Abstract
This study aims to evaluate the impact of adding different items in individually ventilated rat cages on the animal's activity, cardiovascular parameters and faecal stress indicators. The following three cage items made of aspen were compared: a cross made of two intersecting boards, a similar cross where drilled holes were loaded with food pellets (restricted feeding) and a rectangular tube. Male rats of the strains BN and F344 (n = 12) were housed in groups of three; one rat in each group was implanted with a telemetric transponder to measure mean arterial pressure (MAP) and heart rate (HR). In a crossover design, each group spent 14 days with each type of cage furniture, thereafter faecal pellets were collected for faecal analyses. The means of activity and means and coefficient of variation for MAP and HR were calculated for days 2, 6, 10 and 14. As a way of determining which of the statistically significant MAP and HR mean changes were biologically meaningful, the night–day differences of the controls on day 14 were used. Both board types lowered MAP of F344 rats; hence dividing walls seem beneficial for F344 welfare. None of the MAP or HR differences in BN rats were biologically significant. No statistically significant differences in faecal corticosterone or IgA excretion were detected. In conclusion, provision of general recommendations with respect to cage furniture for rat cages is complicated because there is a clear genetic component involved in how animals respond to these structures.
Compliance with the new European regulations mandates the use of nesting material or nest boxes in rodent cages. 1,2 These regulations are based on clear scientific, primarily ethological, rationale. 3,4 Regulatory compliance is easily achieved by adding structures into the cage, but not all of these may function as housing refinements, some may have no value and some may even have a negative refinement or reduction impact. 5–9 The potential value of structures to be placed into rodent cages requires systematic studies to assess and compare the value of any new item or combination of items.
Regulatory texts have a tendency to generalize research findings. Indeed, the current European guidelines on laboratory animal housing and care are identical for all rodent species; and within a species they have to be implemented irrespective of animal strain, and caging system in use. 1,2 One widely used type of housing is the individually ventilated caging (IVC) system, which has been shown to change the physical environment inside the cages compared with that of open cages in terms of illumination, acoustic environment, temperature and relative humidity (RH). 10
When animals are stressed, their attempts to cope with their situation may be evaluated by the activity of the sympathetic nervous system, the hypothalamic–pituitary–adrenal (HPA) axis activity and their behaviour. 11 Common feature of all stress assessment methods is the requirement to sample by using non-disturbing and non-invasive methods. Moreover, single indicators may be misleading, and an evaluation of improvements in cage environment often requires an assessment of a combination of indicators. 12,13
Radiotelemetry is a method for measuring physiological parameters, such as blood pressure and heart rate (HR), in laboratory rodents which allows the animals to move freely during recording, making any restraint unnecessary. Indeed, the values of HR and blood pressure are considerably lower in animals implanted with a radiotelemetry transmitter than the corresponding values obtained with other methods. 14 The effect of the different combinations of added cage items in rats is an active study area, but telemetry assessments have rarely been incorporated into these experiments. The study of Sharp et al. 15 using Sprague-Dawley (SD) and spontaneously hypertensive (SH) male rats showed that their multifaceted enrichment programme reduced HR in both dark and light periods and increased activity during the afternoons for SH rats.
Corticosteroids and their metabolites are excreted into urine and faeces, both of which can be regarded as time-integrated indices of stress. Unlike serum sampling, samples of both urine and faeces can be obtained non-invasively though the changes in the concentrations in the circulation occur with a delay of approximately 10 h in urine and 16 h in faeces. 16 About 20% of the recovered metabolites in rats appear in urine and 80% in faeces. 16,17 In a similar manner to serum corticosterone, 18 concentrations of its metabolites in faeces follow a diurnal rhythm; but conversely to the serum corticosterone, the highest concentrations of the faecal corticosterone metabolites appear to be present in the morning samples. 16,19 If stress persists for a longer time, it may also cause immunosuppression. This can be assessed by quantification of secretory immunoglobulin A (IgA) in faeces. Faecal IgA excretion also exhibits a diurnal variation, but the values detected in morning and evening samples have been somewhat controversial. 19–21 Faecal samples for both assays are easy to collect from the cage without disturbing the animals, enabling long-term, longitudinal studies. 22
Rats are crepuscular and nocturnal animals and their blood pressure and HR are higher during the night than during the day, but there are major differences between different stocks and strains. 15,23–26 In the present study, two rat strains, F344 and BN, were studied in order to elucidate whether there would be any genetic contribution to the variation in the results, and to achieve better applicability within the species. 27 These strains were chosen because they differ in various aspects of their physiology, including locomotor activity level, diurnal rhythm of activity, 25,28 blood pressure, HR 25 and levels of circulating corticosterone. 29,30
Laboratory rodents are commonly fed ad libitum, e.g. food is available all the time, a feeding regime known to cause obesity, increase the incidence of diseases and shorten lifespan in rats. 31–33 On the other hand, restrictive feeding may be difficult to combine with the recommended group housing of rats, 1,2 as there is no practical or effective way to evenly restrict the food intake of all individuals within the group. Rats eat predominantly during the dark period when the food is available ad libitum 34–36 and when the restricted feeding is timed in the light phase in rats, it may impair both natural feeding patterns and gastrointestinal physiology. The diet board, where the access to food requires rats to gnaw wood, has been shown to significantly reduce the weight gain in rats with 15–18% less food being eaten. 28
We hypothesized that a refinement value could be detected via changes in mean arterial pressure (MAP), HR, faecal corticosteroid metabolites and faecal IgA, and a reduction value by changes in the coefficients of variation (CV) of these parameters, this being attributable to various cage items and restricted feeding. This study was designed to evaluate the impact of dividing aspen walls with or without restricted feeding, as well as an aspen tube on laboratory rats using activity, circulatory parameters and faecal stress indicators to determine whether two different rat strains would display variation in their responses, and whether there would be habituation to the items.
