cysts. Post-operatively, in studies I, II and IV, the kidneys were examined without restraint via ultrasound (Logiq e R6, GE Healthcare, USA) using linear (10 MHz) and curvilinear (4 MHz) probes. Kidney length, echogenicity and cortiocomedullary definition were assessed, the renal pelvic region was evaluated for evidence of pyelectasia and colour Doppler was used to evaluate the presence of blood flow in the kidneys. In studies I, II and IV, the urinary bladder was examined one or two times per day with a portable ultrasound machine (Imago 1401MG05, ECM, France). Further, in study II, a semi-automated ultrasound instrument (BladderScan® BVI 9400, Allytec AB, Sweden) intended for urine volume measurement was evaluated for use in non-sedated pigs, and results from the scans were compared to results from ultrasound examinations. Free-flow urine samples were collected with a paper dish once daily in studies I and II.
Euthanasia and post-mortem examinations
At the end of the studies, the pigs were euthanised with pentobarbital sodium (Allfatal vet. 100 mg/mL, Omnidea, Sweden) either IV or intracardially during anaesthesia. All pigs underwent post-mortem examination at the Department of Pathology, SLU.
Results
General health status
The general health status was good throughout the acclimatisation period, with CBC and enzyme activities within the reference range for all pigs. The average daily weight gain during the study was 0.68 ± 0.04 kg, which corresponds to the weight gain of conventional high-health herd pigs of the same age. 23 Post-mortem examinations showed no signs of disease apart from changes related to renal transplantation.
Socialisation and training
All pigs fulfilled the training programme and learned to accept all procedures within 14 days, even though some pigs became accustomed to the different steps faster than others (Figures 1 and 2). The first day, all pigs showed signs of fear when people entered the stable, but became less afraid during the two following days (step 1). Post-operatively, it was possible to palpate the ears and collect blood from the indwelling vein catheters, and