Abstract
In vivo optical imaging enables detection and quantification of light-emitting compounds from the whole body in small animals such as the mouse, but it typically requires the use of anaesthetics for subject immobilisation due to long exposure times. Excessively deep anaesthesia can result in unacceptably compromised physiology, whilst excessively light anaesthesia can result in animals waking up.
Here we report a respiratory monitoring setup for an in vivo bioluminescence and fluorescence imaging device which simultaneously allows real-time adaptive control of anaesthesia depth in multiple animals to (i) potentially increase the consistency between animals, (ii) ensure animals are maintained within minimally intrusive, adequate anaesthetic plane and (iii) provide a valuable refinement strategy for a common challenge within animal-based research.
Introduction
In vivo optical imaging generally allows minimally or non-invasive repetitive in vivo imaging of dynamic biological processes at the molecular level, and has the potential to provide unique insights into disease pathogenesis, drug development and therapy evaluation. Briefly, images are generated by using light in the wavelength range from ultraviolet to near infrared, and a signal is derived via exogenous agents, endogenous molecules with optical signatures, or compounds generated from reporter genes. Whilst fluorescence imaging uses endogenous or exogenous molecules that emit light when activated by an external light source, bioluminescence imaging generally uses a natural light-emitting enzymatic reaction of luciferase (enzyme) with luciferin (one of the substrates) to track cells or follow biochemical processes within the body.1,2 In our department, optical imaging is routinely used in the examination of hypoxia, intra-tumoral viral load, tumour burden and metastatic spread, as well as in sporozoite development and distribution in mouse models of malaria. It also has numerous applications in many other disease areas.
In common with other in vivo imaging modalities, animals are anaesthetised to reduce motion artefacts and achieve acceptable image quality. However, anaesthesia affects both central and autonomic nervous systems inducing muscle reflexes inhibition, loss of skeletal muscle tone, and cardio-respiratory depression.3,4 Inhalation anaesthesia is usually preferred to injectable anaesthesia as it has a lesser effect on the cardiovascular system, it is easily reversible, and anaesthesia depth can be quickly adjustable.3,5 However, inhalation anaesthesia can cause deep respiratory and/or myocardial depression, vasodilation, and hypotension.3,6 Additionally, isoflurane, one of the most generally used inhalation anaesthetics, has been shown to rapidly decrease mean arterial blood pressure with increasing concentrations, 7 and to inhibit bioluminescence signal generation. Additionally, as the luciferin-luciferase reaction is adenosine triphosphate dependent, changes in metabolism will alter reaction kinetics and change bioluminescence read-out.8–10 Hence, depth of anaesthesia can influence luciferin’s biodistribution and kinetics, which will alter the bioluminescence experimental read-out. Lastly, age, sex, and strain are factors known to influence the animal’s sensitivity to isoflurane.6,11
Commercial optical imaging systems are now equipped with an anaesthetic gas delivery system and a heated platform to avoid anaesthesia-induced hypothermia. Unlike other in vivo imaging modalities, such as magnetic resonance imaging and ultrasound, optical systems do not yet provide means to monitor anaesthesia depth. The researcher is unable to confirm anaesthesia depth throughout the imaging session without pausing or interrupting the image acquisition protocol. Animals are at risk of being under- or over-anaesthetised, compromising animal welfare and experimental outcomes. This is particularly important when performing kinetic assays, which can take up to one hour to complete.12,13 Fortunately, respiration rate monitoring is a relatively straightforward method to implement as read-out of anaesthesia depth; 6 if the rate is too high, anaesthesia is too light and the animal is at risk of waking up, but if the rate is too low, anaesthesia is too deep and basic physiology including blood flow and oxygenation can be adversely affected. 7 For that reason, we implemented optical imaging-compatible respiration sensors.
