Abstract
Isolated kidney perfusion forms the bridge between in vitro and in vivo experiments and is mainly performed with rodent kidneys. It enables the controlled investigation of kidney function without systemic influences and is therefore currently, for instance, considered the gold standard for investigating the regulation of renin release, a central regulator of blood pressure. Renin secretion is controlled by several intrarenal signalling cascades and thus cannot be fully modelled in cell cultures. On the other hand, the in vivo investigation of renin release is confounded by systemic counter-regulatory systemic mechanisms. For ethical and logistical reasons, human kidney tissue has not been used for isolated organ perfusion yet. To reduce the use of rodents and to achieve the translation to larger species, including humans, we developed a method for isolated perfusion of slaughterhouse pig kidney segments. This method allows for future use with human kidney segments obtained from nephrectomies. Pig kidney segments were perfused at constant pressure and renin secretion was determined. The sympathomimetic agent isoproterenol, the Ca2+ chelator ethylene glycol tetraacetic acid, the NO-donor S-nitroso-N-acetylpenicillamine and the loop diuretic bumetanide stimulated renin release while angiotensin II suppressed it. These responses were similar to those observed in isolated perfused rodent kidneys, demonstrating that central regulatory mechanisms of renin release are conserved across species and in tissue segments. Moreover, the newly developed model of perfusion of kidney segments of large animals is suitable for the investigation of renin release from human kidneys and provides an ethical method for isolated kidney perfusion in accordance with animal welfare.
Keywords
Introduction
Isolated perfused organ preparations bridge the gap between in vitro and in vivo experiments and are optimally suited for the systematic investigation of the (patho)physiology of an organ without systemic influences that often counterbalance direct organ effects. Isolated perfused rat kidneys have long been established as an inherent part of kidney physiology as well as drug research. The isolated perfused mouse kidney represents a more recent addition to the canon of isolated organ perfusions. While technically more sophisticated it allows the use of a wide variety of genetically modified animals to answer a broad spectrum of pathophysiological questions.1–5 Despite its undeniable scientific significance and the reduced stress compared with alternative in vivo animal experiments, the method also has significant disadvantages. Employment of any model that involves animals has to be extensively discussed for obvious ethical reasons. Next to animal welfare concerns, any research utilizing such models is at best a close approximation of human physiology and its translation can often be realized only at cellular level. 6 Perfusion of human kidneys represents an enticing alternative. While there is a steady supply of human kidneys for research purposes, these mostly stem from tumour-nephrectomies and therefore as a whole organ do not provide adequate physiological conditions for research applications. In addition, a part of the organ including tumour cells is usually needed for further diagnostics, so that no complete kidney is available. In most cases, though, only parts of the organ are affected by tumour cells and healthy tissue is extracted with the organ. The use of kidney sections allows for selection of unaffected healthy tissue and prevents ethical concerns as this tissue is usually discarded after clinical diagnostics. This paper thus aims to establish a method for isolated perfusion of kidney segments that could potentially be sourced from radical or partial nephrectomy in cancer patients. Because of their anatomical similarities to human kidneys, pig kidneys were used for isolated perfusion of tissue segments (isolated perfused pig kidney segments (IPPKSs)). In comparison with isolated perfusion of rodent kidneys the accessibility of pig kidneys from slaughterhouses may prove to be logistically as well as ethically advantageous. Depending on local regulations there also might be less strict requirements for animal testing. Owing to strict standards in livestock cultivation slaughterhouse animals can be compared to those of wildtype laboratory animals. 7 If necessary, experiments carried out on wildtype animals can be transferred directly to genetically modified pigs without need for interspecies translation. 8 Finally, the kidneys of humanized pigs could be an alternative to rodent and human kidneys in the future. However, the advantage of better transferability to humans would then be significantly reduced by the disadvantage of using laboratory animals. The experiments performed with the IPPKSs focused on the regulation of renin secretion, as renin acts as an important player in the regulation of systemic blood pressure as regulatory hormone of the renin–angiotensin–aldosterone system. 9 It is, however, influenced by a multitude of systemic factors which prevent systematic and direct in vivo analysis of the regulation of its release. 10 The isolated perfused kidney model is regarded as gold standard in renin research as it bridges the gap between cellular and in vivo research. Our experiments in IPPKSs largely confirmed the results acquired in isolated perfused rodent kidneys in terms of regulation of renin release. The data presented in this paper demonstrate the suitability of isolated perfusion of partial organs and tissue samples and lay the groundwork for future translation of isolated kidney perfusion to human tissue samples.
Animals, materials and methods
Animals
Pig kidneys were obtained from a local slaughterhouse as a by-product of slaughtering. The organs were obtained from four-month-old crossbreeds between Deutsche Landrasse and Piétrain weighing between 110 kg and 130 kg, of which male pigs had been castrated shortly after birth. The animals were electrically stunned and exsanguinated immediately afterwards.
The whole procedure from slaughtering to storing the kidneys in saline solution on ice took 20–30 min. There was no discernible difference in perfusate flow between organs that were flushed with Ringer’s solution with colloid additive immediately after removal and those that were placed in saline solution on ice without flushing until perfusion. Histologically, there were also no signs of thrombotic events in non-flushed tissues. For this reason, we decided not to flush the organs before storage on ice.
The organs were stored for 1–10 h in saline solution on ice before perfusion. All experiments were conducted in accordance with the Institute for laboratory research guide for the care and use of laboratory animals eighth edition (Washington, DC: National Academy Press, 2011) and German laws on the protection of animals.