examine the renal graft and urinary bladder with ultrasound without restraint. In order to keep the pigs still during ultrasound examinations, one person patted, brushed and offered fruit. The first few times, when collecting free-flow urine samples, the pigs stopped urinating when the tray was put under the belly. However, after training during step 3, urine samples could be collected from all pigs.
After 14 days, all pigs fulfilled the predetermined requirements and were subjected to surgery. The horizontal lines indicate the time (days) for each individual to fulfil the different steps.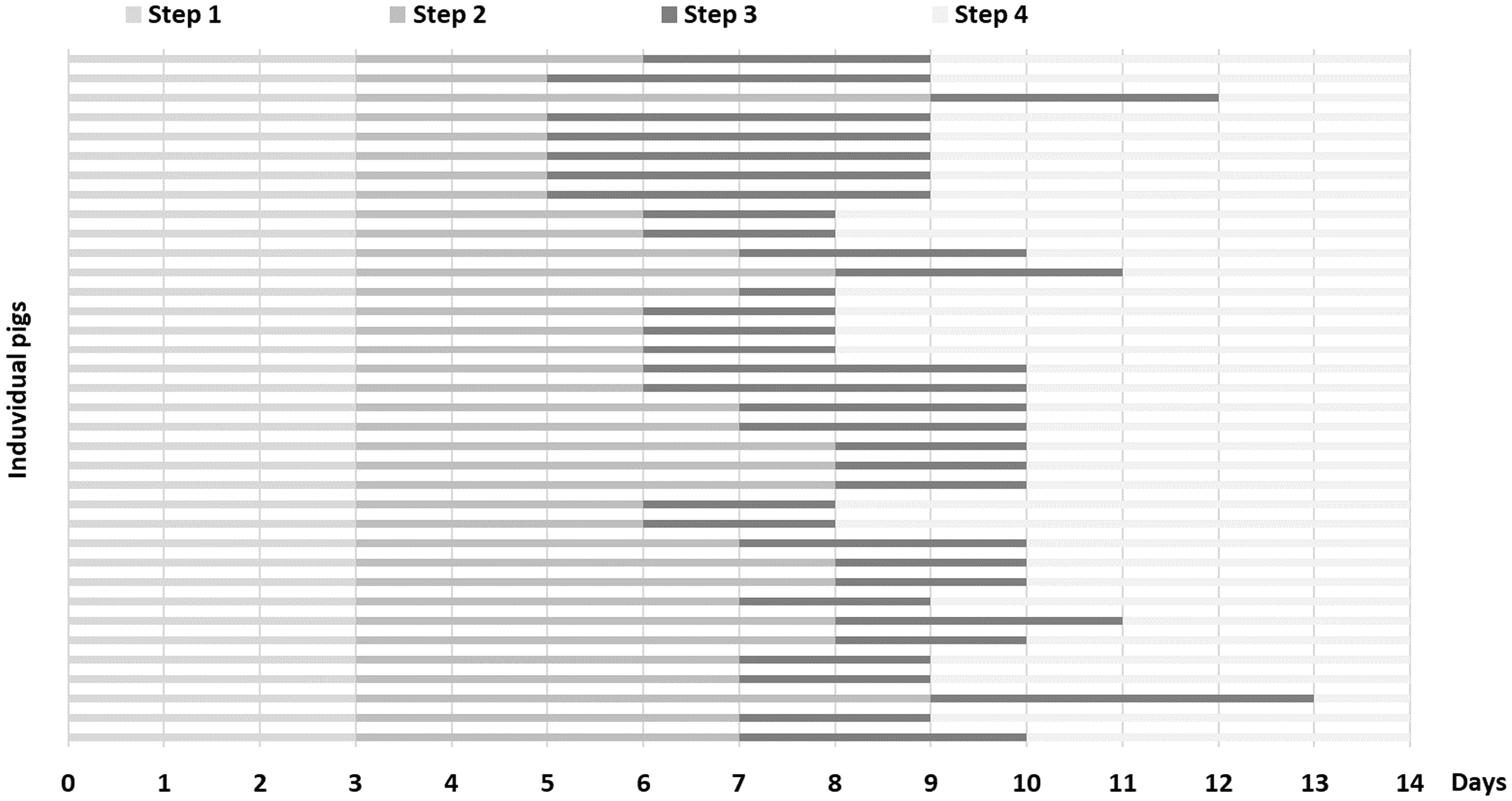
Post-operative care
The anaesthesia and surgery were successful, and the pigs were fully conscious within 1–3 hours following extubation. The general condition of the pigs was moderately affected post-operatively; they were inactive and did not eat all of the feed offered, but commenced eating and drinking to the same extent as before surgery within 24 hours. Two days after surgery, the general condition of all pigs was either unaffected or mildly affected.
On the day after surgery, weakness of the left pelvic limb was observed in three of six transplanted pigs in study I and 1 of 10 transplanted pigs in study IV, and the animals could not regain a standing position even when manually assisted. These pigs were given massage several times during the day, and all pigs had recovered within 24 hours.
Venous catheters and blood analyses
Twenty-five of the 27 catheters used in the included studies could be flushed repeatedly throughout the experimental period. Two catheters could not be flushed due to mechanical damage and were removed. In studies I–III, which used uncoated catheters, blood could not be drawn from 4 out of 15 catheters 24 hours after insertion. In study IV, when MPC-coated catheters were used, it was possible to withdraw blood at all sampling occasions (i.e. for up to 96 hours). Plasma creatinine concentrations after transplantation are presented in Figure 3. Plasma cystatin C concentrations were below the <0.3 mg/L detection limit at all sampling occasions.
