Abstract
The intrathecal (IT) route of administration represents a means to reduce the dose of morphine administered for analgesia, potentially minimizing interactions between opioid effects and experimental outcomes. Perceived technical difficulty, and previously described invasive methods, may limit its use. This report describes a minimally invasive technique for IT administration of morphine by direct transcutaneous lumbosacral puncture in rats; and assesses antinociceptive properties of morphine in anaesthetized rats. Rats (n = 28) anaesthetized with sevoflurane (inspired fraction of sevoflurane: FiSevo = 2.4%) were randomly allocated to receive: IT morphine (0.2 mg/kg); subcutaneous (SC) morphine (3 mg/kg); SC buprenorphine (0.05 mg/kg); or SC or IT sodium chloride (NaCl). After a wash-in period (40 min), thermal nociceptive stimuli were applied at nine locations corresponding to different rostrocaudal dermatomes of the rat. Nociceptive stimulation cycles were repeated at all locations after successive decrement of FiSevo by 15%. Presence or absence of gross purposeful movement (GPM) was recorded for each individual stimulation. IT injection of morphine by direct puncture with a 25 G hypodermic needle is easily performed (successful first attempt: 82%) without complications. IT morphine reduced the frequency of GPM following nociceptive thermal stimulation in a way comparable with SC buprenorphine or morphine. It was not possible to delimit any rostral spinal spread of morphine. This report describes a refined and effective technique of administering morphine IT in rats using readily available materials. IT doses being markedly smaller than the systemic equivalent, analgesia could be provided whilst minimizing the potential interactions of non-analgesic opioid effects with research protocols.
Public perception and acceptance of animal models used in biomedical research fundamentally rely on the application of ethical rules, such as the 3Rs (replacement, reduction and refinement).1–5 In particular, refinement, defined as ‘any decrease in the incidence or severity of inhumane procedures applied to those animals which still have to be used’,6,7 stresses the importance of effective pain relief in animals expected to undergo painful procedures. Although surgical procedures are performed under anaesthesia, only a minority of laboratory animals undergoing surgery receive pain relief to provide postoperative analgesia, 8 including rats.9–11 Non-use of perioperative analgesics is commonly justified by three reasons: concern about interactions between the analgesics and the experimental outcome; concern about potential side-effects from the analgesics themselves; or a failure in recognizing postoperative pain. 10
Morphine has historically been considered the gold standard for opioid analgesia, and is still widely used for the prevention and treatment of severe acute and chronic pain in humans and animals.12,13 Clinically relevant side-effects of morphine (such as pica, respiratory depression) in rodents are very rare when administered at appropriate dose rates for a short duration.13,14 However potential interactions with specific research protocols can still be of concern. For example the immunomodulatory properties of morphine may alter tumour growth profiles,15,16 and so be an issue in cancer studies. Such effects are dose-dependent, and so could be minimized by reducing the dosage required to provide effective analgesia.
Morphine is well absorbed regardless of the route of administration but only 0.1% of morphine administered intravenously penetrates the central nervous system (CNS) at the time of peak plasma concentration in humans. 17 If only 0.1% of the administered dose serves its analgesic purpose, one could therefore assume that up to 99.9% of the total administered amount potentially interacted with the animal model without having the desired analgesic effect. Reasons for the relative poor penetration of morphine into the CNS include its relatively poor lipid solubility and rapid conjugation (metabolism) with glucuronic acid. Morphine is commonly administered neuraxially (by the epidural or intrathecal [IT] routes) in non-rodent species.18–20 In humans, the ratio of potencies between IT and intravenous (IV) routes of administration is 1/200. 21 This suggests that equipotent morphine analgesic effects can be expected with a dose 200 times less when the IT route is chosen, compared with the IV route.
Techniques of neuraxial injection, both IT (i.e. injection in the subarachnoid space) and epidural, have been described in rats.22–25 IT morphine was shown to alleviate pain in rats using nociceptive testing in the Brennan model, 26 or postoperatively.27,28 However, neuraxial administration of morphine is still very rarely used to alleviate postoperative pain in rats. 10 This may be because of the perceived risks and technical difficulties associated with this route of administration. Almost all studies characterizing the analgesic properties of IT morphine in rats involved prior surgical placement of a spinal catheter22,26,27 or undertaken in conscious animals restrained in towels. 28 Both methods are expected to generate stress, increase sources of variation within study data and raise animal welfare concerns. Furthermore, surgical insertion of an indwelling catheter is associated with 5–15% mortality even when carried out by experienced operators, 25 and more than 25% of surviving rats are expected to show some neurological deficit. 29
This report describes a simplified and minimally invasive technique for IT administration of morphine in rats; and compared the antinociceptive properties of morphine administered by this route with subcutaneous (SC) morphine and SC buprenorphine, in anaesthetized rats.
