Abstract
Voluntary wheel running (VWR) behaviour is a sensitive indicator of disturbed wellbeing and used for the assessment of individual experimental severity levels in laboratory mice. However, monitoring individual VWR performance usually requires single housing, which itself might have a negative effect on wellbeing. In consideration of the 3Rs principle, VWR behaviour was evaluated under group-housing conditions. To test the applicability for severity assessment, this readout was evaluated in a dextran sodium sulphate (DSS) induced colitis model. For continuous monitoring, an automated system with integrated radio-frequency identification technology was used, enabling detection of individual VWR. After a 14-day adaptation period mice demonstrated a stable running performance. Analysis during DSS treatment in combination with repeated facial vein phlebotomy and faecal sampling procedure resulted in significantly reduced VWR behaviour during the course of colitis and increased VWR during disease recovery. Mice submitted to phlebotomy and faecal sampling but no DSS treatment showed less reduced VWR but a longer-lasting recovery. Application of a cluster model discriminating individual severity levels based on VWR and body weight data revealed the highest severity level in most of the DSS-treated mice on day 7, but a considerable number of control mice also showed elevated severity levels due to sampling procedures alone. In summary, VWR sensitively indicated the course of DSS colitis severity and the impact of sample collection. Therefore, monitoring of VWR is a suitable method for the detection of disturbed wellbeing due to DSS colitis and sampling procedure in group-housed female laboratory mice.
Voluntary wheel running (VWR) behaviour is a frequently used measure of general activity, exploration and migration and is associated with mood and reward,1,2 but also serves as an indicator of pain in studies investigating inflammatory pain and nerve injury.3,4 Recently, on the basis of VWR behaviour, a cluster model has been established to define individual levels of severity during intestinal inflammation and stress in mice. 5 Here, decreased VWR was observed during the onset and course of dextran sodium sulphate (DSS)-induced acute colitis with graded degrees of inflammation and also in a model of restraint stress. Combining the parameters VWR and body weight change provided a measure to assess the severity experienced by the animal. Analysis of these data with two independent mathematical algorithms revealed three severity levels, which might describe either no, mild, or moderate severity.
The 3Rs principle (refine, reduce, replace) of Russell and Burch 6 is the ethical and, in Europe, legal prerequisite for animal-based research. However, limiting severity for animals under experimentation to a minimum requires the unambiguous and objective definition of severity levels. Although the developed cluster model meets these requirements, mice of the above described study were single housed, which might impact the welfare of these animals. 7 Housing conditions have to be considered not only with regard to wellbeing but also as a potential confounder. Mice are social animals and group housing is recommended to avoid social isolation and to maximize wellbeing. 8 This topic, however, is controversially discussed. Many studies report altered physiological and behavioural responses as a consequence of social deprivation due to single housing. 9 However, there are also studies showing that stress response and results of behavioural tests were not different between group and single housing and that there were no adverse effects resulting from single housing after laparotomy.10,11 In preference tests, mice showed a high demand for social housing. 12 Thus, at least for female mice, social housing is generally recommended. In addition, space restrictions in most conventional housing systems would allow only a limited number of running wheels within one cage and individual performance would be almost impossible to track. In the present study, VWR behaviour in group-housed female mice was analysed in a system providing access to running wheels for each individual mouse.
The main goal of the study was the evaluation of VWR behaviour in socially housed mice as a parameter for disturbed wellbeing. Therefore, a new running wheel system for group-housed mice with integrated radio-frequency identification (RFID) technology and automated monitoring of running performance was tested. Finally, the clinical and behavioural data were entered into a mathematical cluster model to define individual severity levels in mice.
Animals, materials and methods
Ethics statement
This study was conducted in accordance with German law for animal protection and the European Directive, 2010/63/EU. All experiments were approved and permitted by the Lower Saxony State Office for Consumer Protection and Food Safety (LAVES, license 16/2194).
Mice and experimental set-up
Female, 10 to 11 week-old C57BL/6J (B6) mice were obtained from the Central Animal Facility (Hannover Medical School, Hannover, Germany). Routine health surveillance and microbiologic monitoring according to the Federation of European Laboratory Animal Associations recommendations did not reveal any evidence of infection with common murine pathogens.