Plasma creatinine concentrations in samples taken before and 1, 2, 3 and 4 days after renal transplantation in pigs (n = 11, studies I and II). Values are presented in boxplots with first quartile, median and third quartile. Whiskers represent interquartile range × 1.5. Dots represents outliers > interquartile range × 1.5.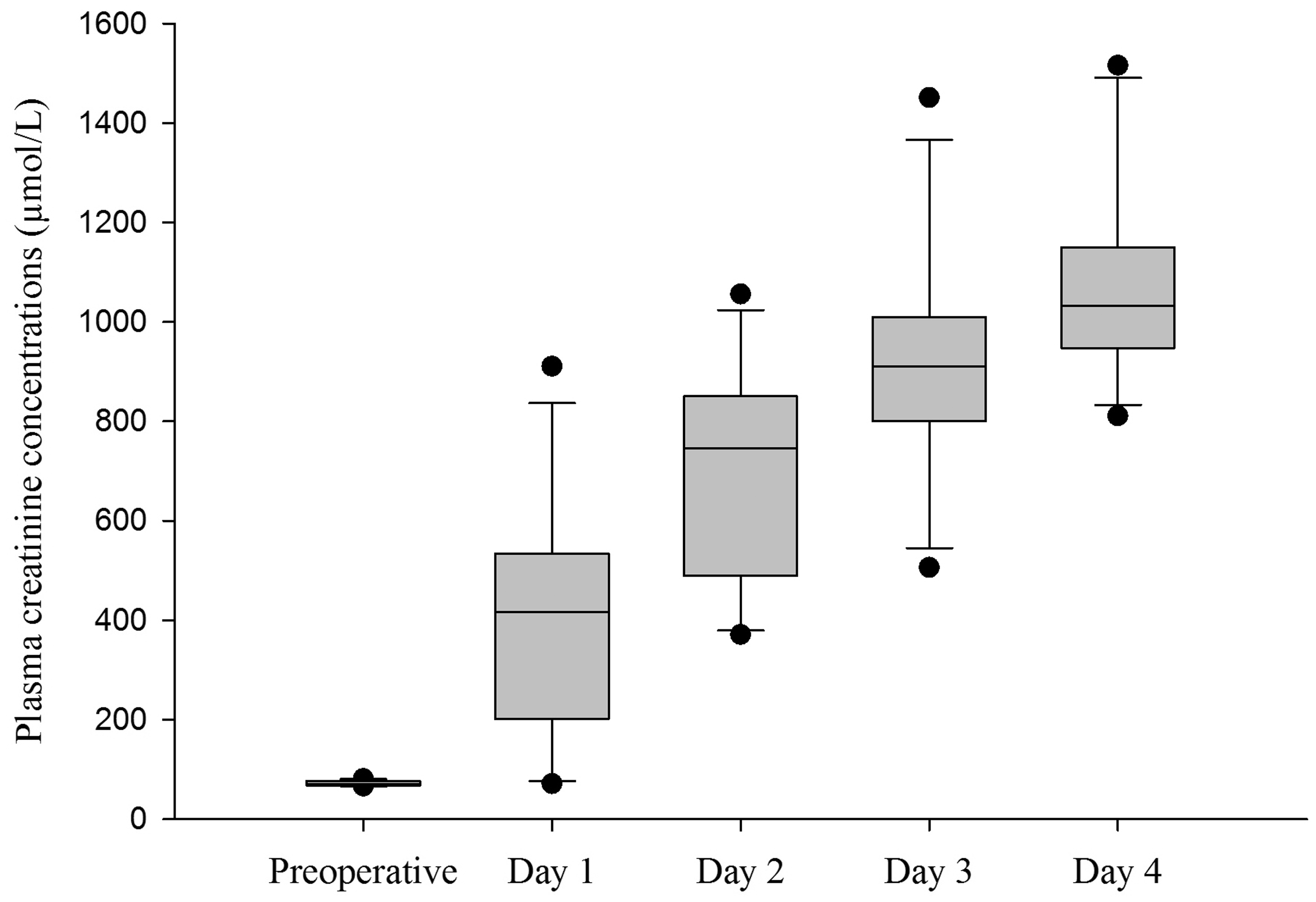
Ultrasound examination
The presence of renal cysts ≥10 mm, diagnosed by ultrasound examination, led to exclusion of 71%, 50% and 0% of the donated organs in studies II, III and IV, respectively. Post-operatively, urine was detected in the bladder on day 1 in 12 out of 21 pigs. Within 3 days, urine was detected in the bladder of all pigs or they were observed to urinate spontaneously. The ultrasound operator subjectively thought that the BladderScan® and ultrasound examinations agreed 30% of the time, that the BladderScan® gave a reading of no urine when urine was detected with ultrasound 30% of the time, and overestimated the volume 40% of the time.
