Abstract
Rat models of lumbar intervertebral disc (IVD) degeneration are widely employed to characterize biologic-based therapeutics, but their anatomy and small size preclude consistent delivery of injectable therapeutics to the lumbar spine via the traditional posterolateral approach. Here, we describe our experience with a repeat ventral transperitoneal approach in female Lewis rats, enabling induction of IVD degeneration and later intervention via an injectable therapeutic. In the initial surgery, the ventral aspect of the L5/L6 IVD was accessed, and an annular defect was created using a #11 scalpel blade. Eight weeks after the initial surgery, follow-up surgery was performed via the same approach, and an injectable gelatin hydrogel was delivered using a 31G needle. A custom injection guard was developed to control injection depth, ensuring consistent delivery to the nucleus pulposus. Notable challenges associated with repeat surgery were increased tissue adhesion, intraoperative bleeding, and difficulty placing the injection guard due to mobile gastrointestinal tissues. Complication rates were 9.4% and 15.6% for the initial and repeat surgeries, respectively. The most frequent complications associated with repeat surgery were transient neuropraxia and significant intraoperative bleeding (6.3% each). The repeat transperitoneal approach is a reproducible method to facilitate both injury and later intervention in a female rat model of lumbar IVD degeneration.
Introduction
Intervertebral disc (IVD) degeneration is a complex and poorly understood pathology, which is difficult to simulate in vitro. Rats are widely employed as preclinical models of IVD degeneration and biologic-based therapeutics due to their low cost, comparable spinal anatomy, and advantageous size.1–8 Acute injury models of lumbar IVD degeneration in rats are achieved via scalpel or needle puncture, traditionally via a posterolateral approach.9–13 However, this approach produces a lateralized, asymmetrical defect. Furthermore, small size, transverse process morphology, and presence of the lumbar plexus make the posterolateral approach challenging in rats, precluding consistent delivery of injectable interventions. Injectable interventions are traditionally modeled using caudal discs7,8,14 due to ease of access and feasibility of percutaneous delivery, but the caudal disc does not accurately simulate the mechanical and biological environment of the lumbar spine.
Recently, a ventral transperitoneal approach to the lumbar spine has been described and applied in rat IVDs in vivo.2,15–17 The ventral approach allows visualization and access to the broader ventral aspect of lumbar IVDs, and enables creation of a symmetrical, medialized defect. The access afforded by the ventral approach theoretically also enables injection of therapeutic agents, though to date this has not been reported in the literature. Our group has recently developed and applied a repeat ventral approach in the rat lumbar spine, consisting of two separate surgeries to induce degeneration and then intervene via intradiscal injection (IDI). The purpose of this article is to report our technique and experience with this model.
Methods
Animal sourcing and husbandry
All procedures were conducted under an Institutional Animal Care and Use Committee-approved protocol for a larger grant-funded study, and the sample size (determined via power analysis) and the use of only female rats were determined based on the needs of this larger study. Standard aseptic techniques were employed as prescribed by the Association for the Assessment and Accreditation of Laboratory Animal Care guide. Female Lewis rats (N = 32, retired breeders, aged five to six months, specific pathogen free) were acquired from Charles River Laboratories (Wilmington, MA). Rats were housed in a 12-hour light/dark facility in a standard barrier-maintained colony, monitored via a sentinel animal program, and allowed ad libitum cage activity with free access to water (via a sipper tube) and food (Teklad global soy protein-free extruded rodent diet; Envigo, Huntingdon, UK).
Our model consisted of two surgeries performed on the L5/L6 disc via the ventral approach: an initial annular puncture injury to induce IVD degeneration, followed by injection of a therapeutic agent 8 weeks post-injury. Rats were followed for 12 weeks following the second surgery and euthanized via CO2 inhalation, in accordance with the American Veterinary Medical Association Guidelines for the Euthanasia of Animals.
Surgery 1: ventral disc puncture injury
Prior to surgery, rats received subcutaneous glycopyrrolate (0.02 mg/kg; American Reagent, Inc., Shirley, NY), enrofloxacin (5 mg/kg, Enrosite; VetOne, Boise, ID), and carprofen (5.0 mg/kg, Rimadyl; Zoetis, Inc., Parsippany-Troy Hills, NJ). Anesthesia was induced via intraperitoneal acepromazine (5.0 mg/kg, AceproJect; Henry Schein, Inc., Melville, NY) and maintained via 1–2% inhaled isoflurane (Forane; Baxter International, Inc., Deerfield, IL) at a flow rate of 1 L/min oxygen, introduced via an induction chamber and maintained via a mask. The abdomen was shaved using clippers and prepped with povidone–iodine and isopropyl alcohol just prior to surgery. Surgery was performed in the dorsal recumbent (supine) position over a covered heating pad to assist in maintaining body temperature. The area surrounding the incision site was covered with surgical drapes, and a fenestrated, transparent rodent drape was used to cover the immediate incision site.