Materials
Perfusion apparatus
The perfusion apparatus is based on a single-pass normothermic pressure-constant (100 mmHg) perfusion setup. The perfusion pressure of 100 mmHg was chosen to closely resemble the physiological mean arterial pressure of pigs of 103 mmHg. 11 It consists of two temperature-controlled vessels (38°C) containing the perfusate (item 2 in Figure 1) and the organ (5 in Figure 1), respectively. Perfusion is maintained by a roller pump (3 in Figure 1) (Model Reglo Analog MS-4/8 by Ismatec, Germany) controlled by an electrical control unit (9 in Figure 1) (STH Pump controller by AD Instruments, New Zealand) connected to a pressure transducer (8 in Figure 1) (SP844 by Memscap, France) between the organ and the pump. A temperature-controlled coil (38°C, 4 in Figure 1) is added to maintain a constant perfusate temperature. The perfusate is constantly gassed with 95% O2 and 5% CO2 (1 in Figure 1). During perfusion the kidney section lays on top of a metal grid above a glass funnel allowing constant drainage of venous effluent. The bottom of the perfusion chamber is covered with water to provide relative humidity of 100%. Drugs (13 in Figure 1) are administered via an in-line membrane injection port (12 in Figure 1) placed between the pressure transducer and the organ. Bubble traps consisting of a 20 ml syringe filled with 10 ml air (11 in Figure 1) between the pump and perfusate container, as well as the heated coil and the pressure transducer, are integrated. A three-way-valve (6 in Figure 1) between the perfusion vessel and the roller pump is installed to enable collection of samples of the effluent. A PowerLab (ADInstruments, New Zealand) serves to establish a digital interface between the pump control unit and the software LabChart (ADInstruments, New Zealand) installed on a PC (10 in Figure 1), allowing to monitor flow rate, systemic and vascular pressure as well as resistance.
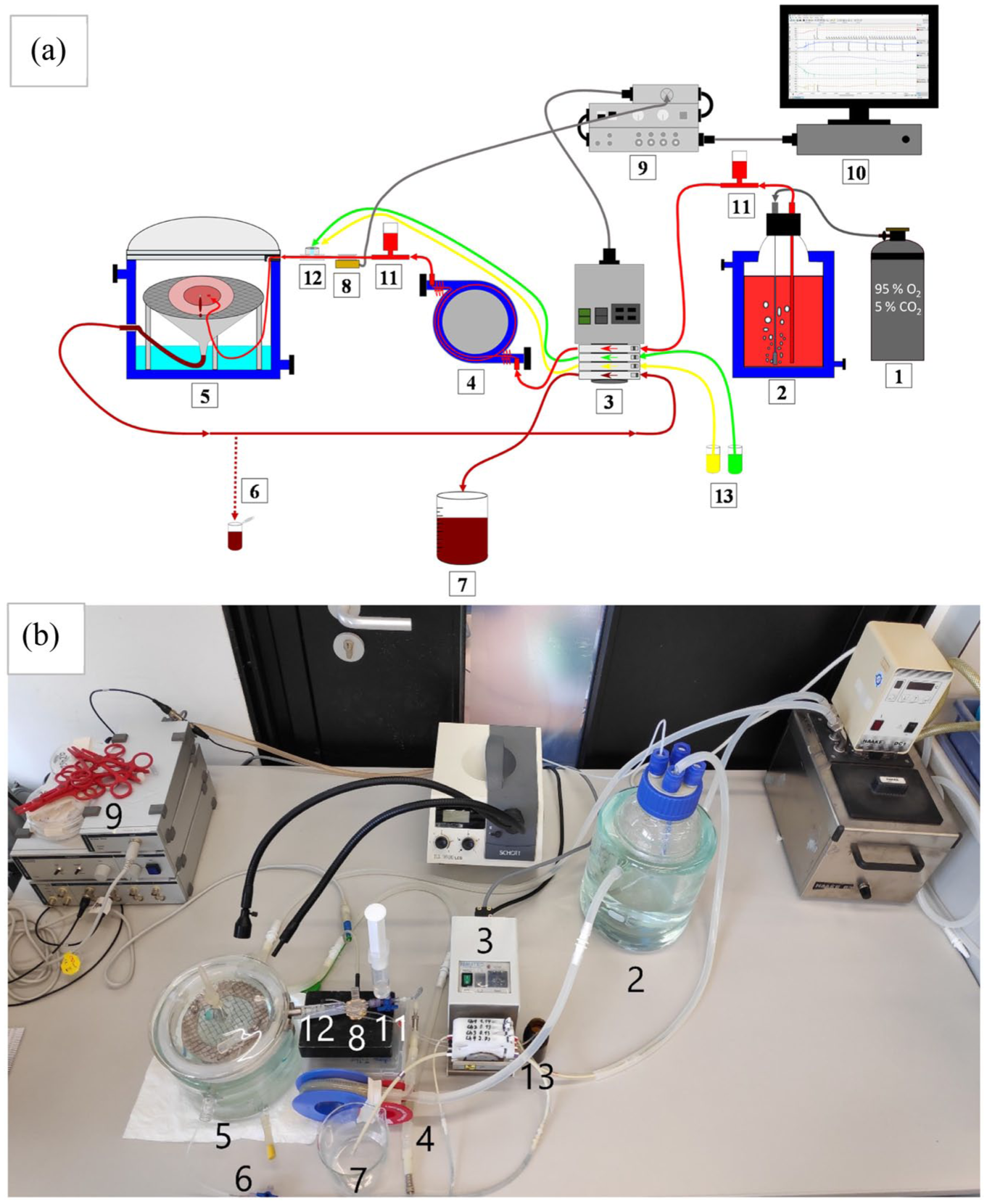
Setup of the perfusion apparatus. (a) Schematic illustration: 1. carbogen tank (95% O2 /5% CO2); 2. tempered perfusate tub; 3. roller pump; 4. tempering coil; 5. perfusion chamber; 6. sample collection; 7. venous effluent waste; 8. pressure transducer; 9. pump control unit; 10. digital interface; 11. bubble traps; 12. In-Line Luer membrane port; 13. administration of experimental substances. (b) Photographic image of the perfusion system.