Discussion
In survival animal studies with major surgery, a number of post-operative examinations (e.g. ultrasound examination) may be necessary. Sedation of the animals in order to carry out such examinations should be avoided as its administration may induce stress, and the drugs may interfere with research results and affect the general recovery of the animals. The aim of the socialisation and training programme was to gradually accustom pigs to the necessary post-operative interventions by pre-operative training and handling. It has previously been shown that more time spent with pigs before surgery is associated with less post-operative stress and difficulty of eye medication. 24 However, in that study, no standardised protocol was used and the pigs needed sedation before clinical examination. The present study shows that the use of a standardised protocol makes it possible for all pigs to accept petting, brushing and voluntary interaction with people during all examinations post-operatively, without any need for sedation or restraint.
To let the pigs settle, the first step was based on our previous experience that research animals can be afraid of unknown people and need to get used to their environment, individual housing and adapt to daily routines before the onset of training time. Interestingly, most of the pigs needed twice as long to adapt to the elements in step 2 than in the other steps, and two pigs needed up to three times as much time to adapt to step 2 as the rest of the group. This is in line with the study by Nicholls et al. (2012), which concluded that touching and handling is the most important, but also the most challenging, part of training. In general, pigs are curious, and very adaptable and willing to cooperate if they are handled carefully. Despite that, individual pigs differ regarding the amount of time needed for learning,8,24,25 which is also true for other research animals such as non-human primates26,27 and dogs. 28 After step 2, the pigs quickly adapted to the remaining interventions. It is known that pigs recognise their handlers and benefit from environmental enrichment, 29 and are more easily handled by trainers that have spent more time with them. Therefore, the training schedule was designed so that all trainers spent the same amount of time with all the animals. Thus, pre-operative training is best performed by the same people that will do the post-operative interventions. One limitation with the study training programme is the absence of a control group. However, based on our long experience, we know that pigs that are not trained are easily scared and difficult to handle. Therefore, due to 3R and animal welfare considerations, it was decided that a control group would not be included.
Central venous access is crucial to enable blood sampling and/or IV administration without discomfort. Additionally, with this method, only one person is required for sampling, while vein puncture in pigs usually requires two people. It is important to note that pigs need to be accustomed to handling of the ears during socialisation and training, otherwise sampling from a catheter in the auricular vein is usually associated with the animals struggling, and thus it is not usually possible to do it aseptically.
In studies I–III, four catheters were partially occluded and, while they could still be flushed, blood could not be drawn. In humans, the most common cause of thrombotic obstruction is fibrin sleeve formation at the catheter tip. Fibrin sleeves cause obstruction when they are pulled over the catheter tip; however, flushing or infusion is still easy. 13 Heparin is frequently used as an antithrombotic agent to maintain venous catheters; however, some studies report no beneficial effect of heparin compared to saline, and there are no universal guidelines for concentrations of heparin solution or frequency of flushing. 30 Concentrations of 100 IU/mL may benefit in catheter patency, 30 and in the present experiment (studies I–III) catheters were flushed once daily with saline and filled with heparin 100 IU/ml in saline; despite this, only 75% of the catheters could be used for blood collection. However, from a 3R perspective, a goal is to use vein catheters for repeated blood sampling in pigs but to avoid occlusion of the catheter. In study IV, the venous catheters were coated with MPC, a polymer that effectively suppresses protein adsorption, platelet adhesion and platelet activation. 31 In comparison with the uncoated catheters, these catheters were flushed with saline without heparin and all of them could be used to collect blood throughout the experimental period. Even if it needs to be evaluated in a larger number of animals, an MPC coating seems promising to eliminate fibrin sheaths on indwelling polyurethane vein catheters used for prolonged implantation.
