Abstract
The use of group-housed rodents in many fields of biomedical research imposes a need to identify individuals in a cage. Few studies have been designed to assess possible negative effects of identification methods of newborn mice on their development and wellbeing. In the present study, three different identification methods were applied to newborn C57BL/6J mice on postnatal day (pnd) 5 (toe clipping, toe tattoo ink puncture and subcutaneous implantation of a small transponder). All identification methods used proved to be effective for long-term marking of individual animals. Newborn mice showed the least reaction to toe clipping followed by toe tattoo ink puncture and transponder implantation was the most distressful individual identification procedure in newborn mice. Importantly, clipped toe tissue proved to be enough for genotyping purposes. No overall consistent differences in somatic and neurological reflex development during the postnatal period were shown as a result of the newborn individual identification procedures used. Further, none of the methods interfered significantly with the adult animals' general normal behaviour (e.g. ability to move, grasp, climb) and sensory–motor functions as assessed with a simplified SHIRPA battery of tests, as well as Rotarod and Elevated Plus Maze tests. Postmortem thymus and adrenal gland weights gave no indication of chronic stress as a consequence of the identification method. We conclude that toe clipping might even be advisable in newborn mice at a very young age, when genotyping is needed. Toe tattoo ink puncture is also a good identification method for newborn mice and transponder implantation should only be used in older newborns or applied at weaning.
Rodents, especially mice and rats, are the most frequently used laboratory animals in biomedical research. Since they are usually group-housed, identification of group members is often necessary to differentiate individuals used for breeding purposes, as a source of tissues, cells or fluids, and in order to maintain health records and to properly manage colonies of laboratory animals in compliance with regulations and research protocol requirements. 1,2
Selection of the best method of animal identification depends on several factors, including species, duration of the study, age and skin pigmentation of the animals, and technical expertise availability. The ideal identification method should not only provide efficient individual identification and be technically easy to apply but should also avoid inducing pain and/or distress. Individual follow-up of group-housed mice might be needed from birth, e.g. when following their development while phenotyping a new mouse model or studying the effects of early life stress in newborn animals. Traditional permanent individual identification methods that can be applied to newborn mice include toe clipping 3,4 and body, tail or foot/paws tattooing. 5–7
Toe clipping has been widely used in the past, but is currently not recommended due to welfare and ethical grounds. 1,8,9 Recommendations and guidelines indicate toe clipping as a method only acceptable for altricial neonates (≤7 postnatal day [pnd]), when no other methods are feasible 9,10 and several Institutional Animal Care and Use Committees (IACUC) only allow toe clipping when strong scientific justification is presented. 11–15
Toe clipping can be applied to newborn mice as early as one day after birth, but it is easier to perform between pnd 5 and 7, when the toes are larger but ossification is not yet complete. 16 An advantage of toe clipping is that the tissue removed can be used for DNA extraction and genotyping, particularly useful when studying transgenes and/or mutations requiring analysis in the early postnatal period. 17
Identification of newborn mice by micro-tattooing can also be used as early as pnd 1 and is relatively easy to apply, but might need specific equipment (electric tattooing gun) and properly trained personnel able to perform the tattooing of very small numbers or marks on the tail or paw of the mouse with minimal distress to the animal. A more simple system using a fine gauge needle full of tattoo ink to inject small dots of ink in the paw, foot or toe of the animal is also available
18
(
Subcutaneously implantable transponders (microchip) using a system of radiofrequency identification (RFID) are routinely used since the early 1980s (Bio Medic Data Systems, Inc:
As for any other procedure applied to laboratory animals, identification techniques should not cause pain and/or distress to animals that can interfere with animal welfare and experimental results since animal welfare and reliable scientific results are inextricably connected. 22 It is, thus, important to know about the possible confounding effects of identification methods applied to mice at a very young age. Toe clipping is a controversial technique and the ethical justification to perform it has been recently discussed, since this identification method is likely to be painful and might impair the mouse's ability to grip, groom and feed, as well as to alter the animal's gait. 2,9 Reports indicate that tattooing newborn mice on their body (dorsal skin) within the first 24 h after birth does not increase mortality or cannibalism 5–7 and only minor short-term negative effects on the animal's welfare have been suggested by the authors in a study using C57BL/6JBomTac mice tattooed on the tail at 4, 12 and 20 pnd. Importantly, no differences in growth or emotionality were found in those animals. 23 Growth, maturation and reproductive capability were also not affected in newborn mice and rats individually identified by palm tattooing. 24 For transponders, a possible interference with the mouse's movements and posture has been suggested as a negative effect in mice. 9 Incidentally, the development of microchip-associated sarcomas in adult mice, which have been implanted in young/adult age (at or after weaning), has been reported in long-term carcinogenicity studies. 20,25,26
Few systematic studies have been designed to directly compare the possible negative effects of identification methods of newborn mice. In the present study, we performed a comprehensive assessment of the consequences of three different permanent identification methods of newborn mice (toe clipping, toe tattoo ink puncture and subcutaneous implantation of a small transponder). We evaluated which of the three methods would be easier to perform and more reliable, and which would cause the least pain and distress to the newborn. Further, we verified the viability of recently developed small transponders for long-term identification of newborn mice. In order to assess the impact of the acute stress/painful experience and/or the physical damage caused by the identification procedure on the normal development of the animals, we measured morphological condition, reflexes and sensory capabilities from pnd 5 until weaning (neurobehavioural neonatal development). Behavioural performance and motor abilities in the adult animals, which had been submitted to an identification procedure at pnd 5, were evaluated by measuring climbing behaviour and using a simplified SHIRPA battery of tests, as well as Rotarod and Elevated Plus Maze (EPM).
In this study we used the C57BL/6J mouse strain, an inbred strain commonly used in the production of genetically-modified animals and widely used in biomedical research.
Animals, materials and methods
Animals
C57BL/6J mice, 9 males and 18 females at seven weeks of age, specified pathogen free, were purchased from Charles River Laboratories, France, kept one week in a quarantine room and then transferred to a conventional maintenance room where they were used for breeding at 11 weeks of age (1 male × 2 females, per cage). The day after mating, females were checked for plugs and subsequently weighed twice a week to confirm pregnancy. Males were removed from the cage as soon as pregnancy was confirmed. Litters were born on gestation days 20–21 (counting the day of plug detection as gestation day 0) and the day of birth was considered pnd 0. The pups were weaned at pnd 21 and housed in groups of 4–5 according to gender. Litter mates were randomly distributed among cages according to their date of birth with animals in the same cage not differing by more than 2–3 days in age.
All animals were maintained under standard laboratory conditions: artificial 12 h light/dark cycle (lights on from 08:00 to 20:00 h), with an ambient temperature of 21 ± 1°C and a relative humidity of 50–60%; they were housed in filter-topped transparent cages 267 × 207 × 140 mm (370 cm2 floor area) (Tecniplast, Buguggiate, Italy), with corncob bedding (Scobis Due, Mucedola SRL, Settimo Milanese, Italy) and nesting material (autoclaved tissue paper); the mice were given standard diet (4RF25 during gestation and postnatal periods, and 4RF21 after weaning; Mucedola SRL) and water ad libitum. Health monitoring was performed according to FELASA guidelines 27 confirming the specified pathogen free health status of sentinel animals maintained in the same animal room.
All procedures were conducted in accordance with European regulations (European Union Directive 86/609/EEC).
Newborn's permanent identification methods
A total of 78 animals (39 females and 39 males) from 17 different litters were assigned randomly to four experimental groups per gender: three experimental groups to test three different permanent identification methods: Toe clip – 22 animals (11 females and 11 males); Toe ink – 20 animals (10 females and 10 males); Transponder – 16 animals (8 females and 8 males) and the Control group – 20 animals (10 females and 10 males).
The experiment started at pnd 5. To avoid a long handling period of the newborn, the identification methods, described below, were performed as rapidly as possible. The immediate reaction of the newborn to the identification procedure was followed by observing sudden movements and registering the presence or absence of vocalization and urination as indicators of pain/distress. 28
Toe clipping (toe clip group)
The newborn was gently held in one hand on top of a soft surface and its paw was cleaned with a 70% ethanol solution and extended between the thumb and index finger. One of the toes was then clipped in one foot per animal, with fine scissors (cat. # 14060-10, Fine Science Tools GmbH, Heidelberg, Germany), removing the distal part of the toe (approximately 1/3, corresponding to the first phalanx). A different toe was clipped in each animal in one of the four feet. Aseptic technique was used and the toe tissue removed from the newborns was collected in 1.5 mL microcentrifuge tubes at 4°C and preserved at −20°C for DNA extraction.
