Abstract
The refinement of surgical techniques represents a key opportunity to improve the welfare of laboratory rodents, while meeting legal and ethical obligations. Current methods used for monitoring intra-abdominal disease progression in rodents usually involve euthanasia at various time-points for end of study, one-time individual tissue collections. Most rodent organ tumour models are developed by the introduction of tumour cells via laparotomy or via ultrasound-guided indirect visualization. Ischaemic rodent models are often generated using laparotomies. This approach requires a high number of rodents, and in some instances introduces high degrees of morbidity and mortality, thereby increasing study variability and expense. Most importantly, most laparotomies do not promote the highest level of rodent welfare. Recent improvements in laparoscopic equipment and techniques have enabled the adaptation of laparoscopy for rodent procedures. Laparoscopy, which is considered the gold standard for many human abdominal procedures, allows for serial biopsy collections from the same animal, results in decreased pain and tissue trauma as well as quicker postsurgical recovery, and preserves immune function in comparison to the same procedures performed by laparotomy. Laparoscopy improves rodent welfare, decreases interanimal variability, thereby reducing the number of required animals, allows for the replacement of larger species, decreases expense and improves data yield. This review article compares rodent laparotomy and laparoscopic surgical methods, and describes the utilization of laparoscopy for the development of cancer models and assessment of disease progression to improve data collection and animal welfare. In addition, currently available rodent laparoscopic equipment and instrumentation are presented.
Keywords
Regulatory and ethical considerations in response to public demands have increased academic, industrial and governmental sensitivity to animal welfare in biomedical research. Investigators utilizing animals in research are required to design experiments with consideration to Russell and Burch's ‘Three Rs’ ... of refinement, reduction and replacement’. 1 Here, refinement is defined as the ‘reduction of the amount of stress imposed on animals being used’, 1 and reduction is defined as the ‘reduction in the number of animals used to obtain information of a given amount and precision’. 1 The initial definition of replacement meant a replacement of a living organism with a non-living organism.