Ultrasound examination of the bladder was valuable since non-invasive techniques to measure urine production in pigs are limited. Catheterisation of the bladder is possible in female pigs, but impossible in male pigs because of many folds in their urethra. 32 The increased risk of infections16,17 and difficulties in fixating the catheters led us to avoid catheterisation of the bladder in the present study. By ultrasound examination, urine was detected in the bladder in about 50% of the pigs from the first day post-operatively. In the remaining pigs, there was no detectable urine in the bladder during the first 2 days after transplantation, which could either be due to a low volume, the urine having recently been voided. The amount of urine was not measured since the aim of the study was to observe when urine production started, and to evaluate whether ultrasound examination was possible on pigs after renal transplantation. In previously published studies where BladderScan® has been evaluated on humans, the device could reliably measure urine volumes with minimal variation between measurements on the same patient.33,34 For these reasons, we decided to evaluate the instrument on pigs. The BladderScan® was perceived as simple and user-friendly and tolerated well by the pigs, but the results showed a large variation between measurements and differed from volumes estimated by ultrasound examination on several occasions. The results are in line with a previous study on anaesthetised pigs from our laboratory (unpublished results), where a low percentage of results from the BladderScan® matched the urine volume measured via cystocentesis. In view of this, the use of BladderScan® on pigs in this type of research cannot be recommended. It was noted during the study that pressing the water nipple in the pen was often an effective way of stimulating the pigs to give free-flow urine samples into a paper dish.
The high frequencies of renal cysts found in studies II and III underscore the importance of examining donors before inclusion in transplantation studies. Congenital renal cysts are frequently seen in pigs 35 with a prevalence of 10–50%.36,37 In general, the occurrence is considered to be sporadic but genetic mechanisms can be involved. 38 Even though renal cysts may not cause alterations in renal function, 38 outcomes in renal transplantation studies might be affected. The majority of cysts are not visible by ocular inspection in pigs, 36 hence inspection during surgery is not enough and the donors need to be examined by ultrasound.
In study I, the weakness of the left hind leg in the early post-operative period could have been a neuromuscular dysfunction due to pigs lying on the same side for a prolonged period post-operatively. In the following studies, the staff turned the pigs from side-to-side every 2 hours and massaged musculus gluteus medius, biceps femoris and gluteus maximus several times. The pigs were also given help to stand up frequently and were let out to walk in the corridor to increase blood circulation. The treatment appeared to be effective, as only one more pig showed signs of weakness in a leg.
Plasma creatinine concentrations were measured to estimate the functions of transplanted kidneys. The true GFR was not calculated in the present study, but increased creatinine levels indicate a gradual decrease of GFR. 39 In studies I, II and IV, the native kidneys were removed and increased plasma creatinine concentrations were to be expected after transplantation. Pre-operative plasma creatinine concentrations were within a narrow range. Post-operatively, plasma creatinine concentrations varied a lot more between pigs, which is likely due to the fact that several studies and treatments were included. However, it is interesting to note that plasma creatinine concentrations could be well above 1000 µmol/L without obviously affecting the animals’ general health status. Pigs have a large relative muscle mass compared to other species such as mice, dogs and humans, and plasma creatinine concentrations can therefore be relatively high. In humans, measurement of plasma cystatin C concentration is considered superior to measurement of creatinine for the estimation of GFR, 20 and in dogs it may be used as an adjunct to creatinine for the diagnosis of decreased GFR. 40 In the present study, plasma cystatin C concentrations were below detectable levels at all sampling occasions despite markedly increased plasma creatinine concentrations. This finding suggests that cystatin C is not a reliable estimation of GFR in pigs, which is in line with a previous study that concluded that cystatin C values in pigs with endotoxemic shock should be interpreted with great care since the cystatin C concentrations did not correlate with GFR measured by inulin clearance. 41
In conclusion, a 2-week period of systematic socialisation and training of pigs resulted in calm animals that accepted blood sampling, urine sampling, and ultrasound examination of the urinary bladder and transplanted kidney without restraint. In addition, when MPC polymer-coated vein catheters were used, clotting was avoided. To assess renal function, diuresis and creatinine can be measured, while analysis of cystatin C was not found to be useful in recently transplanted pigs. The present study is an important contribution to the 3Rs (refine, reduce, replace).
Footnotes
Acknowledgement
Special thanks to Prof. Yuji Teramura for providing MPC polymer-coated catheters.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interests with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The research has received funding from The European Community’s Seventh Framework Programme for project DIREKT (no 602699) and from Torvald and Britta Gahlins stiftelse.
