Abstract
Parasitic infections are a concern in animal facilities, in view of their influence on physiological processes and the immune status of animals. Pinworms are effectively controlled with the anthelminthic fenbendazole (FBZ, [5-(phenylthio)-1
Experimental autoimmune encephalomyelitis (EAE) is a model of neuroinflammation used for investigations of immunopathogenetic mechanisms that may underlie multiple sclerosis (MS).
1
The disease is induced by active immunization with a myelin protein-derived peptide in complete Freund's adjuvant, in susceptible rodents or non-human primates.
2
Protocols also include injection of toxin from
Studies described here are based on two mouse strains: C57BL/6J (Animal Resources Centre, Canning Vale, WA, Australia) and non-obese diabetic (NOD)/ShiLtJ (Walter and Eliza Hall Institute, Kew, VIC, Australia). Our institutional animal facility complies with regulations set by the National Health and Medical Research Council of Australia (NH&MRC, care of animals site: nhmrc.gov.au), which is the national governing body for overseeing animal experimentation. All procedures require approval from the institutional animal ethics committee. Mice are housed in an air-conditioned room with 12:12 h light-dark cycle and are fed with standard rodent chow (Barastoc Rat and Mouse Feed, Ridley AgriProducts, Melbourne, VIC, Australia) and tap water
FBZ is delivered orally by premixing into rodent chow at a rate of 150 mg/kg of diet. This assumes an average daily food intake of 8 mg/kg body weight, sufficient to ensure a therapeutic dose.
6
In some animal facilities, FBZ-medicated chow is administered according to a rolling regimen, while in others it is delivered when required, for periods of up to 14 weeks. Following the discovery of pinworm eggs by perianal tape testing and fecal flotation of caecal contents in mice in the containment room, subsequently confirmed to be of the species
Because our studies are focused on early pathological events, it is crucial to monitor the reproducibility of disease pattern. It became apparent that coinciding with FBZ treatment, reduction in disease incidence, clinical onset, disease duration and severity, within positive control groups of both models, was observed (Table 1). Disease incidence was reduced to about 75% overall. At the time coinciding with the first attack in NOD/ShiLtJ mice (14–16 dpi), only 38.6% of mice demonstrated any clinical scores (Group A), which were low (average of 1) and of short duration (1 day). By 30–40 dpi, a further 7.7% first demonstrated clinical scores (Group B), which were again low (1.5) and of short duration (1 day). However, the single mouse within Group B did demonstrate further attacks. A third subgroup experienced their first attack with a long delay, with 23.1% at 50 dpi (Group C) and 7.7% above 80 dpi (Group D). Surprisingly, in the latter two groups some mice had severe attacks with clinical scores of up to 3–3.5, with lesions of similar histological appearance to those normally observed (not shown). The remaining mice never experienced any attacks (Group E). The effect was less severe in C57BL/6J mice, with 62.5% showing overt disease at the anticipated time of 14–16 dpi and reaching scores of 3 or above (Group F), while 12.5% first demonstrated clinical scores at 30–40 dpi (Group G). The remaining 25% never demonstrated overt disease (Group H). The mice used in these experiments were born prior to FBZ treatment. Their weight relative to age at the time of disease induction was normal and weight increase over the period of FBZ treatment appeared normal.
Effect of FBZ on disease incidence, clinical onset and disease severity on the EAE model
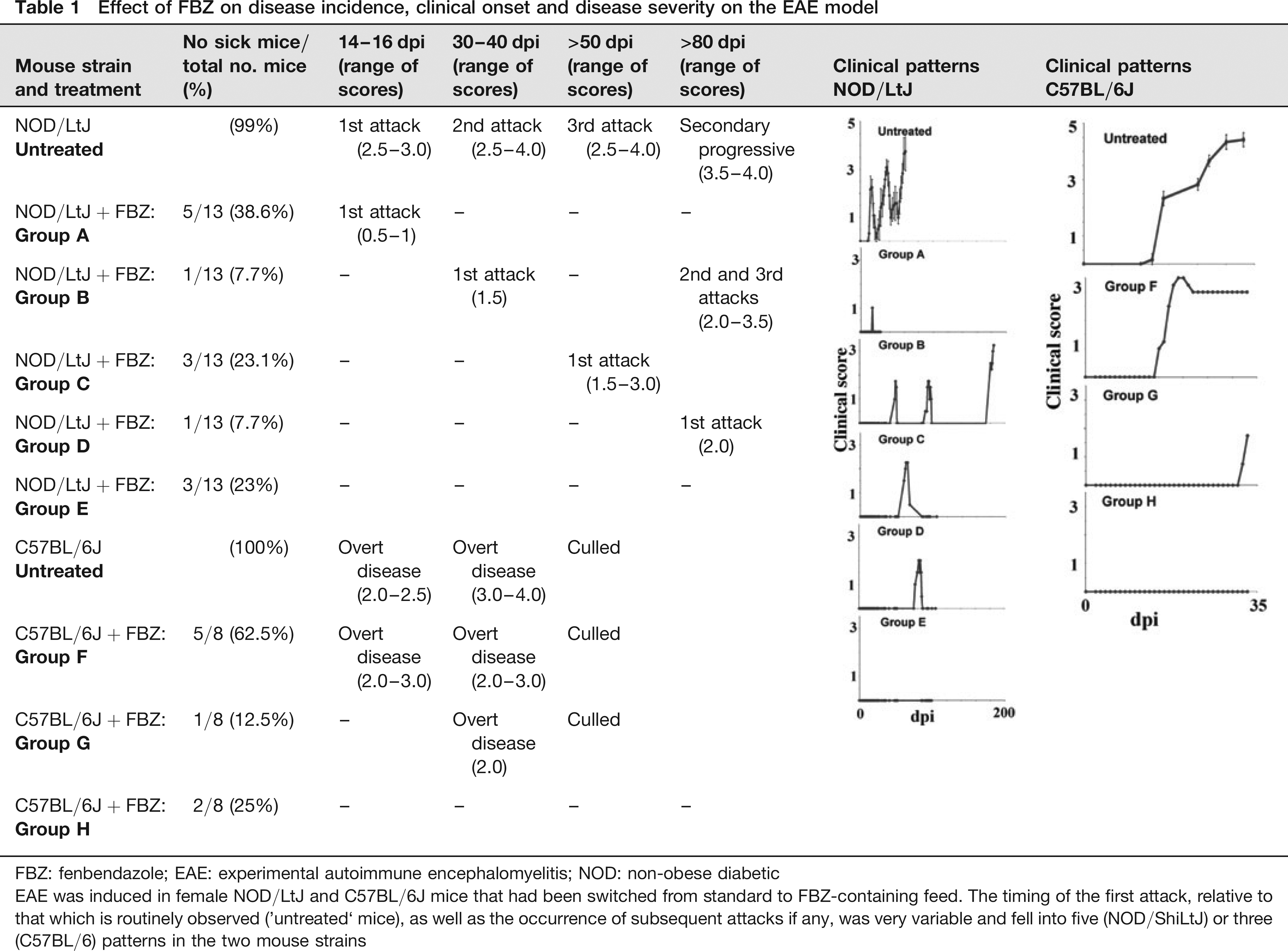
FBZ: fenbendazole; EAE: experimental autoimmune encephalomyelitis; NOD: non-obese diabetic
