Abstract
In order to reduce the number of animal experiments, the use of non-heart-beating donors (NHBDs) from a commercial abattoir has been proposed. Since the use of slaughterhouse organs is legally not defined as animal experiment, this would fulfil international standards as an alternative to animal experiments. The development of intravascular thrombosis after cardiac arrest negatively impacts organ preservation and thus viability during ischaemic storage and reperfusion. A fibrinolytic preflush with streptokinase might overcome these limitations. Therefore, the functional and histomorphological integrity of kidneys preserved immediately with intact circulation (control group A) and kidneys preserved after cardiac arrest with a 30 min period of warm ischaemia (WI) (group B) was compared with kidneys preflushed with 12.5 kU/L (group C) or 50 kU/L streptokinase (group D) after 30 min WI prior to preservation. We could demonstrate that kidneys preflushed with 12.5 kU/L streptokinase (group C) performed better than those without streptokinase pretreatment after WI (group B). Parameters like oxygen consumption, perfusion pressure, laboratory values, lactate dehydrogenase level and lipidperoxidation were closer to that of the control (group A) than in groups B and D. The higher streptokinase concentration of 50 kU/L (group D) resulted histologically in a more pronounced tissue damage and an attenuated renal function, indicating toxic effects. On the basis of our results we believe streptokinase preflushed slaughterhouse kidneys to be an adequate alternative to organs from laboratory animals with the potential to further reduce the number of animal experiments.
A variety of isolated perfused porcine kidney models using non-heart-beating donors (NHBDs) have been developed to investigate electrosurgical therapy approaches 1–5 or to perform pharmacological studies. 6,7 Some of them make use of organs from laboratory animals which is expensive and, moreover, does not meet the ethical standards according to the 3R principle (reduce, refine, replace animal experiments) established by W M S Russell and R L Burch in 1959. Many efforts have been made to replace laboratory animal-derived kidneys by NHBD slaughterhouse kidneys and thereby reducing the number of animal experiments, 6,8–10 but there are still some limitations affecting organ quality. Besides the inevitable warm ischaemia (WI) due to the slaughtering process until explantation of the kidneys, a major concern of NHBD-derived organs is the development of intravascular thrombosis after cardiac arrest. 11 This is followed by inappropriate vascular tissue equilibration with preservation solution and a compromised microvascular perfusion leading to ischaemia/reperfusion injury. 12,13 Some groups applied a heparin flush to eliminate thromboses in NHBD organs. 5,14 But it has been previously shown that heparin has only a limited effect on existing thrombus formations. 15 In contrast thereto, the reversibility of existing thrombus has been demonstrated after in situ flush with streptokinase prior to organ explantation. 13,15–17
The aim of the present study was to investigate the effect of a fibrinolytic preflush with streptokinase in explanted, warm ischaemically predamaged NHBD kidneys on physiological and morphological kidney integrity upon preservation and reperfusion by simulating WI during the slaughtering process in an animal model.
Materials and methods
Animals
All experiments were performed in accordance with the German legislation governing animal studies following The Principles of Laboratory Animal Care (NIH publication 85-23, revised 1996). Official permission was granted from the governmental animal care office (Landesamt für Natur, Umwelt und Verbraucherschutz Nordrhein-Westfalen, Recklinghausen, Germany). Fourteen female German landrace pigs from a disease-free barrier breeding facility were housed in ventilated rooms and allowed to acclimatize to their surroundings for a minimum of seven days before surgery. The animals, weighing 36 ± 8 kg (mean ± SD) were fasted for 12 h prior to the experiments. For premedication, the animals received an intramuscular injection of 4 mg azaperone per kg body weight (BW) (StresnilTM, Janssen, Germany). Anaesthesia was induced by intravenous injection of 3 mg propofol per kg BW followed by oral intubation. The animals were ventilated with 40% oxygen at 20–26 beats per minute (bpm) and a tidal volume of 10 mL kg per kg BW to keep the end tidal partial carbon dioxide tension (pCO2) between 36 and 42 mmHg. Anaesthesia was maintained with isoflurane at a concentration of 1–1.5% (Forane, Abbott, Germany) and fentanyl (Fentanyl, Janssen, Germany) at a concentration of 3–4 μg per kg BW. After midline laparotomy, the kidneys were surgically removed in an en bloc technique with an aortic patch. After nephrectomy and blood collection, the animals were euthanized by intravenous pentobarbital (1 mL per kg BW; Narcoren®, Merial GmbH, Hallbergmoos, Germany) injection.