Toe tattoo ink puncture (toe ink group)
The newborn was gently held, in a similar way as used in toe clipping, the paw was cleaned with a 70% ethanol solution and a 30G needle, previously immersed in animal tattoo ink (green paste, Ketchum Manufacturing Inc, Canada), was used to puncture a different toe in each animal in one of the four feet. The needle containing tattoo ink was inserted into the superficial layer of skin and advanced 1–1.5 mm parallel to the skin surface along the toe length. In order to avoid the tattoo ink coming out of the toe, the needle was left on the toe for 2–3 s after which it was gently removed.
Transponder implantation (transponder group)
The newborn was placed on a clean paper towel with the four limbs facing down and held between the thumb and index finger. The dorsal skin on the back of the neck (injection site) of the newborn was first cleaned with a cotton swab previously immersed in a 70% ethanol solution. The tip of the sterile 18G needle assembly containing the identification transponder (1 mm diameter, 6 mm length and 7.15 mg weight) (NONA06VT02-B, NONATEC™, kindly provided by Lutronic International, Rodange, Luxembourg) was inserted subcutaneously at the back of the neck and the transponder was injected. The needle was removed carefully so as not to withdraw the transponder from the animal and forceps was used to gently massage the transponder under the dorsal skin of the animal, laterally and then caudally. After the loss of some transponders in preliminary procedures, massaging the transponder backwards and to the lateral side appeared to be necessary in order to prevent the mother from removing it from the pup by licking the injection site. A specific reader/writer (NONATEC V1, port USB) was used to detect the transponder code with the support of Nonatec PRO V1.2 software (NONATEC™, kindly provided by Lutronic International).
Injected controls (control group)
Injected control animals were handled and subcutaneously punctured with a 18G needle, in a similar way as was done with transponder subcutaneous injections.
Genomic DNA extraction from toe tissue
Clipped toe tissue samples kept at −20°C were unfrozen at room temperature and 15 µL of ATL buffer (tissue lysis buffer, cat. # 19076, Qiagen; Izasa, Lisbon, Portugal) and 10 µL of proteinase K, at 14–22 mg/mL in 10 mmol/L Tris-HCl, pH 7.5 (cat. # 03 115 828 001, Roche Applied Science; Roche, Lisbon, Portugal) were added. The mixture was vortexed and incubated at 55°C overnight for tissue digestion. Proteinase K was then inactivated at 98°C for 10 min; 100 µL of protein precipitation solution (cat. # PP-350, Citogen; Citomed, Lisbon, Portugal) was added to each sample tube, followed by shaking and centrifugation at 13,000 rpm for 4 min. The supernatant was then transferred to new 1.5 mL microcentrifuge tubes containing 200 µL of isopropanol for DNA precipitation. After inverting the tubes several times, samples were submitted to 10 min centrifugation at 13,000 rpm. The supernatant was then discarded and the DNA pellet was washed with 200 µL of 70% ethanol by inverting the tubes carefully several times, after which they were centrifuged for one minute at 13,000 rpm. The ethanol solution was then removed carefully and the pellet was allowed to dry at room temperature. As soon as the DNA pellets were dried, they were suspended in 20 µL of TE buffer pH 8 (10 mmol/L Tris-Cl, pH 8, 1 mmol/L EDTA, pH 8), mixing up and down with the aid of a micropipette and incubating the samples for one hour at 65°C. Quantification of the DNA in each sample was performed by spectrophotometry (reading optical density at λ = 260 nm), using NanoDrop ND-1000 (NanoDrop Technologies, LLC, DE, USA).
Test procedures with newborns
From pnd 0 to pnd 4, animals were observed for skin appearance, activity and presence of milk in the stomach (milk spot) to confirm feeding (as a wellbeing indicator 9,29,30 ).
From pnd 5 to pnd 21 the cages containing mother and pups were taken daily to an experimental room at the beginning of the light period and all parameters of morphological development and neurological reflexes were measured starting at 08:30, by the same experimenter (MJCC), randomly choosing the order of the litters and animals within litters tested. The experimenter only verified the identification of the animal at the end of the procedures. On pnd 5 all measurements were made before the application of the identification method.
Somatic parameters
As a measure for morphological development, animals were weighed on a precision balance for live rodents (Explorer Pro, Ohaus), and then, holding the pup in one hand between the thumb, index finger and middle finger, with the four limbs facing upwards, anogenital distance was measured in millimetres, using a ruler (previously cleaned with a 70% ethanol solution). Incisor eruption was also verified daily when holding the animals and other parameters were observed, with the pups freely moving on a paper towel. Score 0 or 1 was respectively attributed to absence or presence of fur, erupted incisors, open eyes or ears.
Neurological reflexes
Evaluation of neurological reflexes was performed daily from pnd 5 to pnd 21. Score 0 or 1 was attributed, respectively, to animals not presenting or presenting the reflex or mature response as described below.
Walking (locomotive coordination and muscular strength)
The newborns were allowed to move freely and the development of locomotion was observed during one minute. Mature walking was considered when the animal was able to move with the body completely supported by the four limbs without dragging the belly over the surface. A normal mature response can be expected at pnd 15–16. 31
Grasping (freeing reflex)
The palm of a forelimb of the newborn was stimulated with a thin wire. A mature response was considered as soon as the animal grasped it immediately. A normal mature response can be expected at pnd 8–9. 31
Surface righting reflex (labyrinth and postural response)
The newborns were placed on their back with their limbs pointing upwards and the time needed to turn over and restore their normal prone positions was recorded (maximum time was set at 30 s). Complete acquisition of reflex was assumed when the animals could rotate 180° immediately around its longitudinal axis, standing on all four limbs. A normal mature response can be expected at pnd 6–8. 31
Postural reflex
Newborns were placed in a small plastic open box (10 × 10 mm base and 10 mm height) and gently moved to the left and right, up and down. Those animals that could maintain their original position in the box by extending all four limbs were assumed to have acquired a mature postural reflex. A normal mature response can be expected between pnd 10 and 16. 32
Wire suspension or bar holding test (muscular strength)
A 3 mm diameter metal bar/wire was suspended 35 cm above a soft surface. The animal was held and its forepaws were allowed to touch the bar and the capacity of the animal to hang on to the bar was observed. The mature response, which can be expected at pnd 12–13, 31 was considered when the animal was able to grasp the bar with all four limbs (maximum time was set at 120 s).
Postural stability and mobility test (‘negative geotaxis’) (labyrinth postural reaction)
The newborn was placed on a grid, tilted 45° to the plane, with its head facing downwards. Animals that could rotate a full 180°, face up, and could climb the grid within a maximum time of 30 s were considered to have fully acquired this reflex. A normal mature response can be expected at pnd 6–7. 31
Air righting reflex (statokinetic labyrinth response)
Newborn mice were held on their back (with their four limbs turning upwards), 20 cm above a soft surface, and released. The position in which the animal reached the soft pad was recorded. Reflex was considered mature when the newborn turned its body during the fall and landed on the surface with all four paws. A mature response can be expected after two weeks of age. 33
Test procedures with adult animals
Young/adult animal body weight
After weaning until 15 weeks of age, the body weight of animals was measured once a week using a precision balance for live rodents (Explorer Pro, Ohaus Corporation, Pine Brook, USA).
Home-cage behaviour – climbing
Climbing was scored as a measure for pain/discomfort in the toes due to the identification procedures. At 10 weeks of age, animals were taken out of the cage, pen marked on their tail, put back in their home cage which was placed in the experimental room and left undisturbed for 90 min. The behaviour of the animals in the cage was then recorded during 12 min. The frequency and duration of climbing behaviour for a 10 min observation period was scored by focal sampling with the aid of EthoLog behaviour observation transcription tool (etho 2.2.5 software 34 ).
Elevated Plus Maze
This test, which is used to assess anxiety, was carried out when the animals were 11 weeks of age. Between 09:30 and 13:00, under white light conditions, the animals were placed in the centre of a plus-shaped platform elevated 74 cm above the floor. The maze apparatus consisted of two closed arms (51×10 × 40.5 cm) and two open arms (51 × 10 cm) (NIR plus maze, MedAssociates Inc, St Albans, VT, USA). The animals were tested for 5 min and the number of entries into each of the arms as well as the time spent there was recorded online with the help of an infrared detection and a video tracking system (MedAssociates Inc). The apparatus was cleaned with a 10% ethanol solution between each test. The percentage of time spent in the open arms and the number of entries into the open arms were analysed.