13
Mice were maintained in a room with controlled environment (21–23 ℃; relative humidity 55 ± 5%; 14/10 h light/dark cycle with lights on at 07:00 h and lights out at 21:00 h). Mice were housed in groups of seven mice in connected macrolon cages (360 cm2; Figure 1(a)) with softwood granulate (poplar wood, AB 368P, AsBe-wood GmbH, Germany) and the bedding was changed once a week. Nesting material was omitted to decrease possibly confounding factors of activity apart from social interaction and wheel running. Pelleted diet (Altromin 1324, Lage, Germany) and autoclaved water were provided ad libitum.
Group running system and wheel running parameters during adaptation phase: schematic illustration of the group running system and a close-up picture of cages 2 and 3 (a); preference of running wheels (RW) depicted in a heat map (b); both outer wheels are used with very high frequency by all animals. Course of voluntary wheel running (VWR (c)) and maximum velocity (Vmax (d)) during adaptation phase, showing a slight increase over time and stable baseline values (n = 21). d: day.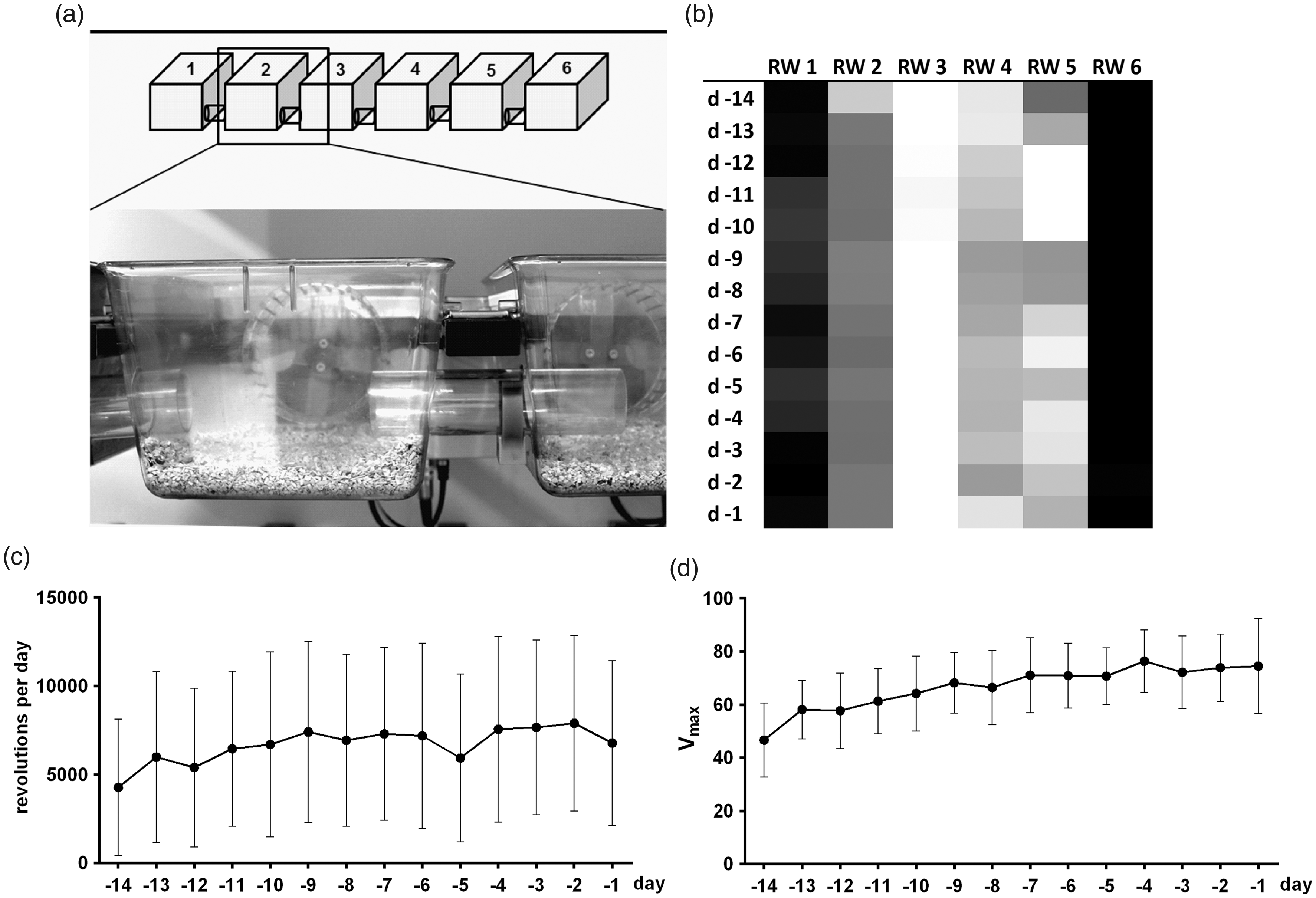
Transponder implantation
After one week of habituation, RFID transponders were implanted under isoflurane anaesthesia on a warming pad after induction with 4 vol% isoflurane in a clear box and confirmed absence of withdrawal reflex. Anaesthesia was maintained at approximately 2 vol% isoflurane. Cornea protection was provided by use of Bepanthen® eye ointment.