Imaging was performed on an IVIS Spectrum upgrade of the IVIS 200 Bioluminescence system (Caliper Life Sciences, PerkinElmer UK). A schematic of respiration monitoring setup is shown in Figure 1. Five piezoelectric vibration sensors (LDT0-028K, Sensor Solutions, TE Connectivity), one per mouse, were placed on the heated imaging platform, under the animal (piezo sensor, Figure 1(b)). These individual sensors converged to a single DA-15 connector on the platform (connector block, Figure 1), which allowed easy connection to a pre-existing and unused electrical services connector at the rear of the instrument, which is labelled as ‘USER I/O’ (chassis connector, Figure 1(a)). Signals are then externalised to a Biopac system (DA100C, Biopac Systems UK) for display and monitoring. Sensors were blacked out with insulation tape and black cables were used to eliminate possible interference with fluorescence signal acquisition (Figure 1(c)). Unlike respiration balloons where small differences in pressure lead to an electrical signal, piezoelectric vibration sensors generate voltage signals at their terminals when the sensor deforms during breathing. These signals are then conditioned by amplifiers (DA100C, Biopac Systems UK) and displayed in real time on a personal computer (AcqKnowledge software, Biopac Systems UK) (Figure 1(d)). Although we opted for the Biopac system to keep the IVIS Spectrum in line with our other imaging modalities, there are cheaper alternatives for respiration rate monitoring such as an oscilloscope. Also, this respiration monitoring system can be useful in other animal experimental procedures that involve general anaesthesia such as in normal surgeries, regardless of the choice of anaesthetic agent.
(a) Schematic overview of setup. (b) Schematic exemplification of sensor location for prone (upper) and supine (lower) positions of a mouse. (c) Five mice (M1–M5) placed on top of five individual piezo sensors for respiration monitoring. White arrows indicate piezo sensor and connector block locations. (d) Individual respiration traces for each mouse (M1–M5). Left panel corresponds to fast respiration rate; right panel to slow respiration rate.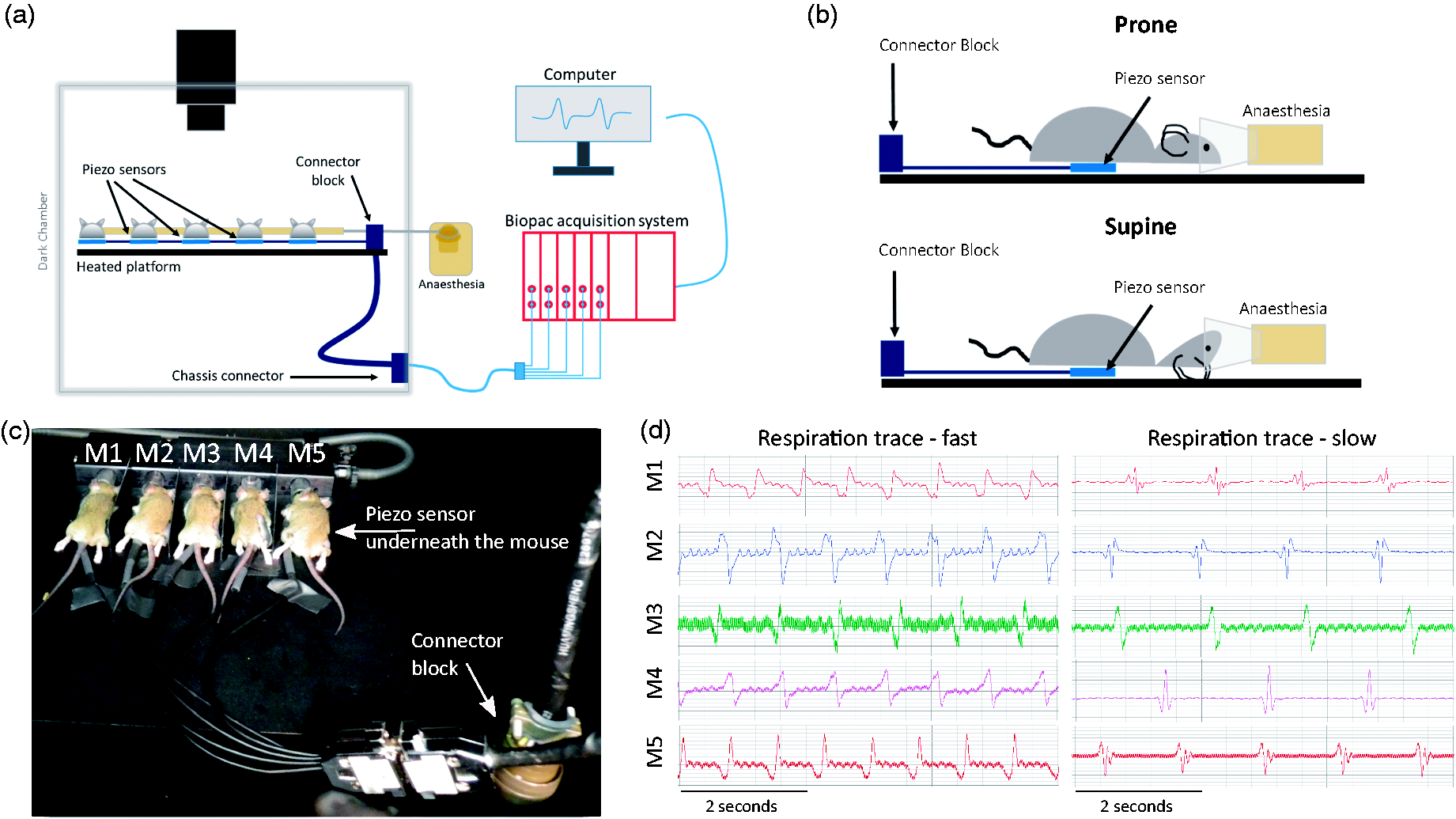