Material and methods
Ethical statement
All procedures were carried out under a project license approved by the Secretary of State for the Home Office (PPL 60/4431), and after animal welfare and ethical review body (AWERB) approval by Newcastle University. All procedures were conducted in accordance with Directive 2010/63/EU and the Animals Scientific Procedures Act (ASPA) 1986.
Animals and husbandry
Twenty-eight female Wistar rats (Charles River, Kent, UK) were used for this study (270 ± 14.7 g, 60.8 ± 3.4 days). The rats were housed in groups of three to five in RC2 cages (North Kent Plastics Company, Erith, UK) with ad libitum access to food and water (SDS RM3 expanded diet; Special Diet Services, Witham, UK), cardboard tubes as enrichment and sawdust bedding. Lighting cycle was on a 12 h basis. Room temperature, humidity and air changes (21 ± 2℃, 55 ± 10% and 15–20 air changes per hour, respectively) were in accordance with Home Office standards. The rats were allowed to acclimatize to the laboratory for seven days prior to the start of the procedure.
Equipment and drugs
A 7 L clear Plexiglas induction chamber (VetTech Solutions Ltd, Congleton, UK) was connected to an anaesthesia machine (Selectatec SM; Cyprane Ltd, Keighley, UK) equipped with oxygen cylinders (BOC, Chester-Le-Street, UK) and a Penlon Sigma Delta sevoflurane anaesthetic vaporizer (Penlon, Abingdon, UK). The gas mixture was delivered to the chamber via a single fresh gas inlet tube located underneath a multi-perforated plastic floor. The scavenging port, located at the top of the chamber, was connected to an active scavenging system (Fluovac; Harvard Apparatus, Cambridge, UK).
Maintenance of anaesthesia was performed using an integrated digital vaporizer designed for small rodents (SomnoSuite; Kent Scientific Corporation, Torrington, CT, USA). The syringe was filled with liquid sevoflurane (Sevoflo®; Abbott, Kent, UK). Oxygen was provided from a cylinder (BOC) to a pump internal to the SomnoSuite. Physiological monitoring of the animals was achieved with a Y-shaped rat-specific pulse-oxymeter and heart rate monitor (SomnoSuite; Kent Scientific Corporation). Body temperature was maintained at 38℃ using a rectal temperature probe coupled to an electric heat pad (SomnoSuite; Kent Scientific Corporation).
The morphine was sterile and free of preservative (morphine sulphate, 1 mg/mL; South Devon Healthcare, Paignton, UK). The sodium chloride (NaCl) solution used was normal (0.9% w/v) and sterile (Braun, Melsungen, Germany). Injectable buprenorphine (Vetergesic, 0.3 mg/mL; Ceva, Bucks, UK) was diluted (1:2) before administration as a positive control.
Thermal nociceptive stimuli were carried out using a handheld miniature thermal probe specifically designed for use in rats (HotProd; Topcat Metrology Ltd, Ely, UK). Baseline probe temperature was set to that of the skin temperature of the rat as measured with an infrared non-contact thermometer (KSC infrared thermometer; Kent Scientific Corporation) at the start of each series of thermal stimulations. The probe was then heated at a rate of 1℃ per second, with a safety cut-off at 55℃. Site and number of thermal nociceptive stimuli are further described below.
Operators
One operator (AAT) was responsible for anaesthetizing, injecting and performing thermal stimulations, as well as euthanizing the rats. This operator was blinded to the substance being injected.
Study design
After induction of anaesthesia and loss of consciousness (absence of righting reflex) in the induction chamber (inspired fraction of sevoflurane [FiSevo] 8% in 4 L/min of oxygen) the rats were connected to the anaesthetic delivery apparatus described above, using a rat-sized low dead space facemask (FiSevo = 2.4% in 250 mL/min of oxygen). This constituted the start of the first wash-in period and is referred to as T0 (start of time recording). The animals were positioned in dorsal recumbency. Abdomen, flanks and sternum were shaved to optimize the contact between the thermal probe and the skin.
Characteristics of the treatment groups.
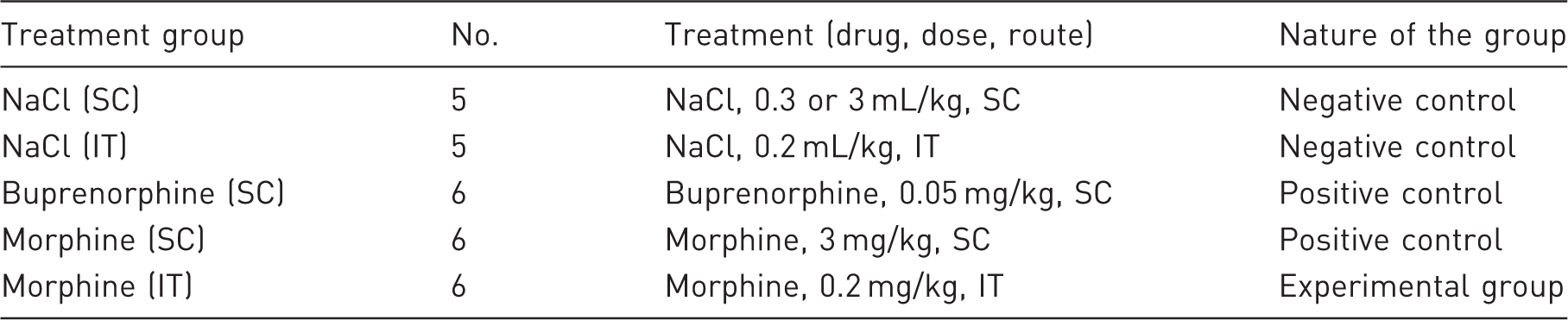
SC: subcutaneously, IT: intrathecally.

