Abstract
Summary
Unintentional infection of laboratory rodents can compromise scientific research as well as the health of the animals and animal handlers. The source of contamination often is unknown, but may be introduced by wild rats from surrounding environments. To determine whether rats in Baltimore, Maryland, USA carry infectious agents commonly found in laboratory rodent colonies, we live-trapped 162 rats during 2005 to 2006 and screened them for a panel of viruses, bacteria and parasites. Antibodies against rat coronavirus/sialodacryoadenitis virus (91.7%),
Unintentional introduction of infectious agents into laboratory rodent colonies may cause serious complications in biomedical research (Baker 1998). In addition to compromising data recovered from infected rodents, the health of laboratory colonies and animal handlers may be at risk during an outbreak. Infection of laboratory rodent colonies occurs worldwide and the mode of pathogen introduction often is unknown (Baker 1998, Jacoby & Lindsey 1998, Zenner & Regnault 2000, Livingston & Riley 2003, Gaertner 2004, Clifford & Cosentino 2006, Schoondermark-van de Ven
Methods
Wild-caught rats
Adult male and female
Serological analyses
Serum samples were submitted to a commercial laboratory (BioReliance Invitrogen Bioservices, Rockville, MD, USA) for CARB, H-1, KRV,
Serological screening programme (Bioreliance, Inc)
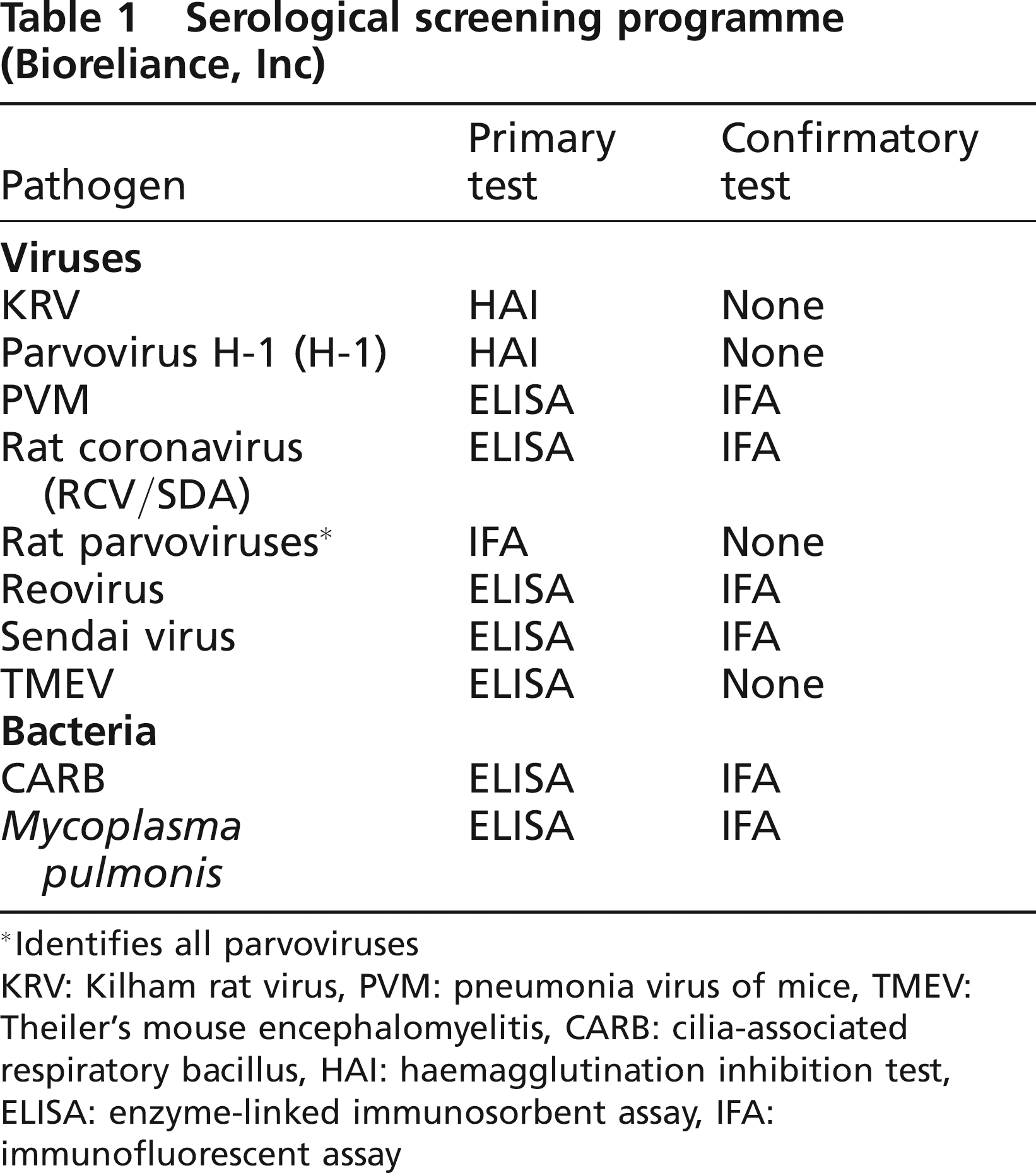
Identifies all parvoviruses
KRV: Kilham rat virus, PVM: pneumonia virus of mice, TMEV: Theiler's mouse encephalomyelitis, CARB: cilia-associated respiratory bacillus, HAI: haemagglutination inhibition test, ELISA: enzyme-linked immunosorbent assay, IFA: immunofluorescent assay
Helminth identification
Helminth ova were identified following faecal and caecum content floats. Briefly, faecal and caecum content samples were homogenized in zinc sulphate buffer (400 g/L) in glass test tubes and filled to the brim with buffer. A coverslip was placed on top for 15 min and transferred to a slide for microscopic evaluation (400 × final magnification). Clear tape was applied to the anus, transferred to a glass slide and evaluated under the microscope for pinworm (
Statistics
Differences in pathogen prevalence by age and seasonality were evaluated by χ2-tests. Body mass was used as a correlate of age as follows: juveniles were <200 g (
Results
Prevalence of rodent-borne pathogens
