Abstract
The Mongolian gerbil (
The Mongolian gerbil (
Direct and indirect virus detection by IFA, HI or PCR in gerbils and mice inoculated with the following viruses
Positives/animals tested are shown
*Two sera showed ambiguous results
†Positives showed very low fluorescence intensity
‡Typical lesions observed in histology
PCR: polymerase chain reaction; IFA: indirect immunofluorescence; HI: haemagglutination inhibition; MNV: murine norovirus; MHV: murine hepatitis virus; PVM: pneumonia virus of mice; MVM: minute virus of mice; MCMV: mouse cytomegalovirus; SeV: Sendai virus; Reo-3: orthoreovirus serotype 3; RV-A: rotavirus A; NT: not tested
Five days after inoculation, gerbils and non-infected sentinel mice (two Ztm:NMRI and two C57BL6/JZtm for each experiment) were placed together (two gerbils with two mice) into a clean cage for 30 days and subsequently euthanized by CO2 inhalation and blood withdrawal. Outcome of inoculation was assessed by serology (IFA and/or haemagglutination inhibition [HI] assay), and, where appropriate, by histology and polymerase chain reaction (PCR) (protocols available on request). The following organs were used for PCR analysis: MHV: colon, liver, lung, spleen and mesenteric lymph node (MLN); MNV: spleen, MLN; PVM: lung, spleen; MVM: spleen, MLN; SeV: lung, spleen. As a positive control, additional mice (NMRI and C57BL/6J) were inoculated oronasally (50 μL oral, 10 μL nasal) with the same virus suspension that was used for gerbils; inoculation (day 0) and euthanasia (day 35) were performed simultaneously with the respective gerbils.
To compare sensitivity of IFA using anti-mouse or anti-gerbil IgG secondary antibodies, gerbil sera were tested by IFA using FITC-conjugated anti-mouse IgG (1:40; Sigma-Aldrich, St Louis, MO, USA) and anti-gerbil IgG (1:40; Immunology Consultant Laboratory, Newberg, OR, USA). To ensure seroconversion for this comparison, additional gerbils were inoculated oronasally and intraperitoneally with one of the viruses listed above as well as with Vaccinia virus strain Lederle (ATTC VR-118; cultured in Vero-cells); treatment with the last of which was repeated on day 30 of the experiment.
This study was conducted in accordance with the German animal protection law and with the European Communities Council Directive 86/609/EEC for the protection of animals used for experimental purposes. All experiments were approved by the Local Institutional Animal Care and Research Advisory Committee and permitted by the local government.
Gerbils inoculated with SeV, Reo-3 and RV-A by the oronasal route seroconverted, but their respective sentinel mice did not, except for one mouse which was maintained together with RV-A inoculated gerbils. However, sera from RV-A-positive animals showed only very low fluorescence intensity. Gerbils inoculated oronasally with MHV, MNV, PVM, MVM and MCMV as well as their respective sentinel mice did not develop specific antibodies against these viruses as assessed by IFA and HI assay (PVM) (Table 1). Viral genomes were not detected by PCR in organs of gerbils or sentinel mice, except for PVM in two of four gerbils and four of four sentinel mice (Figure 1).
Pneumonia virus of mice detected by polymerase chain reaction in lungs and spleen of sentinel mice that were housed with oronasally inoculated gerbils. L: DNA ladder; −: negative control; +: positive control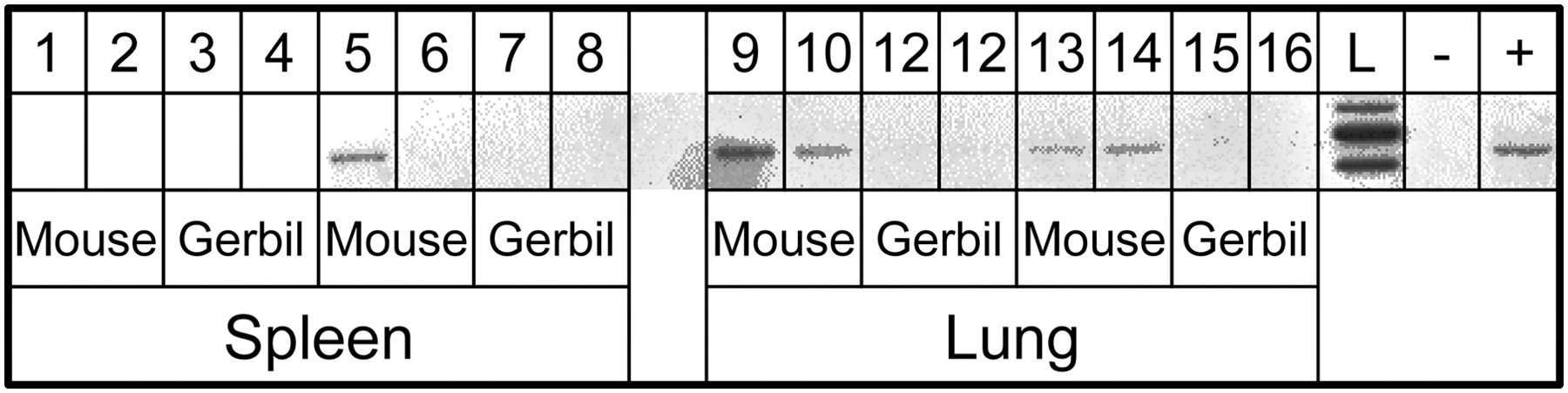
In contrast to gerbils, all mice that were inoculated as positive controls showed seroconversion except for MHV-inoculated mice that were euthanized one week after inoculation because these mice developed lesions typical for MHV infection (necrotizing enteritis and hepatitis). Colons, livers, spleens and lungs of these mice tested positive for MHV by PCR.
Gerbils that were inoculated once by both the oronasal and intraperitoneal routes showed antibody responses to all viruses except for MHV and MCMV. No difference in the sensitivity of IFA was observed using anti-gerbil or anti-mouse IgG antibodies.
In conclusion, seroconversion was observed with SeV, Reo-3 and RV-A in gerbils inoculated by the natural route (oronasally). Based on the data obtained in this study, these pathogens should be included in health monitoring programmes, though response in sentinel mice was only observed with RV-A. Mammalian reoviruses and SeV are known to infect a broad host spectrum, though possible cross-reactivity among parainfluenza viruses might suggest a too broad species spectrum for SeV. 8,9 By contrast, RV-A is generally considered to be host-specific, though bypass of host restriction and limited replication in heterologous species occur under controlled condiditons. 8 In this study, PVM was detected by PCR in sentinel mice, supporting previous findings indicating that PVM might be transmitted by gerbils. 10 Therefore, PVM should also be included in the health monitoring of gerbils. Except for MHV and MCMV, all viruses induced seroconversion after combined oronasal and intraperitoneal inoculation of gerbils. Though these viruses are unlikely to be transmitted by gerbils under natural conditions, care should be taken if gerbils are inoculated with biological materials of murine origin (e.g. antibody preparations). Importantly, no difference in sensitivity was observed using anti-gerbil IgG or anti-mouse IgG as secondary antibody in the IFA.
Footnotes
ACKNOWLEDGEMENTS
This study was sponsored by a grant from the GV-SOLAS. We thank Elena Wiebe for technical assistance.