Intrathecal injection in rats: positioning, anatomical landmarks and injection site (★).
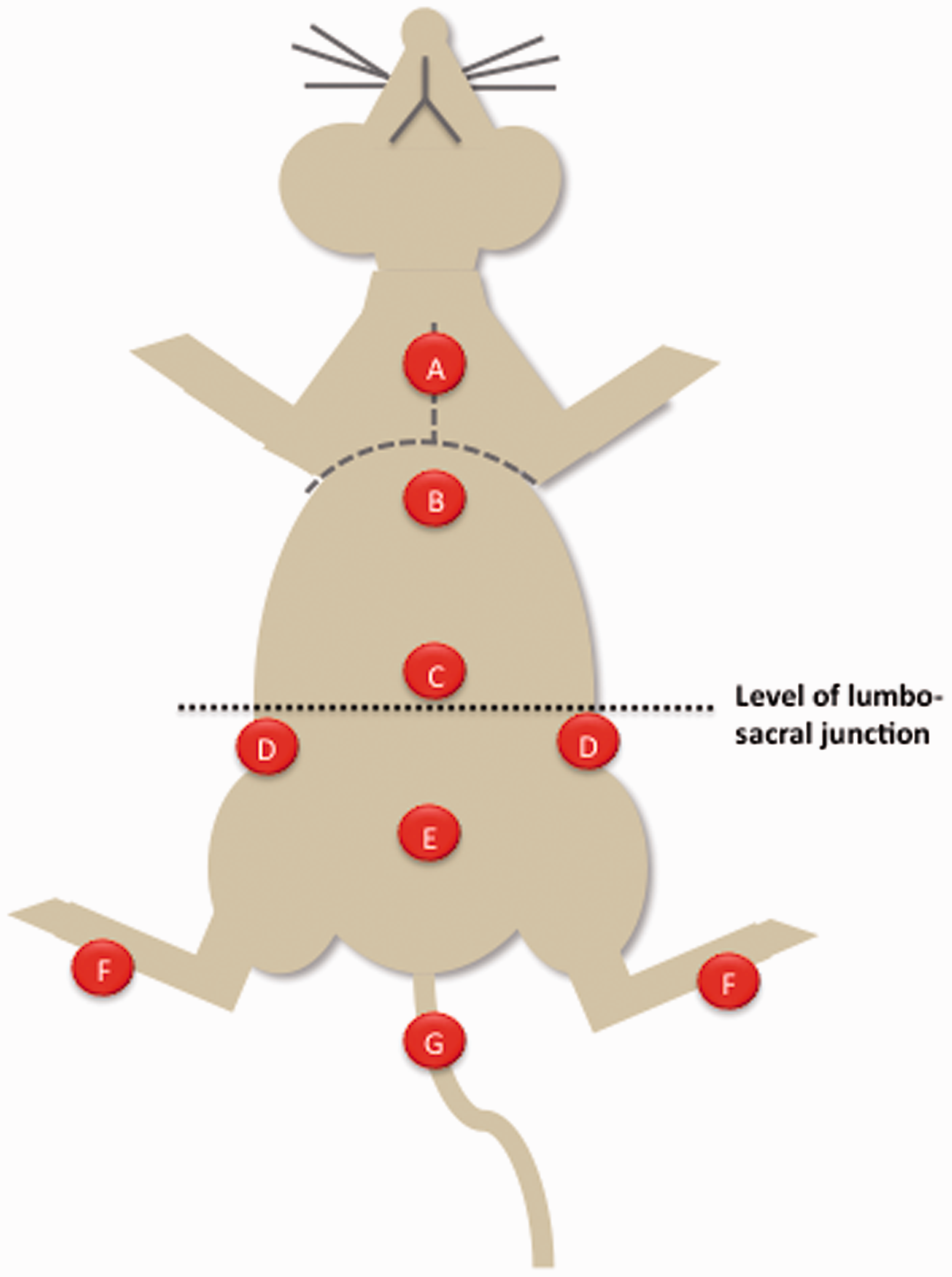
Location of spots for thermal stimulations. A: thorax (mid-sternum); B: cranial abdomen (immediately caudal to the xyphoid appendix); C: mid-abdomen; D: flanks, bilateral; E: caudal abdomen (cranially to the pubic bone); F: hind paws (distal plantar metatarsal area); G: tail (cranioventral aspect of the tail).