Disc puncture injury (DPI) via the ventral approach was conducted similarly to previous reports.15–18 The xiphoid process and pubic symphysis were palpated and marked. A 3–4 cm midline incision was made between the middle and distal thirds between the xiphoid and pubic symphysis. The abdominal wall was lifted with forceps to allow the intraperitoneal contents to fall away. A small incision was made with a scalpel in the midline raphe through the abdominal wall and extended with scissors through the abdominal wall proximally and distally, while directly visualizing the intra-abdominal contents to avoid injury. The cecum and mobile large intestines were removed from the abdominal cavity and surrounded with warm saline-soaked gauze. The small intestines were left in place to minimize risk of gastrointestinal (GI) distress and morbidity15—when this obscured visualization, warm saline-soaked gauze was used to pack and retract the small intestines from the operative field. The dorsal abdominal wall was bluntly dissected with cotton-tipped wooden applicators. Dissection of the psoas off the vertebral body was minimized to reduce bleeding. The iliolumbar vein crosses over just cephalad to the L5/L6 disc space and was used as a landmark (Figure 1(a)). In our experience, the L5/L6 disc could be consistently approached without tying off the iliolumbar vein, preserving its use as a landmark in the second procedure. L5/L6 was located by identifying the “peaks” of the intervertebral disc between the “valleys” of the vertebral body (Figure 1(b)).
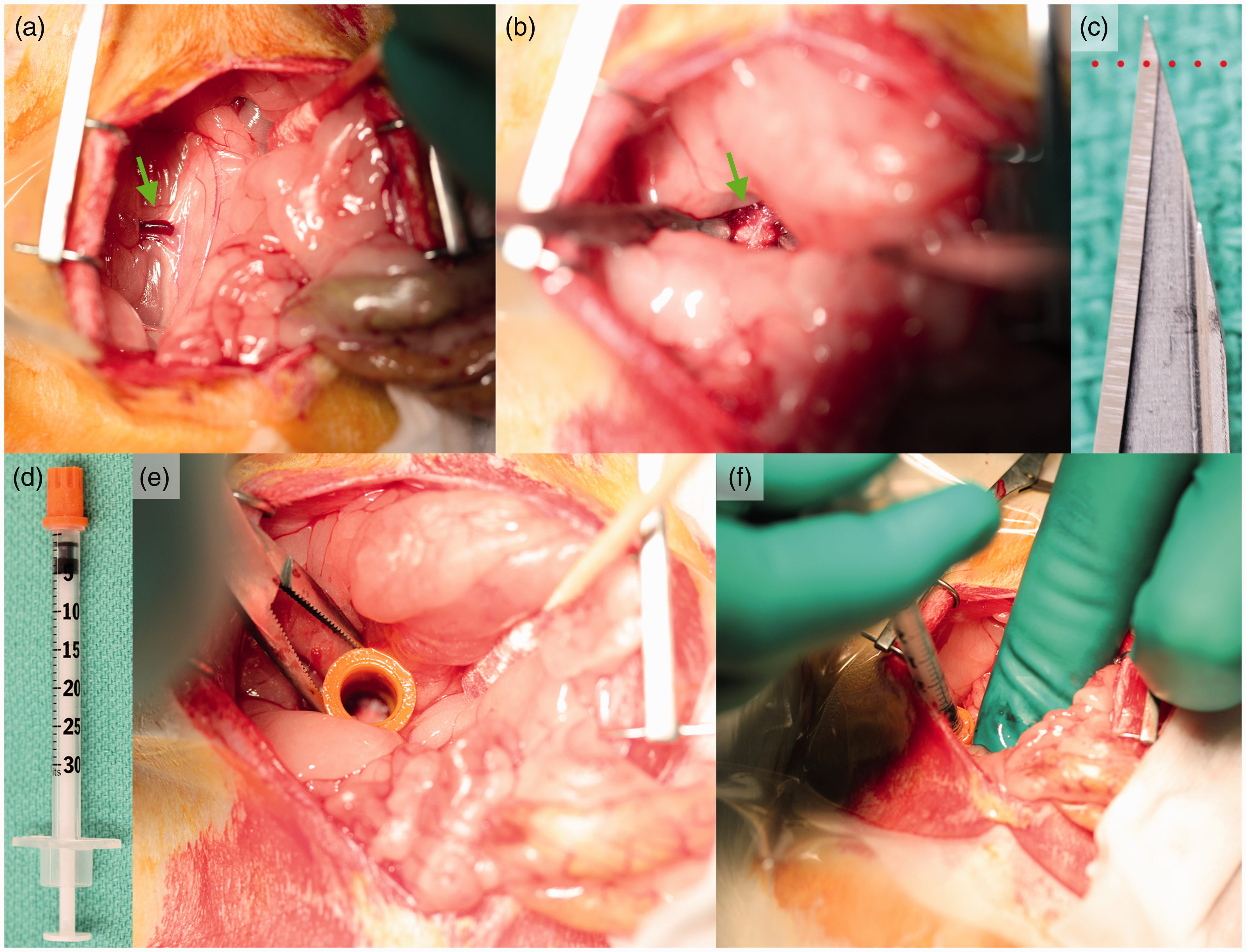
The L5/L6 disc level was located using the iliolumbar (green arrow) as a reference (a). Once the L5/L6 IVD (green arrow) was isolated (b), annular puncture was induced using a #11 scalpel inserted to the first bevel (dotted line) (c). Intradiscal injection was accomplished via the use of a custom injection guard (d). The guard was placed in the surgical site over the L5/L6 IVD (e), and then the needle was inserted and the injection delivered (f). IVD: intervertebral disc.