Perfusate
The organs were perfused using a modified Krebs–Henseleit buffer with the addition of 3% dextran 150 kDa as colloid (detailed description in Supplementary material online).
Drugs
After constant perfusion pressure of 100 mmHg was established, stock solutions of isoproterenol (Sigma-Aldrich), angiotensin II (Sigma-Aldrich), ethylene glycol tetraacetic acid (EGTA) (Merck), S-nitroso-N-acetylpenicillamine (SNAP) (Sigma-Aldrich/Cayman-Chemical) and bumetanide (Sigma-Aldrich) were administered. The concentration of the stock solution was calibrated to achieve final concentrations in the perfusate of 10 nM for isoproterenol, 1 nM for angiotensin II, 3.1 mM for EGTA, 10 µM for SNAP and 100 µM for bumetanide.
Methods
Corrosion preparation
To determine the optimal intrarenal vessel for cannulation as well as to determine a suitable size of kidney segments for sufficient and reproducible perfusion, corrosion casts of the renal vessels of pig kidneys were prepared. To this end, the organs were flushed with saline solution prior to application of 10 ml of a mixture of Biodur E20 Blue Resin and Biodur E20 Hardener (Biodur, Germany) in a proportion of 100 parts resin to 55 parts hardener weightwise as specified by the manufacturer. The resin was allowed to cure for 24 h before the organ was placed inside of a 25% potassium-hydroxide solution on an orbital shaker for 24 h. The organ was then washed with distilled water and soaked in 25% hydroxide solution for another 24 h. After washing with distilled water again the remaining tissue was removed manually.
Haematoxylin–eosin stain
Kidney segments were fixed by immersion in 4% paraformaldehyde and staining with Mayer’s hemalaun and eosin was performed on 10 µm paraffin slices following dewaxing and rehydration. Sections were inspected using a Zeiss Axiovert 200 M Microscope and Zen lite 2012 software.
Surgical procedure
Prior to perfusion the pig kidneys are stored in 0.9% NaCl solution on ice. A kidney pole is dissected at a line drawn in the transversal plane, roughly 3 cm from the kidney hilus. A kidney segment obtained in this way has a size of about 3 cm × 3 cm × 4–5 cm. The tissue is then stripped off its capsule and placed on top of the metal grid inside of the preheated perfusion chamber with the cut surface facing upwards. Under an OP-microscope a suitable intrarenal artery (usually an interlobar artery) is identified and cannulated with a vented flexible polyethylene catheter. The catheter is fixed with a clamp and a ligature distally from the clamp. If the preparation is executed correctly some blood will be flushed out of the corresponding veins upon starting the perfusion and the perfused tissue will turn pale (Figure 2).
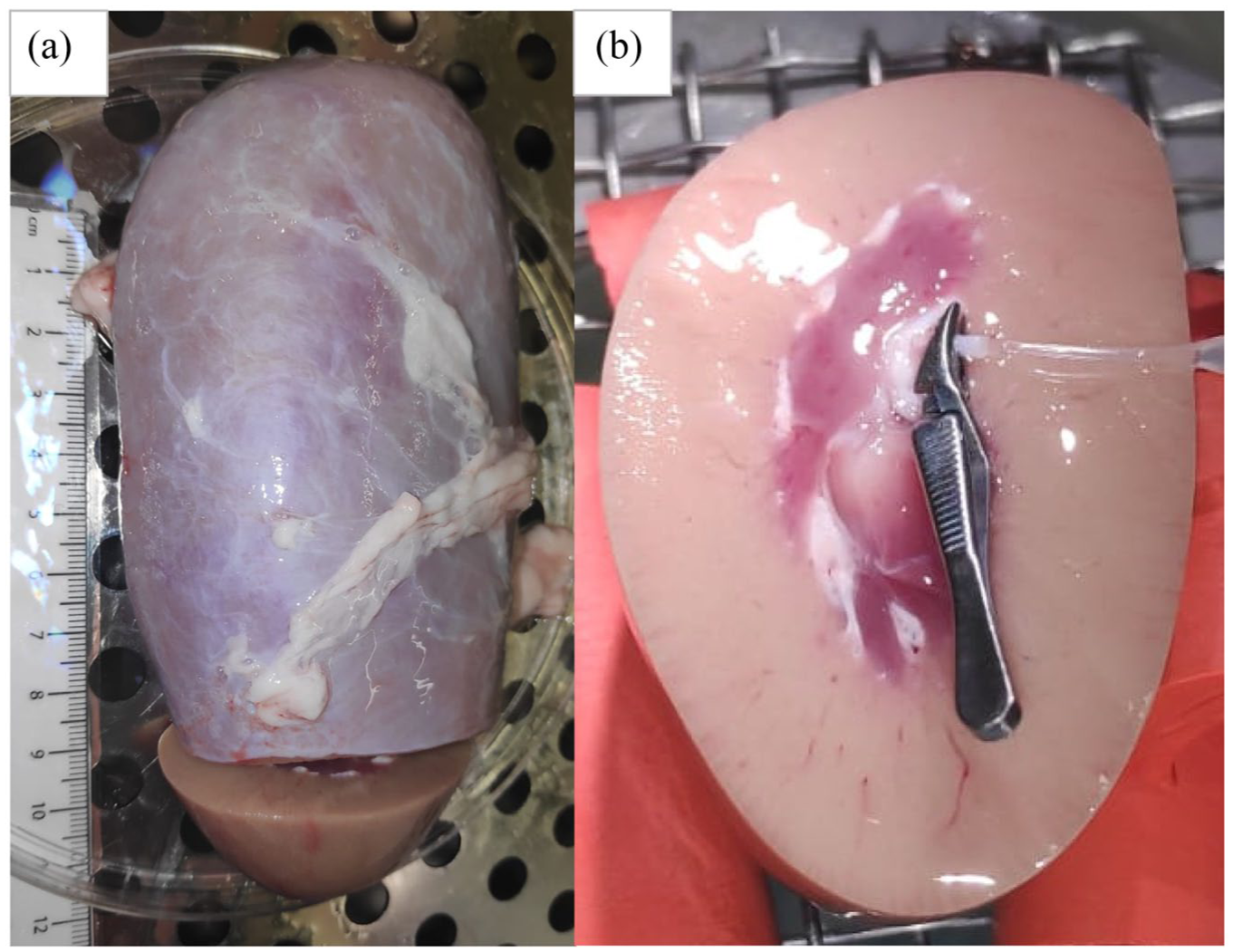
Surgical preparation of a suitable pig kidney segment for perfusion. (a) The caudal kidney pole was dissected in the transversal plane and the capsule removed. (b) A suitable artery vessel was identified, freed from surrounding tissue and cannulated. The clamp was placed and a ligature tied distal to the clamp in a next step.