The animals' fur in the cranial back was clipped, the skin disinfected and the RFID transponder was inserted subcutaneously via a minimal skin opening matching the diameter of the implant (2 mm). To prevent loss, a loosely applied U suture was added. The average duration of anaesthesia was approximately 7 min in total. Animals were constantly monitored during and 1 h after anaesthesia for any adverse clinical signs and daily following implantation. After one week of recovery, the cages were equipped with running wheels.
Group running system
For collection of individual VWR data, a running wheel system with integrated RFID technology was used (IDrevolyzer, by PhenoSys (Berlin, Germany) and Preclinics (Potsdam, Germany)) (Figure 1(a)). Six standard cages (EU Type II), each harbouring a single running wheel, were interconnected via plastic tubes. Therefore, group-housed mice within this system could freely choose between the wheels. RFID sensors were located outside the cages behind each running wheel, exclusively detecting the transponder of the mouse currently active in the wheel. As readout parameter for each individual mouse, number of wheel rotations in total between 13:00 h and 08:00 h were recorded (VWR(19)), therefore omitting 5 h of the light phase, during which procedures took place. Further parameters of running activity were the highest number of rotations within 1 min (maximum velocity (Vmax)) and duration of wheel occupation for each wheel.
Experimental design, sample collection and DSS colitis
Animals of the control group were continuously handled and weighed whereas animals of the 0% DSS group additionally underwent sample collections on days 0, 5 and 14. Sample collection procedure consisted of facial vein phlebotomy (20 G cannula for skin puncture, alternating sides) to collect 15 µl of blood for preparation of dried blood spots, followed by collection of faecal samples, for which animals spent 2 h in separate cages lined with hydrophobic sand (Coastline Global Inc., Palo Alto, USA).
Acute colitis was induced in the 1% DSS group by oral administration of a 1% DSS solution via the drinking water over five consecutive days (day 0 to day 4). The DSS-treated mice were also submitted to sample collection procedure. All mice were euthanized on day 14 of the experiment by CO2 inhalation and subsequent blood collection from the heart followed by dissection for organ harvesting.
Clinical scoring
Starting three days before transponder implantation, the animals' condition was assessed daily around 08:30 h by means of a previously published model-specific clinical score which utilizes parameters such as stool consistency, condition of the fur and behaviour. 14 Changes in body weight were evaluated separately. All handling and scoring procedures were confined to two designated experienced staff members not blinded to experimental group.
Histology
The colon was flushed immediately following removal and prepared as a ‘Swiss roll’ 15 before the colon was fixed in a 4% buffered formalin solution. After embedding in paraffin, the organs were sectioned and H&E stained for microscopic assessment of inflammation. The histological scoring was performed in a blinded manner using a previously published histological score for DSS colitis 14 which separately evaluates the distal and proximal colon.
Statistics
All statistical procedures were run on GraphPad Prism 8 (v8.2.1, GraphPad Software, Inc., La Jolla, CA, USA)®. If not indicated otherwise, all values show mean ± SD. For one animal of the 1% DSS group, the experiment had to be terminated on day 8 due to high body weight loss (humane endpoint). The hypothesis of Gaussian distribution was tested by using the Shapiro–Wilk test. According to the results, comparison between all three groups was performed using one-way ANOVA with Tukey post hoc test in the case of Gaussian distribution. In all other data sets, analysis was performed via Kruskal–Wallis and Dunn's post hoc test. A Mann-Whitney U-test was run on the non-parametrically distributed data of the histological analysis (see also tabular results, Supplementary Material Tables 1 to 4 online). For cluster analysis, a previously developed cluster model was used (https://calliope.shinyapps.io/severity_assessment/).