EAE was induced in female NOD/LtJ and C57BL/6J mice that had been switched from standard to FBZ-containing feed. The timing of the first attack, relative to that which is routinely observed (‘untreated’ mice), as well as the occurrence of subsequent attacks if any, was very variable and fell into five (NOD/ShiLtJ) or three (C57BL/6) patterns in the two mouse strains
Following discontinuation of treatment and a lag period of three weeks on normal chow, EAE was induced in a small group of NOD/ShiLtJ mice (
FBZ acts by inhibiting the polymerization of tubulin into microtubules thereby blocking mitosis, its anthelminth effect resulting from greater affinity for parasite relative to mammalian microtubules.
7
The mode of action of FBZ differs from that of other drugs affecting microtubule dynamics, such as paclitaxel, which enhance microtubule polymerization and interfere with normal microtubule formation during mitosis, migration, Chemotaxis and intercellular transport and have been demonstrated to ameliorate EAE.8,9 The wide use of this drug, resulting from its efficacy as a prophylactic parasite treatment, has caused concern on potential effects on experimental designs. Studies relating to immunological parameters have shown apparently contradictory outcomes. In one,
10
prolonged FBZ exposure (23 weeks) of NOD/ShiLtJ mice, susceptible to spontaneous development of type 1 diabetes, had no effect on the onset, progression or incidence of the disease. Furthermore, various lymphocyte sub-populations, as well as the proliferative response of T-cells to concavalin A appeared unaffected. Another study
11
using BALB/cN mice reported severe effects on the humoral immune system. mRNA and protein levels for the transcription factor E2A, which regulates expression of several B lineage genes as well as immunoglobulin rearrangements, were significantly reduced in bone marrow precursor B lymphocytes, resulting in decreased function of activated B lymphocytes. In a recent study, a direct effect of FBZ on T-cell proliferation and activation
EAE is mediated by activated T-cells, but parasitic infections promote a T helper 2 (Th2)-type response, which would counterbalance the pathogenic Th1 cells responsible for inflammatory demyelination. It is unlikely, however, that the parasitic infection (or parasitic infection alone) was the basis for the altered profiles, for the following reasons. Firstly, the change in disease profile was clearly coincident with the introduction of medicated feed. By the time this was implemented infection of the whole rodent colony was likely, yet no alterations in clinical profiles was observed. Secondly, the infection was under control within four weeks, but aberrant profiles were observed for the full duration of FBZ treatment. Thirdly, in view of the reported studies in other species, it is likely that FBZ adversely affects selected immune responses in mice.
Variants of EAE clinical profiles result from specific antigen/mouse strain combinations.2,3 Our data show that NOD/ShiLtJ mice were more severely affected in terms of disease severity and lag time before the first appearance of clinical disease, relative to C57BL/6J mice. The sum of our observations, therefore, suggests that FBZ treatment causes or contributes to an altered EAE profile, with different severity in different mouse strains. This may explain the inconsistent reports on the effects of this drug on the murine immune system and argues in favour of full evaluation of the effect of the drug on the disease course in specific experimental models prior to data collection.
Footnotes
Acknowledgements
The authors would like to thank the National Multiple Sclerosis Society and the Trish MS Research Foundation for their support.