Materials and methods
The study was carried out in the Laboratory Animal Centre, University of Helsinki. The protocol of the study was reviewed and approved by the Animal Ethics Committee of the University of Helsinki.
Animals
A total of 12 BN (BN/RijHsd) and 12 Fischer344 (F344/NHsd) male rats (Harlan, Horst, The Netherlands) were used in this study. The rats were 25 weeks old and weighed 280–370 g (BN) or 350–460 g (F344) at the beginning of the study. Upon arrival from the commercial breeder at the age of 10 weeks, the animals were housed in IVCs with the same bedding material and the same social groups as during the study.
Animal housing and care
All rats were housed in the same room in groups of three rats per cage in polysulphone IVCs (Tecniplast, Buguggiate, Italy). The cage type used was 1500U Eurostandard IV S (44.5 × 33.5 × 21.0 cm – floor area 1500 cm2) with a solid bottom and IVC-double lids. The cage floor was covered with 3.0 L aspen chip bedding (size 4 × 4 × 1 mm, 4HP, Tapvei Oy, Kaavi, Finland). The cages were changed once weekly. The room temperature was 21.2 ± 0.3°C (mean ± SD) and the RH was 53.5 ± 7.7%, but actual values inside the IVCs were 1–4°C and up to 6% RH higher. Artificial lighting with fluorescent tubes (light colour warm white) was on from 06:00 to 18:00 and the light intensity in cages 1 m above the floor was 6–9 lx. The sound level with R-weighting (adjusted for the hearing sensitivity of rats), in empty IVC cages, was 20–25 dB(R), with the corresponding A-weighting (adjusted for the hearing sensitivity of humans) being 45–47 dB(A). 37 Tap water was provided in polycarbonate bottles, changed once a week and refilled once in between. Feeding was restricted or ad libitum depending on treatment as outlined below. For a more thorough description, see Kemppinen et al. 10
Cage furniture and study design
Animals were housed in permanent groups of three. The experiment utilized a crossover design with two-week periods and a rotational order between control and the following three cage furniture items (Figure 1):
Cage without furniture (control); Cage equipped with a cross made of intersecting two aspen boards (34.0 × 14.7 × 3.2 and 21.1 × 14.7 × 3.2 cm, plain board); The same as (2) but holes (12 mm) were drilled into the boards and then were loaded snugly with food pellets; rats had to gnaw wood to gain access to the food, no other food source was available (diet board); Cage provided with a rectangular aspen tube (20.0 × 12.0 × 12.0 cm, external dimensions). Pictures of the cage items used: the diet board above, the plain board on the left below and the tube on the right below. Both rat strains (BN and F344) had an added furniture item for two weeks

The order of the cage items was arranged at random, and the first item in each group was randomly allocated. The illustration of the item order can be seen elsewhere. 38
The items were made of aspen because this was the same material as the bedding and they can be assumed to have the same volatile compound emissions, in this case especially the absence of α- and β-pinenes, 39 but also to be able to endure several bouts of sanitation. 40 The day when the rats were introduced to the new cage furniture was designated day 1 in all study periods. Rats were changed to clean cages on day 8 at noon during every period.
Irradiated (25 kGy) pelleted feed (2016 Global Rodent Maintenance, Harlan Teklad, Bicester, UK) was available to the three groups (control, plain board, tube) ad libitum, while the diet board group had the food pellets in holes drilled into the aspen board. When fed with the diet board, rats have been shown to eat 12–18% less and gain weight slower. 28
Surgical procedure
Eight rats were implanted with a radiotelemetry transmitter (TA11PA-C40; Data Sciences International, St Paul, MN, USA). Anaesthesia was induced with a mixture of fentanyl/fluanisone (Hypnorm®, Janssen Pharmaceutica, Beerse, Belgium; one part), midazolam (Dormicum®, Hoffmann-La Roche AG, Grenzach-Wyhlen, Germany; one part and two parts of sterile water) at the doses of 0.15–0.20 mL/100 g subcutaneously. The ventral abdomen area was shaved and scrubbed with MediScrub®, 1% triclosan solution (Medichem International, Sevenoaks, UK) and disinfected with chlorhexidine solution (Klorohexol® 5 mg/mL, Leiras, Turku, Finland). Ocular lubricant (Viscotears®, Novartis Healthcare, Copenhagen, Denmark) was applied to both eyes. After a small incision on the ventral abdominal midline had been made, the presoaked transmitter was placed into the abdominal cavity and the catheter into the abdominal aorta. The transmitter was secured into the abdominal wall with 4–0 Ethicon® Ethilon® II sutures (Johnson & Johnson Intl, St Stevens-Woluwe, Belgium) and the incision was closed with 5–0 Ethicon® Vicryl® (Johnson & Johnson Intl). The surgery procedure lasted about 20–30 min. Postoperative pain alleviation was carried out with 0.01–0.05 mg/kg subcutaneous buprenorphine (Temgesic®; Schering-Plough Europe, Brussels, Belgium) twice a day and once a day a dose of 5 mg/kg subcutaneous carprofen (Rimadyl®; Vericore Ltd, Dundee, UK) for at least three days, and parenteral fluids were given for three days. On these three days, the implanted rats were housed alone and then placed back into their home cages. The pain medication for each rat was titrated against the individual response. The animals were allowed to recover for 10 days before the experiment was started.