Experimental groups
In group A, kidneys were preserved immediately after explantation from heart-beating donors serving as control without no WI. Groups B–D comprised NHBD organs explanted and preserved after a standardized 30 min period of WI by blocking the blood supply with a vessel clamp at the renal artery. This procedure simulates the situation of WI in the slaughterhouse where explantation of kidney is usually performed within 8–30 min after stunning of the animal. 8,9,14
After nephrectomy the renal artery, the vein and the ureter of the kidneys were cannulated. The organs of group A (control) and B (WI) were subsequently rinsed with 500 mL of room temperature (RT, 22°C) isotonic NaCl solution (B Braun Melsungen AG, Melsungen, Germany), transferred on ice and infused with 250 mL 4°C histidine-tryptophan-alpha-ketoglutarate (HTK, Custodiol; Koehler Chemie, Alsbach, Germany) solution. Kidneys of groups C and D were flushed with 500 mL NaCl solution (RT) containing 12.5 or 50 kU/L streptokinase (Sigma-Aldrich Chemie, Steinheim, Germany), respectively, before preservation with HTK and storage on ice as described. All perfusions were performed by gravity flow (100 cm). The kidneys were stored on ice in HTK solution for 3 h until isolated reperfusion for 60 min. The groups are indicated as follows: group A: control (n = 10), group B: WI (n = 5), group C: WI + 12.5 kU streptokinase (n = 5) and group D: WI + 50 kU streptokinase (n = 4).
Blood collection and perfusion solution
After nephrectomy, autologous blood was collected from the vena cava and anticoagulated with 3000 IU/L heparin (Liquemine; Roche, Mannheim, Germany) and 3 g Fibrisol/L (Muscalla, Viernheim, Germany). For organ reperfusion the blood was diluted with Krebs–Henseleit buffer (Sigma-Aldrich Chemie) in a ratio of 1:4.
Perfusion system and perfusion procedure
The perfusion system is based on isolated perfused kidney models described elsewhere. It was adapted with a closed perfusion circuit and an oxygenation system to supply the kidney with sufficient oxygen (Figure 1). The kidneys were transferred in a plastic chamber containing perfusion solution and placed in a water bath adjusted to 38°C. To guarantee