Abstract
Much remains to be understood with regards the effects of prolonged anaesthesia on maternal and fetal haemodynamics and oxygenation. With the aim of improving anaesthetic management of pregnant sheep undergoing recovery surgery under anaesthesia, paired maternal and fetal arterial blood samples were collected during caesarean delivery of twin preterm lambs to document the blood gas status of the ewe and fetus. Twenty-one Merino twin pregnant ewes at 126 (±1) days of gestation were anaesthetized for caesarean delivery of their fetuses. Arterial blood samples were collected from the radial artery of the ewe and umbilical artery of the fetus at the point of delivery. There was a significant difference between maternal PaCO2 and end-tidal CO2 and alveolar and arterial PaO2, indicating ventilation perfusion mismatch. Interestingly, the ewes were anaemic but the fetuses were not. These data underscore the need to undertake further work to determine the optimal anaesthetic regimen for twin pregnant ewes at different gestational ages in a biomedical research setting.
Pregnant sheep and their fetuses are commonly utilized as animal models for investigations into human pregnancy and the causes and consequences of preterm birth.1–4 As elegantly argued in Russell and Burch’s landmark work, The principles of humane experimental technique, the continual refinement of practice in animal-based research is key to minimizing the impact of research on animal welfare and to ensuring the integrity of scientific data. 5
General anaesthesia causes major physiological alterations to the respiratory, cardiovascular, neurological, immune and endocrine systems and specific considerations exist for anaesthesia of pregnant patients. 6 These considerations relate primarily to the global increase in oxygen consumption and decreased functional residual capacity of the lungs during pregnancy. 7 In addition, mean arterial blood pressure decreases and the heart rate and cardiac output increase in pregnancy. Pregnant patients also have an increased blood volume and a dilutional anaemia. 7 These changes all contribute to an increased risk of hypoxaemia and hypotension during general anaesthesia. However, it is essential that maternal hypoxaemia and hypotension are avoided to maintain fetal viability. 6 In the context of research focusing on the fetus it is especially important that these physiological alterations are managed to optimize the condition of the fetus, thus avoiding experimental confounding.
Uterine blood flow is a major determinant of fetal perfusion but is maintained in the face of decreasing maternal arterial blood pressure during increased isoflurane anaesthesia in sheep. 8 Nevertheless, the development of maternal hypoxaemia or hypotension may decrease uterine blood flow and exacerbate fetal acidosis and secondary fetal cardiac dysfunction.8,9 Maternal hypoxaemia can invariably be managed by optimizing the inspired oxygen concentration, administering intermittent positive pressure ventilation (IPPV) and strategic positioning of the recumbent ewe. 9 Likewise, maternal hypotension can be managed with intravenous fluid therapy, anaesthetic drug and dose selection and the administration of vasopressors. 9
Singleton pregnant ewes are most commonly used in a biomedical research environment but twin pregnancies enable an overall reduction in the number of animals used in research as the sacrifice of one ewe yields two fetuses. The blood gas status of twin lamb fetuses under anaesthesia is not well documented in the literature. Accordingly, we aimed to measure the blood gas status of paired maternal and fetal samples during anaesthesia for caesarean delivery. These data are presented herein.
Materials and methods
The data were collected as part of another project that was approved by the Animal Ethics Committees at the University of Western Australia and Murdoch University in accordance with the Australian code for the care and use of animals for scientific purposes. 10
Animals
Twenty-one Merino ewes carrying twin fetuses at 126 (±1) days of gestation (term ∼ 150 days) underwent anaesthesia and surgery for caesarean delivery of preterm lambs as part of another study investigating gentle ventilation strategies for preterm lambs as a model for preterm babies. The sheep were held at the Large Animal Facility at the University of Western Australia in raised group pens for at least one week prior to surgery. Rooms were controlled for temperature (20.5–21.5℃) and relative humidity (40–60%). Ewes were weighed on the morning of surgery.