However, it has been updated and subdivided into absolute replacement, which means complete replacement of animals, and relative replacement which involves utilization of ‘animals of lower neurophysiology sentience or animal tissues used’. 2 Within the last two decades, experimental study design has received significant attention in regard to implementation of biomethodologies, animal husbandry, pain assessment and humane endpoints to maximize rodent welfare in biomedical research.3–6 In addition to elevating the standards for rodent care and use to match those mammalian species covered under the Animal Welfare Act, specific surgical procedures have been refined to meet the challenges presented by these smaller species. 6
Rodent tumour models are becoming more complex with the widespread adoption of genetically-modified mice and rats.7–10 Prior to the 1970s the majority of tumour experiments in rodents were performed by growing neoplastic cells subcutaneously or under the renal capsule.11,12 During the late 1960s and 1970s orthotopic xenograft models were introduced, such as the co-injection of human breast carcinoma and stromal cells into the mammary fat pad of immunocompromised mice.13–15 These models reproduce the host and tumour cellular interactions more accurately and thereby support the study of human disease progression in rodent models with higher dependability. By providing appropriate organ microenvironments, the chemosensitivity and metastatic behaviour of tumour cells are better preserved, allowing for the
In addition to providing animal models for investigating the complexities of intra-abdominal tumour biology (e.g. tumour development, metastasis, effects of immunity), rodents are widely used for toxicological, metabolic and inflammatory disease studies. A current requirement of such studies involves the euthanasia of rodents at various time-points to allow for gross, histopathological and molecular analyses of tumour progression. This experimental design requires a larger number of animals per study to obtain statistically significant data. Increased numbers of rodents at each time-point are also required to overcome the increased interanimal variability, morbidity and mortality associated with laparotomies. 