Simplified SHIRPA primary screen
A simplified version of the SHIRPA primary screen protocol, a comprehensive three-stage screening protocol of behaviour and morphology originally developed by Rogers et al. in the ENU Mutagenesis Consortium (
Simplified SHIRPA primary screen protocol tests and respective scoring system used and measurements performed
After weighing, the animals were transferred to an arena made of clear Perspex with its floor measuring 55 cm × 33 cm and marked with 15 squares and the immediate reaction of the mouse to the novel environment was observed (transfer arousal and fear reaction). The mouse was then left in free movement in the arena and locomotor activity was measured. Holding the tail between the thumb and index finger, the animal was removed from the arena and allowed to grip a 3 mm hanging wire, elevated 35 cm above a soft surface (hanging wire test). The righting reflex was tested by holding the tail and forcing the animal to turn on its back and observing the animal's reflex to turn to the normal prone position.
A 3 mm diameter wire and a normal cage grid were adapted on top of the arena to perform the wire manoeuvre, visual placing, body position (trunk curl, limb clasping, limb grasping), grip strength and toe pinch tests. For the wire manoeuvre, the mouse was placed on top of the wire and allowed to grip it with its four limbs. For the visual placing, trunk curl, limb clasping and limb grasping, the mouse was held by the tail between the thumb and index finger, and approximated to the surface of the horizontal cage grid, head facing down. After the animal grasped the bars of the cage grid with the front limbs, the toe pinch test was performed using 10 cm long straight serrated forceps, with 1 mm diameter tips, to pinch one hind toe (mid-digit). This was followed by SHIRPA ‘negative geotaxis’ test, for which the animal was held by the tail and placed on the horizontal cage grid that was immediately turned into a vertical position with the animal facing with the head down. The capacity to turn 180° and the time it took to perform that reaction was scored and described in Table 1. Grip strength was tested with the animal grasping the bars of the cage grid with its forelimbs, held by the tail and pulled gently backwards.
For the vertical pole test the animal was placed on the centre of the pole (40 cm long × 2 cm diameter), which was held in a horizontal position 30 cm above the floor, and then gradually lifted to a vertical position, with the animal facing down.
For the footprint (gait evaluation) test, the paws of the animals were painted (front paws in red and hind paws in black) with a non-toxic water-based ink (tempera paste, Lefranc & Bourgeois, France) and the animals were allowed to walk on a 70 cm long, 4.5 cm wide runway covered with white paper (with 10 cm high walls), tilted 45°, into an enclosed dark box. The footprints were analysed for four-step parameters (all measured in cm): stride length was measured as the average distance of forward movement between each stride; hind paw-base width and front paw-base width were measured as the average distance between left and right hind footprints and left and right front footprints, respectively. Distance between left or right front footprint/hind footprint was used to measure uniformity of step alternation.
After each animal had completed the simplified SHIRPA primary screen protocol, all equipment was cleaned with a 10% ethanol solution.
Rotarod
In order to evaluate their motor coordination and balance, the animals were tested in a rotarod apparatus (TSE RotaRod System Advanced, TSE Technical & Scientific Equipment GmbH, Bad Homburg, Germany), at age 13.5–15 weeks. On the day before testing, mice were individually marked with stripes on their tail (with a non-toxic pen Sharpie, Banford®, USA) in order to be followed and tested always on the same place on the rotarod apparatus.
The protocol consisted of three days of training, at a constant speed of 15 rpm, for a maximum period of 60 s in four trials, with a 15 min interval between each trial. On the fourth day, animals were tested for each of six different fixed velocities (5, 8, 15, 20, 24 and 31 rpm), for a maximum of 60 s in two trials, with a 15 min interval between each trial. Animals were then left undisturbed in their home cage for a period of 1.30 h after which they were submitted to a continuous acceleration protocol from 4 to 40 rpm, for a maximum of 60 s in four trials (with 15 min intervals between them). The latency to fall off the rod was scored. All tests were performed between 10:00 and 16:30 h. The rotarod apparatus was always cleaned with a 10% ethanol solution between trials.
Postmortem procedures
At the end of the experiment, at 20 weeks of age, the animals were euthanized by decapitation; adrenal glands and thymus were collected and weighed in an analytic balance (AUW120D, Shimadzu Europe GmbH). The site of transponder implantation in the adult animals was observed and transponders were recovered.
Statistical analysis
Data on the reaction of animals to the identification procedures as well as qualitative data from the simplified SHIRPA protocol were statistically analysed using χ2 test or Fishers's exact test, when appropriate. The median pnd at which parameters on physical development and reflex acquisition by newborn mice was scored as 1 (present) was compared between groups. Since these data were not normally distributed, Kruskal-Wallis test was used to compare groups. Mann-Whitney test, with Bonferroni correction, was used to make multiple comparisons between pairs of groups. Body weight curves, anogenital distance/body weight ratio, wire suspension, surface righting reflex time and latency to fall off an accelerating rotarod were analysed using a general linear model of repeated measures analysis of variance (ANOVA). Quantitative data obtained during home-cage behaviour focal sampling (climbing duration and frequency), EPM, SHIRPA protocol (including number of squares in the arena and footprint gait) and postmortem organ weight were statistically analysed comparing mean values by one-way ANOVA, after confirming the normal distribution of variables. Whenever appropriate, post hoc multiple comparisons between experimental groups were performed using Tukey's honestly significant difference (Tukey HSD) test. Independent sample t-test was used when comparing genders. Quantitative data obtained during adults' tests, which did not follow a normal distribution (e.g. obtained with the fixed velocities rotarod protocol and SHIRPA hanging wire and vertical pole time), were analysed by Kruskal-Wallis test.
In all cases, statistical significance was set at P ≤ 0.05. Statistical analysis was performed using SPSS 15.0.
Results
Handling of the animals for experimental procedures was always performed in the same way and by the same experimenter for all experimental groups, in order to minimize the differences in results due to handling.
Newborns' permanent identification methods
The identification methods were performed as rapidly as possible to avoid a long handling period of the newborn: the quickest method was the toe clipping (1–1.5 min), followed by the toe tattoo ink puncture (1–2 min), with the transponder injection being the most laborious method (3–4 min).
General observations
No nest disruption, rejection or cannibalism was observed after any of the identification methods applied to the newborn mice.
The newborns' reaction to the identification procedure, scored as urination and vocalization reactions to toe clipping, toe ink puncture and transponder subcutaneous injection or just subcutaneous injection is presented in Table 2. The percentage of animals urinating and/or vocalizing during transponder implantation (Transponder) was higher than for other identification procedures.
Newborn mice urination and/or vocalization reaction to permanent identification procedures
Results are presented as percentage of animals showing these reactions. Significant differences between Transponder and other groups are indicated: *P < 0.005 and **P = 0.041 (analysed by Fisher's exact test)
Toe clipping
No problems occurred regarding haemostasis after toe clipping (there was almost no bleeding). This identification method could be applied very quickly and little or no reaction was observed in newborns during the procedure (Table 2). In adulthood, there was no case of toe tip/nail regeneration observed in toe clipped animals and the identification was easy to see by experienced personnel.
Genomic DNA extracted from the clipped toes yielded a concentration of 154.8 ± 30.6 ng/µL (mean ± SEM) (about 3 µg of DNA per toe).
Toe tattoo ink puncture
Newborn animals had to be held for about 1–2 min in order to perform the puncture procedure, including cleaning the animal's paw for excess of tattoo ink. As in toe clipping, only minor bleeding was observed with this procedure with no haemostasis problems. One of the males marked with this technique showed a defect in the ink punctured toe while growing: the punctured toe became twisted in the direction opposite to the animal, but this animal did not show any particular difficulties in moving or performing the tests used in the study. The individual identification of the adult animals in their cage by observing a green toe was very easy.
Transponder implantation
The subcutaneous injection of an identification transponder was the longest of the permanent individual identification procedures applied in this study and seemed to cause more pain. When we started to apply this identification method, from 14 injected animals six newborns lost the transponder and after adapting the technique by pushing the device backwards and laterally, only one out of 14 new injected transponders was lost. Moreover, in a total of 28 transponders injected, four transponders were rejected through an inflammatory process in the first 2–3 days after injection, and in one case a transponder became non-readable on the day after its injection. The successfully implanted animals were all used for the subsequent tests. The individual identification of cage mates with the help of the reader (NONATEC V1, port USB) was very easy to perform.
Effects of identification methods on the development of newborns
Somatic parameters and neurological reflexes
In the nest, until pnd 5, all animals presented normal skin colour (pink ventral and pink to light grey dorsal skin), had milk present in their stomach (milk spot) and presented normal wriggling activity, hence all animals used in the study were in good condition at the moment of the identification procedure.