sufficient oxygen saturation, a carbogen mixture (95% O2, 5% CO2, flow 4 L/min) was used for oxygenation of the perfusate. The perfusion solution was transported from a reservoir into the cannulated artery of the kidney by a pulsatile roller pump. After passage of the organ the perfusate was recirculated through the cannulated vein via the oxygenator system (Lilliput 2 [D902], Dideco, Germany) into the perfusate reservoir.
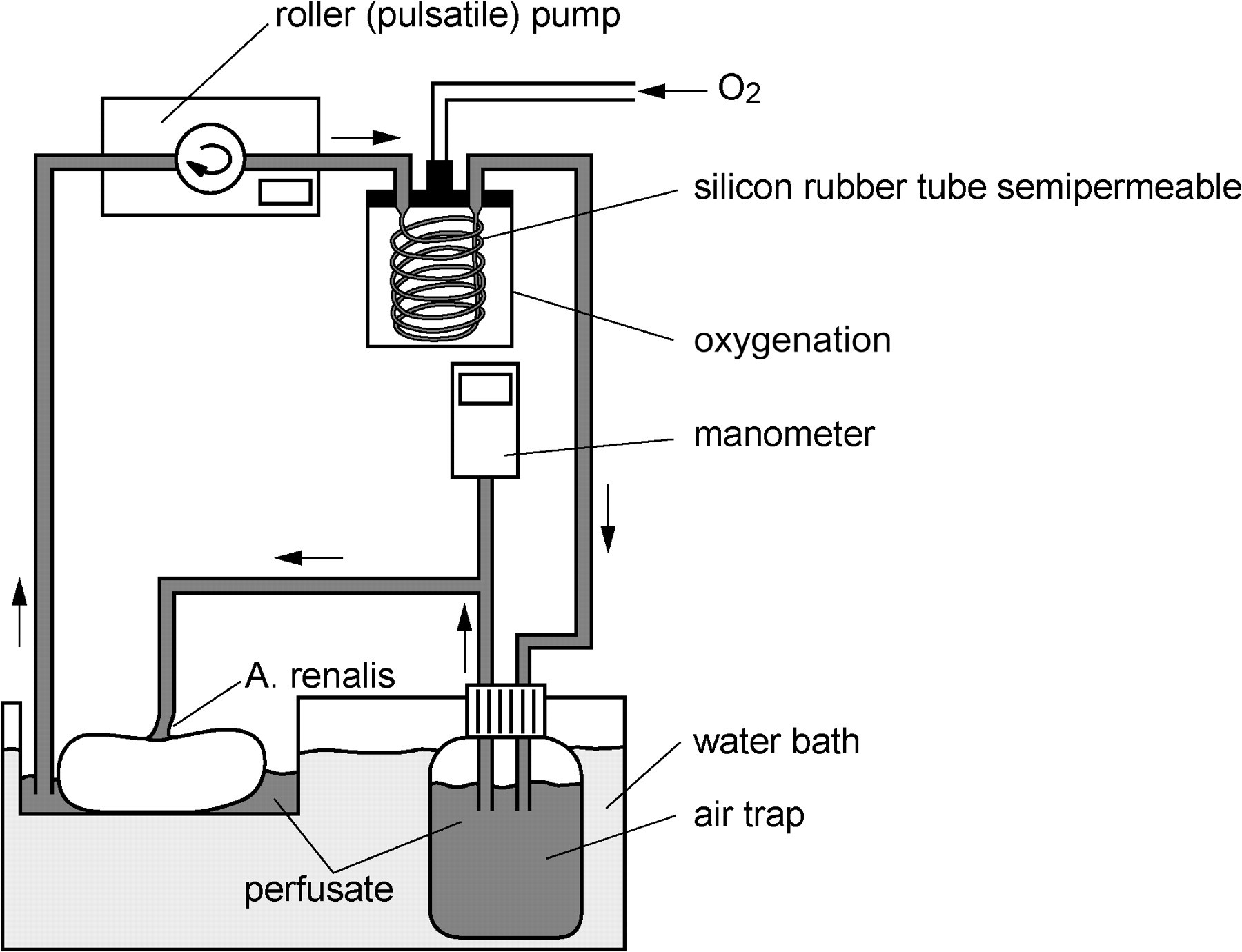
Perfusion system: closed perfusion circuit driven by a pulsatile roller pump, an oxygenation system and a manometer to control the systolic and diastolic perfusion pressure
Flow-controlled reperfusion of the organs was started with an initial arterial flow of 30 mL/min. The pressure was gradually increased within 5 min to 1 mL/g/min and an arterial pressure of 100 mmHg at maximum. Systolic and diastolic perfusion pressure was monitored from the start until the end of the 60 min perfusion period (Digital-pressure-meter, GMH 3161-13; Greisinger electronic GmbH, Regenstauf, Germany). The relative perfusion pressure was calculated by adding up the systolic and diastolic pressure divided by two.
Measured values and histology
Organ weight was assessed immediately after explantation, after preservation (flushing with NaCl and Custodiol solution) and at the end of the reperfusion. Arterial and venous blood samples were taken at the beginning and every 15 min for blood gas analysis (Radiometer ABL-700, Copenhagen, Denmark). Oxygen consumption, plasma creatinine (Vitros 250 Chemistry System, Ortho-Clinical Diagnostics, Johnson & Johnson, Neckargemuend, Germany) and urea (Vitros 250 Chemistry System) were used as markers to estimate organ vitality and renal function. Plasma malondialdehyde (MDA) measured by using fluorometry (Tecan, Infinity 200; Tecan Gmbh, Crailsheim, Germany) served as a biomarker to assess the level of oxidative stress. Lactate dehydrogenase (LDH) (Vitros 250 Chemistry System) was used to quantify cell damage in the isolated system. Urine was collected by a urethral catheter to estimate the relative urine density with a Urinprober (Eickemeyer Medizintechnik für Tieraerzte KG, Tuttlingen, Germany).