25 Advances in non-invasive bioluminescent and fluorescent imaging allow longitudinal assessment of the growth and metastatic dissemination of orthotopic tumours but are limited by the inability to collect serial tumour samples.26–31 Laparoscopy can augment these technologies by providing direct observation and assessment of gross and macroscopic morphology, and access to collect biopsies with high precision through direct visualization of target organs. Most importantly, the scientific community has a moral and legal responsibility to develop and implement methods that refine experimental models, minimize potential pain and stress, while providing quality data that meet study objectives.4,32
Overview of laparoscopic surgery
Laparoscopic surgery, also referred to as minimally invasive surgery of the abdomen, is a surgical technique that involves making small incisions through which ports are placed. The camera and laparoscopic instruments are then introduced through the ports that allow access to the abdomen. The camera transmits an image of the organs onto a monitor, and the surgeon uses the image from the camera to perform the surgical procedure. Laparoscopy in human and veterinary medicine provides a powerful method to examine the abdominal cavity for neoplastic and inflammatory changes and to collect biopsies. Laparoscopy has become the gold standard in human medicine for many abdominal surgical procedures (e.g. cholecystectomy).33–36 Laparoscopic surgery has several benefits over laparotomy (open surgery). Animals recover faster, as the potential for postoperative pain, inflammation and infection are reduced.37–40 There is a quicker return to normal metabolic and behavioural activities, including intestinal transit recovery and subsequent normal food and water intake, than when the same procedure is performed by laparotomy. The laparoscopic approach allows for repeated visualization, biopsy collection and intervention within the same rodent which eliminates interanimal variability. 25 Biopsies of the liver, spleen, kidney, mesenteric lymph nodes and gonads can be performed safely, swiftly and easily in all rodents including mice and rats. Laparoscopic techniques can be easily standardized and reproduced, decreasing user bias. Laparoscopy increases the sensitivity of disease assessment relative to other techniques.41–45 Laparoscopy can be especially useful in diseases such as alcoholic steatohepatitis, where it can be difficult to assess disease progression due to variations in disease severity between individual rodents.25,46–48 Laparoscopic procedures allow the serial collection of biopsies, facilitating longitudinal studies of disease progression within the same animal, rather than relying on postmortem samples collected from replicates. This also permits the monitoring of early stages of diseases, when pathological changes are usually limited or non-existent. 