Anaesthesia and surgery
Food and water were withheld from the time of premedication. Premedication, induction and maintenance of anaesthesia were performed as previously described. 11 Following physical examination the ewes were premedicated with a combination of acepromazine (0.03 mg/kg, ACP 2 injection, 2 mg/mL; Ceva Delvet Pty Ltd, Glenorie, NSW, Australia) and buprenorphine (0.01 mg/kg, Temgesic, 0.3 mg/mL; Reckitt Benckiser, Melrose Park, NSW, Australia) by intramuscular injection 30–40 min prior to the induction of anaesthesia. Anaesthesia was induced with a combination of midazolam (0.25 mg/kg, Midazolam injection, 5 mg/mL; Pfizer Australia Pty Ltd, Melrose Park, NSW, Australia) and ketamine (5 mg/kg, Ketamil, 100 mg/mL; Troy Laboratories, Glendenning, NSW, Australia) by intravenous injection and the trachea was subsequently intubated (endotracheal tube 8.5 mm internal diameter, cuffed; Portex Ltd, Ashford, Kent, UK). Anaesthesia was maintained with isoflurane (1–2%, Attane Isoflurane 1 mg/mL; Bayer Australia Ltd, Pymble, NSW, Australia) in 100% oxygen delivered through a circle breathing system. The isoflurane vaporiser was adjusted as judged by an experienced veterinary anaesthetist to maintain an adequate depth of anaesthesia. A line block of ropivacaine (100 mg, Naropin 1%; Astrazeneca, Macquarie Park, NSW, Australia) was performed along the laparotomy incision site prior to surgery. IPPV was commenced immediately following endotracheal intubation to maintain normocapnia (end-tidal CO2 35–45 mmHg) using an Aestiva 5 anaesthetic machine with a Smartvent ventilator (Aestiva 5 with Smartvent 7900; Datex-Ohmeda, GE Healthcare, Bromma, Sweden). Volume controlled ventilation was utilized with a set tidal volume of 8–12 mL/kg and a respiratory rate between 8 and 12 breaths/min. The animal was positioned in dorsal recumbency in the Trendelenburg position with the table tilted 25°. Physiological monitoring included electrocardiogram, oxyhaemoglobin saturation (SpO2, with the pulse oximeter probe positioned on the pinna or tongue), side stream capnography (sample rate 150 ± 20 mL/min), temperature (nasopharyngeal) and invasive blood pressure (radial artery) using a multiparameter monitor (Surgivet V9203; Smiths Medical, Norwell, MA, USA). Intravenous fluid therapy was administered during anaesthesia with a balanced isotonic electrolyte solution (compound sodium lactate solution; Baxter Healthcare Pty Ltd, Toongabbie, NSW, Australia) at 10–20 mL/kg/h and succinylated gelatin solution (Gelofusine 4%; Braun Australia Pty Ltd, Bella Vista, NSW, Australia) at 2–5 mL/kg/h to maintain normotension (mean arterial blood pressure 60–90 mmHg). A ventral midline laparotomy and hysterotomy was performed so each fetus could be exteriorized and delivered for neonatal care as dictated by the project protocol. Following delivery of the second fetus the ewe was euthanized with pentobarbitone (160 mg/kg by intravenous injection). The procedures were always performed during the day.
Blood gas analyses
Samples for blood gas analysis were collected from the ewe’s radial artery via the catheter that had been placed for invasive blood pressure measurement. The first one millilitre of blood drawn from the catheter was discarded and the sample proper was collected into a heparinized syringe (RapidLyte 3 mL/L Syringe with filter cap and dry lithium heparin [∼21 IU]; Siemens Healthcare Diagnostics Inc, Tarrytown, NY, USA). Fetal samples were collected into the same syringes by venepuncture of an umbilical artery immediately following clamping of the vessel and before the first breath. The samples were analysed within 5 min using a benchtop arterial blood gas analyser (RapidLab 1265; Siemens Healthcare Diagnostics Inc) which was calibrated daily and underwent regular quality control (Automatic QC Cartridge, Ref 05293926; Siemens Healthcare Diagnostics Inc). The temperature of the ewe was recorded at the time of sample collection and the data presented are corrected for temperature. The partial pressure of oxygen in the alveoli was calculated according to the alveolar gas equation where PAO2 = (Patmospheric − PH2O) × FiO2 − PaCO2/0.8 (Patmospheric was 760 mmHg at sea level, PH2O was assumed to be 47 mmHg and FiO2 was 1.0). 12 The alveolar–arterial oxygen difference was calculated as PAO2 − PaO2.
Data analysis
Normally distributed data were compared with a student’s t-test. Otherwise a Mann–Whitney Rank Sum test was used. All tests were performed with Sigmaplot 12.5 (Systat Software Inc, San Jose, CA, USA). Data are presented as mean (±SD) or median (25–75%) as appropriate. P < 0.05 was considered to be statistically significant.
Results
Physiological data of the ewes (n = 21) at the time of delivery of twin 1 and twin 2.

Data is median (range) or mean (±SD). *P < 0.001 compared with end-tidal CO2.
Arterial blood gas data for the ewe and fetus (n = 21), and the differences between them, at the time of delivery of twin 1.