Sampling
Mean blood pressure, HR and locomotor activities were recorded telemetrically every 75 s for 24 h on days 2, 6, 10 and 14. In order to conduct the locomotor activity measurements, the telemetric receiver had two antennae, located at two sides, like an x- and y-axis, and the receiver detected the difference in signal strength when the animal moved in relation to these antennae.
At the end of each period, the rats were housed singly for 6 h (06:00–12:00) and all faecal pellets voided from each individual were collected and frozen (−18°C). All samples were analysed individually.
Faecal corticosterone and faecal IgA quantification
The extraction of both corticosterone and IgA was performed as described by Pihl and Hau. 19 The corticosterone enzyme-linked immunosorbent assay was performed using a commercial corticosterone kit (DRG Diagnostics, Marburg, Germany) using the manufacturer's instruction manual. The quantification of IgA was performed using the assay also described by Pihl and Hau 19 and reagents were obtained from AbD Serotec (Kidlington, Oxfordshire, UK; purified rat IgA standard, PRP01), concentrations 0–1000 ng/mL; coating antibody (mouse anti rat IgA heavy chain, MCA191); and detection antibody (mouse anti rat kappa/lambda light chain: HRP, MCA1296P; diluted 1:1000).
Data processing and statistical analysis
The number of animals needed in the study was estimated with the resource equation method. 27 The cage was used as an experimental unit, and with the crossover design used, this resulted in 12 degrees of freedom for error per strain, well within the optimum range, i.e. 10–20. Means and CV for MAP, HR and mean for locomotor activity were calculated in the light and dark periods for days 2, 6, 10 and 14 in each period for 30 min intervals from the raw data. The 12 h mean values for MAP and HR of the controls were subtracted from those of plain board, diet board and tube groups for the same days, and CV values of four furniture items were used as such. Mixed-model repeated measures analysis of variance (ANOVA) on SPSS for Windows (version 14.0) was used, followed by Bonferroni correction for post hoc comparisons. For locomotor activity, furniture item was used as the main effect and age as covariate. For MAP, HR and all CVs, furniture item was used as the main effect and age and activity as covariates. Analogous