5 Direct, magnified laparoscopic visualization allows for the precise placement of tissue or tumour cells into target organs, promoting the study of disease development in its natural or xenogenic microenvironment.
Rodent laparoscopy
Rodent laparoscopic procedures have been reported in the literature but are not routinely used by the vast majority of biomedical researchers. This resistance to adopting this superior surgical approach has been mainly due to the need for specialized surgical skills and lack of available training, and the large expense and size of laparoscopic equipment needed to perform laparoscopic procedures in rodents. Laparoscopic towers typically include a transillumination unit for the laparoscope, a power source and monitor for a charge coupled-device camera, as well as an insufflation unit and tank. These stacks of equipment utilized in human and veterinary surgeries have traditionally been quite large and require spacious surgical suites for proper placement and use. This presents a challenge, since most institutions do not have dedicated rodent surgical suites, and procedures are often performed in areas that serve other purposes.49,50 In addition, the use of existing veterinary and human laparoscopic instruments that typically range between 5 and 10 mm in diameter for rodent surgeries is inappropriate, as mouse and rat instrumentation should ideally be no wider than 3 mm in diameter.51–54
Recent advances in the laparoscopic field have led to a significant reduction in the size and cost of equipment. New rodent laparoscopic towers are compact and mobile. They can easily fit on a bench top and be transported between procedure rooms or facilities (Figure 1). Specialization of instruments and miniaturization of optics have made many rodent procedures using laparoscopy now practical. Rodent-specific scopes provide high-quality imaging and are available as either viewing or working laparoscopes. Viewing laparoscopes allow only for visualization of the cavity without the ability to manipulate tissues and are available in diameters as small as 0.4 mm (images of various small sizes) (Figure 2). Whereas, a working laparoscope has a channel, or built-in working channel, through which laparoscopic operating instruments are introduced into the abdomen (Figure 2). These working laparoscopes are available