Perfusion protocol
The perfusion is carried out in a normothermic constant-pressure single-pass setup with a directly oxygenated modified Krebs–Henseleit buffer. The organ is perfused until a steady pressure plateau has been reached and subsequently samples of venous effluent are collected every 2 min for the rest of the experiment. Following a 15 min control period drugs are administered for 15 min each. After the last intervention the tissue is either flushed with Biodur resin to make a corrosion cast or with an Evans Blue solution to determine the perfused tissue weight. In the latter case the perfused kidney segment is afterwards frozen to determine the perfused area by scratching off any tissue that has not been stained, weighing only the perfused tissue.
Immunoassay
To determine the renin secretion, three samples of venous effluent were collected at the end of every intervention period, stored on ice and frozen at −20°C until needed for the immunoassay. Renin activity was determined by the capacity of the sample to generate angiotensin I from angiotensinogen. Angiotensin I concentration was determined by a commercially available Enzyme-linked Immunosorbent Assay EIA (IBL International, Tecan, Switzerland). Perfusate samples that had not passed the kidney were used as blanks. Angiotensin I concentrations (ng angiotensin I/(h * ml)) were multiplied by perfusate flow (ml/(min * g)) to calculate renin secretion rates (ng angiotensin I/(h * min * g)).
Statistical analysis
Values are given as mean ± SEM. In the isolated perfused kidney experiments, the last three values obtained within an experimental period were averaged and used for statistical analysis. Student paired t-test was used to calculate significance levels when a control period was compared with a single stimulation period. In experiments in which the control phase was compared with a stimulation phase and a recovery phase, one-way analysis of variance for repeated measures was used. For multiple comparisons, Dunnett’s multiple comparisons test was generally applied. Statistical calculations were performed using GraphPad Prism 7.05. p <0.05 was considered statistically significant.
Results
Corrosion casts of intrarenal vessels
In order to determine which arterial vessels were best suited for cannulation in terms of diameter and position, the intrarenal vasculature was visualized using corrosion casts (Figure 3(a)).

Direct measurement of the vessel diameters using digital callipers revealed the following values: renal artery: 5.35–6.25 mm; cranial segment of renal artery: 4.24–5.07 mm; caudal segment of renal artery: 3.7–4.48 mm; segmental arteries: 2.33–2.83 mm; lobar arteries: 1.76–2.21 mm; interlobar arteries: 0.78–1.43; arcuate arteries: 0.51–0.81 mm. The interlobar artery proved to be the smallest vessel that was easily cannulated, with an inner diameter of 0.78–1.43 mm, while running a radial course. It was easily located in most kidney segments while still providing a sufficient tissue seam around the perfused tissue to prevent leakage.
Perfusion control
Corrosion casts as well as histologic images served as controls of the preceding perfusion. The corrosion casts enabled a retrospective view at perfusate leakage as well as proficiency of preparation.
Figure 3(b) to (d) shows the dense bed of glomeruli among the fine capillary network. As shown in Figure 3(b) and (c), even the finest vessels, such as the vasa vasorum, providing blood flow to the branches of the renal artery are properly perfused. The glomeruli are entangled in the capillary bed and are neither deformed nor show any signs of damage due to embolism. An interesting attribute of the Biodur resin used for the corrosion casts is its lighter appearance when cured in a diluted state. As seen in Figure 3(b), (c) and (e), this aids in the identification of vascular leakage, but it is also seen in venous vessels. More importantly, though, leaks can be detected by improper perfusion of smaller vessels (Figure 3(e)) as the resin is taking the way of least resistance through the leak instead of perfusing the capillary network, requiring the build-up of pressure.
The haematoxylin–eosin-stained histologic images show well defined cell borders without any visible blood clotting or thrombus formation (Figure 4). However, the partial loss of the tubular brush border membranes indicates tubular cell damage after prolonged perfusion (Figure 4(b)).