calculations were carried out to compare data within a furniture item between the days; the day was used as the main effect instead of furniture item. We assumed that for a cardiovascular effect of housing to be biologically meaningful, it would have to be at least as large as the difference between night and day values. Therefore, mean night–day difference was calculated from day 14 data for the control group of both rat strains.
Significant CV differences between groups were processed further to point estimates [=(CV1/CV2) 2 ]. Significance was set at P < 0.05 for mean value calculations, but at P<0.01 for CV statistics to increase the reliability of the findings.
The faecal corticosteroids and faecal IgA results of three rats from the same cage were averaged and calculated with repeated measures mixed-model ANOVA using the age as a covariate.
Results
F344 rats were far more active than the BN rats during the dark phase, whereas the activity during the light phase was similar in these strains (Figure 2). In the dark phase, F344 rats were significantly (P < 0.01) more active in cages with diet boards than in the control cage on day 6, while on day 10, they exhibited significantly (P < 0.05) higher activity with the tube compared with those living with the diet board and controls (Figure 2a), but these differences disappeared by the end of the two-week period. The BN rats exhibited no differences in activity between the cage furniture items (Figure 2b).
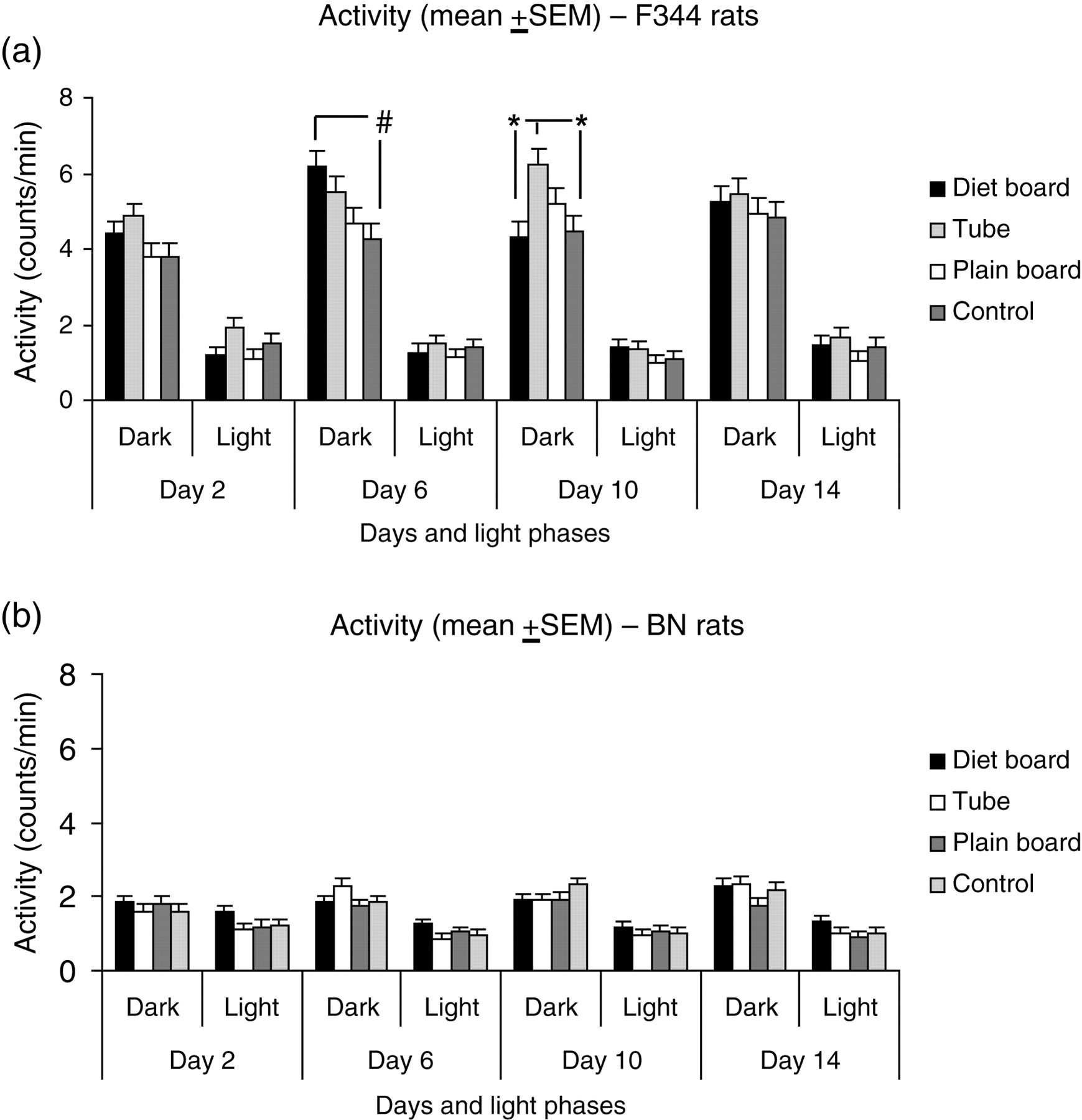
Activity (SEM) of F344 (a) and BN (b) rats with different cage items and controls during the dark and light periods in individually ventilated cages (IVC). *P < 0.05; # P < 0.01. Number of experimental units within a strain = 16
When calculated from data of both strains and all the groups, F344 rats displayed higher values of MAP and HR than BN rats throughout the study. In both dark and light phases, MAP exhibited a significant (P < 0.001) group × strain interaction on all observation days. HR showed a significant (P < 0.05) group × strain interaction during the dark phase from day 6 onwards. Due to the multiple interactions encountered, the following results will be presented separately for both strains and for each light phase.
Effect of housing item
F344 rats