Preweaning body weight was significantly influenced by the day of analysis (P < 0.005), as expected, but no significant differences were detected when comparing the body weight of male and female animals. The weight of transponders (0.24% of the weight of the newborns at pnd 5) was subtracted from the total body weight of the animals in the Transponder group. For newborns with transponders, a slightly (non-significant) lower weight could be observed between pnd 11 and pnd 19, when compared with Toe clip, Toe ink and Control animals (Figure 1).
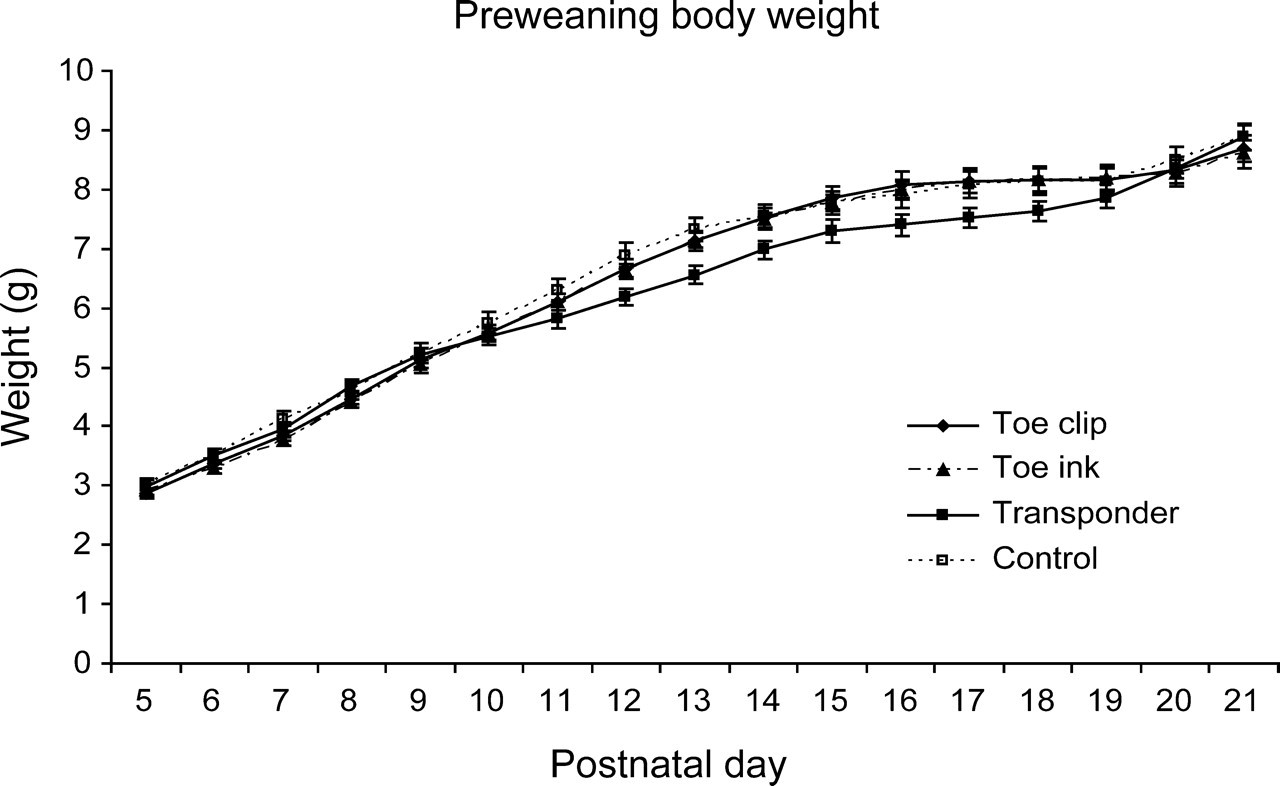
Preweaning body weight from postnatal day (pnd) 5 to pnd 21. Body weight gain (in grams) for both genders is plotted together according to experimental group. Values are mean ± SEM
Anogenital distances are typically different between males and females. Transponder-implanted males presented significantly smaller anogenital distances when compared with Control, Toe clip and Toe ink animals (P < 0.005, P = 0.022 and P = 0.018, respectively), in accordance to what was observed for preweaning body weight. As body weight might influence anogenital distance, the ratio between anogenital distance and animal preweaning body weight was calculated; no significant differences could be detected between different experimental groups, indicating that the differences detected in anogenital measurements were related to body weight and not caused by the identification procedure (data not shown).
In the parameters examined to evaluate physical maturation of the newborn mice, no significant differences were found between experimental groups in which an identification method was used and the control group.
A gender effect was only verified for ‘negative geotaxis’, using Mann-Whitney test (P = 0.035). Even though a gender effect was not detected in the other developmental parameters evaluated, it was decided to keep the analysis of genders separately for all parameters. No significant differences were observed in fur appearance that occurred at pnd 7 to pnd 8 (first hairs were observed on pnd 7, but fur was scored as present on pnd 8) for all animals. A milk spot could be clearly observed in the abdomen of all animals until pnd 7, after which it became more difficult to identify (data not shown). Opening of the ears and eyes was observed between pnd 12 and pnd 14 and incisor eruption was observed between pnd 8 and pnd 9 without significant differences between newborns of individually identified and control groups. No significant differences were found between identified and non-identified animals for the acquisition of postural reflex (mature response observed between pnd 9 and pnd 12) with most animals presenting the reflex at pnd 10 or pnd 11, and for acquisition of grasping reflex (ability to grasp a thin wire was present at pnd 8 to pnd 9) (Figures 2a and b). Acquisition of the reflex of ‘negative geotaxis’ was observed between pnd 5 and pnd 11 without differences between Toe clip, Toe ink, Transponder and Control groups, indicating that the identification procedure did not interfere with the newborns' ability to turn to their upright position and climb a 45° inclined grid (data not shown).
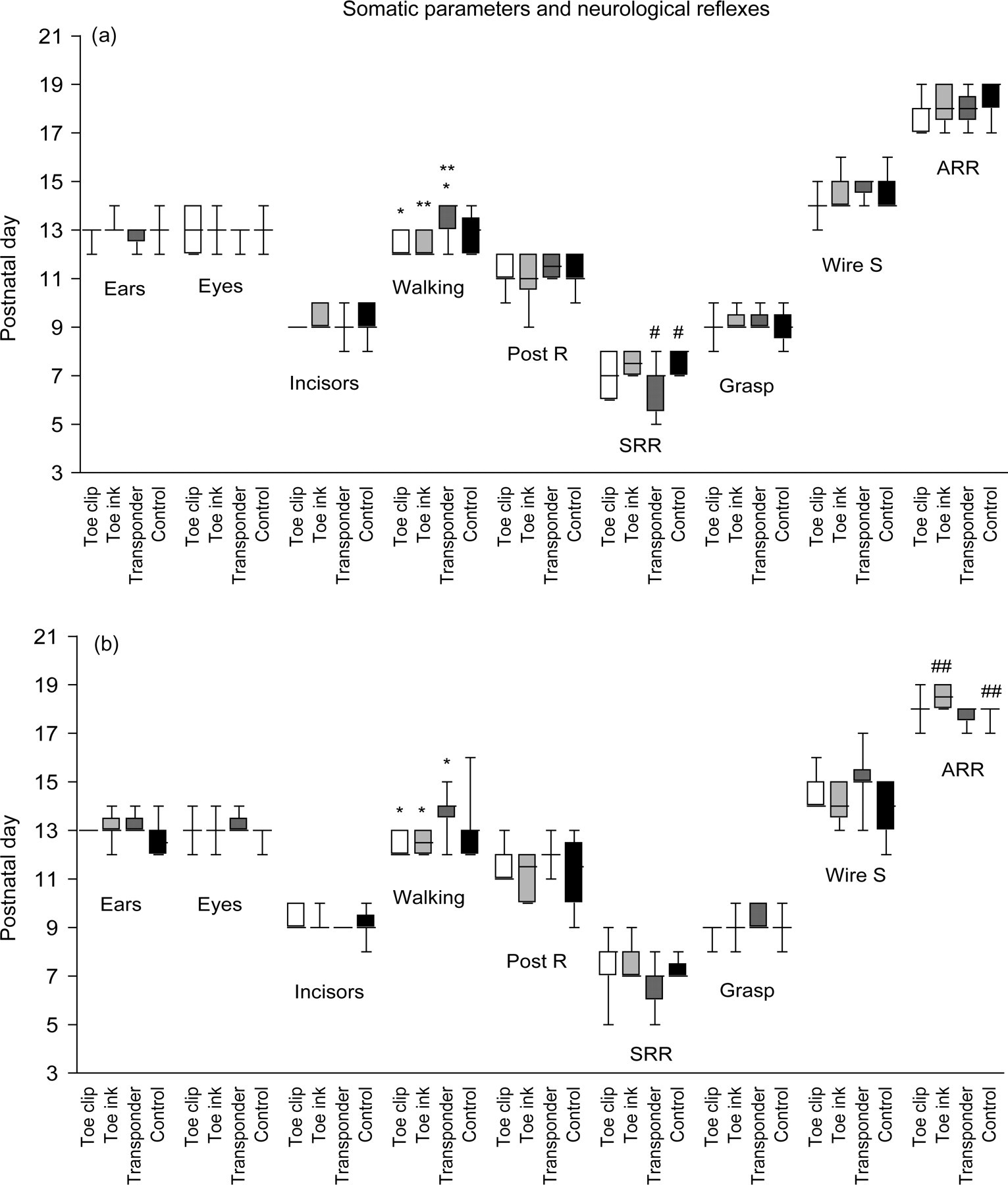
Effects of permanent identification methods on newborns' somatic development and acquisition of neurological reflexes. The median postnatal day (pnd) at which female (a) and male (b) animals presented a mature response (scored as present = 1) is shown for: ears opening (Ears), eyes opening (Eyes), incisor eruption (Incisors), mature walking (Walking), postural reflex (Post R), surface righting reflex (SRR), grasping (Grasp), wire suspension (Wire S) and air righting reflex (ARR). *P ≤ 0.005 (a) and **P ≤ 0.017 (b) (analysed by Kruskal-Wallis test and Mann-Whitney test with Bonferroni correction)