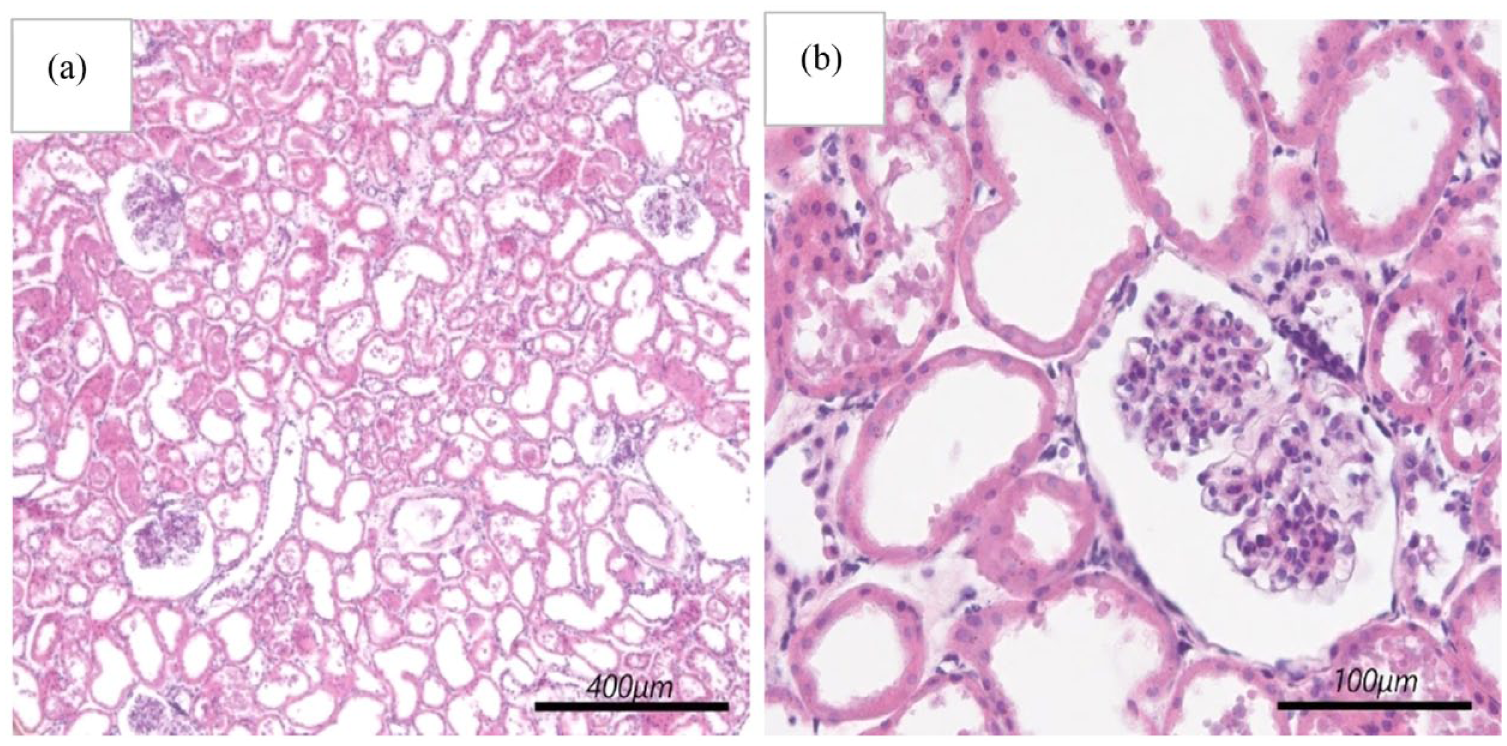
Haematoxylin–eosin-stained sample of kidney tissue following 115 min of perfusion. (a) 10× magnification; (b) 40× magnification.
Colloid and ischaemia
Dextran 150 kDa is added to the perfusate as a colloid as it provides superior maintenance of various physiological functions in comparison with other synthetic colloids such as Pluronic F108, Haemaccel or HES. 12 Bovine serum albumin is often used for perfusion of rat and mouse kidneys, is the main oncotic protein in vivo and would therefore be the most physiological solution.2,13 However, direct oxygenation of a solution containing albumin results in massive foam formation and therefore requires additional technical efforts. To keep the perfusion setup as simple as possible, dextran was used instead.
To test whether hypothermic storage might induce renal damage and disturbance of physiological renin release, additional experiments were conducted on isolated perfused mouse kidneys that had been stored on ice for 1 h before perfusion. Renin secretion rates and renal perfusion were normally regulated in these stored kidneys, indicating that hypothermic storage per se does not affect regulation of these parameters of kidney function (Supplementary material).
Control of renin secretion and perfusate flow by classical regulators
Isoproterenol
Ten IPPKSs were perfused at 100 mmHg. After a control period 10 nM isoproterenol was administered and resulted in a significant increase in renin secretion rate from 76.3 ± 24.7 to 208.9 ± 74.7 ng angiotensin I/(h * min * g) (p <0.05), while no noticeable vasoreaction was observed (Figure 5, top row, and Figure 7 in Supplementary material, panel A).