Results
Adaptation to the running wheels and frequencies of wheel occupation
To estimate the time seven group-housed female mice need to reach a stable running performance, individual VWR was monitored from 13:00 h to 08:00 h (VWR(19)) over 14 days. The mice started to run immediately after getting access to the running wheels and demonstrated an average of ∼4000 revolutions (r) on the first day (Figure 1(c)). Over the next five days the animals accomplished a stable running performance of approximately 7000 r/day, which remained at this level until end of given adaptation time. Detected maximum running velocities (Vmax) showed a similar course, in which mice accomplished a stable Vmax of ∼72 r/min on day –6 (Figure 1(d)).
To analyse whether individual mice demonstrated spatial preferences for particular running wheels, frequencies of wheel occupation were assessed. Quantification of spatial preferences revealed that mice most readily used the peripherally located running wheels 1 and 6 with approximately 35% of total usage, respectively (Figure 1(b)). Running wheels in the centre (wheels 2–5) were used with frequencies of only 5–8% of total usage.
Reduction of wheel running behaviour due to DSS colitis and phlebotomy
To assess the change of VWR behaviour mice were treated with 1% DSS via drinking water and additionally submitted to sampling procedures on day 0, 5 and 14 (1% DSS, Figure 2(a); Supplementary Figure 1(a)).
Wheel running parameters during experimental phase: comparison of voluntary wheel running (VWR) between all three groups ((a); 1% dextran sodium sulphate (DSS) n = 6–7; 0% DSS n = 7; control: n = 7) showed significant differences on day 0 (first sample collection) as well as on days 5 to day 8 under and following DSS treatment (1% DSS vs. control: * = p < 0.05, ** = p < 0.01; 0% DSS vs. controls: ## = p < 0.01; 1% DSS vs. 0% DSS: $ = p < 0.05). Statistically significant impact of DSS treatment on maximum velocity (Vmax) (b) is seen on day 7 and day 8 while sample collection fails to have any effect (1% DSS vs. control: * = p < 0.05). Distribution of running wheel (RW) preferences (c) shows no variation in comparison with the adaptation phase and is not affected by DSS treatment or sample collection. (For detailed results of statistical analyses see Supplementary Material Tables 1 and 2 online).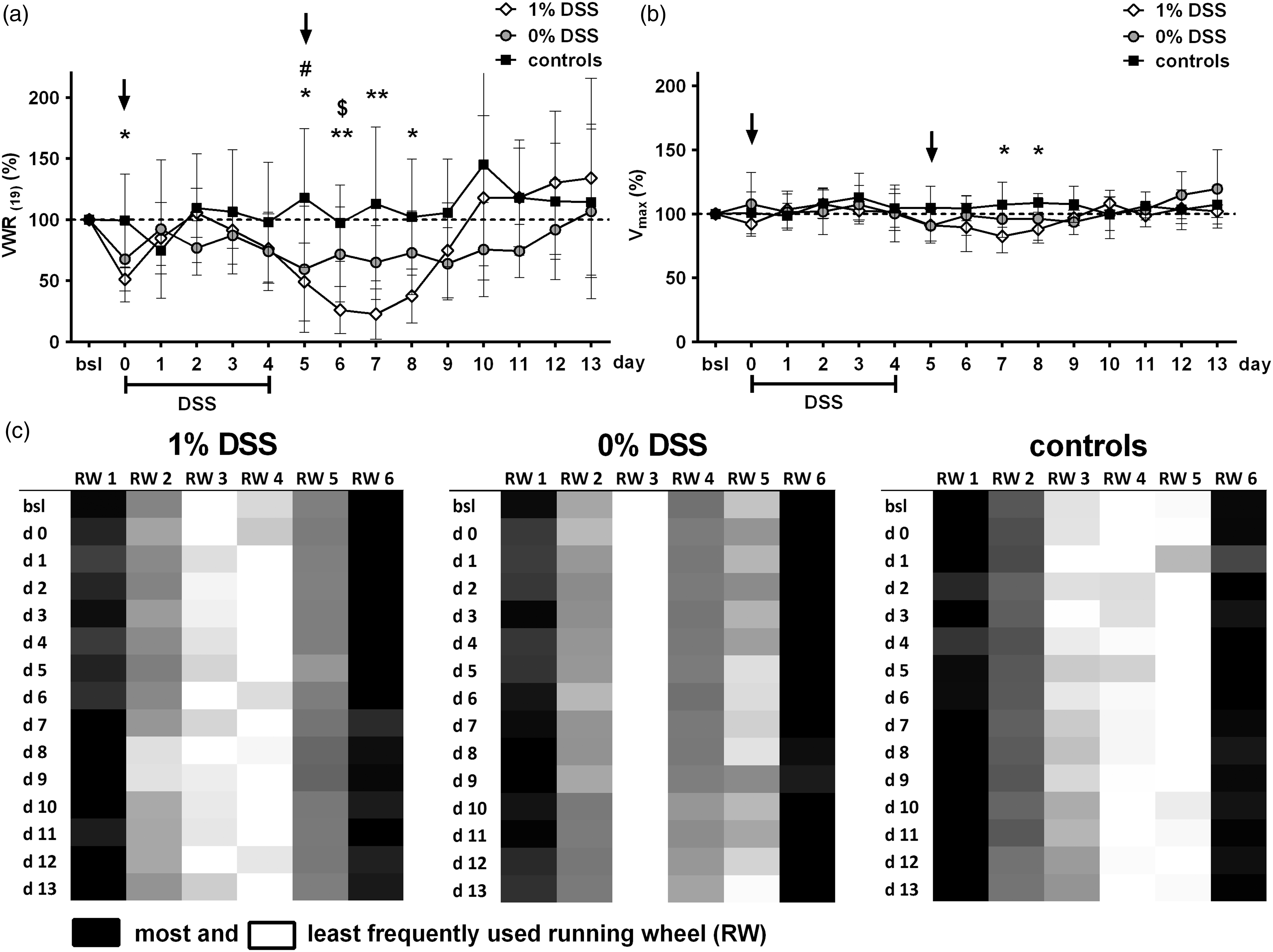