At T = 40 min (end of the first wash-in period), the first set of thermal stimulations was performed (Figure 3) at each of the nine locations. The presence or absence of gross purposeful movement (GPM) was recorded in a binomial manner (0 = absence, 1 = presence) following each thermal stimulation. GPM was defined as any of the following movements: retraction of the limb(s), contraction of the abdominal muscles, movement of the tail, or attempt to regain sternal recumbency. Twitch reflex of the skin or tail was not considered to be a GPM. Immediately after the last stimulation, the FiSevo was decreased by 15%, and a wash-in period of 15 min was observed before repetition of thermal stimulation. This cycle was repeated until the rat regained its righting reflex (which rendered further assessments impracticable). The rat was then placed in the induction chamber (FiSevo = 8%) before proceeding to euthanasia using an intraperitoneal injection of 2.5 mL of 20% pentobarbital (Euthatal; Merial, Harlow, UK).
Graphic representation of the study design: thermal stimuli were applied at different FiSevo % after dedicated wash-in periods. Thermal stimuli are applied at locations explicated in Figure 2.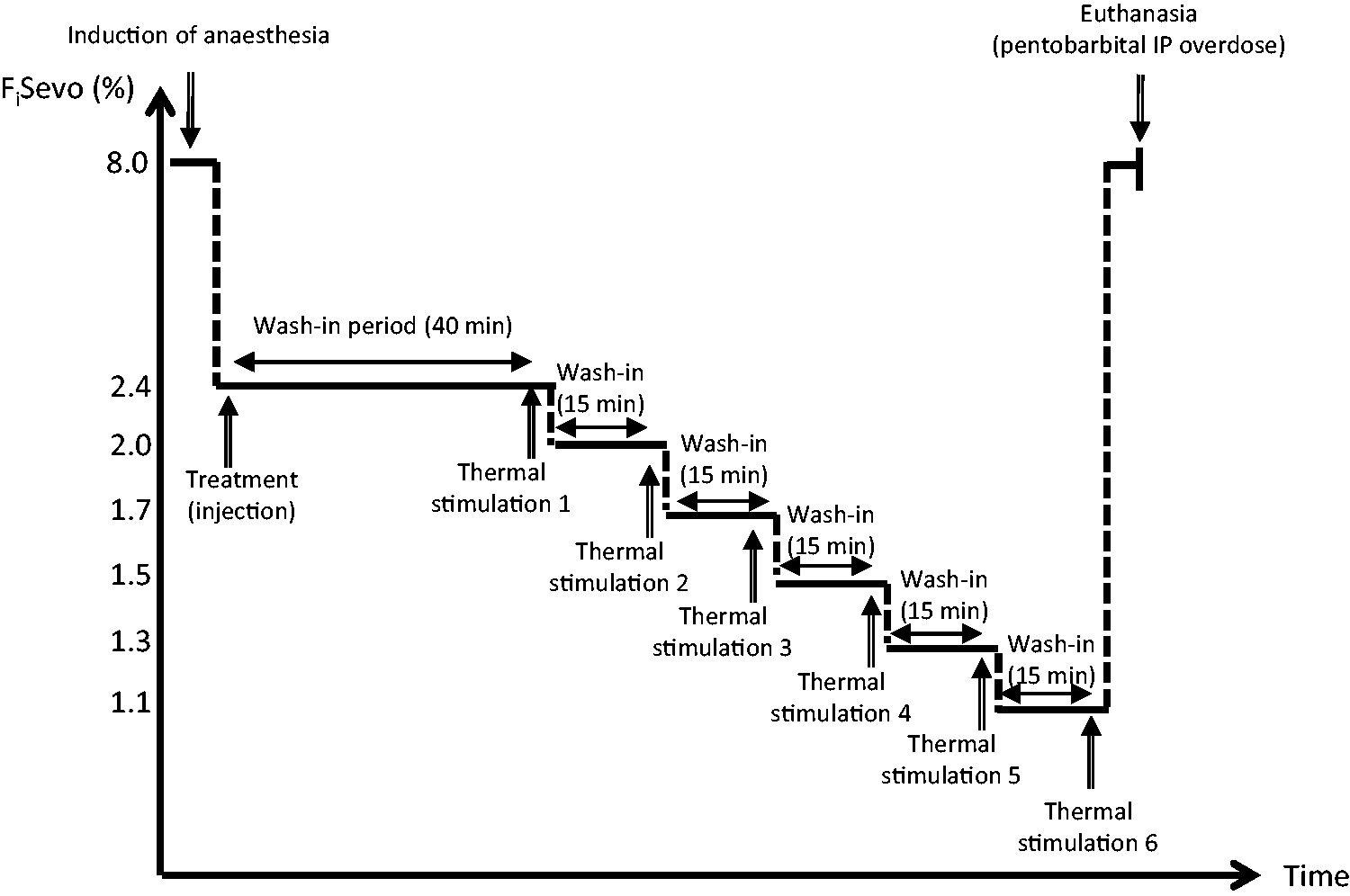
Statistical analysis
A mixed model for repeated measures (i.e. with both fixed and random effects) was used to analyse the results of this study. The random effects accounted for the covariance between repeated measurements. The fixed effects accounted for the difference within treatments, FiSevo, and the stimulated area on the percentage of GPM (SAS v9.3; SAS Institute Inc, Cary, NC, USA). An autoregressive (first order) covariance structure was assumed between repeated measures.