A total of 162 wild rats were trapped as part of the study. The panel of pathogens for this study included the panel used for sentinel rats at the Johns Hopkins University Medical Institutions (JHMI) in Baltimore, Maryland. Pathogens that could be visually identified (i.e. nematodes and ectoparasites) also were included. For those pathogens that were screened for the sentinel panel of pathogens, cost constraints limited the numbers of individuals that could be tested; for those agents a subsample of individuals (
Prevalence of pathogens in wild-caught Norway rats
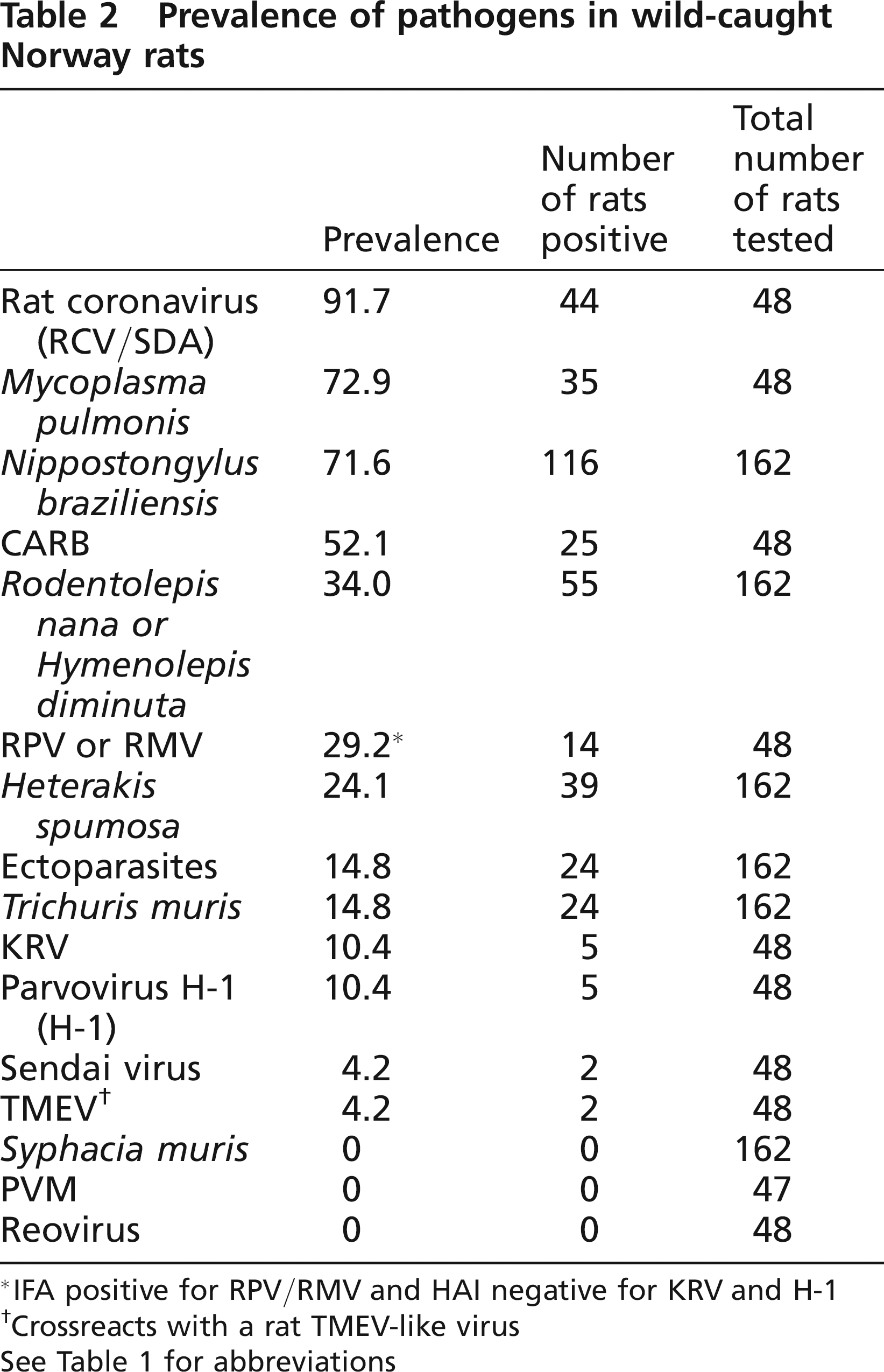
IFA positive for RPV/RMV and HAI negative for KRV and H-1
Crossreacts with a rat TMEV-like virus
See Table 1 for abbreviations
Age
The distribution of rats by age was variable for the helminth panel (juveniles,
Season
Rat trapping success was not evenly distributed across seasons due to a rainy fall in 2005 (spring,
Correlations
There was a correlation between infection with
Discussion
Historically, pathogens reported in laboratory rodent colonies reflected those found in the wild because rodents were housed in open cages in large rooms with no barriers against the spread of infection between laboratory rodents or from wild rodents entering the facility (Lindsey & Baker 2006). Initial attempts were made to eliminate pathogens that caused apparent morbidity or mortality in rodents. Due to technical advances, several non-pathogenic infections were discovered in laboratory rodent colonies (Baker 1998). Although not pathogenic to rodents, infections were reported to alter research data (e.g. cellular and humoral immune responses); thus, modern research facilities have much lower prevalences of pathogens due to the availability of pathogen-free rodents from vendors, improved infection control procedures in research institutions, and routine use of surveillance programmes to diagnose colony infections (Compton
As a consequence, recent reports indicate a major reduction in pathogens in laboratory colonies (Lindsey & Baker 2006). The most commonly reported rat-borne pathogens detected in laboratory rat colonies in the United States remain pinworms (i.e.
Although outbreaks may be primarily caused by introduction of an infected laboratory animal or animal product (i.e. transplantable tumour or serum), the source of contamination often is unknown. Wild rodents may contribute to laboratory outbreaks and have been implicated in prior outbreaks of infectious agents in laboratory colonies (Skinner