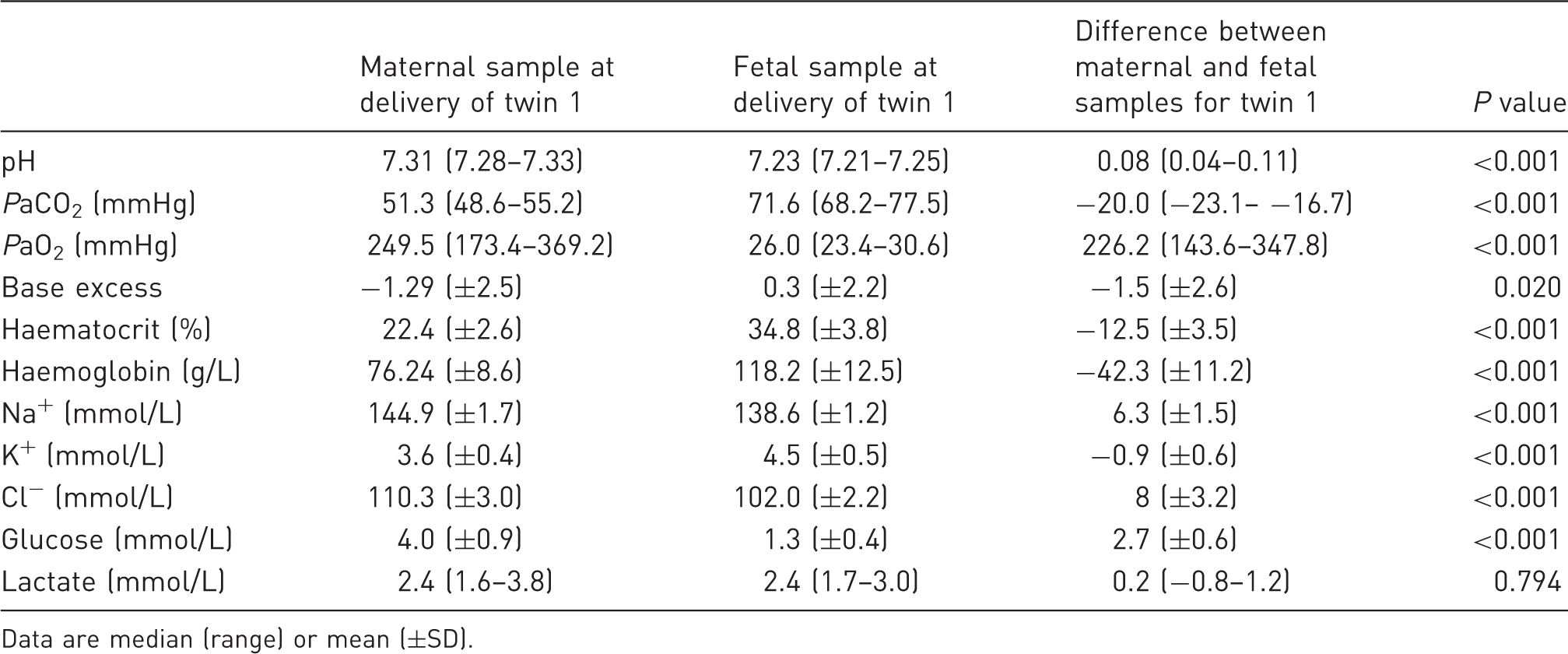
Data are median (range) or mean (±SD).
Arterial blood gas data for the ewe and fetus (n = 21), and the differences between them, at the time of delivery of twin 2.