Mature walking could be observed in newborn animals from pnd 12 to pnd 15. A slight (but non-significant) delay in the development of mature walking was observed for the Transponder animals when compared with Control animals, but this small delay was significant when compared with that of Toe clip and Toe ink animals, both in females (P < 0.005 and P = 0.006 respectively, Bonferroni α = 0.01) and males (P ≤ 0.005, Bonferroni α = 0.01) (Figures 2a and b). In the period tested, surface righting reflex was present already at pnd 5 in some animals, but at pnd 9 all animals had acquired the mature reflex. A statistically significant difference was found when comparing the median pnd at which Transponder and Control females presented this mature reflex (P = 0.016, Bonferroni α = 0.017), but no significant differences were found between groups in the case of males (Figures 2a and b). When analysing the latency time to perform the surface righting reflex, significant differences were found between postnatal days, with latency time to perform the reflex being reduced with ageing (P < 0.005) as expected, but no significant differences were detected between groups by repeated measures ANOVA (data not shown). All animals were able to turn and stand on their four limbs in about one second from pnd 12 of age.
The air righting reflex was acquired between pnd 17 and pnd 19 in all animals. The only significant difference found was between Toe ink and Control males (P = 0.01, Bonferroni α = 0.017), showing a slight delay for Toe ink newborns (Figures 2a and b).
Wire suspension ability appeared between pnd 12 and pnd 16, but most of the animals acquired the ability to sustain their own body weight between pnd 14 and pnd 15 of age and no significant differences could be found between individually identified and Control animals (Figures 2a and b). As soon as the reflex was acquired, no significant differences were found in the latency time to fall off the wire (data not shown).
Effects of identification procedures on adult behaviour and motor abilities
Body weight after weaning
Body weight of individually identified C57BL/6J mice and control animals, from week 4 to week 15 after weaning, is presented in Figures 3a and b. No differences in body weight gain of young/adult animals were observed after the use of individual identification methods, such as toe clipping, toe tattoo ink puncture or transponder implantation, when they were five days old. A body weight of 22.9 ± 0.2 g and 29.2 ± 0.3 g (mean ± SEM) was reached at week 15 by females and males, respectively.
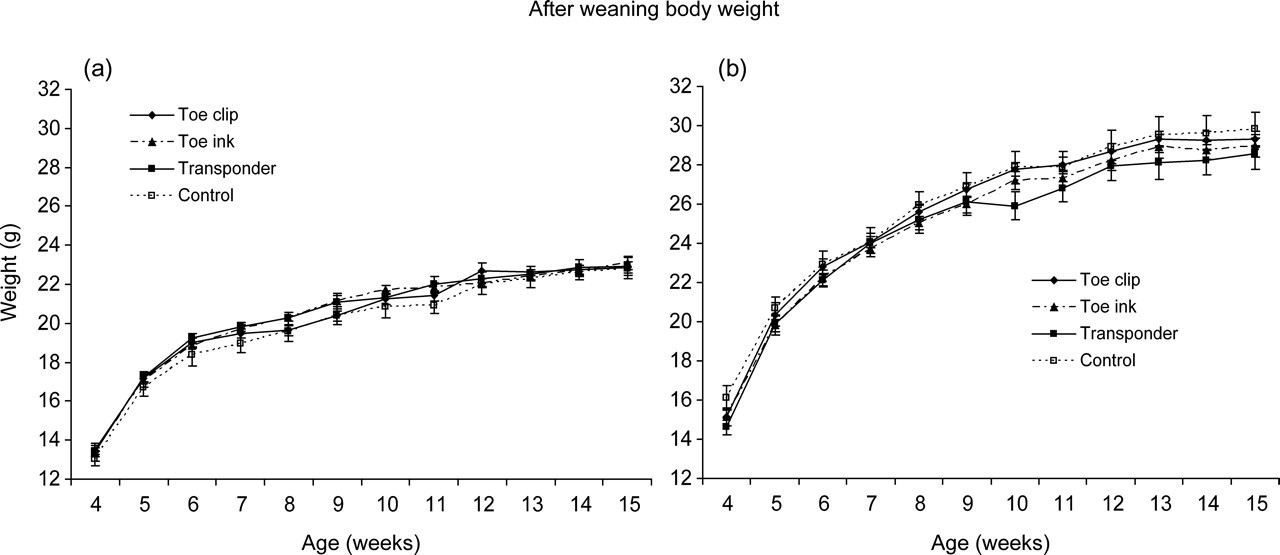
Body weight gain (in grams) after weaning (at 4 weeks of age), until 15 weeks of age, for female (a) and male (b) mice. Values are mean ± SEM
Home-cage behaviour – climbing
When analysing climbing behaviour, significant differences were revealed between male and female animals, but no effects of identification method were detected (Figures 4a and b). Climbing frequency in females was 13.1 ± 1.1 and males had a lower climbing frequency of 4.9 ± 0.6 (mean ± SEM; P < 0.005). The duration of climbing was also higher in females when compared with males: 153.3 ± 13.0 and 53.0 ± 8.3 s, respectively (mean ± SEM; P < 0.005).