in diameters as small as 1.9 mm and allow for the collection of intra-abdominal biopsies of sufficient size for histopathological and molecular analyses through a single incision (~2 mm) (Figure 3). This specialized instrumentation can also be used to perform various procedures such as ovariectomies through a single port. Additional minimally invasive techniques can be performed with this same instrument to produce renal or ovarian ischaemia models. The development of lighter and shorter instrumentation adapted for smaller species also reduces user fatigue, leading to a steadier hand. This is especially important in rodents, where a slight movement can result in inadvertent tissue trauma and poor surgical outcome. Traditional human and veterinary laparoscopes can be 30 cm or longer, whereas rodent laparoscopes are usually about 10 cm in length (Figure 4). These shorter instruments allow for better control, thereby decreasing the risk of complications. Improvements in rodent laparoscopic optics yield high-quality videos and images, which can be used for data collection, publication and teaching purposes. This can be especially important for good laboratory practice studies that have strict requirements for data collection.55,56 Rodent laparoscopy allows for high precision injection of test compounds and neoplastic cells under direct visualization. These procedures can be performed serially in the same animal with minimal complications. Historically, many of these procedures were not feasible in these species and animals were euthanized to visualize the intra-abdominal organs or collect tissue samples. As a consequence, only indirect evidence of tumour development and disease progression, such as weight loss and poor hair coat, could be obtained prior to euthanasia. These clinical signs can also occur in response to stress or as a result of unrelated infections, especially when using immunocompromised mice and rats, rather than cancer-induced cachexia.
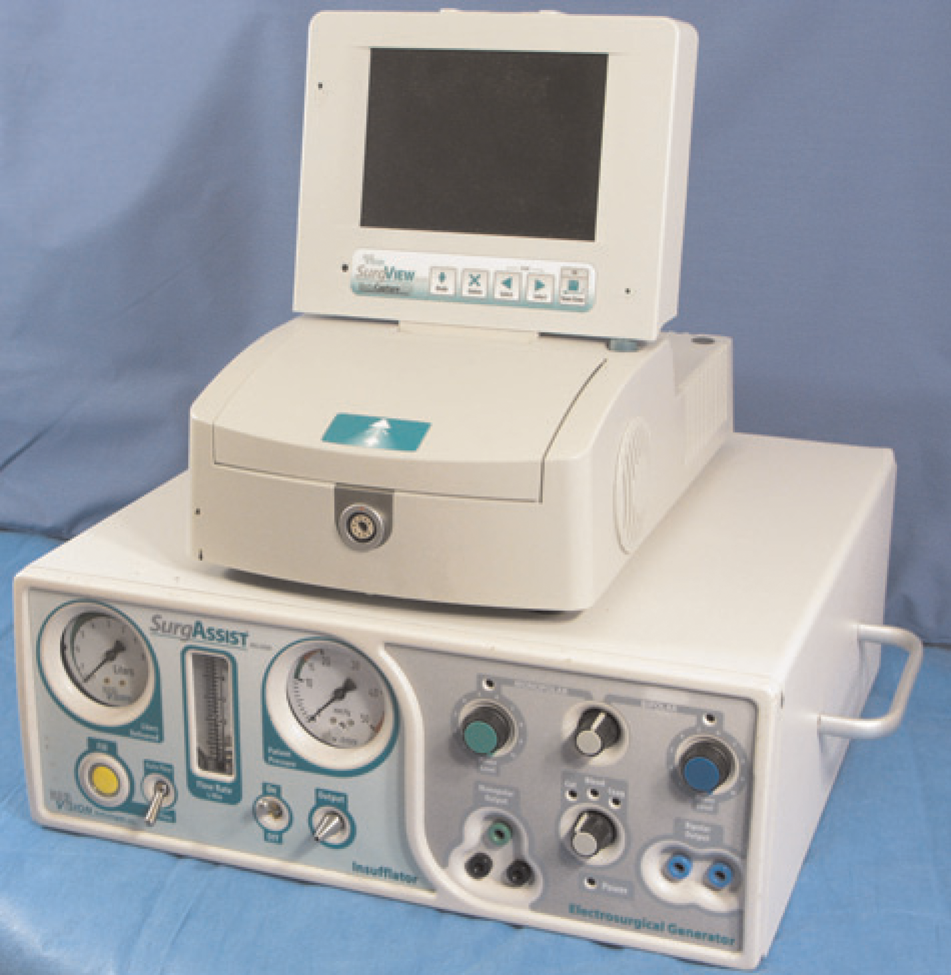
Compact laparoscopic tower (BioVision Technologies, Denver, CO, USA)
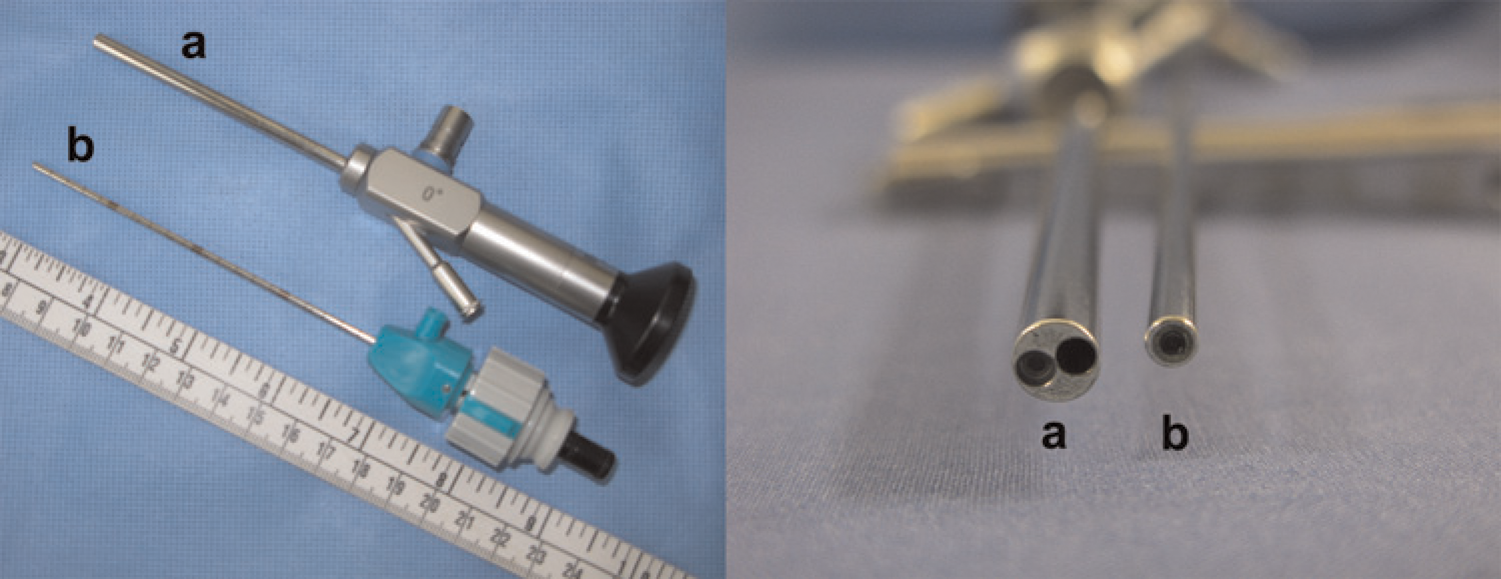
Laparoscopes with (a) working channel and (b) without working channel (BioVision Technologies)
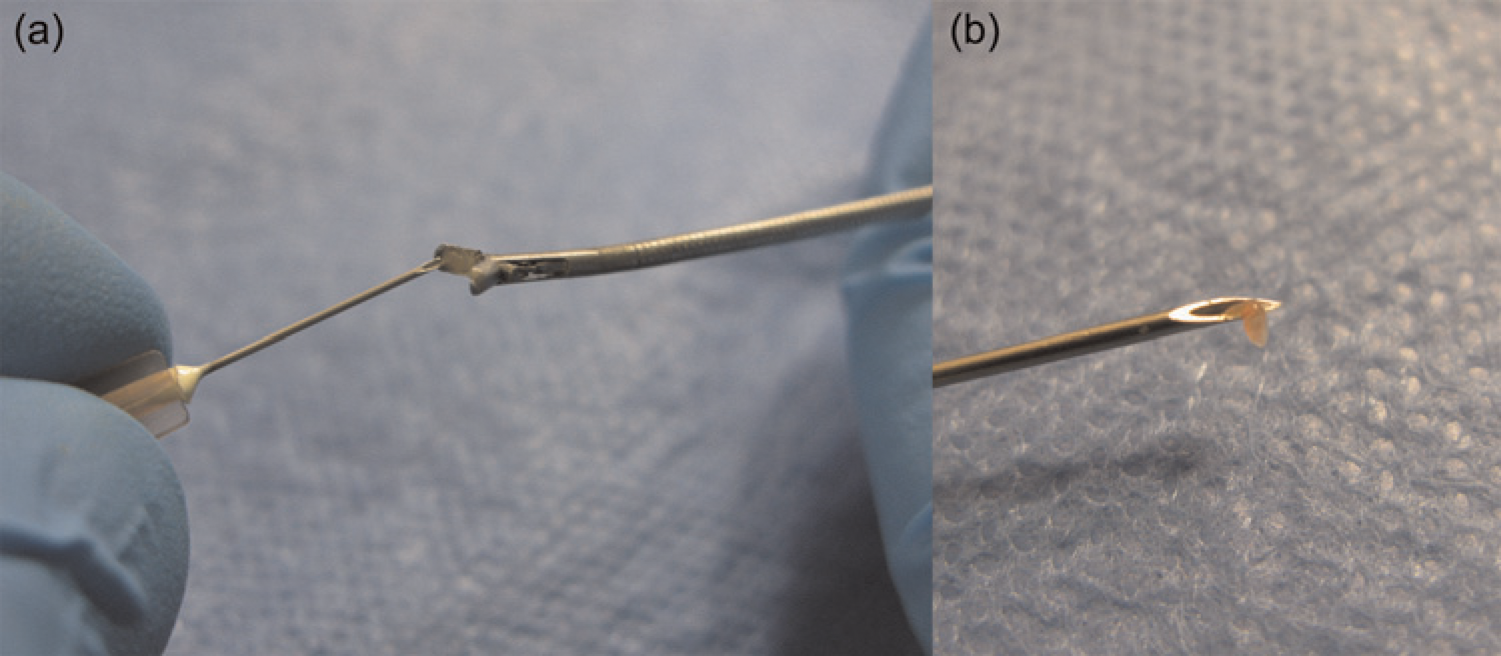
Liver biopsy obtained with (a) 2 mm biopsy graspers (BioVision Technologies) and (b) removed with a 30-gauge needle (Fisher Scientific, Waltham, MA, USA)
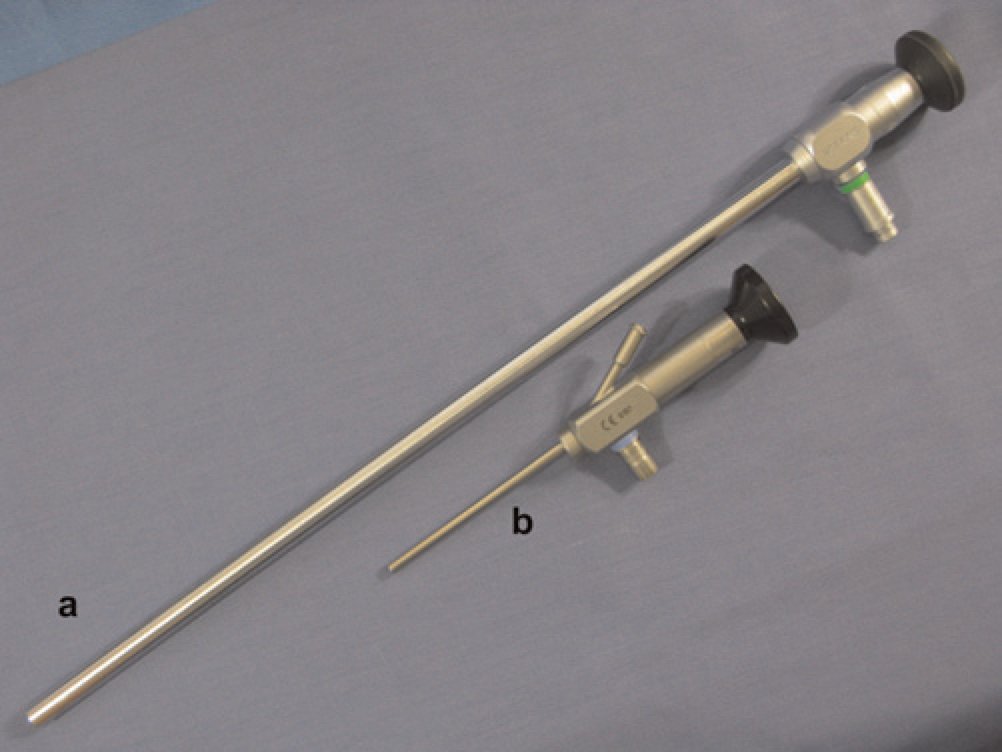
Comparison of a 33 cm laparoscope and a 10 cm laparoscope (BioVision Technologies)