MAP and HR night–day differences for F344 controls were 5 (±2) mmHg and 52 (±13) BPM at day 14. Throughout the two-week periods, both during the dark and light phases, F344 rats living with the tube exhibited increased MAP, while the other two items decreased MAP compared with the baseline of the controls; all these comparisons being statistically significant (P < 0.001). When housed with the plain board, rats had lower MAP on day 6 light phase (P < 0.001), on day 10, dark phase (P < 0.01) and on day 14 all throughout the 24 h (P < 0.01–0.05). In most cases, these differences were also biologically significant, i.e. exceeded 5 mmHg (Figure 3a). HR changes are shown in Figure 3b with statistical significances, but none of the differences came even close to the HR night–day difference of the F344 control group, i.e. 52 BPM.
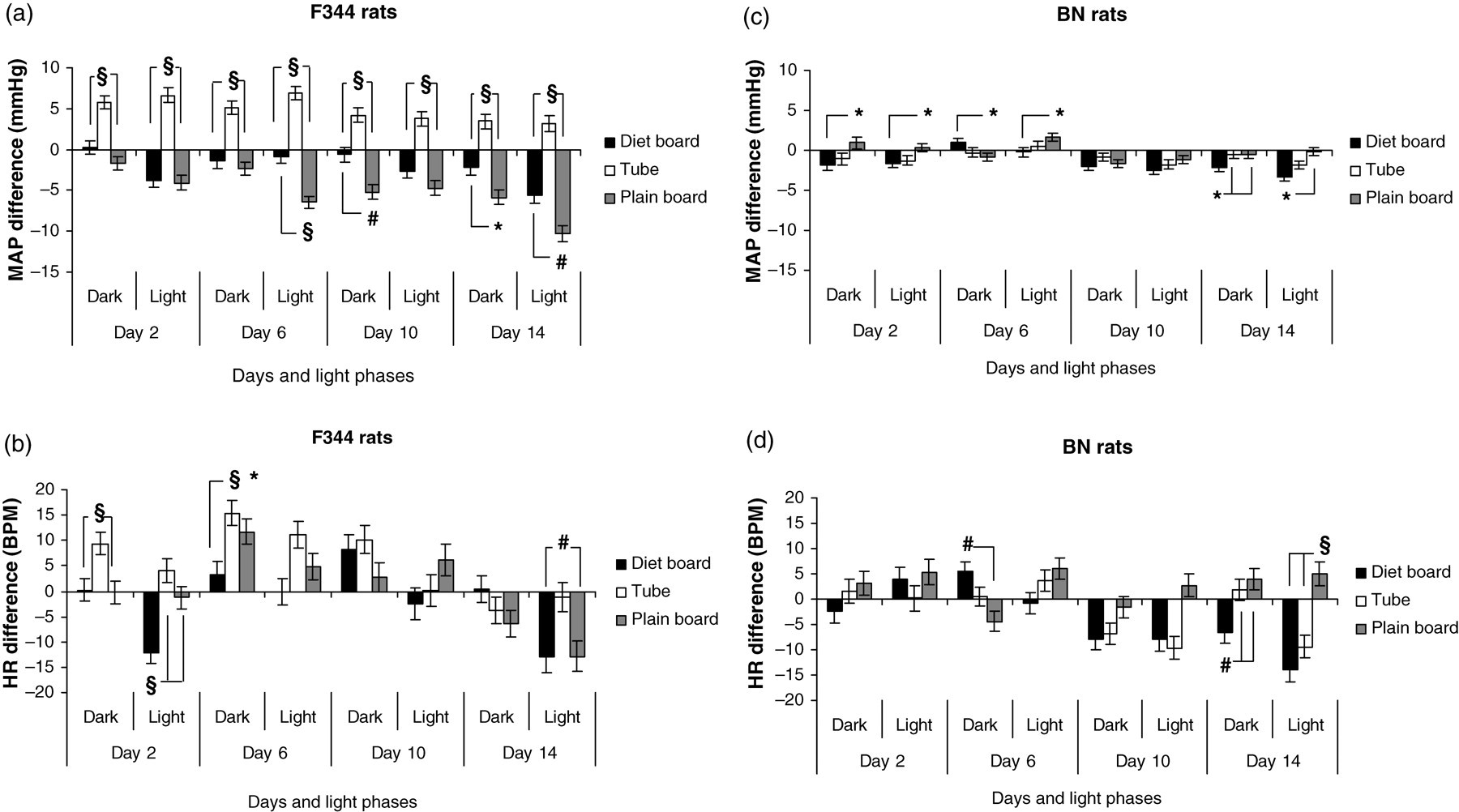
The mean arterial pressure (MAP) and heart rate (HR) differences (SEM) of F344 (a and b) and BN (c and d) rats to controls with three cage items during the dark and light periods in individually ventilated cages (IVC). BPM: beats per min; *P < 0.05; # P < 0.01; § P < 0.001. Number of experimental units = 12
In F344 rats on day 2, the CV of MAP was significantly higher when the animals were housed with the plain board than with the two other items and the control cages during both the dark (all P < 0.01) and the light (all P < 0.001) phases. On day 10 in the light, the CV of HR was significantly lower in the diet board animals than in the control cages (P < 0.01). On day 14, the CV of MAP in the light phase was significantly (P < 0.01) higher with the plain board compared with the tube (P < 0.01), while CV of HR in the dark phase was significantly lower with the plain board cages than in the tube cages (P < 0.01). All these results are shown graphically in Figures 4a and b and in table format with the corresponding point estimates in Table 1.
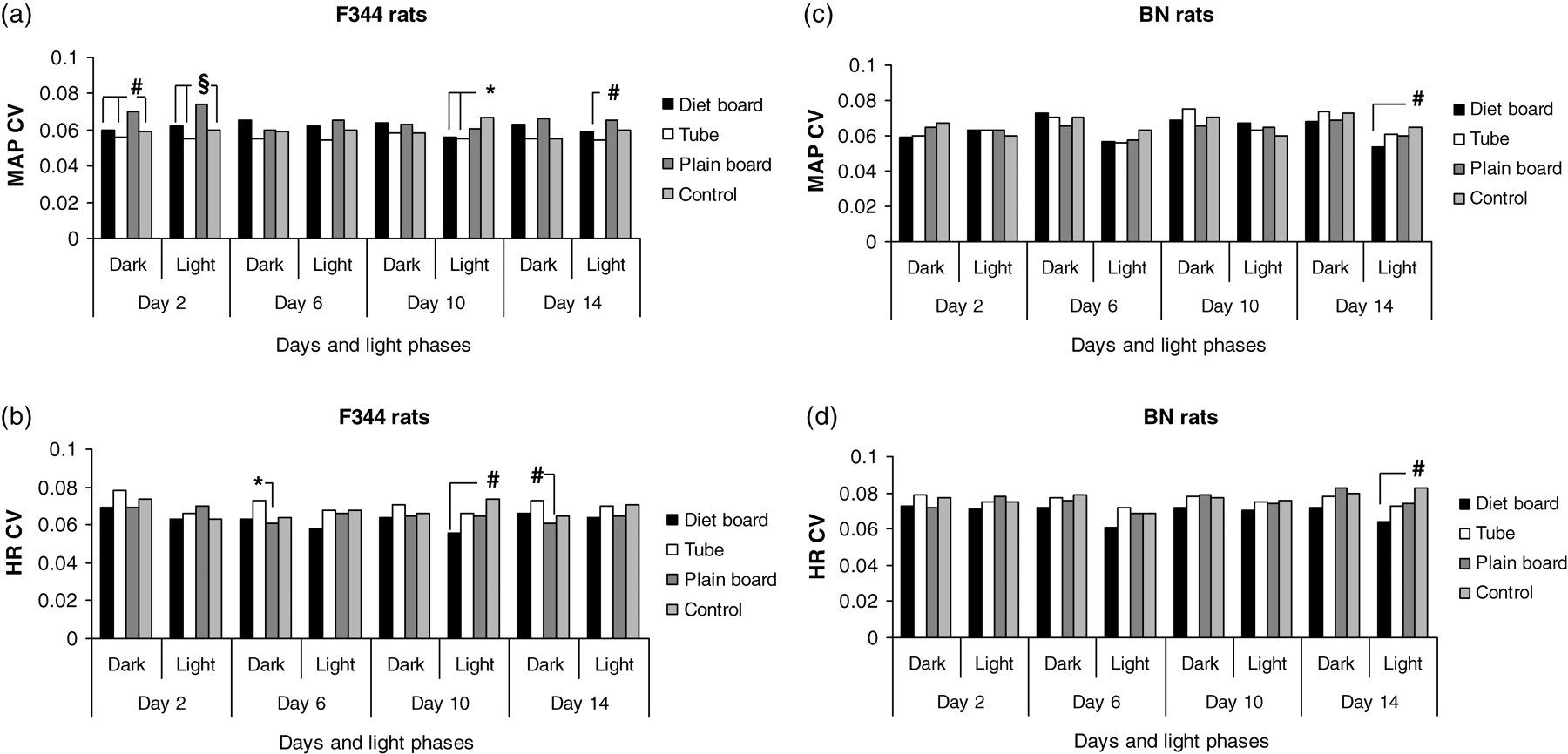
Coefficient of variation (CV) of the mean arterial pressure (MAP) and heart rate (HR) for F344 (a and b) and BN (c and d) rats with three cage items and control groups during the dark and light periods in individually ventilated cages (IVC). *P < 0.05; # P < 0.01; § P < 0.001. Number of experimental units = 16
P values for significant mean arterial pressure (MAP) and heart rate (HR) coefficient of variation (CV) comparisons between the groups and corresponding point estimates (PE) for both F344 and BN rats and both light phases on the observation days