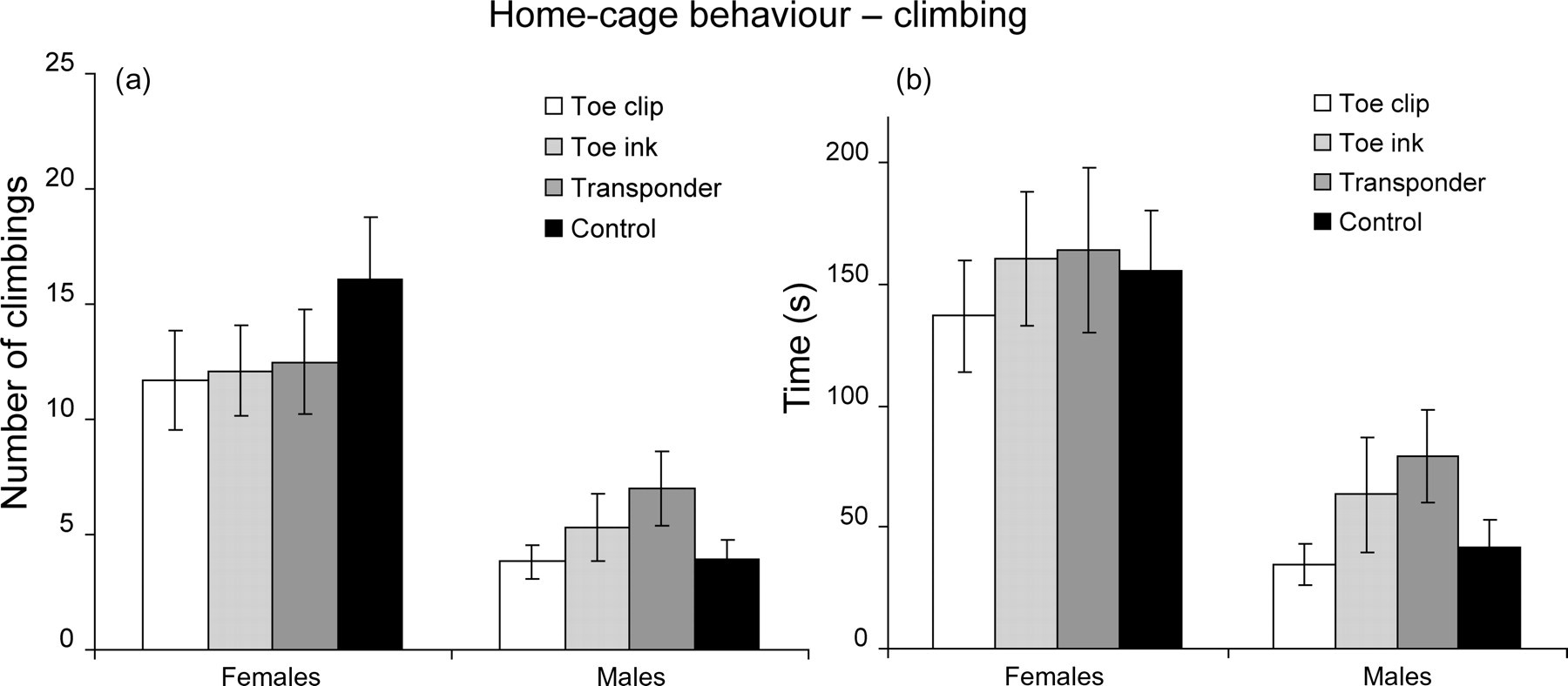
Home-cage behaviour analysis: climbing frequencies (a) and duration (b) are presented for each experimental group for females (left) and for males (right). Values are mean ± SEM
Elevated Plus Maze
No significant differences were found between groups and gender in the EPM test. The overall percentage of time spent in open arms was 14.4 ± 0.8 s (mean ± SEM), while the number of closed arms entries revealed an average of 12.5 ± 0.5 entries (mean±SEM).
Simplified SHIRPA primary screen
As previously shown, body weight after weaning showed no differences between groups and was not expected to influence possible differences between groups in the simplified SHIRPA primary screening used in this study. No differences were found between groups for transfer arousal or walking behaviour in the arena, with all animals presenting a normal behaviour. The same was true for visual placing, grip strength and reaction to toe pinch, as well as for the wire manoeuvre test and vertical pole test (animals were able to keep holding to the pole and moving moderately up/down and the average latency time to fall from the pole was 106.7 ± 4.8 s for females and 94.7 ± 6.3 s for males; values are mean ± SEM).
No differences between groups were shown in the hanging wire test, righting reflex, ‘negative geotaxis’ behaviour and time to turn and move up, or in the animal's reaction to restraint, urination or defaecation during the simplified SHIRPA primary screen protocol (data not shown).
For the locomotor activity in the arena, measured as number of squares the animal walked through during a 30 s period, Tukey HSD test showed a significant difference between Toe clip and Transponder males when compared with Toe ink males (P = 0.036 and P = 0.012, respectively) which presented a better performance, but no significant differences were found when comparing identified animals with controls (Figure 5). No differences were found in the number of squares walked through by different groups of females, although they were more active than males in this test (P = 0.018). The footprint test revealed a normal walking gait without differences between groups, revealing that stride length, hind footprint width, front footprint width and uniformity of step alternation were not affected in mice having a clipped toe or an ink tattooed toe or carrying a transponder since pnd 5 (data not shown).
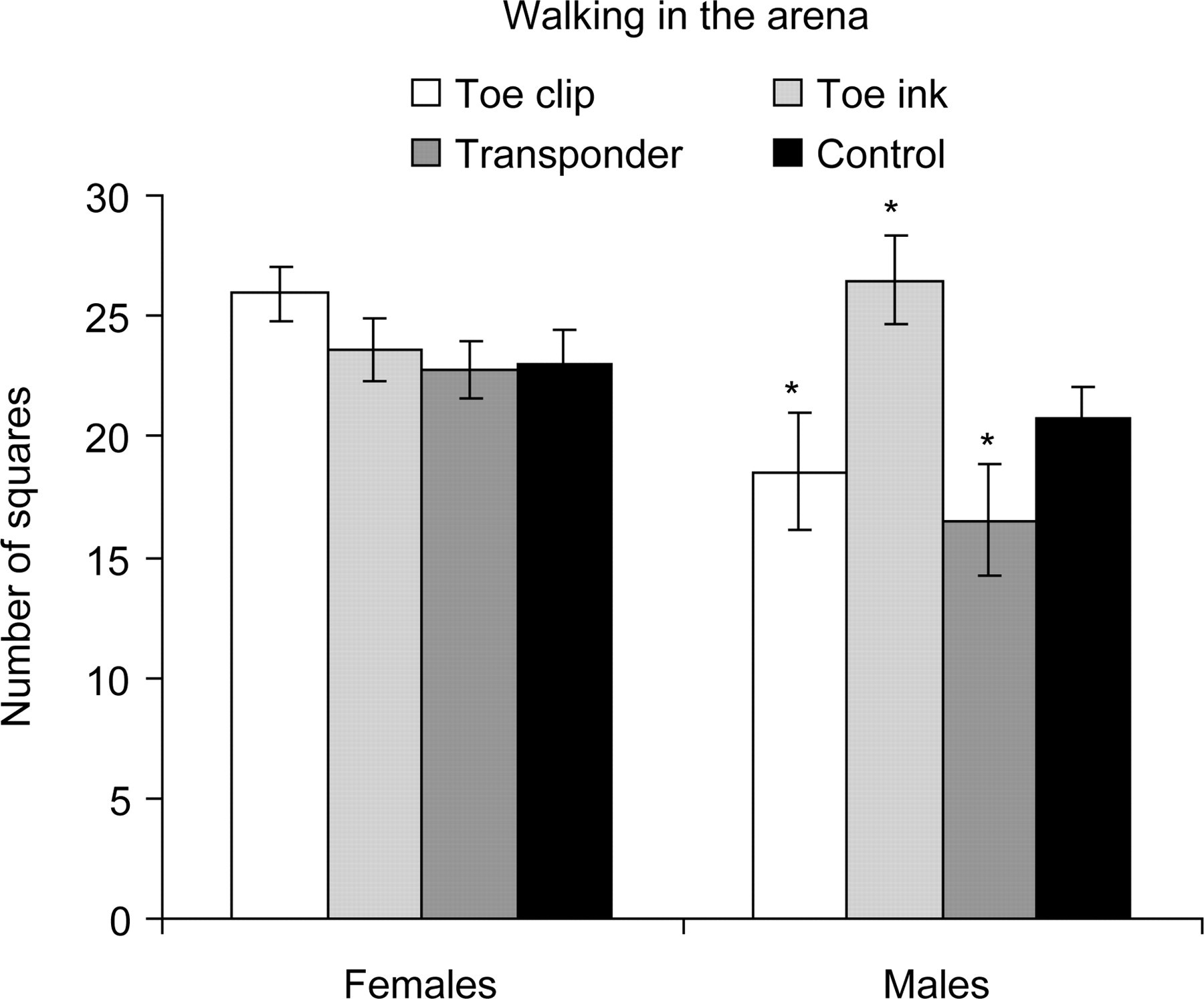
Locomotor activity of C57BL/6J mice measured in the arena (as number of squares walked through) during simplified SHIRPA primary screen protocol. Values are mean ± SEM. *P ≤ 0.05 (analysed by Tukey's HSD test)
Rotarod
The analysis of the rotarod test performances using a set of six constant velocities did not show any differences between males and females. Toe ink females presented a shorter latency time to fall from the rod at a constant velocity of 15 rpm (Figure 6); no significant differences were found when comparing permanently individually identified animals with controls, but multiple comparisons using Mann-Whitney test with Bonferroni correction showed a significant difference between Toe ink and Toe clip groups (Bonferroni α = 0.01; P < 0.005). No differences were detected at any other rod velocity. When the acceleration protocol was applied, no differences were found between groups or between genders for both latency time to fall from the accelerating rod and velocity at which the animals fell from the rod (data not shown). Even though no significant differences were found between groups, an improvement in the latency to fall and velocity at which the animals fell off the rod was shown, reflecting a motor learning capacity (P < 0.005).