Harm–benefit and financials–benefit
Laparoscopy can and should be considered to replace or augment common procedures such as laparotomies and terminal tissue collections. Toxicology and longitudinal studies of neoplastic, inflammatory and metabolic diseases can greatly benefit from incorporating serial laparoscopic assessments. Doing so will improve surgical recovery, reduce postsurgical pain and preserve immune function. Performance of serial observations and biopsy collections within the same animal has the added benefits of reducing study variables and potentially reducing costs by increasing animal survival. The same test subject is utilized repeatedly, serving as its own control. Although an initial investment will need to be made to purchase the necessary laparoscopic equipment, the costs for rodent instruments and laparoscopic equipment are significantly less than for typical companion animal and human systems. Such initial expenditure is well worth the investment and the benefits to the animals and the researcher are many.
Adoption of laparoscopy in appropriate rodent models may allow for the cost-effective replacement of traditional large animal models. Mammalian species such as pigs or non-human primates have commonly been used for laparoscopic studies because their size closely approximates that of humans, and because human laparoscopic instruments could readily be used in these species. However, large animal models are expensive to maintain and limited by the number of institutions with appropriate housing.57–62 Furthermore, species-specific reagents are necessary to carry out physiological, immunological and oncological assays. The availability of these assays and reagents for large species is quite limited, whereas they are usually readily available for mice and rats.59,60,62 The ability to genetically alter rodents allows for development of lines with human genes. Such technology at present is limited in larger species.63–66 Even for tumour studies, the number of cultured tumour cell lines developed for large animals is scarce, making it challenging to conduct such
Consequently, there is a learning curve with any newly performed surgical technique, and surgeons should be proficient no matter what surgical modality is utilized. 69 Overall, it is important to keep in mind that a specific procedure, such as laparoscopy, will have a positive impact on three Rs, and the authors believe that laparoscopy would improve animal welfare.