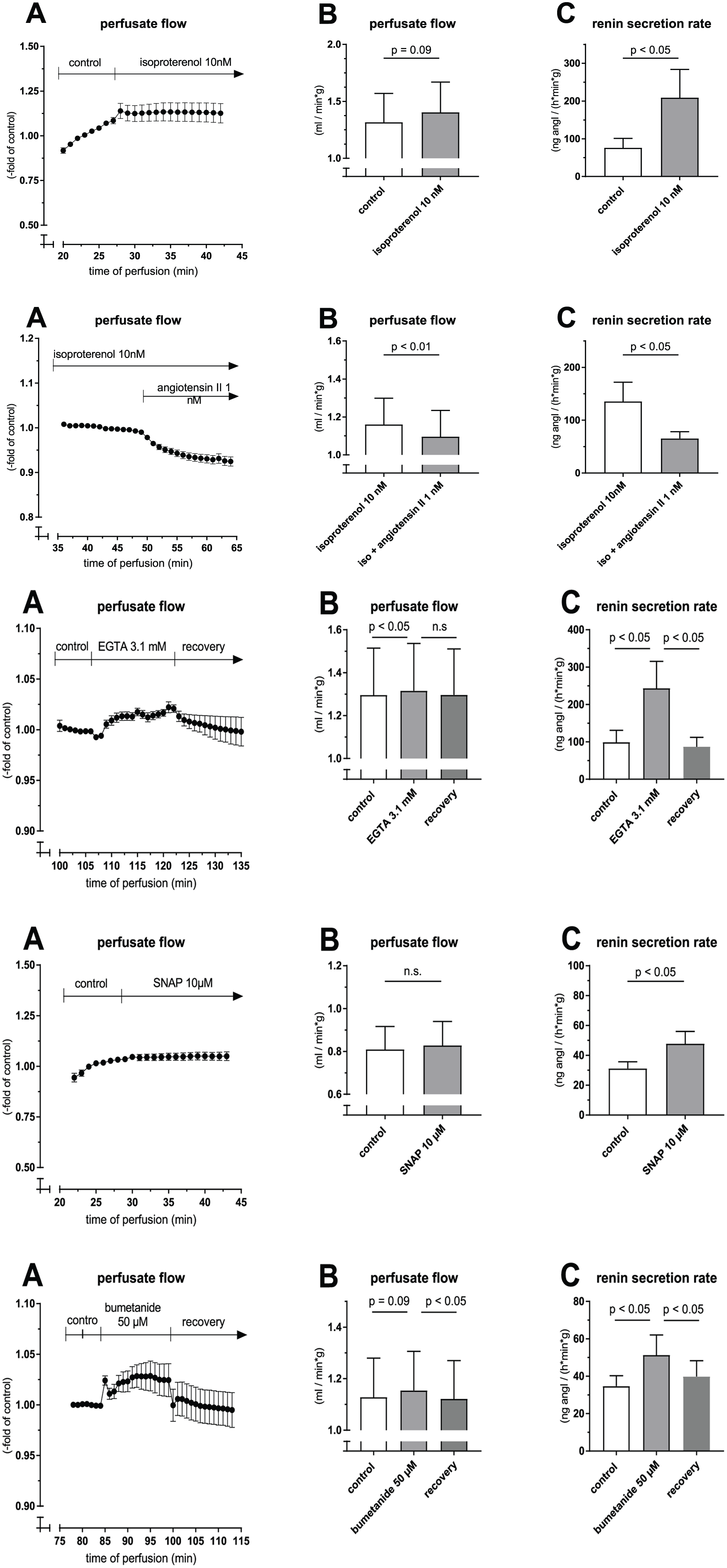
Response of perfusate flow and renin secretion to different pharmacological interventions. Top row: perfusate flow A + B and renin secretion rate C in 10 IPPKSs following administration of 10 nM isoproterenol; second row: perfusate flow A+B and renin secretion rate C in 23 IPPKSs following administration of 1 nM angiotensin II; third row: perfusate flow A+B and renin secretion rate C in 13 IPPKSs following administration of 3.1 mM EGTA; fourth row: perfusate flow A+B and renin secretion rate C in nine IPPKSs following administration of 10 µM SNAP; bottom row: perfusate flow A+B and renin secretion rate C in eight IPPKSs following administration of 50 µM bumetanide.
Angiotensin II
Twenty-three IPPKSs were perfused until steady perfusate flow was established at 100 mmHg for 15 min. Since renin release rates are rather low under control conditions and it was expected that angiotensin II would further suppress renin secretion, 10 nM isoproterenol was used to prestimulate renin release rates. In fact, 1 nM angiotensin II significantly suppressed prestimulated renin secretion rates from 135.4 ± 36.5 to 65.6 ± 12.6 ng angiotensin I/(h * min * g) (p < 0.05). Additionally, perfusate flow mildly decreased from 1.16 ± 0.13 to 1.10 ± 0.13 ml/min per g (p < 0.001) (Figure 5, second row). As shown in Supplementary material Figure 7 (panel B), all but one kidney responded with a decrease in perfusate flow.
EGTA
It has been shown in rodent kidneys that an inverse relation between calcium and renin release exists, the so-called calcium paradoxon. To test whether such a relation also exists in porcine kidneys the calcium chelator EGTA was used to reduce the free extracellular calcium concentration. Administration of 3.1 mM EGTA in 13 IPPKSs stimulated renin secretion rates from 99.3 ± 31.6 ng to 243.4 ± 72.0 ng angiotensin I/(h * min * g) (p < 0.05). There was also a slight increase in perfusate flow from 1.29 ± 0.22 to 1.32 ± 0.22 (p < 0.05; Figure 5, third row and Supplementary material Figure 7(c)). The effect on renin secretion but also vasodilation was fully reversible upon the stopping of EGTA infusion (p < 0.05 for renin secretion rate; Figure 5, third row).
SNAP
Administration of the NO-donor SNAP in nine IPPKSs stimulated renin secretion from 31.2 ± 4.5 to 47.8 ± 8.3 ng angiotensin I/(h * min * g) (p < 0.05) but did not alter the perfusate flow (Figure 5, fourth row and Supplementary material Figure 7(d)).
Bumetanide
The loop diuretic bumetanide stimulated renin secretion from 34.7 ± 5.6 to 51.3 ± 10.8 ng angiotensin I/(h * min * g) (p < 0.05, n=8). Perfusate flow tended to be higher under bumetanide treatment but did not reach statistical significance. Stopping bumetanide infusion rapidly restored both parameters to control levels (Figure 5, bottom row and Supplementary material Figure 7(e)).
Discussion
Regulation of renin secretion and flow rate in IPPKS compared with isolated perfused kidneys
The main objective of the present experiments was to develop a model for the isolated perfusion of kidney segments with a view to applying it to segments of human kidneys in the next step. In addition to the advantage of being able to investigate the regulation of certain parameters of kidney function directly in human tissue, this method would also lead to a reduction in the number of experimental animals used for isolated kidney perfusion. Since only segments will be available for perfusion rather than whole human kidneys, surgical separation of the segment will inevitably result in transection of renal tubules. Complex investigations of tubule function and urine collection will therefore not be possible. However, our results show that the effect of known regulators of renal blood flow and renin release is preserved in the fragmentary preparation of IPPKSs, so that isolated perfusion of human kidney segments could become the gold standard for the investigation of these parameters in the future.