For evaluation by light microscopy, biopsies of the renal cortex and the medulla were fixed in 4% buffered formaldehyde and embedded in paraffin. These samples were cut into 4 μm cross-sections and stained with haematoxylin and eosin. A semiquantitative scoring system was used to assess tissue damage like protein accumulation, vacuolization, haemorrhage and cellular integrity: 0 = no abnormalities, 1 = mild, 2 = moderate, 3 = severe changes. The histopathology score and the erythrocyte count were evaluated at each sample with at least four non-overlapping high-power fields.
Statistics
Data were analysed by means of descriptive statistics (mean and standard deviation). Normal distribution of data was assessed by the Kolmogorov–Smirnov test. One-way analysis of variance was used to test for differences among more than two independent groups. Tukey's multiple-comparison test was applied to compare the individual groups. P values <0.05 were considered statistically significant.
Results
Weight gain
The mean kidney weight after explantation did not differ significantly among the experimental groups (Table 1). After preservation the weight increased in all groups (A–D). The highest weight gain was found in the control group A (51.9 [9.2%]) and in group B (52.1 [13.8%]) compared with groups C (32.9 [15.1%]) and D (16.4 [12%]) (Table 1). A further increase in weight was detectable after reperfusion which was similar in groups A, B and D (21.4 [6%], 20.2 [13%] and 25 [9.9%], respectively) and more pronounced in group C (38.6 [6.8%]).
Procedure-related weight gain in mean (SD) upon kidney preservation and reperfusion
WI: warm ischaemia (30 min); strep: streptokinase; HTK: histidine-tryptophan-alpha-ketoglutarate solution (Custodiol)
Perfusion pressure
The perfusion pressure varied over the time and was initially significantly higher in groups B–D compared with the control (P < 0.001, Figure 2). In group C (WI + 12.5 kU streptokinase) the pressure declined after 15 min to reach the same perfusion pressure as the control group at 60 min, while a continuous increase in pressure was visible in group D (WI + 50 kU streptokinase) (Figure 2). The mean perfusion pressure in the control group A was 110.9 (10.3) mmHg. A higher perfusion pressure was observed in groups B and D with 178.8 (22.1) and 173.4 (11.2) mmHg, respectively (P < 0.0035).
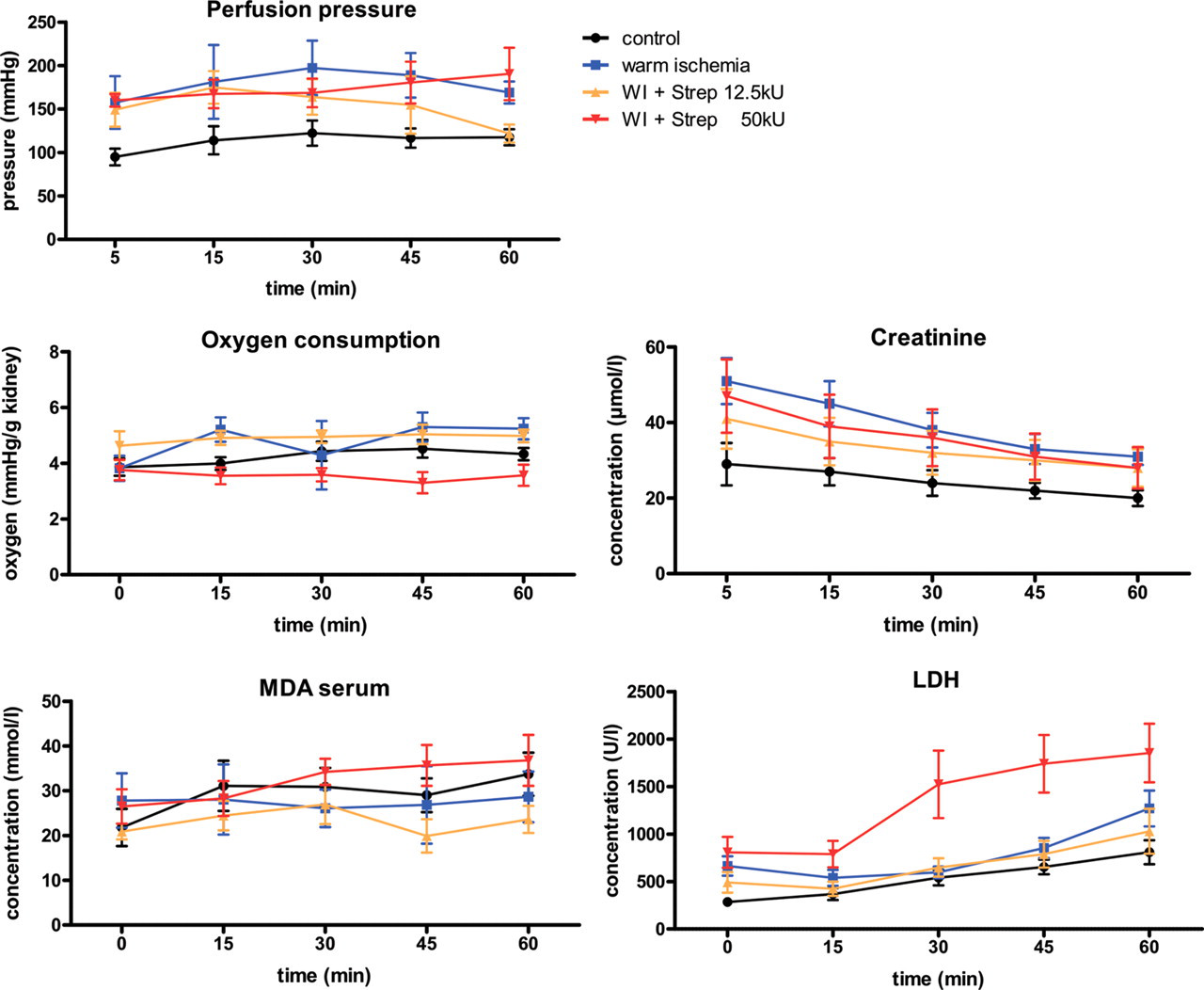
Time course of perfusion pressure, oxygen consumption, malondialdehyde (MDA), creatinine and lactate dehydrogenase (LDH) levels. WI: warm ischaemia; Strep: streptokinase
