Abstract
Experimental autoimmune encephalomyelitis (EAE) is a frequently used animal model for the investigation of autoimmune processes in the central nervous system. As such, EAE is useful for modelling certain aspects of multiple sclerosis, a human autoimmune disease that leads to demyelination and axonal destruction. It is an important tool for investigating pathobiology, identifying drug targets and testing drug candidates. Even though EAE is routinely used in many laboratories and is often part of the routine assessment of knockouts and transgenes, scoring of the disease course has not become standardized in the community, with at least 83 published scoring variants. Varying scales with differing parameters are used and thus limit comparability of experiments. Incorrect use of statistical analysis tools to assess EAE data is commonplace. In experimental practice the clinical score is used not only as an experimental readout, but also as a parameter to determine animal welfare actions. Often overlooked factors such as the animal’s ability to sense its compromised motoric abilities, drastic though transient weight loss, and also the possibility of neuropathic pain, make the assessment of severity a difficult task and pose a problem for experimental refinement.
EAE: a model for multiple sclerosis
Experimental autoimmune encephalomyelitis (EAE) is the most commonly used animal model for multiple sclerosis (MS), an autoimmune demyelinating disorder of the human central nervous system (CNS). MS affects 2.5 million people worldwide with a preponderance in higher latitudes and developed countries. Usually MS commences in early adulthood and is more common in females. Afflicted individuals develop motor impairment and cognitive dysfunction.1,2 Disease severity can be assessed using the expanded disability status scale (EDSS), 3 a scale based mainly on a combination of functional systems and ambulation. It serves to document the course of the disease and to escalate therapy if necessary. It is also used in clinical trials where it helps to assess the efficacy of the therapeutic agent. To ensure inter-rater standardization, training in the use of the EDSS score is delivered by an independent online platform. 4 Although MS is a uniquely human disorder not observed spontaneously in other species, animal models have helped greatly in increasing our knowledge of MS. They served as useful tools in investigating the dynamics of both the immune system and the CNS during neuroinflammation. Accordingly, many of the MS drugs in use and under testing in humans have been developed on the basis of experimental data coming from EAE. 5 EAE is, however, not a single model but consists of a family of animal models induced through different protocols, each serving a different experimental purpose. It was first described in the 1930s while investigating the neurological complications arising after the rabies vaccination. 6 After a series of studies that showed myelin destruction and perivascular infiltration in the CNS 7 in 1947, similarities between EAE and human MS were described. 8 Since then, EAE was established in a variety of mammals such as monkeys, guinea pigs, cats, goats, primates, rats and mice, and was used to investigate the pathobiology of MS. 9 In mice, active EAE is induced by subcutaneous immunization with myelin components and adjuvants. Self-tolerance is broken and encephalitogenic effector T cells migrate into the CNS to attack the myelin sheath. 10 The first clinical signs of disease, characterized by ascending flaccid paralysis, appear 7–8 days after immunization, with disease peaking often between days 14 and 15. The most commonly used antigens, derived from myelin, are proteolipid protein (PLP), myelin oligodendrocyte glycoprotein (MOG), and myelin basic protein (MBP). In both MS and EAE, the CNS is infiltrated by T cells, B cells and macrophages. 11 Nevertheless, other aspects of the disease differ between patients and can be modeled in EAE. For instance, induction in C57BL/6 mice using MOG35-55 peptide emulsified in complete Freund’s adjuvant (CFA) and followed by Pertussis toxin (PT) injection usually results in chronic disease. 12 On the other hand, induction in SJL/J mice using PLP131-151 peptide in CFA results in a relapsing–remitting pattern. Adoptive transfer EAE, or passive EAE, is a model in which encephalitogenic T cells are transferred from myelin-immunized or diseased mice to naïve recipient mice. 10 This method allows the direct assessment of the effector phase of EAE, or the particular study of transferred cells types or hosts’ backgrounds. Apart from active and passive EAE, transgenic models were developed in which EAE develops spontaneously (reviewed by Croxford and colleagues). 13 One example are T cell receptor transgenic mice crossed with IgH knock-in mice, both specific for MOG,14 which develop disease within 28 days.
Scoring scales for EAE
Examples of experimental autoimmune encephalomyelitis (EAE) scoring systems.