For system validation, five female CBA/CaCrl mice (10–12 weeks of age, 20.2 g ± 1.3, Charles River, UK) were used, as this is the maximum number of mice that can be imaged at one time in this system. The experiment was conducted in accordance with the Animals Scientific Procedures Act of 1986 (United Kingdom), and approved by the Committee on the Ethics of Animal Experiments of the University of Oxford. Mice were housed (n = 5 per cage) in individually ventilated cages with 12-hour dark/light cycle, maintained at 22℃ in 50% humidity, and were provided with certified rodent diet, filtered water ad libitum, autoclaved bedding material and cage enrichment. Anaesthesia induction was performed using 4% isoflurane in oxygen. Adequate anaesthetic depth was determined by lack of labyrinthine righting reflex, lack of pedal reflex, and breathing rate of 60–80 breaths per minute (bpm). Isoflurane concentration was then reduced to 2% and mice placed inside the IVIS imaging chamber, on top of the respiration sensors (Figure 1(b) and (c)). Overall average respiration rate (mean ± standard deviation) was 58.6 ± 7.1 bpm for prone (Figure 2(a)) and 75.7 ± 9.4 bpm for supine position (Figure 2(b)). Whilst in supine position, two mice were at risk of waking up as breathing rate quickly accelerated (Figure 2(b), grey arrows). This was promptly resolved by opening the imaging chamber door and correcting mouse position relative to the anaesthesia nose cone. Thoracic movement in supine position can cause the whole mouse to start moving away from the nose cone. This increases the risk of the mouse waking up mid-scanning, especially in the absence of a respiration monitoring system. This may be resolved with a modified gas delivery system that would incorporate a tooth-bar. At the moment, the main limitation of this respiration monitoring system is that, when used with transillumination, the piezoelectric sensor might be in the light path and prevent the light source being excited. Alternative respiration monitors could be developed for that specific application.
Breathing rate throughout time of five mice (M1–M5) placed in prone (a) and supine (b) positions (bpm: beats per minute). Grey arrows on panel (b) show two separate situations in which mice were at risk of waking up.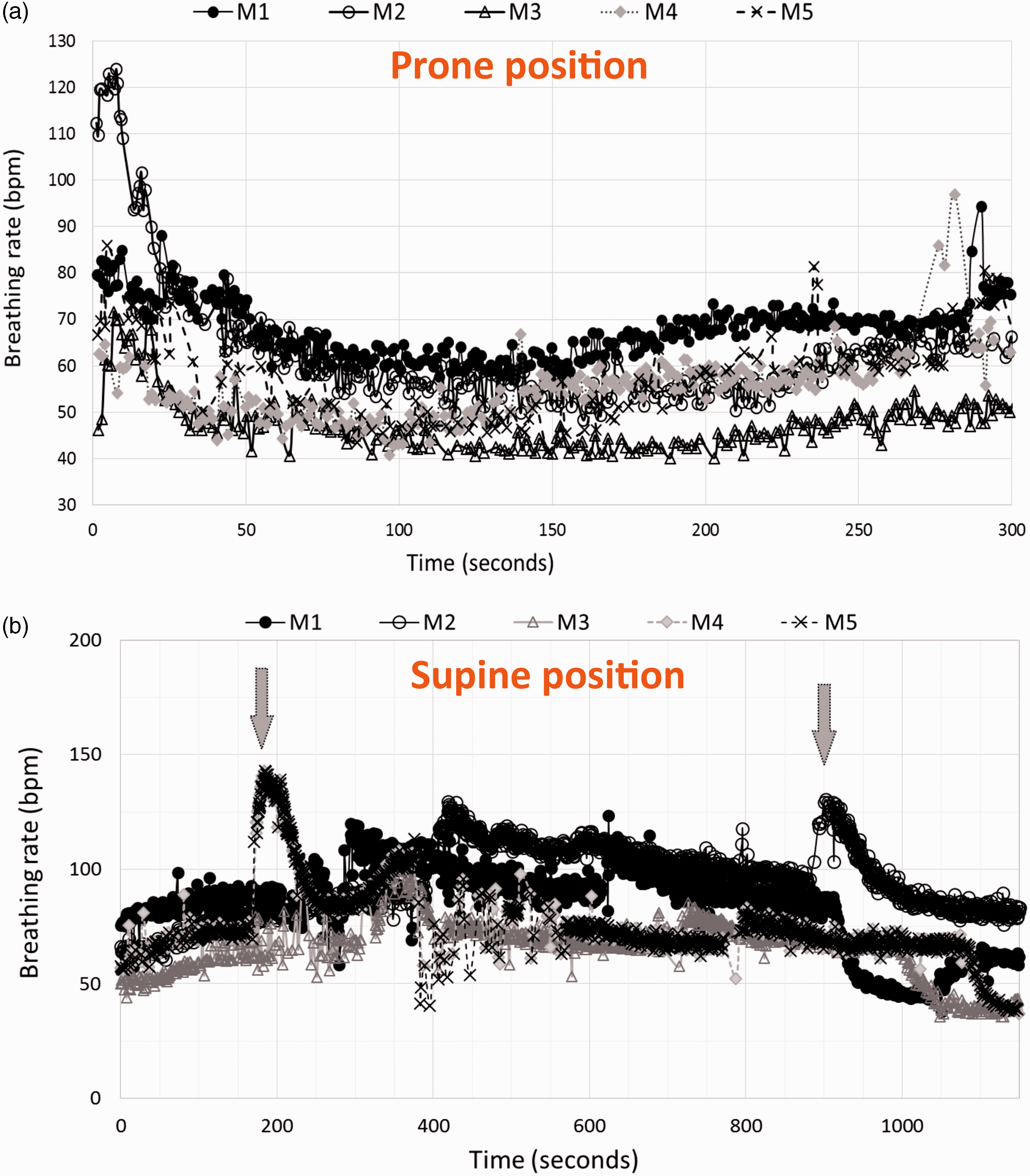