Organ vitality and renal function
Oxygen consumption was nearly constant over 60 min in all groups (Figure 2). In groups B (WI) and C (WI + 12.5 kU streptokinase) the mean oxygen consumption was higher than in the control group A (4.77 [0.30] and 4.90 [0.07] versus 4.23 [0.13] mmHg/g kidney weight, respectively), while it was lower in group D (3.55 [0.07] mmHg/g kidney weight). The serum creatinine dropped in all groups during reperfusion and was elevated in group B (WI) (P < 0.01, Figure 2). Blood urea was almost constant in groups A–C but significantly increased in group D (WI + 50 kU streptokinase) compared with the control (P < 0.05, Table 2). Urine density at 60 min was within the physiological range and comparable in groups A, C and D (1.022 [0.004], 1.024 [0.013] and 1.015 [0.006], respectively). An increase in urine density compared with the control was found in group B (WI) with 1.050 (0.006) (P < 0.05, Table 2). Calcium and chloride values did not differ among the groups and a slight increase over the reperfusion course was observed (Table 2).
Laboratory values at different time points of reperfusion in mean (SD)
WI: warm ischaemia (30 min); strep: streptokinase; MDA: malondialdehyde. The time point 0 min was used only for MDA assessment. ND: not done. *P < 0.05 compared with the control
Reperfusion injury and oxidative stress
LDH release steadily increased over the time in all groups (Figure 2). Group C (WI + 12.5 kU streptokinase) was nearly indistinguishable from the control (group A). Significant (P < 0.0083) higher LHD levels were observed in group D (WI + 50 kU streptokinase) with a marked, almost two-fold increase at 15 min. Plasma MDA was relatively constant in groups A–C, whereas a constant increment was seen in group D (WI + 50 kU streptokinase) (Figure 2). In groups B (WI) and C (WI + 12.5 kU streptokinase) the values were below that of the control.
Histology
The structural integrity of the kidneys of group C (WI + 12.5 kU streptokinase) was comparable to that of the control group A with only mild to moderate changes concerning protein accumulation, vacuolization and cellular integrity in the cortex (Table 3, Figure 3). These changes were moderate to severe in groups B (WI) and D (WI + 50 kU streptokinase). In the medulla, the changes were less pronounced with the exception of a distinctive cellular destruction in the kidneys of group D (WI + 50 kU streptokinase) (P < 0.05) (Table 3).