Pairwise comparisons were examined using Bonferroni correction. Differences were considered significant when P < 0.01.
Results
Success of IT injection by direct puncture in L6/S1 intervertebral space
Physical manifestations associated with intrathecal injection in rats (n = 11).

In n = 10 rats, tail twitch was observed during needle placement. In n = 1 rat, tail twitch was observed during drug injection. **The presence of visible blood in the needle hub prompted withdrawal of the needle and repeat attempt of L6/S1 injection. In one rat, the second attempt in L6/S1 was successful. In one rat, the second attempt in L6/S1 was unsuccessful (blood visible in needle hub). A third attempt in L5/L6 was successful.
Treatment effect: GPM and anatomical spread
Number of rats undergoing thermal nociceptive stimulation at each inspired fraction of sevoflurane and time at which the stimuli were started.

FiSevo: inspired fraction of sevoflurane.
Overall, no significant differences were found between the two negative control groups (SC NaCl and IT NaCl) (P = 1.00). The route of administration (SC vs IT) did not in fine influence GPM or sevoflurane requirements. All opioid groups were significantly different to the negative control groups; but the presence/absence of GPM was not overall significantly different between the opioid groups (P = 0.0135 to 1.00).
For all areas of stimulation and all treatment groups, average GPM was not significantly different if FiSevo was 2.4 or 2.0% (P = 0.0497). Results obtained for FiSevo of 2.4% were however different to those obtained at FiSevo of 1.7 and 1.5% (P < 0.0001, both). Across-the-board, it was also verified that symmetrical stimulation locations were equivalent: left/right hind paws; left/right flank were not significantly different (P = 1.00, both). Interestingly, average GPM recorded on hind paws were significantly different to all other areas of stimulation, with the exception of the mid-abdominal location. Conversely, all locations produced significantly different reactions to the left and right hind paw stimulations, but not to each other. There was no effect of the FiSevo on the area of stimulation (P = 0.0478).
There was a significant effect of the area of stimulation on average GPM (P < 0.0001). In each opioid group, thermal stimulus of the left and right hind paws triggered significantly less GPM than in both NaCl groups (P = 0.0003 or <0.0001 in all cases). When rats received SC buprenorphine or IT morphine (but not SC morphine), the mid-abdominal location reacted in a similar way to the hind paws (P = 1.00, all). When rats received IT morphine, stimulation of the cranial abdominal location triggered significantly less GPM than in the SC NaCl group (P = 0.0081), but not in the IT NaCl or any other treatment group (P = 1.00, all). Within each opioid group, each stimulation location resulted in the same degree of purposeful movement. By contrast, in the negative control groups stimulation of the left and right hind paws showed significant differences from most other locations, but were not different from each other.
Due to the small sample size, it was not possible to analyse the effect of the three opioid treatments on each stimulated area in relation to the FiSevo (insufficient degrees of freedom). A graphic representation of the results is shown in Figure 4 for FiSevo = 2.4, 2.0 and 1.7%. When average GPM were considered for each inspired concentration of sevoflurane, negative control groups appeared to react more often than IT morphine and the positive control groups (SC morphine and SC buprenorphine), especially for the hind paws and the cranial abdominal areas. It was not possible to detect the geographical spread of morphine administered intrathecally.