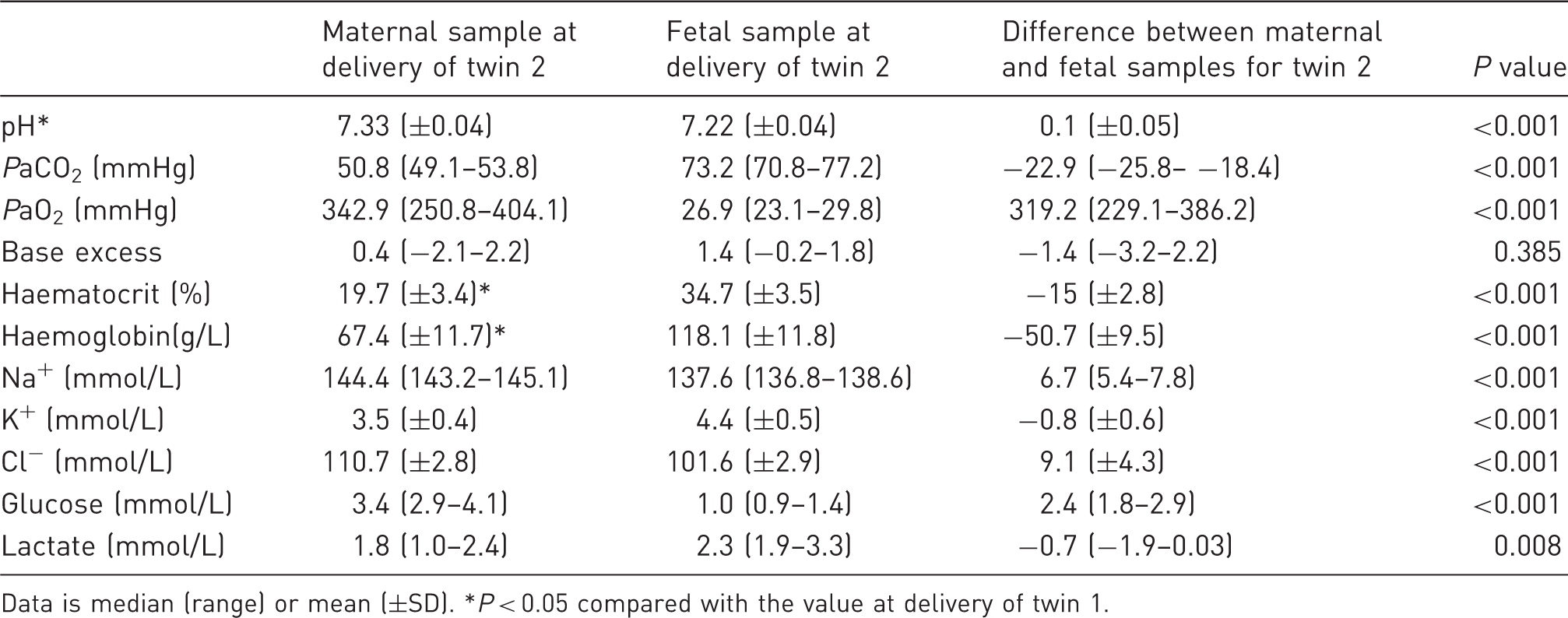
Data is median (range) or mean (±SD). *P < 0.05 compared with the value at delivery of twin 1.
Discussion
Simultaneous arterial blood gas sampling from the maternal and fetal circulation revealed that fetal pH, PaO2, sodium and chloride ion concentrations and glucose concentrations were consistently lower than maternal values and that fetal PaCO2, base excess, haematocrit, haemoglobin concentration and potassium ion concentration were consistently higher than maternal values. The differences between end-tidal and maternal PaCO2 and between alveolar and PaO2 indicate ventilation and perfusion mismatching with the presence of both alveolar dead space and physiological shunt. Lastly, despite maternal anaemia, the fetuses had a haematocrit and haemoglobin concentration higher than the ewe.
The physiological parameters of the ewe during anaesthesia were stable and within the expected range for anaesthetized sheep.9,11 IPPV settings created a mild respiratory acidosis and intravenous fluid therapy was sufficient for maintenance of the mean arterial blood pressure as planned. The ventilator parameters were successfully adjusted to maintain normocapnia based upon capnography but the arterial–end-tidal CO2 difference indicates a degree of ventilation and perfusion mismatching in the lungs and specifically the presence of physiological dead space. 13 The discrepancy between the arterial and end-tidal CO2 values is generated by relatively poorly perfused alveoli and to a lesser extent by right-to-left pulmonary shunting. This difference is not reported in sheep but is usually 10 mmHg in horses and may increase up to three fold if ventilator settings are inappropriate or cardiac output decreases. 13 Again, in horses, the difference is reported to be 12 (±) 6 mmHg in one study which also demonstrated that alveolar dead space was influenced positively by weight, dorsal recumbency, IPPV and duration of anaesthesia. 14 Pregnant sheep are likely to be prone to the development of alveolar dead space during anaesthesia because of the low functional residual capacity of the lungs and placement in dorsal recumbency for surgery. Although capnography is non-invasive and gives continuous information about the adequacy of ventilation it may be prudent to collect arterial blood samples intermittently to confirm the PaCO2 is within the target range.
Although the ewe was not hypoxaemic the difference between alveolar and arterial PO2 indicates physiological shunt. In this study the provision of IPPV and an FiO2 of 1.0 likely prevented hypoxaemia in the ewe. However, physiological hypoxaemia was evident in the fetuses in this study but the fetal PaO2 is normally much lower than after birth. 15 These results are consistent with other studies but the fetus can survive because of specific adaptations enabling high oxygen delivery to fetal tissues and organs.15,16
Factors contributing to the development of ventilation and perfusion mismatching include the pregnancy status of the sheep, the supine position and the Trendelenburg position. 6 Although the pulmonary complications of anaesthesia and recumbency in sheep contribute to the risk of anaesthetic-related complications they can be managed and did not appear to cause any detrimental effects in either the ewe or the fetuses in this study. Further investigations into the implications of ventilation and perfusion mismatching are required to determine the significance of this abnormality.