Comparison with perfused human kidneys shows that the perfusate flow in IPPKSs and human kidneys are similar.14–16 For ethical reasons, healthy human kidneys are not used for basic scientific experiments and, accordingly, there are no studies on the regulation of renin release or blood flow by hormones such as angiotensin II or experimental substances such as EGTA. However, comparisons with human transplant kidneys that have been perfused at normothermic temperatures for organ preservation can be used. Here, perfusate flows of approximately 1–3 ml/min per g kidney weight,15,16 or 660 ml/min per kidney, 14 were reported. These values are in the same range as the perfusate flows of IPPKSs in the control phase, which were between 0.5 and 3.2 ml/min per g. Moreover, the release of renin from normothermic perfused transplant kidneys was also investigated.17,18 The determination methods used in these studies differ from those used in our study, and the lack of information on perfusate flow and kidney weight allows for only an approximate comparison. According to this, renin release in the IPPKSs under control conditions would be about five times higher than in perfused transplant kidneys. Whether this is due to the different species or differences in perfusate composition cannot be answered at this time. It should also be noted that the relative proportion of renin-producing cortical tissue is greater in IPPKSs than in the whole kidney, which has a large proportion of medullary tissue in which there are no renin producing cells. The reference of renin release to kidney weight therefore leads to lower values in perfused transplant kidneys. Given the overall large variation in renin release from perfused human kidneys, IPPKS, and also rodent kidneys, the calculated difference between transplant kidneys and IPPKS is within the range of variation. In addition, in studies on the regulation of renin release the effect of a certain hormone or substance is put into relation to the control value of the respective kidney in a before-after comparison, so that the absolute value in the control phase is of secondary importance.
In contrast to perfused human kidneys, in which no data on the regulation of blood flow or renin release by hormones or other interventions are available, there are numerous corresponding studies in isolated perfused kidneys from mice and rats. To verify whether the central regulatory mechanisms known from rodent kidneys are intact in IPPKSs, the results of this study will be compared with existing data from rodent kidneys. Administration of the β-adrenoreceptor agonist isoproterenol stimulated renin secretion in both isolated perfused mouse kidneys (IPMKs) and IPPKSs, albeit at different magnitudes. In stored mouse kidneys (see Supplementary material, Hypothermic ischaemic storage) in this study and previous studies in untreated mouse kidneys renin secretion rates were stimulated approximately 6× versus control and therefore more than twice as pronounced as in IPPKSs in this study. 19 IPMKs also showed a slight increase in perfusate flow rate that could not be observed in IPPKSs. While the effect of isoproterenol on renin secretion rates in IPMKs was more pronounced, the general direction shows an adequate response to β-adrenergic stimulation via isoproterenol in IPPKSs. 19
These observations translate to all the drugs that have been administered in IPPKSs in this study. Angiotensin II markedly decreased the renin secretion rate of both IPPKSs and IPMKs, 20 in the same manner that EGTA,19,21,22 SNAP 23 and bumetanide21,23,24 led to statistically significant increases in renin secretion rates of both models examined. The effect of EGTA and bumetanide in IPPKSs was fully reversible upon recovery, indicating that the observed preceding stimulation of renin secretion was not an effect of progressive kidney damage. There was no significant change in perfusate flow when administering isoproterenol, SNAP and bumetanide. Angiotensin II mediated statistically significant vasoconstriction while EGTA induced vasodilation in both models.
The non-selective beta-adrenoreceptor agonist isoproterenol is known as one of the most potent stimuli of renin secretion rate in isolated perfused mouse and rat kidneys and mimics the stimulatory effects of the sympathetic nervous system on renin release in vivo.4,21,22,25,26 Available data suggest an involvement of adenylyl cyclases AC5 and AC6 in mediating the stimulation of renin secretion through the sympathetic nervous system in mice.27,28 So far, this mechanism has been investigated only in isolated perfused rodent kidneys. The data presented in this paper show that IPPKSs react in a similar manner to rodent preparations to isoproterenol, allowing further investigation of the underlying mechanism in pig and, in a next step, potentially human kidneys.
The effect of angiotensin II on renin secretion rate is manifold. For one it acts as vasoconstrictor and stimulates aldosterone secretion and therefore renal sodium and water retention and activates the sympathetic nerve system, all leading to an increase in blood pressure resulting in an inhibition of renin release. Moreover, angiotensin II can inhibit renin release independent of its effects on tubular function and renal perfusion pressure, implying a direct effect on renin producing juxtaglomerular cells. 19 The effect of angiotensin II on renin secretion rate in pig kidney segments that are perfused at constant pressure and a perfusate with fixed composition indicate that the inhibitory effects of angiotensin II on renin release are directly mediated and not dependent on its systemic effects.
The cytosolic calcium level is the main inhibitor of renin exocytosis while cyclic adenosine monophosphate (cAMP) is the main stimulus for renin secretion. There is evidence for a crosslinking of both pathways through calcium inhibited adenylyl cyclase 5 and 6. Thus, an increase in intracellular Ca2+ mediates an inhibition of the calcium-inhibited adenylyl cyclases 5 and 6, resulting in a reduction of cytosolic cAMP, while a decrease of cytosolic calcium functions as stimulus to renin secretion rate by increasing intracellular cAMP levels. 27 Administration of the calcium chelator EGTA reduces the free cytosolic calcium concentration, providing a stimulus to renin secretion rate in isolated perfused rodent kidneys.19,21,22 Similarly, the data presented in this paper suggest potent stimulation of renin secretion rate in isolated perfused pig kidney segments through calcium chelation and might therefore provide the basis for further evaluation of the underlying mechanisms in human kidneys.
Similarly to angiotensin II the effect of the NO-donor SNAP on renin secretion is based on direct and indirect mechanisms. The indirect mechanism is based on a decrease in systemic blood pressure and as such is excluded in a pressure constant perfusion setting. The direct mechanism works via cyclic guanosin monophosphate (cGMP) mediated inhibition of phosphodiesterase 3, subsequent increase in cAMP levels resulting in a stimulation of the renin secretion rate. 23 As found in isolated rodent perfusions, SNAP led to an increase of renin secretion rate in IPPKSs at pressure constant conditions.