The advantages of respiration monitoring shown here are numerous. Not only does it improve animal welfare by tightly controlling respiration rate and minimising adverse effects on the animal’s physiology, but it also has the potential to significantly reduce animal numbers by minimising biological variance. 6 Moreover, it allows real-time decision on whether to continue or stop ongoing experiments; animals that are respiratorily depressed can be recovered whilst animals that are waking up can be intercepted before their micturition reflex returns. This is very important as an animal that wakes up mid-scanning will either need further dosing, repetition of the imaging session or even be killed and eliminated from study. Assessing the depth of anaesthesia via respiration rate monitoring also becomes an important safety consideration when radioisotopes are involved. A urine or faeces spill from a waking animal that has been dosed with radiotracer for Cerenkov, positron-emission tomography or single-photon emission computed tomography imaging, for example, presents a safety hazard for the operator and could render the system unfit for use until the spill is cleared. Additionally the recording of respiration monitor traces allows post hoc auditing and reporting of respiration rates.
In summary, we report a simple addition to the in vivo optical imaging protocol by enabling real-time respiration monitoring. It is not only an animal welfare refinement, but it also allows standardisation of anaesthesia depth between imaging sessions. This has the potential to reduce inter- and intra-mouse variability in bioluminescence read-outs and thus animal numbers. Similarly to what already exists in other in vivo imaging modalities, respiration monitoring is the first step to enable respiratory gated acquisitions that would help localise emission sources more precisely.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MW is an employee of PerkinElmer UK LTD. The other authors have nothing to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Cancer Research UK (C5255/A12678, C2522/A10339) and a Medical Research Council Unit Grant for Oxford Institute for Radiation Oncology. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