Non-significant comparisons are not shown in the table. NS = not significant
BN rats
The night–day differences in MAP and HR for BN controls were 3 (±1) mmHg and 25 (±7) BPM at day 14. In BN rats, there were few statistically significant changes in MAP and HR between the different cage items. Overall MAP was significantly (P < 0.001–0.05) lower in the diet board cages than in the plain board cages early in the period; this became less and finally it stabilized as the lowest of all the groups at the end of the two-week period (Figure 3c). Nonetheless, it was only by day 14 that the MAP decrement of the diet board, as compared with the plain board, exceeded the limiting night–day difference. The relationship of the HR between the cage furniture items was rather similar (Figure 3d), but in analogy to F344 rats, none of the differences reached a biologically significant value. In BN rats on day 14 in the light phase, the CVs of the MAP and HR were significantly lower in the diet board cages than in the control cages (P < 0.01). All possible comparisons are illustrated graphically in Figures 4c and d and those statistically significant in table format with the corresponding point estimates in Table 1.
Between days comparison
When the days were compared within the furniture item during the two-week period in both strains, there was a decreasing trend both in MAP and HR compared with the control baseline, and although between-day differences were small, they were in most cases statistically significant (P < 0.001–0.05), particularly in the F344 rats (Figure 5).
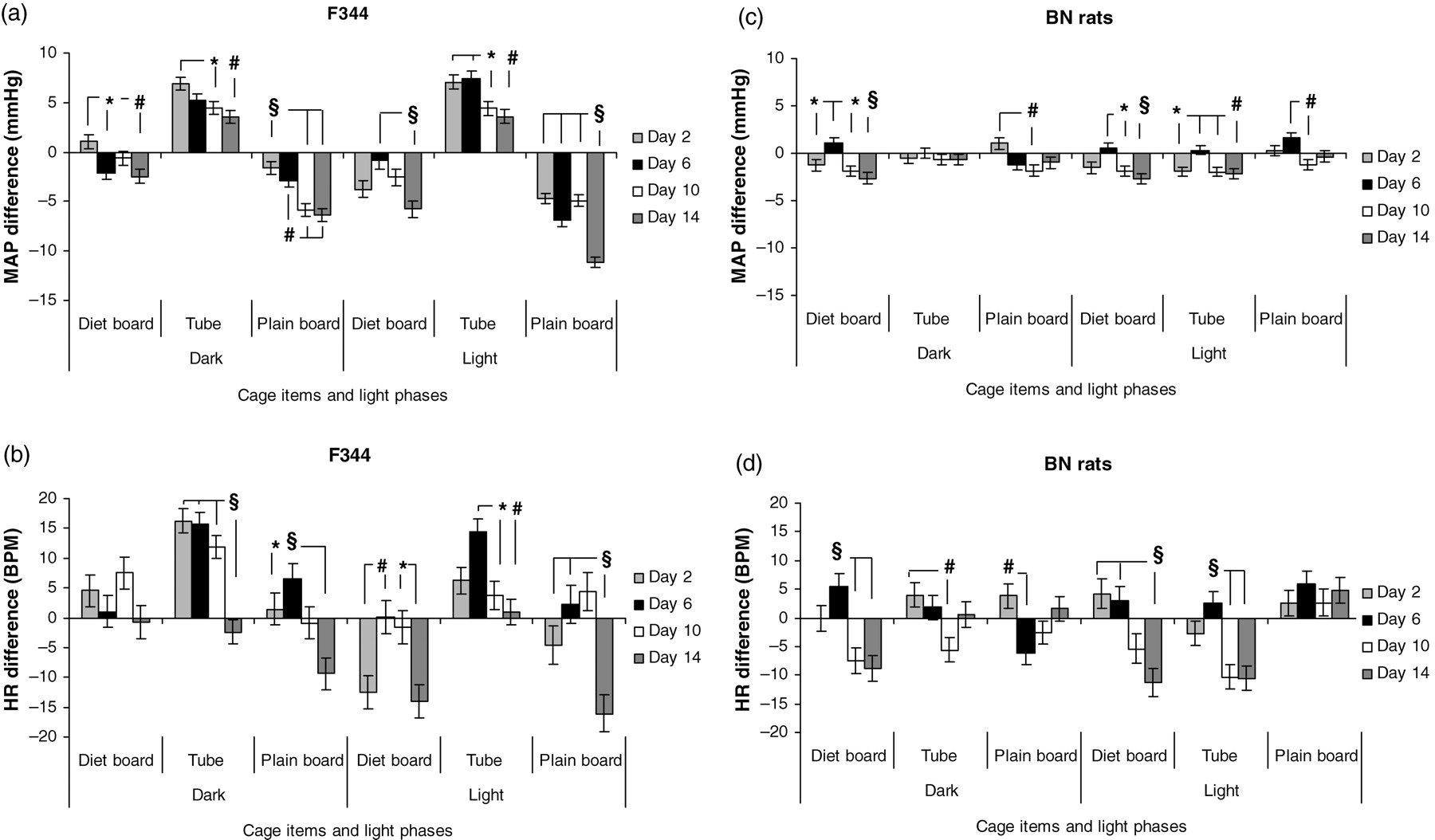
The mean arterial pressure (MAP) and heart rate (HR) differences (SEM) to controls of F344 (a and b) and BN (c and d) rats with three cage items during the dark and light periods in individually ventilated cages (IVC). BPM: beats per min; *P < 0.05; # P < 0.01; § P < 0.001. Number of experimental units = 16
Faecal assays
There was a large variation in the number of faecal pellets voided, from none up to more than 10 pellets from one animal. No group × strain interaction was seen, and none of the two strains exhibited any between-furniture item differences in the faecal corticosterone or in faecal IgA (results only in F344 rats) excreted (Figure 6).