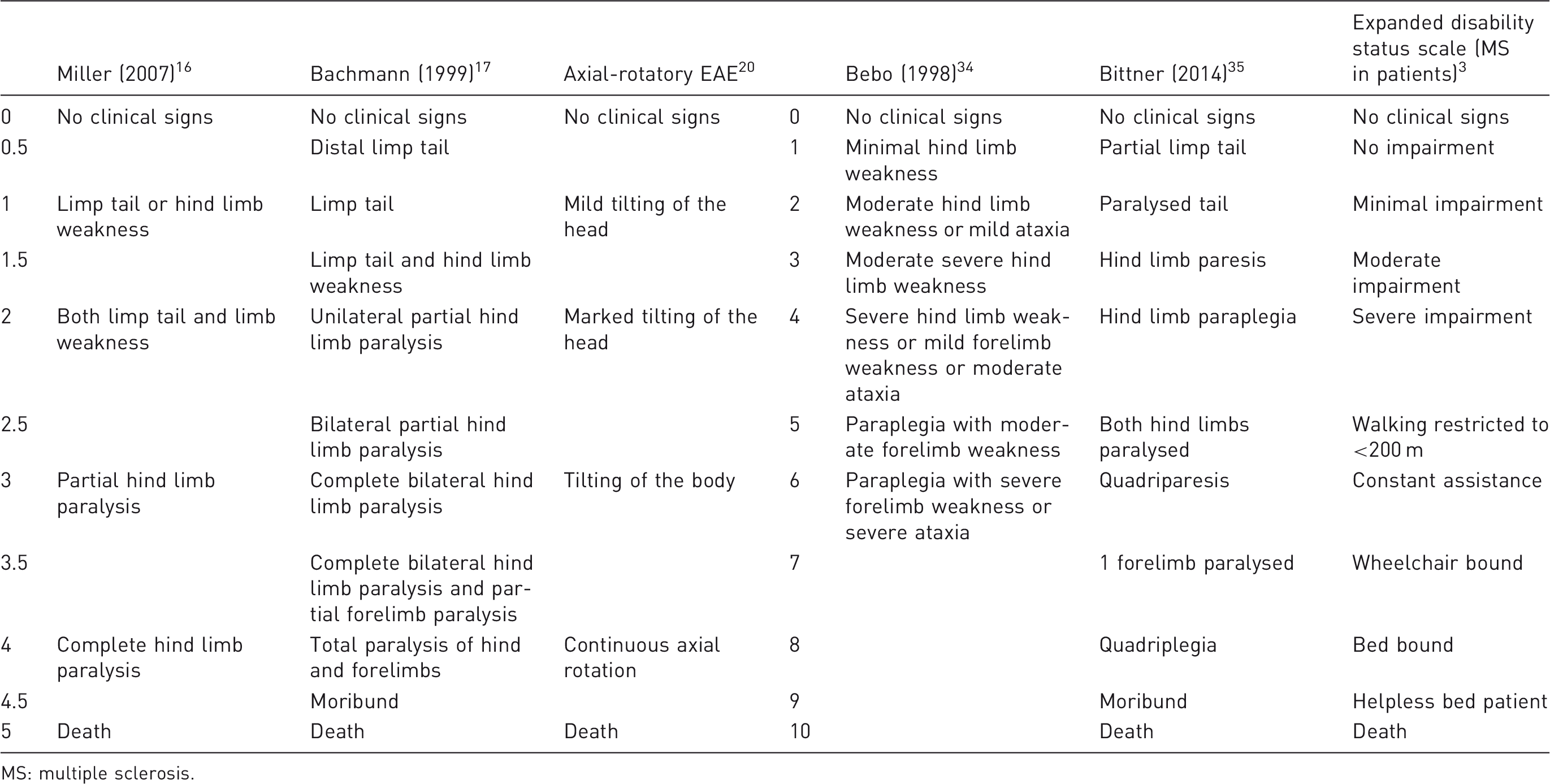
MS: multiple sclerosis.