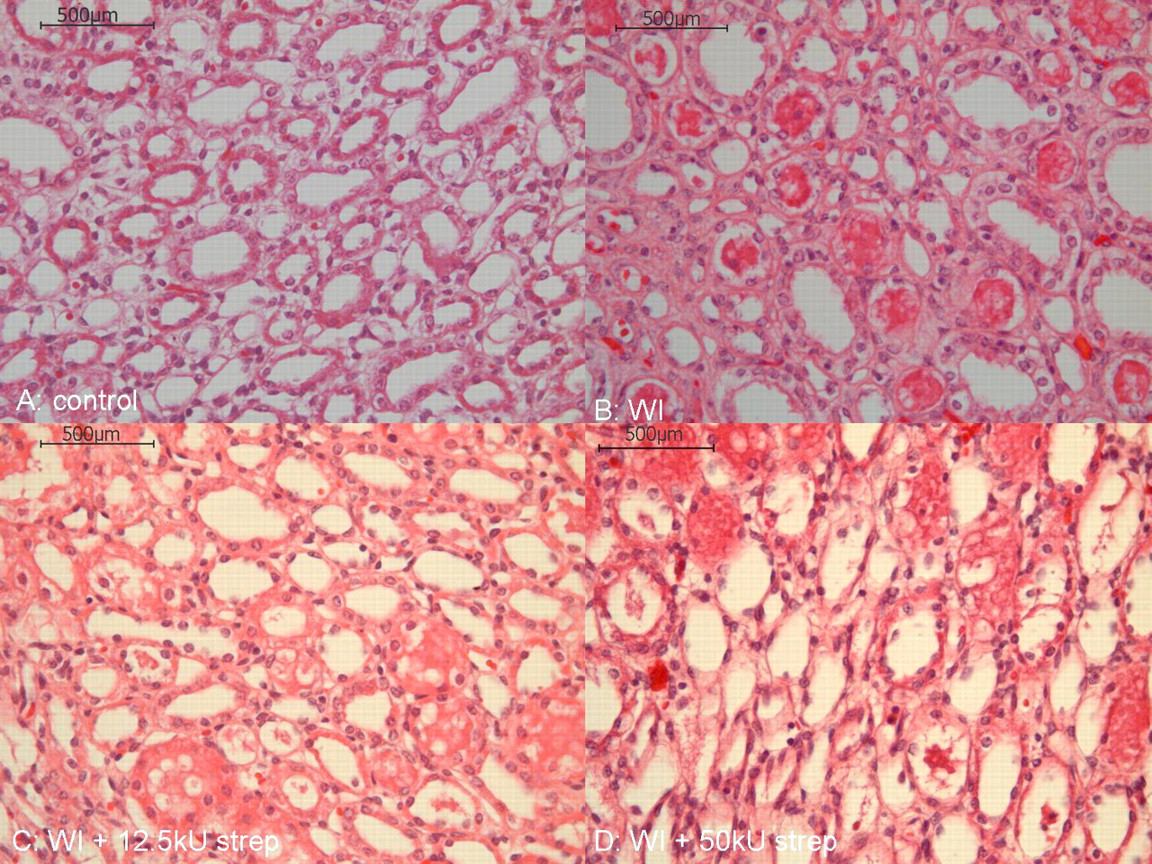
Haematoxylin and eosin stained histological cross sections from the medulla of kidneys: control group A, groups B (WI), C (WI + 12.5 kU strep) and D (WI + 50 kU strep) (magnification: ×400). WI: warm ischaemia; strep: streptokinase
Histological analysis of tissue damage based on a semiquantitative scoring system: 0 = no abnormalities, 1 = mild, 2 = moderate and 3 = severe changes
Values are given in mean (SD). *P < 0.05 compared with the control. WI: warm ischaemia (30 min); strep: streptokinase
The erythrocyte count, applied to estimate the flushing efficacy, was highest in group B without fibrinolytic preflush and could be reduced by streptokinase irrigation in groups C and D (Figure 4). The use of the higher streptokinase concentration of 50 kU (group D) resulted in a large number of desintegrated erythrocytes.