Prevention of ventilation and perfusion mismatching should be attempted in every case. The factors that contribute to this physiological alteration may be fixed (pregnancy status) or not (duration of anaesthesia, provision for IPPV, positioning during surgery). Minimizing the duration of anaesthesia, and especially the period before delivery of the fetus, may limit physiological deterioration during anaesthesia while IPPV from the outset may prevent alveolar collapse. Positive end-expiratory pressure (PEEP) during ventilation is part of a lung protective ventilation strategy to keep the lungs open 17 but the ideal PEEP is not known in pregnant sheep. The most appropriate ventilation strategy for pregnant sheep is also not known but pressure-controlled ventilation may provide superior oxygenation at lower inflation pressures when compared with volume-controlled ventilation. 11 Manipulating the position of the ewe during surgery can be problematic as good surgical access is important to minimize the duration of surgery and anaesthesia. In this study surgical access was facilitated by positioning the ewe in dorsal recumbency in the Trendelenburg position. Further investigations into the ideal position of the ewe during surgery for caesarean delivery of a fetus are warranted. Bronchodilators such as salbutamol have been used in horses to treat intraoperative hypoxaemia but the safety and efficacy of these drugs are not known in pregnant sheep. In horses the risk of occurrence of clinically relevant unwanted cardiovascular side-effects (tachycardia and major alterations in arterial blood pressure) is significant so medical bronchodilation may not be appropriate in pregnant sheep. 18
The anaesthetic management of the ewes in this study ensured that arterial blood pressure was measured and managed to maintain normotension. Uterine arteries have minimal autoregulation to maintain uterine perfusion pressure and uterine blood flow so it is essential to avoid maternal hypotension, minimize the duration of supine positioning and prevent frequent uterine contractions. 19 In this study maternal blood pressure was successfully maintained with a combination of an isotonic balanced electrolyte solution and a colloid by intravenous infusion. The concentration of isoflurane was also adjusted to ensure an adequate depth of anaesthesia and minimize hypotension associated with this potent vasodilator.
The ewes in this study were anaemic and although this is a normal finding in pregnancy when there is a disproportionate increase in plasma volume compared with red blood cell volume this abnormality did not translate to anaemia in the fetus.6,19 The ewes in this study were considered healthy and free of disease based on thorough pre-operative clinical assessments and preventative health strategies so the anaemia was not attributed to disease or chronic blood loss. Anaesthesia and fluid therapy may contribute to a decreased haematocrit but the significance of these factors in this study is unknown. Nevertheless, acepromazine and ketamine have both been associated with a decrease in haematocrit in other species. 20 Intravenous fluid therapy can cause dilutional anaemia which may have contributed to the decline in maternal haematocrit and haemoglobin concentration over time. 21 The decrease in the maternal haematocrit and haemoglobin concentration between the delivery of the first and second fetuses was statistically significant but was not clinically significant. The fetal arterial blood gas results are difficult to compare with other studies as the anaesthetic regimen and duration are likely to have an effect on these values. After 60 min of anaesthesia with desflurane the fetal pH of singleton ewes was higher than in our study. 9 And during 1.0 MAC isoflurane and sevoflurane anaesthesia the fetal pH was also higher than in our study. 8 This higher pH is caused by a relatively low PaCO2. Fetal PaO2 values were comparable between our study and the aforementioned studies but other data are not included. Lastly fetal base excess and lactate concentration were similar to other studies.
To minimize the risk of maternal hypoxaemia and hypotension and to optimize the condition of the fetus, especially in twin pregnancies, IPPV and the ability to monitor and manage arterial blood pressure is important. The data presented here documents maternal and fetal arterial blood gas values from preterm twin pregnant ewes during general anaesthesia where maternal hypoxaemia and hypotension did not occur. Maternal anaemia and ventilation and perfusion mismatch were the most prominent abnormalities detected in this study. Further work is required to determine the optimal anaesthetic regimen for twin pregnant ewes at different gestational ages in a biomedical research setting.
Footnotes
Acknowledgements
The authors would like to thank the Animal Care Services staff at the Large Animal Facility for expert care and husbandry of the pregnant ewes. The authors also wish to acknowledge the generous donation of Rapidlab 1265 reagents and consumables by Siemens, Australia.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