Assessment of welfare
In most studies the EAE score, alone or in combination with other parameters such as weight, defines animal welfare actions. These commonly constitute provision of food and water on the cage floor, mostly in jellified form, and termination by euthanasia. In EAE, animal ill-being results from a combination of neurological deficits such as reduced motor control, the possibility of nausea or neuropathic pain, and features associated with any severe disease like weight loss and dehydration. Increasing loss of motor function clearly impairs the animals’ access to food and water, participation in social activities and their ability to fend off cage mates. Whether the animal realizes its disability and suffers purely from comprehending this remains an open and probably unanswerable question. In humans, hedonistic adaptation appears to allow paraplegic patients a similar quality of life as the healthy population. 22 Weight loss and dehydration, which are both easy to assess during daily monitoring of the mice, have direct welfare relevance and their assessment is often required by the responsible animal welfare authorities. Mice can adapt to a deprivation of up to 50% of water for one week. 23 Nevertheless, due to daily scoring in EAE, dehydration could be detected rather acutely by adopting the scale established by Bekkevold and colleagues. 23 In case of dehydration, an intraperitoneal administration of saline (maximum 80 mL/kg) 24 should be enough for full recovery. In contrast to dehydration, critical weight loss, often defined as a reduction of 20% in body weight, leads to termination of the experiment because mice must be euthanized to abide with humane endpoints. Although to the best of our knowledge there is no direct causal relationship between weight loss and disease progression; in EAE mice frequently lose weight transiently, correlating with higher scores and paralysis. 25 Thus, they are able to recover their weight when disease ameliorates. This raises the question of whether a 20% weight loss constitutes a good termination point. A report in ABH mice shows that these are highly susceptible to weight loss without any corresponding increase in disease severity. 26 In this study, mice lost around 26% of weight during acute disease, followed by almost complete recovery. Thus, if the authors had rigidly applied the frequently pre-set guidelines of 20% body weight loss as the endpoint, they would have killed most of their experimental subjects, without gaining insight into the relapsing–remitting phase of the disease that is characteristic of this strain and which can be a source of valuable clinical information. 26 Consequently, in EAE critical weight loss may have to be defined on a case-by-case basis in order to overcome the problem of losing statistical power and scientifically important data. In this regard, a European Union document with practical guidelines on how to implement Directive 2010/63/EU in EAE studies suggests using 35% weight loss as the humane endpoint, whenever applicable, in order to maximize 3R practice. 27 For ABH mice, an endpoint based on body temperature was suggested in one study: when body temperature decreases below 31°C, recovery is unlikely. 26 An even more stringent suggestion was made in a pertussis infection/vaccination study, where a lower body temperature limit of 34.5°C was shown to be a humane endpoint. Nevertheless, future research in EAE using C57/BL6 also has to show whether temperature could be a useful endpoint, and whether it justifies the additional handling of animals. 28
Refinement opportunities
Current clinical scoring of EAE suffers from some obvious problems, most of which can be easily overcome. Firstly, the community needs to come to agreement on a commonly accepted scoring scheme that allows comparison of experiments. This may well be based on a frequently practised 10-step scale. Also, since data from clinical EAE scoring are generated within non-linear scales, they must be analysed using non-parametrical statistical tests such as the Mann–Whitney U or Wilcoxon rank sum test. Even though this is long-known, 50% of reports feature parametrical statistics. 29 Application of correct tests and the corresponding power calculations would strongly increase reproducibility of EAE experiments. Blinding of EAE studies is rarely reported, making inadvertent biases highly likely. Since circadian rhythm affects immune responses and vice versa, induction and scoring should be performed at identical times during an experiment.30,31 Though only suitable for mice not yet paralysed, the use of quantitative motor function tests such as the grip strength and rotarod have been shown to help decrease bias. 25 Such motor assessment facilitates statistical analysis with parametric tests and thus increases power. Treating weight loss as a humane endpoint criterion should be questioned critically, as discussed above.
Littermate controls should be preferred over wild-type mice bought from commercial suppliers to ensure the genetic background differs only by the factor in study. Lastly, environmental stress and gut microbiome may influence disease outcome, 32 and should thus be similar between experimental groups. Hence, preference should be given to mixing mice of different experimental groups in the same cage from early age on and allowing them to adapt to housing conditions. 33 In conclusion, a unifying system capable of efficiently and objectively inducing and accessing disease progression and animal welfare without causing extra discomfort to the animals should be sought.
Recommendations and further 3R research
A common measurement in most EAE studies is the assessment of clinical symptoms using scoring scales, which not only yield experimental data but also define welfare actions. EAE experiments would profit greatly, if researchers were to implement the following points:
One common scale. Induction and measurement of disease at identical time of the day. Induction and measurement of disease in a blinded fashion. Use of littermate controls of the same genetic background and hosting similar microbiomes. Use of non-parametric statistics for data analysis and power calculation during experimental planning. Allowance of up to 35% transient weight loss, according to characteristics of the strain, EAE induction paradigm and aim of the study. Consistent use of jellified food/water as welfare action. Assessment of dehydration paired with respective actions.
When the aim of a study is to describe small differences at low scores, motor tests such as rotarod and grip strength should be used and may increase the power of the study. Future research has to show whether neuropathic pain constitutes a relevant animal welfare problem in EAE. In conclusion, EAE scoring scales are a good example of tools that represent well-established and common practice, but which need to be re-evaluated with a critical eye. Currently, the variety of scoring scales and their analysis may contribute to irreproducibility and failure in translation of animal experiments.
Footnotes
Acknowledgement
We thank Phillipe Bugnon for his helpful comments on the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Hertie Foundation, grant number P1140090, but otherwise received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