The JHMI in Baltimore, Maryland are located in an urban environment and Norway rats are commonplace in the surrounding neighbourhoods (Easterbrook
Pinworms were not found in wild rats in Baltimore; therefore, pinworm outbreaks at JHMI and other research institutions are most likely being perpetuated by environmental contamination or laboratory rodents.
Antibodies against
The effect of age on the serological analyses may be an artefact because only one juvenile rat was included. Juveniles were not selected for the serological screening because maternal antibodies are likely present in young animals and may overestimate seropositivity in juveniles and young rats have less time to have been exposed to a pathogen, thus underestimating the overall prevalence. Increasing age-related prevalence of
Ectoparasites were collected more frequently during the summer and fall than spring and winter. Whether temperature contributes to the prevalence of ectoparasites in wild-caught rats requires additional consideration.
Presence of antibodies to CARB and
Introduction of zoonotic pathogens into laboratory rodent colonies may not only compromise research, but also the health of the rodents and laboratory personnel. Infectious agents currently found in laboratory rodent colonies do not produce overt pathology in immunocompetent rodents, but still may alter results obtained for scientific research (Baker 1998). Increasing numbers of genetically altered rodents are utilized in research and may be more susceptible to morbidity and mortality caused by pathogens commonly identified in laboratory rodent colonies (Rozengurt & Sanchez 1993, Baker 1998). Laboratory animal handlers also are at risk to zoonotic pathogens carried by laboratory rodents. Antibodies against zoonotic agents, including Seoul virus (the hantavirus carried by Norway rats; 57.7%), hepatitis E virus (HEV; 73.5%),
Wild populations of Norway rats harbour many rodent-specific and zoonotic pathogens (Webster & Macdonald 1995, Easterbrook
Footnotes
Acknowledgements
We would like to thank Darren Kaw for help with trapping the rats during summer 2005 and Andrew Glenn and Bruce Baldwin for help in identification of helminth adults and ova. Financial support was provided by NIH grant R01 A1054995 (SLK), Research Animal Resources (Johns Hopkins University) and NSF grant EF-0525751 (GEG).