A midline plane between the psoas muscles was created, approaching the spine from the left while sweeping the great vessels to the right. The L5/L6 disc was located and confirmed fluoroscopically (OEC 9800; GE Healthcare, Inc., Chicago, IL). DPI was induced using a #11 scalpel blade inserted to the first bevel (Figure 1(c)) in an “X” pattern. After intervention, a running, locked, 4-0 Vicryl absorbable suture (Johnson & Johnson, New Brunswick, NJ) was used to close the abdominal wall, and interrupted 4-0 Ethilon sutures were used for the skin.
Immediately following surgery, rats received subcutaneous injections of long-acting buprenorphine (1.2 mg/kg, Buprenex SR; ZooPharm, Fort Collins, CO) and warmed lactated Ringer’s solution (3 cc). Intraoperative blood loss was determined by the surgeon’s visual estimation, and rats estimated to have lost >1 cc received an additional 2 cc of lactated Ringer’s solution to combat dehydration. Rats were single-housed for the first postoperative week and monitored closely for signs of neuropraxia, GI distress, and other issues. Rats were returned to group housing at 1 week postoperatively. At 2 weeks postoperatively, skin sutures were removed by a trained veterinary technician under inhaled isoflurane.
Surgery 2: ventral intradiscal injection
IDIs were delivered via a 31-gauge, 6 mm, 0.3 mL insulin syringe (Beckton-Dickinson, Franklin Lakes, NJ) using 10 µL gelatin hydrogel vehicle (Sigma–Aldrich, St. Louis, MO). To facilitate consistent delivery to the nucleus pulposus (NP), we developed an injection guard consisting of an insulin needle cap modified to a length of 10.4 mm, restricting injection depth to roughly the first 2 mm of the needle tip (Figure 1(d)). This guard length consistently delivered the contents of the needle to the NP (determined in preliminary studies using dye and dissected cadaveric spines). Guards were disinfected via ethanol sonication prior to each surgical date and wiped/soaked in isopropyl alcohol before and after individual surgeries.
Preoperative anesthesia, analgesia and preparation, and postoperative management were conducted as described above for DPI surgery. The transperitoneal approach was performed as described in the first procedure, with the following modifications and considerations. When going through the ventral abdominal wall, the initial incision was made just proximal of the previous approach to prevent inadvertent damage to any peritoneal contents adhered to the undersurface of the abdominal wall. The incision was carried distally with scissors, and adhesions on the undersurface were released via a cotton-tipped wooden applicator. During the dorsal abdomen approach, the surgeon (S.G.) observed that elevating the psoas muscle off the ventral spine was more difficult compared with the first approach—the muscle was more adherent and bled more often. Bleeding could always be adequately controlled with direct pressure with a cotton-tipped wooden applicator. The injection guard was placed over the L5/L6 IVD prior to injection (Figure 1(e)) to ensure consistent delivery of the hydrogel into the NP. The needle was then inserted into the positioned guard (Figure 1(f)), and the injection was delivered, followed by the same closure procedure as used in the initial surgery.
Results
In our series, no technical reasons prevented the repeat ventral approach from being used. Complication rates for DPI and IDI surgeries (Table 1) were 9.4% (3/32) and 15.6% (5/32), respectively. No specific complication occurred in more than two animals in either procedure. No rats suffered postoperative GI distress. IDI was associated with slightly higher incidences of intraoperative bleeding, as measured by the surgeon’s visual estimation, and transient postoperative neuropraxia, which manifested as altered gait and delayed foot placement of a rear limb and/or abnormal hip/knee extension. Both cases of neuropraxia improved gradually and resolved within 3–4 days. There were two instances of necessitated euthanasia in our data set: one due to a rat repeatedly opening its skin incision, and one due to a missed IDI—the surgeon noted that the needle had drifted into the L5/L6 cranial endplate rather than the annulus and was thus not delivered intradiscally. In both cases, the animal was immediately euthanized via CO2 inhalation as described above.