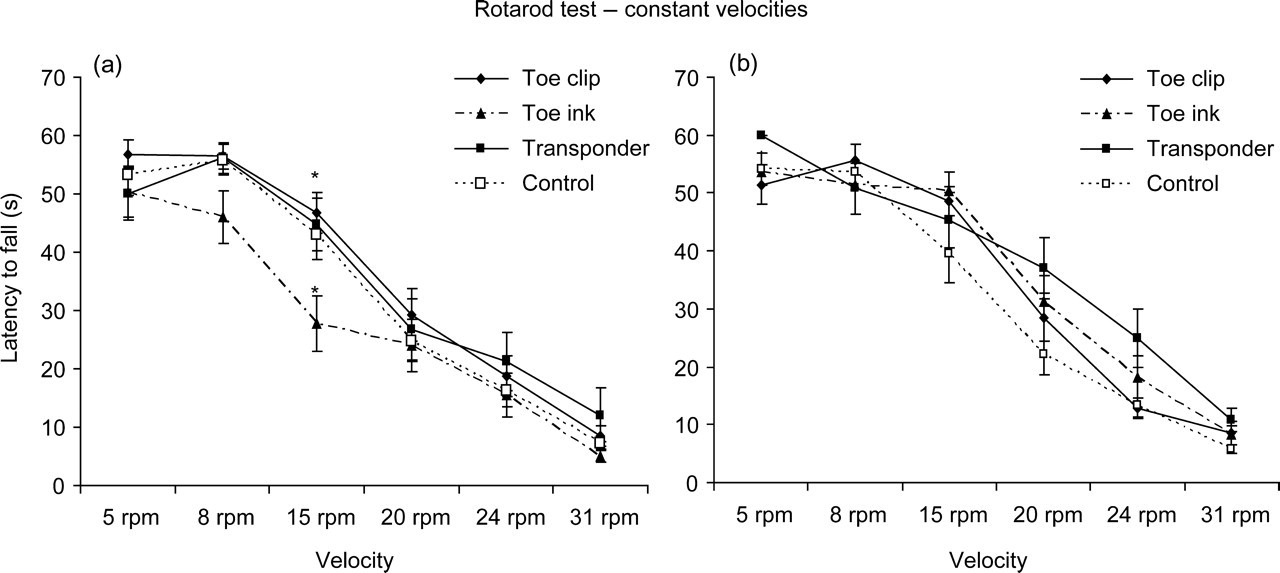
Rotarod test: latency to fall off the rod (in seconds) at different constant velocities: 5, 8, 15, 20, 24 and 31 rpm (a: females, b: males). Each line represents a group of animals, either individually identified at pnd 5 or control. Values are mean ± SEM. *P ≤ 0.005 (analysed by multiple comparisons with Bonferroni correction)
Postmortem analysis
Analysis of variance did not show any significant difference between adrenal glands and thymus weights of individually identified and control animals; however, differences between genders were present (P < 0.005 and P = 0.008 for adrenal glands and thymus weights, respectively). Adrenal glands of females and males weighed 5.24 ± 0.19 and 3.04 ± 0.15 mg, respectively, and thymus weights were 35.12 ± 1.83 mg in females and 27.37 ± 2.19 mg in males (values are mean ± SEM).
In the case of animals carrying a transponder, careful dissection of the dorsal skin was performed in order to expose the place of implantation of the transponder. No fibrous tissue or tumours were found in association with the tissue capsule surrounding the transponders that could easily be removed from the animals. After recovery and autoclave sterilization inside a microcentrifuge tube (vapour sterilization, for 20 min, at 121°C and 15 psi), transponders were still readable.
Discussion
Identification methods for marking newborn mice
The impact of three identification methods of newborn mice was systematically assessed in the present study in order to identify possible negative effects on development, behaviour and wellbeing of the animals. Toe clipping has been used in several animal facilities as a reliable method for permanent identification of very young rodents and it proved to be an efficient marking method in our study. It was the easiest and fastest method when compared with the other two identification procedures used and, surprisingly, newborns showed little reaction to the fast clipping of a toe.
Overall, it appeared that the longer the identification procedure lasted, the more signs of pain or discomfort 28 were shown by the animals. Additional distress was caused when injecting the identification transponder subcutaneously as the technique had to include a step of massaging the transponder laterally and backwards in order to avoid transponders being lost, which obviously caused pain in the newborns. Toe tattoo ink puncture was relatively easy to apply, although requiring more precision and skills than toe clipping, and a slightly longer time of restraint, which was reflected in a slightly higher percentage of animals urinating and vocalizing during this method of marking. In one case, the ink puncture procedure affected the normal growth of the toe, but the behaviour of this animal was not different from the other animals used in the present study. After successful marking of the young animals, all identification methods used were equally reliable and efficient throughout the study.
Toe clipping, although not being recommended due to humanitarian and ethical grounds, 9 is still a reliable individual identification method and is the only one of the three methods used here providing tissue for genotyping. Tail biopsies are usually performed for this purpose, which also interferes with the animals' physical integrity. Our results show that the amount of DNA recovered from the clipped toes is enough to perform several polymerase chain reactions (PCR), but not for Southern blot analysis. 37 This result is in accordance with previous studies using mouse toe lysates for genotyping large colonies of mice, where toe DNA lysates were proven to give reliable PCR results even after being stored at −20°C for over a year, allowing assays to be confirmed at a later date if necessary. 17
Transponder identification has the advantage of being associated with the registration of data in computerized tracking systems. The transponders used in the present study were recovered from the animals after euthanasia and proved to be in good condition to be reused after sterilization at high temperatures, which can reduce the costs associated with this identification method. Rejection of transponders in the first 2–3 days after their injection in newborn pups, or transponders losing the capacity of radiofrequency transmission, should be taken into account when using this identification method. An advantage of transponder use is that they can be kept within tissues collected from the animals to keep tracking data from the original animal. In contrast to some long-term studies, 20,25,26 no tumours were detected in association with the transponders in 20-week-old C57BL/6J mice in the present study.
Postnatal development following individual marking
To assess wellbeing of newborns and possible negative consequences of the newborn individual identification methods on their development and behaviour, we used a systematic approach based on standardized behavioural screening protocols. We observed normal development and behaviour in all mice used from the day of birth until the day of the identification procedure (pnd 0 to pnd 4), suggesting that their wellbeing was not impaired. When we individually identified the pups at pnd 5, no nest disruption, cannibalism or rejection by the mother was observed, which was in accordance with what had been previously reported when tattooing newborn mice. 5,7
Standardized protocols of neurobehavioural tests for non-invasive analysis/scoring of physical growth and neurological reflex maturation have been extensively used to phenotype several mouse strains and lines and assess newborn and post weaning laboratory rodents' wellbeing. 30,31,33,38–44 When comparing growth in Toe clip, Toe ink and Transponder groups, with Control animals, no negative impact on growth of the newborns was found by either of the individual identification methods. At the time of weaning, the animals' body weight means were similar for all groups of animals and presented normal values. 8 Parameters indicating wellbeing in mouse pups, such as physical growth, feeding, fur appearance and presence of a milk spot, were unaffected by the identification methods, indicating that the acute distress caused by these procedures did not interfere with the animals' wellbeing and development in the following postnatal days. Physical maturation as assessed by timing of ear and eye opening, and incisor eruption was also normal in individually identified animals and comparable to control animals.
A modified version of the battery of tests developed by Fox 33 and Altman and Sudarshan 38 has been used to assess the general maturation of the nervous system, welfare and stress effects in several studies using neonatal rodents. 31,33,38,41,45 In our study, no significant differences were detected between individually identified and control animals in the acquisition of postural reflex and ‘negative geotaxis’. In the case of the surface righting reflex, a statistically significant difference was detected only when comparing the median postnatal day at which female Transponder and Control animals presented the mature reflex, but this was not verified for males. These tests require the integrity of muscular and motor functions being dependent on the acquisition of symmetrical coordination between the left and right sides of the body. 46 ‘Negative geotaxis’ has been used as a test for an unlearned orientation response in opposition to gravitational cues, used to assess vestibular function (as the postural and righting reflexes 38,47 but has recently been argued to be more useful for evaluating postural stability on an inclined plane. 48 Grasping reflex was acquired by individually identified animals in the same postnatal period as Control mice, showing that no negative effect on grasping could be attributed to toe clipping as has been previously suggested. 2,9
More complex locomotor skills are needed for mature walking, wire suspension and air righting reflex, which appear later in the postnatal period. 33,38 Individually identified C57BL/6 mice generally showed good motor coordination and muscular strength development in these behavioural tests, comparable to Control animals; the only exception was that male Toe ink animals presented a slight delay in the development of the air righting reflex, but this delay was not confirmed in females. A delay in the presentation of mature walking was detected in Transponder animals when compared with the other groups of individually identified animals, but the delay was not statistically significant when comparing with Control animals. The wire suspension test assessing balance control and muscular strength again showed no differences between groups. All parameters assessed from pnd 5 until weaning were present in the normal timing as shown in other studies. 31,33,38,41 The present study showed no consistent differences in most parameters of somatic and neurological reflex development during the postnatal period as a result of the newborn individual identification procedures used. Neuromuscular function development was, in general, similar in Toe clip, Toe ink or Transponder C57BL/6 mice when compared with Control animals; and motor abilities, coordination and balance were not affected by a toe missing a distal phalanx or by carrying a transponder. The small, but statistically significant, delay in mature walking development detected for Transponder animals in comparison to Toe clip and Toe ink animals, in both genders, might possibly be due to carrying a dorsolaterally implanted transponder.