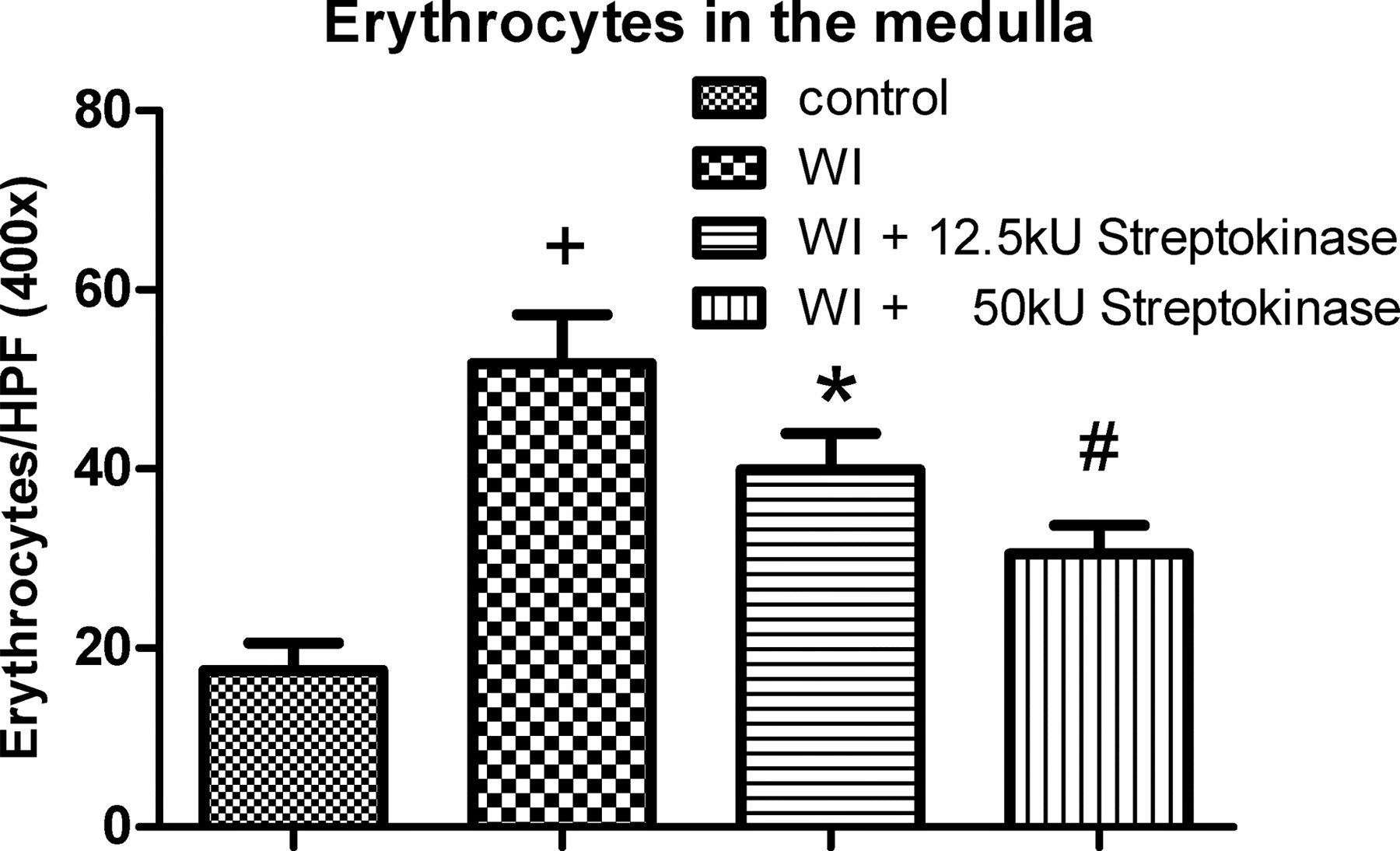
Erythrocyte count in the medulla of kidneys of group A (control), groups B (WI), C (WI + 12.5 kU strep) and D (WI + 50 kU strep) of at least four non-overlapping high-power fields, respectively. + P < 0.05, *P < 0.01, # P < 0.001 compared with the control. WI: warm ischaemia; strep: streptokinase
Discussion
The isolated perfused porcine kidney is an established model used in a variety of pharmacological or electrosurgical studies due to structural und functional homology of the porcine kidney to that of humans. 5–7,14 In order to reduce the number of animal experiments, the use of NHBD from a commercial abattoir has been proposed. 5,7 The exploitation of slaughterhouse organs would be a crucial step forward in reducing the number of animal experiments since the use of slaughterhouse kidneys is not legally defined as an animal experiment and would fulfil international standards as an alternative to animal experiments. 18 The subjection to warm or cold ischaemia- and reperfusion-related injuries impairing the functional and structural integrity of donor organs is still a general problem. Especially the development of intravascular thrombosis after cardiac arrest negatively impacts vascular tissue equilibration with preservation solution, which is a prerequisite for optimal maintenance of organ viability during ischaemic storage as well as to reduce reperfusion injury. 11,12,19
Flushing of organs with heparin after stunning and desanguinating of the animals prior to organ preservation have frequently been used so far. 5,14 But since heparin is unable to resolve existing microthromboses, 16 heparin appears to be useful only in the prevention of microthromboses when applied in situ prior to organ explantation. This procedure is not compatible with the slaughtering procedure. Slaughterhouse organs have to be explanted in accordance with the official meat protocol law resulting in a delay of up to 30 min from stunning of the animal up to organ retrieval, thereby increasing the risk of intravascular thrombus formation. In this case, enzymes with fibrinolytic and thrombolytic activity like streptokinase or urokinase appear to be more useful. In situ flushing with fibrinolytic enzymes has been shown to be effective in dissolving thromboses in animal models as well as in clinical settings. 13,17,20,21 Besides, the otherwise compromised microvascular perfusion with preservation solution protecting the tissue during cold ischaemic storage and against reperfusion injury could be restored. 12,13,19
Our investigations on the effect of streptokinase in warm ischaemically predamaged NHBD kidneys indicate that the physiological and structural integrity can be restored by preflushing the kidneys with a streptokinase concentration of 12.5 kU/L.