Average gross purposeful movement (absence = 0, presence = 1) per treatment and body location at different inspired fraction of sevoflurane (FiSevo). N = 5–6 for FiSevo 2.4% and 2.0%. N = 3–6 per treatment for FiSevo 1.7%. Significant differences are explained in the text.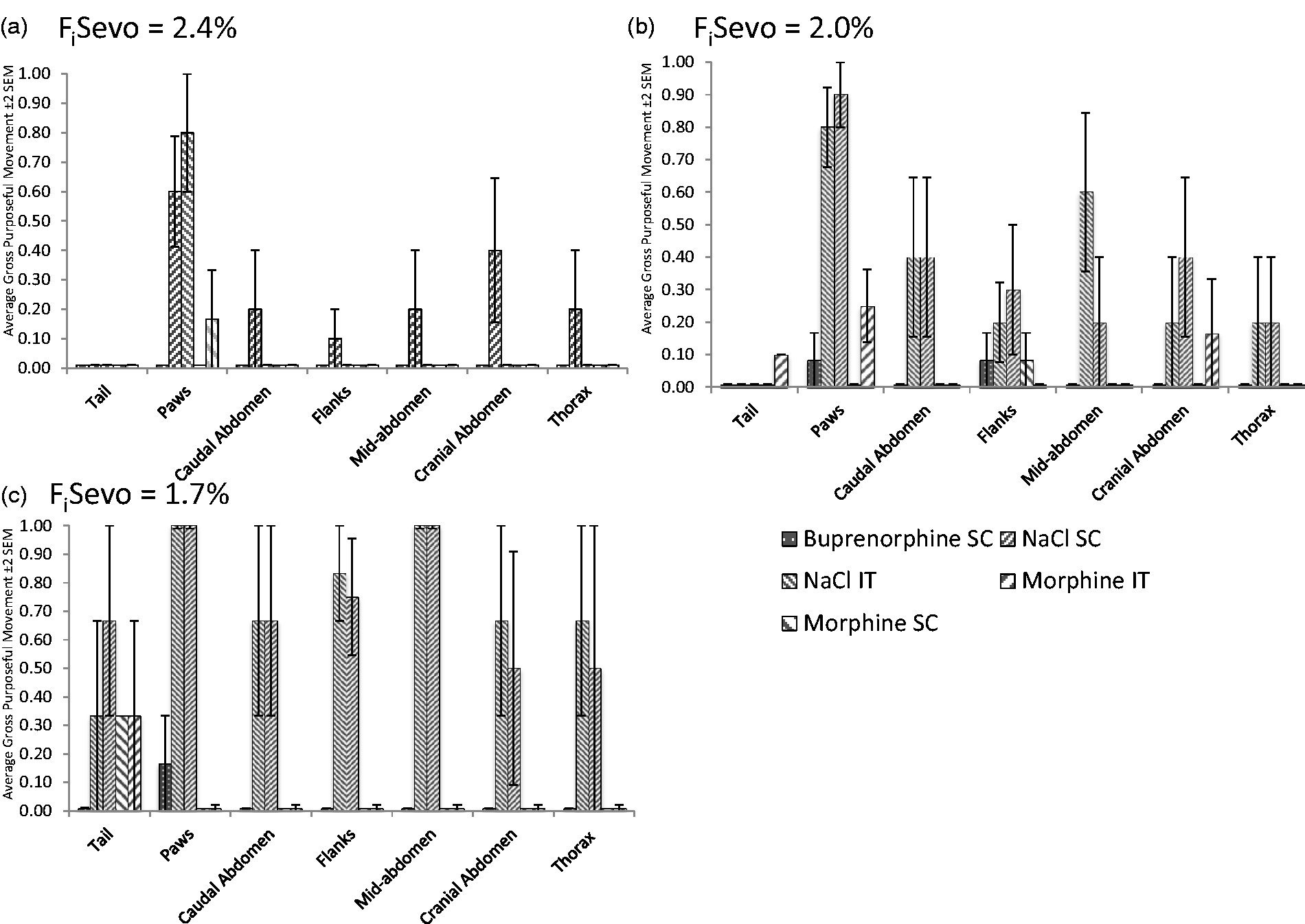
Sevoflurane sparing effect of opioids: number of experimental cycles
The number of experimental cycles completed was not significantly different between groups (P = 0.737, Figure 5), therefore the amount of sevoflurane required in the fresh gas flow to prevent GPM following thermal nociception was not significantly different between treatments. As a result, at the doses used in this study, there was no detectable sevoflurane sparing effect over time between treatments. All rats successfully completed the first two experimental cycles (FiSevo = 2.4 and 2.0% respectively). All rats that received buprenorphine successfully completed the third experimental cycle (FiSevo = 1.7%), as well as 67% of rats that received IT morphine, 50% of rats allocated to the SC morphine group, and 60% of the saline groups.
