Abstract
The extent of placental transfer of medetomidine and ketamine is unknown in pregnant ewes. Date-mated singleton (n = 8) and twin (n = 8) pregnant merino cross ewes were anaesthetized for Caesarean delivery of preterm lamb fetuses. A combination of medetomidine (20 μg/kg) and ketamine (10 mg/kg) was administered by intravenous injection and surgery performed immediately thereafter. Blood samples were collected from the ewe at one, five and 10 min after intravenous injection and from the umbilical vein of the fetus at delivery. Non-pregnant ewes were also anaesthetized (n = 8). There was no difference in the plasma concentration of medetomidine or ketamine when comparing singleton and twin ewes or pregnant and non-pregnant ewes for the short duration of the study. Fetal plasma concentrations of each drug were comparable to the maternal concentrations at the same time. We conclude that both drugs cross the placenta readily and provide anaesthesia and analgesia for the fetus when it is delivered.
Pregnant ewes and their fetuses have been extensively utilized as a model in biomedical research for some time. 1–5 These studies require general anaesthesia of date-mated pregnant ewes and Caesarean delivery of the preterm lamb. A popular combination of drugs utilized for the induction of general anaesthesia in this setting has been an α 2 adrenoreceptor agonist such as xylazine, combined with ketamine. 6–9
Alpha-2 adrenoreceptor agonists are potent sedative drugs that provide muscle relaxation and analgesia. 10 They are however associated with dose-dependent cardiovascular effects: peripheral vasoconstriction and bradyarrhythmias; and a biphasic arterial blood pressure response. 11–13 Furthermore, hypoxaemia is well documented when these drugs are administered to sheep. 14–16 Medetomidine is a newer drug within this class, with the greatest α 2/α 1 selectivity (1620) of all the α 2 adrenoreceptor agonists currently used in veterinary practice. 17 Despite fewer α 1 effects, decreased heart rate, decreased cardiac output and PaO2 are features of the physiological changes occurring after the administration of medetomidine to sheep. 18 Blood flow to the brain, heart, lung, skeletal muscle and intestines is also decreased. 18
Ketamine is an N-methyl-
Despite their extensive use in the pregnant sheep model, to our knowledge there is no information about the passage of this combination of drugs across the placenta to the fetal circulation. The aim of the present study was to determine the behaviour of medetomidine and ketamine after administration to pregnant ewes for short-term terminal anaesthesia.
Materials and methods
The experimental protocol was approved by the Animal Ethics Committees at the University of Western Australia and Murdoch University, according to the guidelines of the National Health and Medical Research Council of Australia code of practice for the care and use of animals for scientific purposes. 20
Animal handling
Date-mated pregnant merino sheep with lambs at a gestational age of 128–130 days (term = 150 days) were used for the study. The ewes were housed in group pens in the research facility and had had at least two weeks to acclimatize to the environment after transport from the farm of origin. They were fed a prescribed ration and had a minimum body condition score of 2.5. Preanaesthetic physical examination did not detect any abnormalities. Singleton ewes (n = 8), twin pregnant ewes (n = 8) and non-pregnant merino ewes (n = 8) were used to study the distribution of medetomidine and ketamine following intravenous injection. Sheep were restrained in the shearing position for placement of a catheter in the right cephalic vein. The catheter was secured in place and flushed with 5 mL of heparinized saline (10 IU/mL). Anaesthesia was induced with a combination of medetomidine (Domitor 1 mg/mL; Pfizer Animal Health, West Ryde, NSW, Australia) 20 μg/kg and ketamine (ketamil 100 mg/mL; Troy Laboratories, Glendenning, NSW, Australia) 10 mg/kg by intravenous injection into the left cephalic vein.
Following induction of anaesthesia the ewe was placed in right lateral recumbency. Pregnant ewes were prepared for Caesarean delivery of the fetus and a subarachnoid injection of lidocaine was administered at L5–L6. Non-pregnant ewes remained in right lateral recumbency for 10 min. Venous blood samples were collected from the catheter at one, five and 10 min after induction of anaesthesia. The catheter was flushed after each sample collection and the first 3 mL of blood aspirated during collection was discarded.