Evaluation of adult behaviour and wellbeing: impact of individual identification
Video recording the animal's behaviour is a method of evaluating animal wellbeing through non-invasively observing the animal in its home environment. Home-cage behaviour has been used, for instance, to search for alterations in behaviours (locomotion, grooming, social behaviour, climbing, etc.), as a welfare indicator and to evaluate the consequences of husbandry procedures, group and cage sizes in laboratory mice. 49–51 We used this method to assess possible differences in climbing, a normal behaviour of mice, that can be impaired in painful conditions. 52 At 10 weeks of age, frequency and duration of climbing of individually identified animals was comparable to their controls with females being more active than males, as shown in other studies using C57BL/6 mice. 49 Toe clipping, for instance, could be expected to have an influence in the climbing ability, 9 but our study clearly demonstrated that this behaviour was not affected by this identification procedure. This result is of note as this concern has been put forward as one of the disadvantages of this identification method.
The SHIRPA protocol, a semi-quantitative three-stage protocol developed by Rogers et al. 35 in 1997, including a battery of neurobehavioural tests, has been extensively used, in its original version as well as in new modified ones, for behavioural assessment in large-scale ENU mutagenesis programmes in order to screen for dominant phenotypes and neurological dysfunction in mice as well as to characterize behavioural strain differences (e.g. see references 36, 53–55). In the present study, muscle and lower motor neuron and spinocerebellar functions were not affected in toe clipped, toe ink marked or transponder implanted mice. Concerning locomotor activity females actually showed a better performance when compared with males, and Toe ink males moved more than other groups of identified males, but no difference was detected in comparison to controls. The neuromuscular function of individually identified adult animals was shown to be normal, and no impairment of the ability to grip a wire/bar and sustain the body weight on the hanging wire test could be detected, independently of the animals missing part of a toe or carrying a transponder. The animals' ability to hold and move without falling in the vertical pole test as well as the sensory–motor function was not affected either. Even when the pinched toe was a clipped one, the ipsilateral flexor reflex of withdrawal was the same, indicating that sensory–motor function was not affected. In the ‘negative geotaxis’ test, most animals turned up and climbed the grid without any signs of postural instability with the application of rotational forces while they were holding the cage grid. Similar responses were again obtained when comparing groups of individually identified and control animals. No signs of neuropsychiatric dysfunction were detected in any animals during this study, but some vocalizing and irritability were shown, which is a normal response to handling. 54 Vocalization, urination and defaecation during handling were similar for all animals reflecting a normal autonomic function and usual motor-affective response in a restraint situation. Our results were comparable to previous ones using SHIRPA primary behavioural screening protocol to phenotype C57BL/6J inbred mice. 36,54
Footprint gait assesses walking patterns and has been used to evaluate movements and motor coordination in rodents, providing information on cerebellar function. 40,56,57 In this study the mice did not show abnormalities in the walking patterns as a consequence of the marking procedure when compared with control animals.
The rotarod test, measuring locomotor function and coordination, 35,40,44,56,57 confirmed that there were no major differences in the locomotion abilities of individually identified animals except in a particular constant velocity test, at 15 rpm, in which Toe ink females presented a shorter latency time to fall off from the moving rod, but the same was not detected in males. Moreover, when using an accelerating rod (0.6 rpm/s), no differences could be detected between the different groups. The overall results obtained in the rotarod did not allow us to conclude that a real impairment of motor coordination and balance in Toe ink females at a constant velocity of 15 rpm could be due to the fact that those females had a toe marked with tattoo ink. Motor learning capacity, a measurement of cerebellar learning, was also verified by the improved performance shown on rotarod with repeated testing, without an influence of individual identification.
The EPM is a standard test to assess anxiety-like behaviour and screening anxiolytic drugs in rodents. 45,58–60 It is also used to behaviourally phenotype rodents 35,40,44 by measuring the conflict between exploration of novelty and avoidance of brightly lit open areas as an anxiety indicator, also giving information on locomotor activity. In the present study, no group differences were found in EPM performance. Moreover, our results are in accordance with previous publications comparing the performance of different strains of mice on the EPM test. 59,61
Behavioural assessment is a complex issue and behavioural measures are usually used in conjunction with physiological data. 62 Even though no behavioural signs of distress were found in the present study, an additional and retrospective evaluation of the wellbeing of the animals was carried out at the end of the study by means of thymus and adrenal weights. Thymus weight is known to decrease under the influence of stress, caused by increased apoptosis of thymus cells 63,64 and adrenal weight reflects the hypothalamic–pituitary–adrenal (HPA) system activity in response to stress, being increased in animals submitted to chronic stress. Postmortem organ weight confirmed the behavioural observations indicating that no long-term effects or distress resulted from the individual identification of animals by clipping or ink puncturing a toe, or by subcutaneously implanting a small transponder.
In conclusion, it can be stated that the methods for identification of newborn mice studied were proved to be effective for long-term marking of individual C57BL/6 mice. Implantation of a transponder, even being small sized, was shown to be more difficult to perform and to cause greater distress to the newborn pups during implantation. Toe clipping, a method usually referred as a non-advisable method on a welfare and ethical basis, was actually shown to elicit the smallest pain/distress related response in newborn pups at pnd 5. The use of this identification method would even be advisable when studying genetically-modified animals in which tissue is usually collected for DNA extraction in order to genotype the animals. In this case, we would advise the use of this method at a young age (between pnd 3 and pnd 7), before ossification is completed, but when sufficient tissue can be collected for genotyping purposes. Green tattoo ink puncture of a toe was also proved to be applicable in young newborn mice, being particularly easy to observe in young/adult C57BL/6 mice as a consequence of the green colour contrasting with the black skin and fur. The toe ink puncture procedure was slightly more distressful to apply than toe clipping, but in cases where tissue is not needed, this method could be preferable and ethically justifiable because it is not interfering with the integrity of the animal.
Present results showed no overall consistent differences in most parameters of somatic and neurological reflex development during the postnatal period as a result of the newborn individual identification procedures used, with the exception of a small delay in mature walking development that was detected for Transponder animals. This result, together with the signs of pain/distress during the procedure of the subcutaneous injection and massage needed for successful transponder implantation, suggests that this individual identification method should not be applied in five-day-old C57BL/6 newborns. Transponder identification has the advantage of allowing the registration of data in computerized tracking systems useful for large colony management and facilitating the construction of databases containing well organized and easily accessible research data from individual animals, but this method should be considered for use only in older pups.
No discomfort or poor welfare was detected in adult animals. In general, our results indicate that neuromuscular, spinocerebellar and sensory functions of adult animals, including reaction toe pinch, walking patterns, motor coordination and balance and climbing behaviour are not impaired as a consequence of missing a distal part of a toe or having an ink-marked toe or a dorsally-implanted transponder. We did not detect any alteration in adult anxiety-like behaviour, and no obvious negative impact and welfare disturbances could be related to any of the three identification methods studied.
Finally, for any experiment that requires the long-term identification of mice at a young age, the advantages and disadvantages of each method should be considered in order to choose the most adequate and less distressful identification method at the age of interest. In addition, proper training must be provided to individuals performing the techniques in order to minimize unavoidable animal pain and/or distress during the identification procedure.
Footnotes
Acknowledgements
We thank Lutronic International (Rodange, Luxembourg) for providing the identification transponders, as well as the respective reader/writer and software. We also thank Professor Pedro Oliveira for the helpful comments on the statistics. As this study was performed within the framework of the first author's master thesis, thanks are due to Laboratory Animals Ltd for providing financial support.