Complication rates of repeat ventral approach stratified by surgery and reported as number of cases (percent of cases).
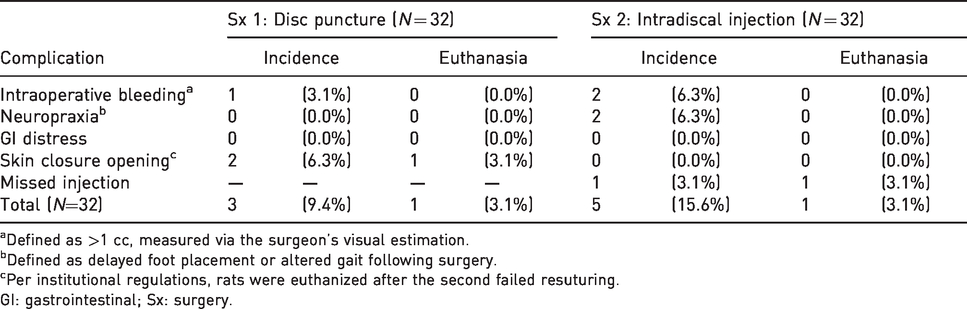
aDefined as >1 cc, measured via the surgeon’s visual estimation.
bDefined as delayed foot placement or altered gait following surgery.
cPer institutional regulations, rats were euthanized after the second failed resuturing.
GI: gastrointestinal; Sx: surgery.
Discussion
Despite significant interest in biologic therapies for IVD degeneration, most commonly used preclinical models do not easily enable creation of a chronic IVD degeneration model and subsequent intervention at a later time point—this is particularly challenging in small rodent models. Our experience using the repeat ventral approach indicates that this approach enables induction of IVD degeneration, with subsequent intervention, within the anatomically relevant rat lumbar IVD.
IDI was associated with a mild increase in the incidence of intraoperative bleeding. The surgeon observed that the plane between the psoas and lumbar spine was more difficult to dissect due to the presence of fibrous adhesions from the previous surgery, and bleeding was encountered more frequently than in DPI surgery. Direct pressure with cotton-tipped wooden applicators adequately controlled this bleeding, and when deemed necessary, rats were administered additional subcutaneous fluid during recovery to prevent dehydration. Intraoperative blood loss was visually estimated by the surgeon based on his past experience with clinical and rodent surgery, and was not quantitatively measured—our data should be interpreted in light of this limitation.
Dissections were kept within 1.5mm of the midline to avoid lumbar plexus damage and neurologic complications. However, we noted two cases of acutely altered gait following IDI surgery. Meloxicam SR (4.0 mg/kg, subcutaneous; ZooPharm) was administered to these animals to combat nerve compression related to tissue inflammation and to resolve acute neuritis. Given that both cases were transient and resolved within 3–4 days postoperatively, they may have been related to increased intraoperative bleeding during the second surgery leading to transient hematoma and pressure on the nerve. Another possible explanation is that the increased presence of fibrous adhesions near the nerve during IDI required increased dissection and retraction and may have led to traction neuropraxia. Application of lateral pressure may help mitigate this complication.
During preliminary cadaveric practice injections, the most significant challenge for IDI was controlling the injection depth to ensure consistent delivery to the NP. In our experience, visual feedback and tactile feedback were inadequate to ensure consistent delivery, particularly given the very small size of the target tissue. The injection guard we developed was a straightforward and effective means to standardize the injection depth, and preliminary optimizations using dye indicated consistent delivery into the NP. Correct placement of the guard and needle within the surgical site required great care due to the mobility of surrounding tissues and the fact that the needle itself blocks injection site visualization. The surgeon found that correctly positioning the guard before attachment of the needle maximized consistent placement. Injection was nonetheless challenging, and a missed IDI was responsible for one of the two euthanasias in our dataset.
The described experiment was carried out using female Lewis rats only, and we acknowledge that complication rates in male rats could be slightly different. The length of the injection guard may also have to undergo minor re-calibration to account for the slightly larger size of male Lewis rats. Our study should be interpreted in light of this limitation.
Our experience in more than 30 rats has shown that the repeat ventral transperitoneal approach is well tolerated and can be utilized to allow creation of a chronic disc degeneration model at an anatomically relevant disc level, followed by intervention at a later time point.
Footnotes
Acknowledgements
The authors wish to acknowledge Heather Brzezinski, BA, LVT, Estrellita Morrison, LVT, LAT, Michele McGonagle, BIS, LVT, LAT, and Susan Cook, DVM, MS, DACLAM, for animal care and husbandry, surgical preparation, anesthesia, intraoperative monitoring and recovery, and veterinary consultation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this work was provided by a 2016 Resident/Fellow Research Grant from the Cervical Spine Research Society.