Ten minutes after the induction of anaesthesia, non-pregnant ewes were euthanized and the pregnant ewes were continued according to protocol: the lamb was delivered and an umbilical venous sample of blood was collected. The time of delivery was noted and the ewe was euthanized immediately after delivery of the fetus(es). An intravenous injection of pentobarbitone was administered for euthanasia (100 mg/kg, Valabarb, Pentobarbitone 320 mg/mL; Jurox, Rutherford, NSW, Australia). All blood samples were collected into glass ethylenediaminetetraacetic acid vials, centrifuged and separated within 30 min. Plasma was stored at −80°C in Teflon-coated glass vials.
A 3.5–4.5 mm (internal diameter) cuffed oral endotracheal tube was placed in the trachea of each lamb to facilitate mechanical ventilation according to the study protocols. The depth of anaesthesia required to allow oral intubation was assessed and was, without exception, found to be sufficient. Assessment of anaesthesia was based upon gross purposeful movement, withdrawal responses to a toe pinch and withdrawal of the tongue during intubation. The eyelids were still fused so ocular reflexes could not be assessed.
Sample analysis
A SPE-LC-MS/MS method has been developed for the simultaneous determination of both ketamine and medetomidine. 21 Five hundred microlitres of plasma samples were extracted using a 30 mg Bond Elut Plexa SPE cartridge (Varian Inc, Palo Alto, CA, USA). Samples were analysed using a Pursuit XRs diphenyl analytical column (Varian Inc) with dimensions of 150 mm × 2.0 mm × 3 μm. An Agilent 1100 HPLC was coupled to an Agilent Classic Series ion trap mass spectrometry (Agilent Technologies, Santa Clara, CA, USA) and samples were analysed in positive ion mode. Protonated pseudomolecular ions were isolated at m/z 238 and m/z 201 for ketamine and medetomidine, respectively, while quantifier product ions were isolated at m/z 207 and m/z 95, respectively.
Results
All ewes were merino cross between five and six years of age. The body condition scores were 2.5–3 (maximum 5). Twin pregnant ewes were heavier than singletons (P = 0.039). There was no difference in the blood pH, PaCO2 and PaO2 of the lambs at delivery (Table 1).
Ewe weight, time of delivery and blood gas results for the fetus
*P = 0.039 compared with singleton ewes
There was no difference in the plasma concentration of either medetomidine or ketamine between twin and singleton ewes (Figures 1a and b). At the time of delivery the plasma concentration of each drug in the fetus was comparable to the maternal concentration at the same time. Furthermore, there was no difference in the plasma concentration of either medetomidine or ketamine between pregnant and non-pregnant ewes at the three time-points (1, 5, 10 min) (Figures 2a and b).
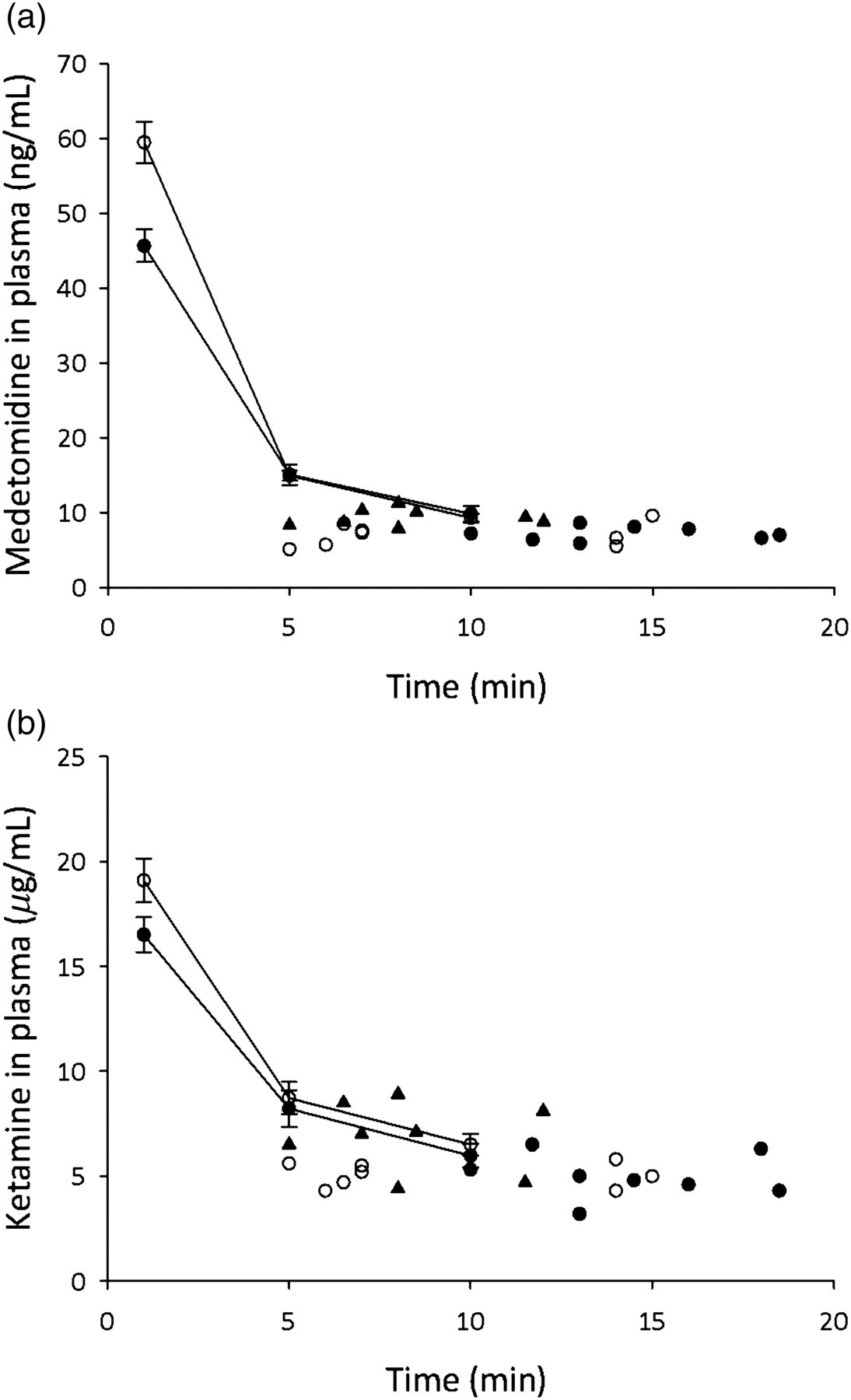
Anaesthetic drug plasma concentration in twin and singleton ewes and their fetuses at delivery. (a) Concentration of medetomidine (ng/mL) in the maternal plasma at 1, 5 and 10 min after intravenous injection and in the fetal plasma at delivery. ○ = singleton ewe or fetus, • = twin ewe or second fetus delivered, ▴ = first fetus of twin ewe. (b) Concentration of ketamine (μg/mL) in the maternal plasma at 1, 5 and 10 min after intravenous injection and in the fetal plasma at delivery. ○ = singleton ewe or fetus, • = twin ewe or second fetus delivered, ▴ = first fetus of twin ewe