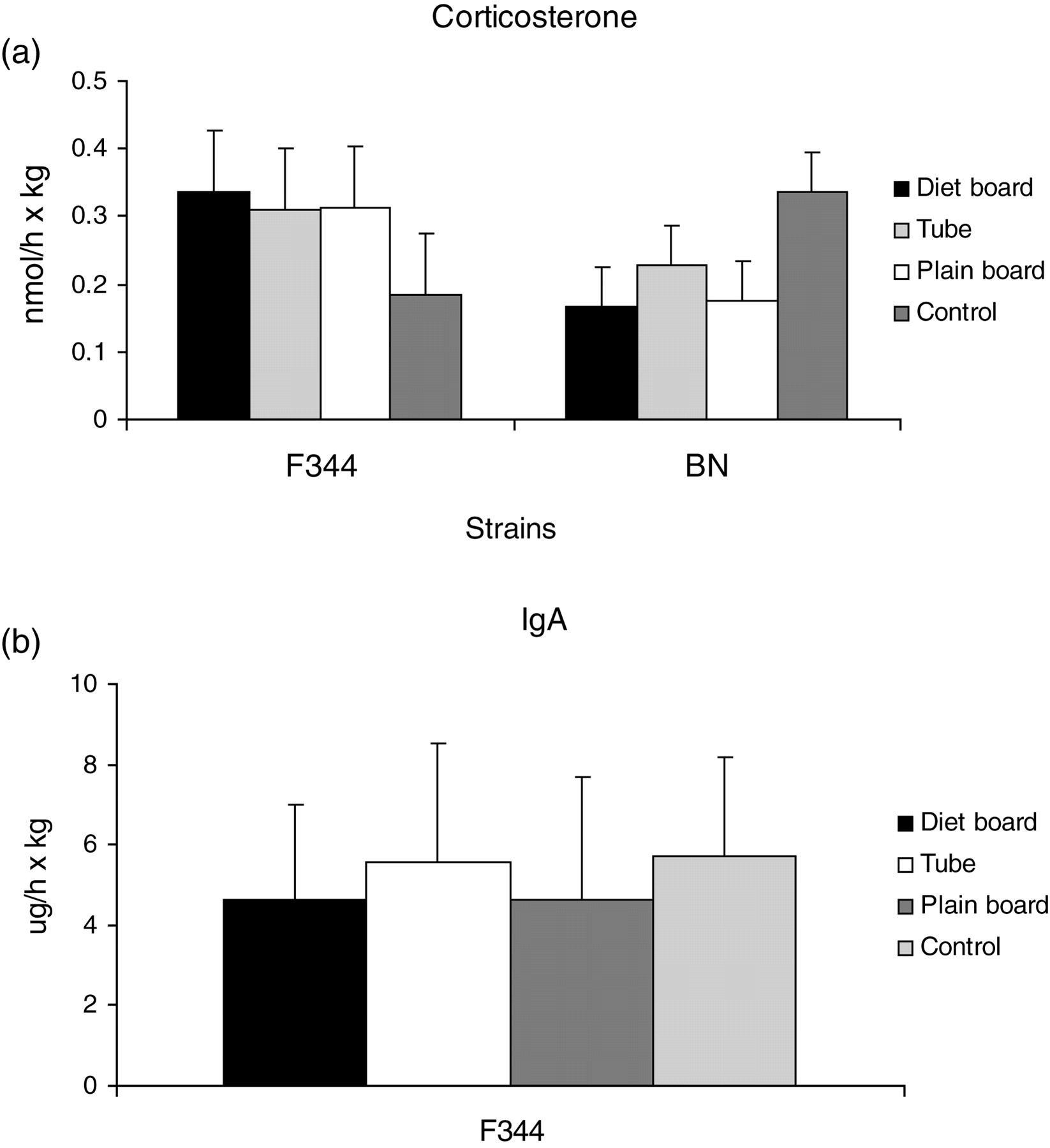
Corticosterone (a) and immunoglobulin A (IgA) (b) excreted (SEM) via faeces for F344 and BN rats with three cage items during the dark and light periods in individually ventilated cages (IVC). Number of experimental units within a strain = 16
Discussion
The F344 rats consistently had higher MAP and HR than BN rats, and the dark activity levels of the F344 rats were also higher than those of BN rats (Figure 2). This is well in line with the results of van den Brant et al. 25 who concluded that the BN strain no longer possesses the typical rodent nocturnal activity. The present study showed that in comparable environments, F344 rats display larger night–day differences both in MAP and HR values compared with the BN rats. This agrees with the results of van den Brant et al. 25 who categorized the F344 as a ‘hypertensive’ and the BN as a ‘hypotensive’ rat strain. We compared all the MAP and HR level differences to the corresponding night–day difference of the controls, and propose that statistically significant differences in means should exceed the normal night–day difference, in order to be considered to be biologically meaningful. The results of this study suggest that a fixed percentage, e.g. 6–7% as suggested by Krohn et al. 41 may not be applicable for all strains, and that a strain-specific measure, such as night–day difference, may be more valid for this purpose.