There are multiple ways loop diuretics such as bumetanide might act on renin secretion rate. In vivo, a decrease of extracellular volume can lead to an activation of the sympathetic nervous system as a countermeasure to hypotension providing a stimulus to the renin secretion rate. 20 Loop diuretics inhibit not just tubular but also macula densa salt transport, leading to prostaglandin release from macula densa cells and in turn an increased renin release.4,25 This effect of loop diuretics on salt transport is independent of systemic regulation and can be observed in isolated perfused rat and mouse kidneys.4,21,24 Tubular function of the IPPKS is impaired owing to the fragmentary nature of its preparation. 29 Yet administration of bumetanide leads to an increase of renin secretion rate as expected for fully functional kidney preparations. The effect of loop diuretics on renin secretion rate does not seem to be mediated only via NKCC2 leading to lower tubular salt load at the macula densa, but also by direct inhibition of NKCC1 in renin producing juxtaglomerular cells.19,30 Pharmacological blockade of NKCC1 can therefore provide a stimulus to renin secretion despite impaired tubular function. 30 Data from IPPKSs seems to support the hypothesis of predominantly direct and macula densa independent stimulation of renin secretion rate further reflecting the proposed mechanism of blocking NKCC1. 20
As mentioned above, some interventions showed a less pronounced effect on renin secretion and especially flow rate in IPPKSs than in previously cited IPMKs. In the case of angiotensin II, decrease in renin secretion rate was 1.5 times more pronounced in IPMK than in IPPKSs, while decrease in perfusate flow was more than 13 times more pronounced in IPMK than in IPPKSs. The potential reasons for these differences are discussed in detail in the Supplementary material.
In conclusion, agents known to regulate renin release in other species, such as isoproterenol, angiotensin II, EGTA, SNAP and bumetanide, had qualitatively the same effects on renin secretion rates in the new established IPPKS model. Stimulation of renin release by isoproterenol was completely reversed by angiotensin II, as were the effects of EGTA and bumetanide by recovery. In direct comparison with IPMK, 12 the observed effects were generally smaller, which may partially result from the fragmentary nature of the preparation as well as interspecies differences.
Optimization of the setup
The perfusion experiments described in this paper have been carried out on a basic apparatus for pressure constant single-pass perfusion. Keeping the setup as simple as possible was a conscious decision, to enable easy replication, encouraging researchers looking for an alternative to rodent based isolated perfused kidney experiments. For that reason, several points of potential adaptations of the setup according to the needs of the investigators are discussed in the Supplementary material.
Limitations and chances
The results indicate a possible use of IPPKS as an alternative to IPMK and, from a long-term perspective, human kidney segments regarding questions concerning renin secretion rate. While there are still advantages of isolated perfused rodent kidneys, the preparation is laborious and takes a skilled researcher to carry out consistently. This may prevent small scale experiments involving isolated perfused kidneys. Isolated perfusion of pig kidney segments, on the other hand, is much more straightforward, can easily be taught and a trained researcher can carry out 3–4 perfusions per day while IPMK is usually limited to two perfusions, depending on the length of the perfusion protocol.
The primary aim of this study was to establish a model that could serve as fundament for a method of perfusing human kidney tissue obtained from partial or total nephrectomies – thus evading the need for any animal tissue to be used at all. The data we have collected speak for a realistic implementation of such an endeavour when it comes to the examination of renal hormone regulation and the regulation of renal cortical blood flow.
One limitation of perfusion of surgically dissected kidney segments is that urine cannot be collected for further analysis. This means that direct examination of the glomerular filtration rate or tubular function is not possible, which limits the application of the described method. A partial solution to this problem would be to combine the method with multiphoton microscopy. Multiphoton microscopy is a very helpful and powerful method for investigating, among other things, glomerular filtration rate and the glomerular filtration barrier. 31 However, the technical and financial costs of operating the necessary equipment are high, which might prevent its widespread use.
As for the technical aspect of the preparation: caudal segments of human and pig kidneys share similarities in regard to anatomical as well as physiological aspects. Large urology departments often perform more than 100 nephrectomies per year, ensuring a constant supply of tissue. Furthermore, this tissue is in many circumstances discarded as excess material and perfusion would lend a purpose to this valuable resource otherwise potentially wasted.
Summary
A model for isolated pressure-constant perfusion of kidney segments in a single-pass setup was developed. Isolated perfusion of pig kidney segments is a suitable alternative to isolated perfusion of complete organs when investigating renal hormone secretion and cortical blood flow. This opens up ways to examine the renal hormone regulation of a wide array of different species, including human kidneys. Anatomical and physiological aspects speak for a possible translation of the preparation as described in this paper on human tissue obtained from nephrectomies. With concerns regarding animal welfare in research the work presented in this paper might very well encourage researchers seeking an easily established alternative to rodent isolated perfused kidneys to make use of precious human tissue otherwise frequently discarded.
Supplemental Material
sj-docx-1-lan-10.1177_00236772251369180 – Supplemental material for Isolated perfusion of pig kidney segments to study renal function: a translational model for human kidneys
Supplemental material, sj-docx-1-lan-10.1177_00236772251369180 for Isolated perfusion of pig kidney segments to study renal function: a translational model for human kidneys by Thomas C Beham and Frank Schweda in Laboratory Animals
Footnotes
Author contributions
Both authors were fully involved with carrying out the experiments, doing the statistical analysis and interpretation of the results.
Consent for publication
Not applicable.
Consent to participate
Not applicable.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Ethical considerations
Ethical approval was not required as the perfused kidneys were acquired as a waste product of slaughtering.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation), Project TRR374 (grant number 509149993).
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