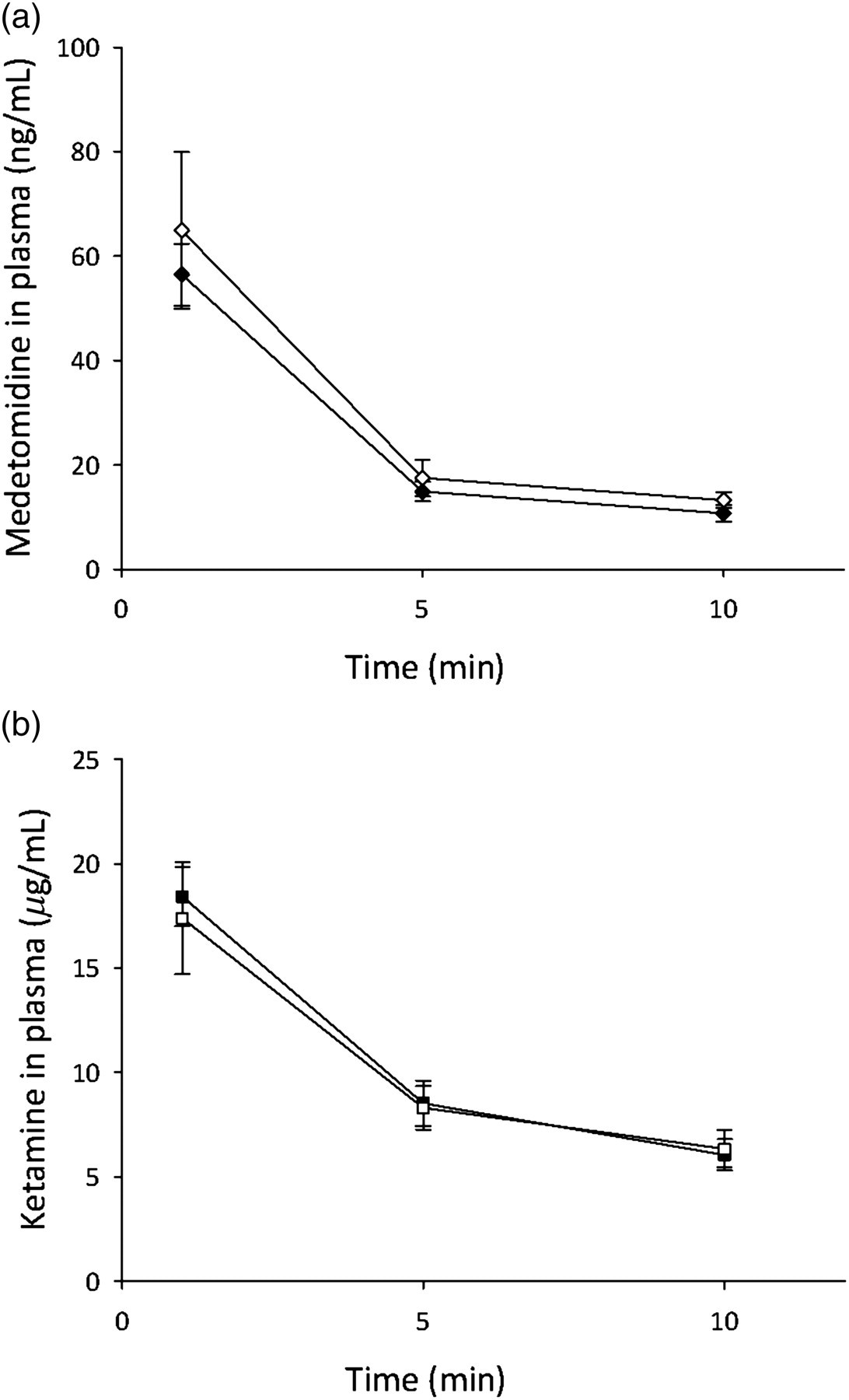
Anaesthetic drug plasma concentration in pregnant and non-pregnant ewes. (a) Concentration of medetomidine (ng/mL) in ovine plasma at 1, 5 and 10 min after intravenous injection. ⧫ pregnant ewes, ◊ = non-pregnant ewes. (b) Concentration of ketamine (μg/mL) in ovine plasma at 1, 5 and 10 min after intravenous injection. ▪ = pregnant ewes, □ = non-pregnant ewes
The lambs delivered in this study were enrolled in research projects approved by the Animal Ethics Committee at the University of Western Australia. These projects involved mechanical ventilation of the lamb for up to 3 h. They were euthanized at the end of those studies. At delivery, the lambs were all sufficiently anaesthetized to tolerate endotracheal intubation without further chemical restraint. The time to requiring supplemental anaesthesia ranged from 3 to 77 min (mean [SD]: 42 [6] min).
Discussion
General anaesthesia of pregnant sheep is often performed to facilitate medical research that focuses on the lamb fetus. Drugs given to the ewe may cross the placenta from the maternal circulation to the fetal circulation and cause anaesthesia or sedation of the fetus. In a research environment it is prudent to aim for placental transfer of anaesthetic and analgesic drugs if the fetus is the subject of the study. As assessment of pain and suffering is difficult in preterm neonatal lambs, it is preferable to err on the side of caution and ensure they receive anaesthetic and analgesic drugs. On the other hand, getting the dose right may be a challenge.