This study shows that cardiovascular responses of F344 rats and BN rats to the added furniture items differed. The F344 rats had the highest MAP in the tube cages all the time throughout the two-week period (Figure 3a). It has been shown that albino Wistar rats prefer a cage with a shelter rather than a barren environment 42,43 and that they tend to stay inside the tube during the light phase. 44 The elevated MAP, as seen in this study, indicates that this type of covered structure does not serve the purpose for which it was intended; a result that contradicts earlier findings. 44,45 However, it is possible that the tubes used in the present study may have been too small to comfortably accommodate three large rats, and that the finding is applicable to the F344 strain only.
Both board structures lowered the MAP values of F344 rats throughout the two-week period and towards the end, the periods with the plain board seemed to be most effective in this respect (Figure 3a). Based on this observation, it is likely that the dividing walls represent a suitable cage item for F344 rats, but also the diet board appears better than the tube, and certainly also better than no furniture at all. It is interesting to note that both boards, unlike the tube, could not be used to provide cover, a feature mandated by the European guidelines. 1,2 The diet board involves the concept of work for food, a feature that would be expected to raise at least the active period MAP, but nonetheless the difference was minor compared with the plain board. For routine animal observation purposes, dividing walls are clearly better than covered structures that obscure the animals. This seems to challenge the concept that all albino rats need to be provided with structures for hiding places and privacy. 44,45 The BN rats exhibited no biologically significant MAP or HR between-furniture item differences.
Only a handful of earlier studies on the effect of new cage items have utilized telemetry. Sharp et al. 15 evaluated their enrichment programme in rats with telemetrically recorded HR, systolic blood pressure and activity. They showed that HR of SH rats was significantly reduced by their combination of cage items, but for the SD rats they could detect no effect. Their programme consisted of several smaller items added to the cage between three and seven days. There were some similarities between these items and those used in our study, such as the simulated burrow, which is comparable to the tube used in the present study, and a gnawing and food foraging item, which is comparable to the diet board of our study. A detailed comparison between the two studies is not relevant because we used one item at a time for 14 days, as opposed to their combination of items and much shorter exposure time.
When the processed days were compared within the furniture item during the two-week periods, there was a decreasing trend both in MAP and HR levels in both strains (Figure 5), but statistical and biological significant findings were only found for MAP of F344 plain board rats when comparing days 2 and 14. We interpret this to mean that the F344 rats had habituated to the plain board, but not to the other items. Conversely, no such effects were detectable in BN rats.
The CV of MAP in F344 rats was highest in the plain board groups on day 2, both in the dark and light phases. This may simply be a novelty effect of this cage item which was introduced into the cage one day before (Figure 4a). The corresponding point estimates showed that with the plain board and MAP as the result parameters, the number of animals needed would be 1.36–1.81 times greater than those with other items including controls. Similar results were found between plain board and the tube on day 14 dark (Table 1). The high MAP seen in the F344 rats living with the tube was not associated with excessive variation but rather the opposite. In the BN rats, the CV values of both MAP and HR were higher in the control groups compared with the diet board groups (Figure 6) and the corresponding point estimates were 1.45 and 1.68, respectively (Table 1). Cage furniture may thus have marked, strain specific, effects on the within-group variation and this could be anticipated to influence the number of animals needed in blood pressure studies.
In the present study, the cage furniture items used exhibited no effect on faecal corticosterone or IgA excretion and there were no differences compared with the rats housed in control cages. Sarrieau and Mormède 30 have shown significantly higher plasma corticosterone levels in F344 rats compared with BN rats, but we could not detect any significant differences in faecal corticoids between the strains (Figure 6a). A recent study by Siswanto et al. 46 showed that injection of at least of 100 µg/kg adrenocorticotropin hormone is required to detect a subsequent change in the faecal corticosteroid level in rats. Hence, it was not surprising that the HPA axis was not sufficiently stimulated by the changes in cage environment for this to be detectable as changes in faecal corticosteroid excretion. Faecal IgA concentrations in rats have also been measured in response to housing in metabolic cages 20 but not to cage furniture. Royo et al. 21 stated that stress-induced changes in excreted IgA concentrations are slower and perhaps less pronounced than those of corticosteroids; hence, IgA may be useful only for assessing long-term animal wellbeing. Obviously, the lack of significant changes in this study suggests that two weeks may not have been long enough or alternatively the response was simply too small to be detected with this assay.
In conclusion, cardiovascular parameters can be used to assess the welfare value of cage furniture, whereas changes in faecal corticosterone and faecal IgA excretion would appear to be too small to be quantifiable. Furthermore, covered structures may not be any better than no structure at all in the cage, and the establishment of general recommendations may be difficult because there is a clear genetic component involved, resulting in major between-strain differences.
Footnotes
Acknowledgements
This study was supported by the Academy of Finland, the Finnish Ministry of Education, ECLAM and ESLAV Foundation, the North-Savo Cultural Foundation, the Finnish Research School for Animal Welfare, the Oskar Öflund Foundation and the University of Kuopio. We are thankful to Mr Heikki Pekonen for surgical procedures and to Ms Virpi Nousiainen for daily care of the rats.