This was reflected by the fact that parameters like oxygen consumption and perfusion pressure as well as laboratory values such as creatinine, urea, calcium, chloride and urine density were closer to that of the control than in the WI group (B) or the group receiving a 50 kU/L streptokinase preflush (group D) (Figure 2, Table 2). Furthermore, LDH levels (an indicator for parenchymal injury) in group C (WI + 12.5 kU streptokinase) were quite similar to that of the control. Interestingly, plasma MDA, a marker for lipidperoxidation to assess oxidative stress, was lower (even though not significant) in group C than in the control group. These data indicate that the preflush with 12.5 kU/L streptokinase has the potential (a) to improve tissue equilibration with preservation solution by dissolving microthromboses, (b) to restore structural and functional integrity and thereby (c) reduce reperfusion injury. The histological analysis is in line with this assumption since the structural integrity of the kidneys in group C (WI + 12.5 kU streptokinase) was comparable to that of the control group A with only mild to moderate changes of the cellular integrity in the cortex and moderate to severe changes in groups B (WI) and D (WI + 50 kU streptokinase) (Table 3).
Although lysis of microthromboses by fibrinolytic preflush is supposed to be more effective when using higher streptokinase concentration, the higher dose of 50 kU/L used in our experiments may have had toxic effects leading to tissue damage and an attenuated renal function. This assumption is in accordance with a strong increase in LDH, elevated urea levels and higher creatinine concentrations compared with the control (group A) and group C (WI + 12.5 kU streptokinase) (Figure 2, Table 2). Furthermore, cellular destruction was significantly more severe in group D (WI + 50 kU streptokinase) compared with the control (Figure 3, Table 3) as well as a pronounced disintegration of erythrocytes in group D.
In conclusion we have shown that by streptokinase preflush the functional and morphological integrity of warm ischaemically predamaged kidneys can be restored resulting in a stable organ function compared with kidneys explanted immediately after cardiac arrest. On the basis of our results we believe streptokinase preflushed slaughterhouse kidneys to be an adequate alternative to organs from laboratory animals with the potential to replace or reduce animal experiments.
Footnotes
ACKNOWLEDGEMENTS
We would like to thank Thadeus Stoppinski and Mareike Schulz for their technical support.