The anaesthetic technique used in the present study has been employed in studies where acute delivery of a preterm lamb is required. 1,22 The duration of anaesthesia for the pregnant ewe is short as non-sterile Caesarean delivery of the lamb is performed expediently. The dose of medetomidine and ketamine was determined from a pilot study (unpublished data) and was sufficient to induce general anaesthesia and conditions suitable for surgery. The ewe was euthanized as soon as the fetus(es) were delivered and for the purposes of the study in which the lambs were enrolled it was necessary for them to be anaesthetized as well. The ewes did not show evidence of recovery from anaesthesia in the short period of this study and no adverse effects of anaesthesia were noted. Pulse rate, respiratory rate and character and mucous membrane colour were observed and despite the potential for hypoxaemia in this species after the administration of an α2 adrenoreceptor 14,16 we did not observe any untoward side-effects. The lambs were sufficiently anaesthetized for oral endotracheal intubation and mechanical ventilation.
The factors that affect the ability of a drug to cross the placenta include lipid solubility, molecular weight and the degree of ionization. 23 Medetomidine is highly lipid soluble 24 and as demonstrated by the similarity between fetal and maternal plasma concentrations, it readily crossed the placenta. Ketamine has been reported to cross the placenta in pregnant ewes and rapid equilibration between the maternal and fetal circulation occurs. 25 Our results on the disposition of medetomidine and ketamine across the placenta of singleton and twin pregnant sheep showed that both drugs can pass through the placental barrier and reach the ovine fetus immediately after an intravenous injection to the ewe. By the time the fetus was delivered the concentration of each drug was very similar to the maternal concentration at the last data point 10 min after induction of anaesthesia. These findings suggest rapid equilibration between the maternal and fetal circulatory compartments. Calculating the fetal/maternal concentration ratio for each drug would enable quantification of the partitioning across the placenta, but our study design prevented simultaneous collection of maternal and fetal blood samples.
Uterine and umbilical blood flow will also influence the transfer of drugs across the placental barrier. 26 Increased uterine blood flow is likely to decrease the extraction of drugs by the placenta, and maintain maternal plasma concentrations at a higher level for a longer period of time. Conversely, increased umbilical blood flow facilitated the placental transfer of propofol in vitro. 26 The drugs in this study are known to alter blood flow: medetomidine will decrease blood flow to a range of organs 18 while ketamine has little effect on uterine blood flow. 27,28 The interaction of these effects in this study is unknown.
Our comparison of the plasma concentration of medetomidine and ketamine in pregnant and non-pregnant ewes after intravenous injection revealed little difference in the distribution of these drugs. Pregnancy causes a multitude of physiological alterations including increased cardiac output, increased heart rate, decreased systemic and pulmonary vascular resistance, increased blood volume and decreased haematocrit. 29 These changes create a hyperdynamic circulation and we expected a difference in the shape of the curve for the pregnant versus the non-pregnant ewes. Although the same dose rate of each drug was administered, there was no difference in the distribution of the drugs during the short study period.
The fetal blood gas analysis results indicate acidaemia, hypercapnia and hypoxaemia when compared with normal adult values. These results are consistent with other studies where preterm lambs are delivered during general anaesthesia of the ewe. 9,30 Altered acid–base status may influence the distribution of drugs across the placenta as some drugs may accumulate in the relatively acidic environment of the fetus when the ionization status changes in response to the new pH. This is likely to be a theoretical concern, as opposed to a clinically significant issue.
We conclude that medetomidine and ketamine by intravenous injection is suitable for induction of anaesthesia in pregnant sheep. The lamb fetus was not severely compromised by the concentration of medetomidine and ketamine that crosses the placenta in this short-term study. Further work is required to determine the impact of prolonged anaesthesia of the ewe on the lamb fetus and the ability of the fetus to eliminate the drugs given immature hepatic metabolic pathways.
Footnotes
Acknowledgements
This work was made possible by a grant from the Women and Infants Research Foundation. We would like to thank the Ovine Research Group in the School of Womens' and Infant's Health at the University of Western Australia for the opportunity to collect the samples for this study.