Graphic representation of the average number of cycle repetition (wash-in period + thermal nociception) underwent by the rats, for each treatment groups. FiSevo 1st cycle = 2.4%; FiSevo 2nd cycle = 2.0%; FiSevo 3rd cycle = 1.7%; FiSevo 4th cycle = 1.5%.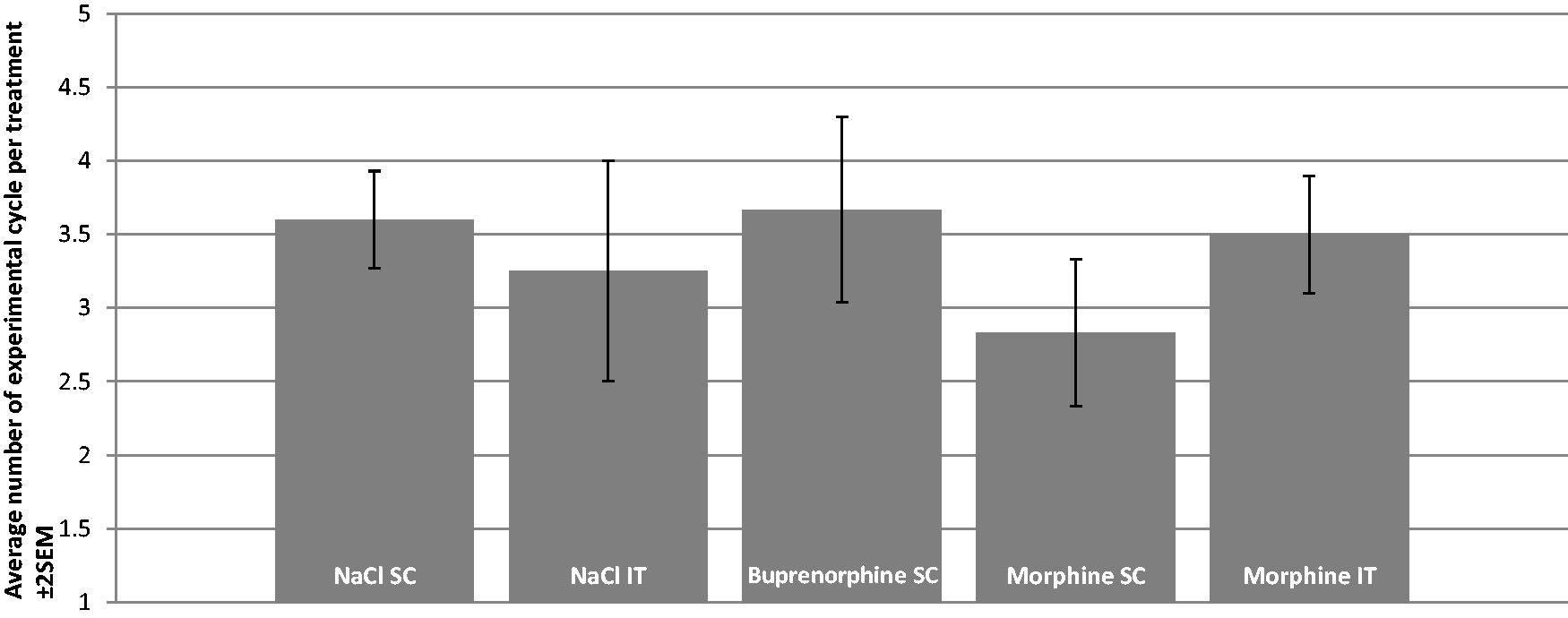
Discussion
The main results from this study suggest that IT injection by direct puncture with a 25 G hypodermic needle can be easily performed, with a success rate at first attempt of 82%. No major visually detectable complications, such as apnoeic episodes or severe respiratory depression, were recorded. Morphine, delivered intrathecally at a dose of 0.2 mg/kg, reduced the frequency of GPM following nociceptive thermal nociception in a way that was not significantly different to either buprenorphine or morphine administered subcutaneously (0.05 mg/kg and 3 mg/kg, respectively). It was however not possible to identify the caudocranial spread of morphine with stimulation of different dermatomes. Furthermore, it was not possible to identify any sevoflurane sparing effect across the different treatment groups.
The apparatus used for thermal nociception in this study was equipped with a rodent-sized probe and a security cut-off at 55℃. Polymodal nociceptors are typically activated from 40℃ (C fibres) and 45℃ (Aδ fibres).30,31 The heating probe used was therefore expected to successfully identify opioid-mediated antinociceptive effects. The heating rate of the thermal probe was slow (1℃/s), selectively activating C fibres and increasing the sensitivity and specificity for detection of antinociceptive properties of morphine whilst minimizing heating of the skin.35,32 The nine locations stimulated corresponded to different spinal dermatomes, from the base of the tail to the sternum. The tail failed to produce reliable GPM across the treatment groups and the experimental cycles unlike the hind paws, which were the most reactive locations. Activation of C fibres with a given thermal probe varies depending on skin properties such as reflectance, diffusivity and initial temperature,30,33,34 and this may account for these differences on the tail. Other locations such as the flanks, the abdominal and thoracic areas produced more uniform responses on anaesthetized rats. Despite these limitations, morphine, when injected intrathecally at a dose of 0.2 mg/kg, was shown to have similar antinociceptive properties, as assessed by a reduction in the frequency of GPM, as buprenorphine, the most commonly used opioid for rodent analgesia,9,10 and morphine (3 mg/kg) injected subcutaneously.