Scientific aspects
Gas exposure
One aspect of laparoscopic procedures that is often not addressed is the decreased exposure of intra-abdominal organs and tissues to atmospheric air. Exposure to room air leads to exaggerated physiological alterations, e.g. interleukin (IL)-6 response. 70 The acidification of the peritoneum via CO2 insufflation leads to attenuation of the inflammatory response through up-regulation of anti-inflammatory cytokines such as IL-10 and down-regulation of proinflammatory cytokines such as TNFΑ.70–74 Acidaemia induced by CO2 can be minimized by using the lowest intra-abdominal pressure necessary to expand the abdomen for visualization. 75 CO2 pneumoperitoneum preserves cellular immune functions, lowers abdominal discomfort (feeling of retained gas in the abdomen) and offers better visibility during laparoscopy when compared with an open surgery. 76
Immune function
Laparotomy results in significant postoperative immune suppression, due to decreases in lymphocyte count and lymphocyte/macrophage interactions, neutrophil Chemotaxis, natural killer cell activity, alpha-2-macroglobulin release and delayed hypersensitivity responses.77–82 This immune suppression compromises the rodent's defences at the time, when the risk for introducing pathogens is maximal and can lead to increased morbidity and mortality. This effect can last up to nine days after surgery, and is a specifically important confounding variable in cancer models, as immune suppression may effect tumour cell proliferation and engraftment rates.76,79,83,84 Such immune suppression is directly proportional to the degree of surgical trauma. The higher the trauma, the greater the effect. Thus, the minimal invasiveness of laparoscopy, when compared with open surgery tends to preserve immune function by preserving T-cell mitogen response and monocyte release of tumour necrotic factor and superoxide.85–87 Often the incision is smaller with laparoscopic surgery versus laparotomy, and larger incisions lead to postoperative decreases in animals’ abilities to mount effective cell-mediated immune responses.83,88
Faster return to normal physiological status
Recovery of normal gastric emptying and normal intestinal motility is faster with laparoscopic surgery when compared with laparotomy. 37 Radiographic analyses demonstrate increased intestinal gas postlaparotomy, and histological analyses of the gastrointestinal tract postlaparotomy show pronounced serosal layer thickening in comparison with laparoscopy. 37 Postoperative adhesion formation has an impact on recovery since adhesions can cause intestinal obstructions and chronic pain.89–93 In general, laparoscopy leads to less adhesion formation than laparotomy. A potential mechanism for this difference may be due to the introduction of room air during laparotomy. Increases in intra-abdominal partial oxygen tension above physiological levels is detrimental to serosal cells promoting the formation of reactive oxygen species and inflammatory reactions. 94 Transient introduction of room air into the abdomen by laparotomy can introduce a profound effect on subsequent tumour growth.
Tumour development and metastasis
Rodents often serve as intra-abdominal tumour models for studying hepatic, splenic or renal cancers. Most commonly cancer cells are injected into the liver or spleen or under the kidney capsule in mice and rats, after these target organs are visualized and/or exteriorized by laparotomy.95–97 All of these techniques expose tissue to room air which induces an inflammatory response.
98
Jacobi
Surgical trauma can also impair cell-mediated immune responses and increase the risk of infection and metastatic spread.101,102 Tsuchiya
Multiple surgeries
The Animal Welfare Act and the
Addressing laparoscopic limitations
The principal disadvantages of laparoscopy are the need for specialized equipment and training. Adequate training on how to use laparoscopic equipment effectively is imperative to perform laparoscopic procedures successfully. The cost of rodent laparoscopic equipment has decreased dramatically in the last few years, and training is becoming more available during annual meetings such as the Academy of Surgical Research and American Association for Laboratory Animal Sciences.109,110 As to anaesthesia, the preferred method for rodent laparoscopic procedures is isoflurane through a calibrated vaporizer. This significantly decreases the anaesthesia time compared with injectable anaesthesia protocols thereby allowing mice and rats to recover quickly. The amount of analgesia required for laparoscopic procedures is significantly less relative to laparotomies.111–115 It has been suggested that laparoscopy may lead to port site tumour seeding due to the intra-abdominal pressure;116–119 however, this is more likely the result of intraoperative instrument contamination.
120
Most reports of metastases associated with laparoscopy are clinical cases with many variables and lacking adequate controls.121,122 Mutter
Conclusion
Rodents serve as invaluable experimental models for investigating the pathogenesis of and evaluating potential therapeutic treatments of neoplastic, inflammatory and metabolic diseases. The scientific community has made considerable refinements in how rodents are used in research. These refinements have improved rodent welfare, while allowing researchers to reach their study objectives more reproducibly. However, we must continually strive to refine experimental techniques and identify new methodologies to improve animal welfare as required by regulatory and accrediting bodies, and as part of our ethical responsibility.104,109
In this article, we have tried to provide the most compelling evidence that demonstrates the benefits of rodent laparoscopic surgery. Rodent laparoscopy presents an opportunity for significant refinement of rodent surgical procedures by reducing tissue damage and inflammation, postsurgical pain and recovery time, and by preserving the immune system when compared with open surgery. Manipulations such as direct injection of reagents or the taking of biopsies from target organs or tissues can easily be performed with precision, allowing the researcher to assess and manipulate disease progression in mice and rats