The onset of action of IT morphine in rats is 20–30 min. 22 IT morphine is expected to provide shorter lasting analgesia in rats (i.e. approximately 120 min)26,27,35,36 compared with most species.35,37–40 Although the present study was not designed to measure onset/duration of IT morphine, our results (Table 3 and Figure 4) suggest that onset and duration of action were compatible with previous findings.
Stimulating different dermatomes along the spinal axis did not enable the detection of caudocranial spread of IT morphine. Multiple factors influence subarachnoid spread: baricity, volume, concentration and lipophilicity of the solution.41,42 The dose and volume of injection chosen for this study (i.e. average 54 µg of morphine per rat) was selected after pilot studies carried out in our laboratory as being the highest dose deliverable spinally without inducing observable side-effects (unpublished data). This dose is comparable to the highest doses used by Yaksh,22,23 and is substantially higher than doses used in recent fundamental studies.36,43 The injected volume (i.e. 0.05 mL per rat) may have varied slightly between animals due to the simple apparatus used (disposable hypodermic needles and syringes), but sits within the commonly quoted range.23,36,44 Morphine has relatively low liposolubility compared with other opioids. 45 While all opioids migrate within the CSF and the neural tissue, lipophilic opioids are expected to display a narrower band of spinal analgesia than hydrophilic ones. Morphine is therefore more likely to lose its anatomical specificity shortly after IT injection. 21 Additionally, long wash-in periods may also explain why it was not possible, in our study, to delimit anatomical dermatomes showing antinociception after IT morphine injection.
The aim of the present study was to document the antinociceptive properties of a refined method of IT morphine injection to assess the potential for its use in providing postoperative pain relief in rats used in biomedical research. It is therefore important that the apparatus and materials used are easily available and relatively inexpensive (disposable hypodermic needles and syringes). Despite meticulous preparation and manipulation, this apparatus does not allow for precise measurement of the volume injected, in comparison to use of Hamilton glass syringes. It is important to note that preservative-free morphine is recommended for IT injections in order to prevent toxic neurological lesions.46,47 Use of the only commercial preservative-free morphine formulation available locally (1 mg/mL) resulted in relatively large volumes of injection, both subcutaneously (average 0.81 mL/rat) and intrathecally (average 0.05 mL/rat). While this did not result in any side-effects in adult-sized rats, a more concentrated preservative-free morphine formulation, if available, might be preferable. The maximal volume to be safely injected intrathecally depends on factors such as physicochemical properties of the solution and speed of injection. 48 Injecting 1 mL of solution (pH = 7, 300 mOsm/kg H2O) at 1 mL/min can be fatal in rats. 48 IT volumes injected in our study remained well below 1 mL. The speed of injection was approximately 1 mL/min and was achieved without any complications.
In this preliminary study opioids failed to decrease the FiSevo required to prevent rats from regaining their righting reflex. This finding was unexpected. Morphine and buprenorphine have been reported to decrease the minimal alveolar concentration (MAC) of inhalational agents by up to 50%.49,50 The present study was however not an MAC reduction study. Calculation of MAC values for inhalational agents usually requires the use of validated nociceptive stimuli 51 and endotracheal samples to determine the end-tidal concentration of volatile anaesthetics. The rats in the present study were allowed to ventilate spontaneously and despite the use of a high-precision digital vaporizer, our methodology did not meet the requirements for MAC determination. This preliminary study was aimed at characterizing antinociceptive properties of intrathecally-injected morphine. Thermal nociception was selected as a means of assessing the antinociceptive properties of opioids, and was successful at doing so. A small sample size (n = 5–6) was used. A sample size calculation based on effect size and standard deviations in our populations (www.sample-size.net, power 80%, α = 0.05; β = 0.20) suggested that over 300 rats per group would be needed to document a significant reduction in sevoflurane requirements between IT NaCl and IT morphine groups. We did not attempt this for obvious ethical reasons.
All the rats used in this study were female. Gender influences nociception and analgesia. 52 Females are generally more sensitive to noxious stimuli whereas males are generally more sensitive to mu-opioid receptor agonists (e.g. morphine). 53 Given the preliminary nature of the study, it was decided to keep the sample size to the lowest relevant size and to work with single-sex groups. Consequently, caution should be applied when extrapolating our results to male rats.
In conclusion, this preliminary study describes a refined technique for administering morphine intrathecally in anaesthetized rats using inexpensive and readily available materials. The antinociceptive properties of morphine injected, using this technique, were similar to those of systemic buprenorphine, without surgical intervention or stressful handling. Using the IT route of administration, the amount of opioid administered is markedly reduced, thus minimizing the potential interactions of non-analgesic opioid effects with research protocols, whilst providing analgesia. Further studies are warranted to investigate the analgesic properties of morphine injected using such techniques in research models such as pelvic limb orthopaedic surgery or urogenital procedures.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