Analysis of proportional change from baseline revealed a first drop in VWR on day 0, after phlebotomy and faecal sample collection on this day. Changes in VWR under DSS treatment appeared from day 4 with a continuous decrease of wheel running until day 7. After reaching a maximum reduction of 77%, an increase of VWR was observed from day 8, exceeding baseline level on day 10. Mice submitted to phlebotomy and faecal collection alone (0% DSS group; Figure 2(a); Supplementary Figure 1(b)) demonstrated a slightly decreased VWR behaviour on day 1 after sampling procedures, which sustained until day 4, and was followed by a further decrease after phlebotomy on day 5 with a reduction of VWR to 40% of baseline level. VWR started to increase again from day 10, reaching baseline VWR on day 13. In contrast, the animals in the control group (control; Figure 2(a); Supplementary Figure 1(c)), which were solely weighed each day, demonstrated wheel running behaviour varying around baseline level over the whole observation time.
Level of Vmax remained at baseline for all but one time point in the 1% DSS group, which coincided with the day of maximum drop in running activity and showed a statistically significant reduction of 9% compared with the control group (Figure 2(b)). Both 0% DSS and control group displayed constant levels of Vmax (Figure 2(b)).
Preferences of running wheels were displayed similarly throughout all groups and showed no variation during the experiments (Figure 2(c)).
Change of clinical score and body weight
Body weight course of 1% DSS-treated mice showed a slight increase until day 3 followed by a continuous reduction from day 4 to day 7, similar to the observed course of VWR (Figure 3(a)). After a maximum drop of 10.4% on day 7, body weight restored baseline level on day 9. Both 0% DSS and control group maintained a slow overall increase of body weight above baseline (Figure 3(a)).
Body weight, clinical score and histological results: change of body weight during the experiment showed a statistically significant reduction in mice treated with 1% dextran sodium sulphate (DSS) (n = 6–7) compared with all other groups (0% DSS: n = 7; control: n = 7) from day 4 onwards with a maximum drop of 10.4% on day 7; (1% DSS vs. control: * = p < 0.05, ** = p < 0.01, **** = p < 0.0001; 1% DSS vs. 0% DSS:$ = p < 0.05,$$ = p < 0.01,$$$$ = p < 0.0001). Clinical signs (soft and/or bloody faeces) were subtle and demonstrated only in DSS treated animals ((b); * = p < 0.05). Histological analysis (c) revealed a statistically significant increase of score in the DSS treated animals compared with untreated mice (** = p < 0.01). For detailed results of statistical analyses see Supplementary Material Tables 3 and 4 online.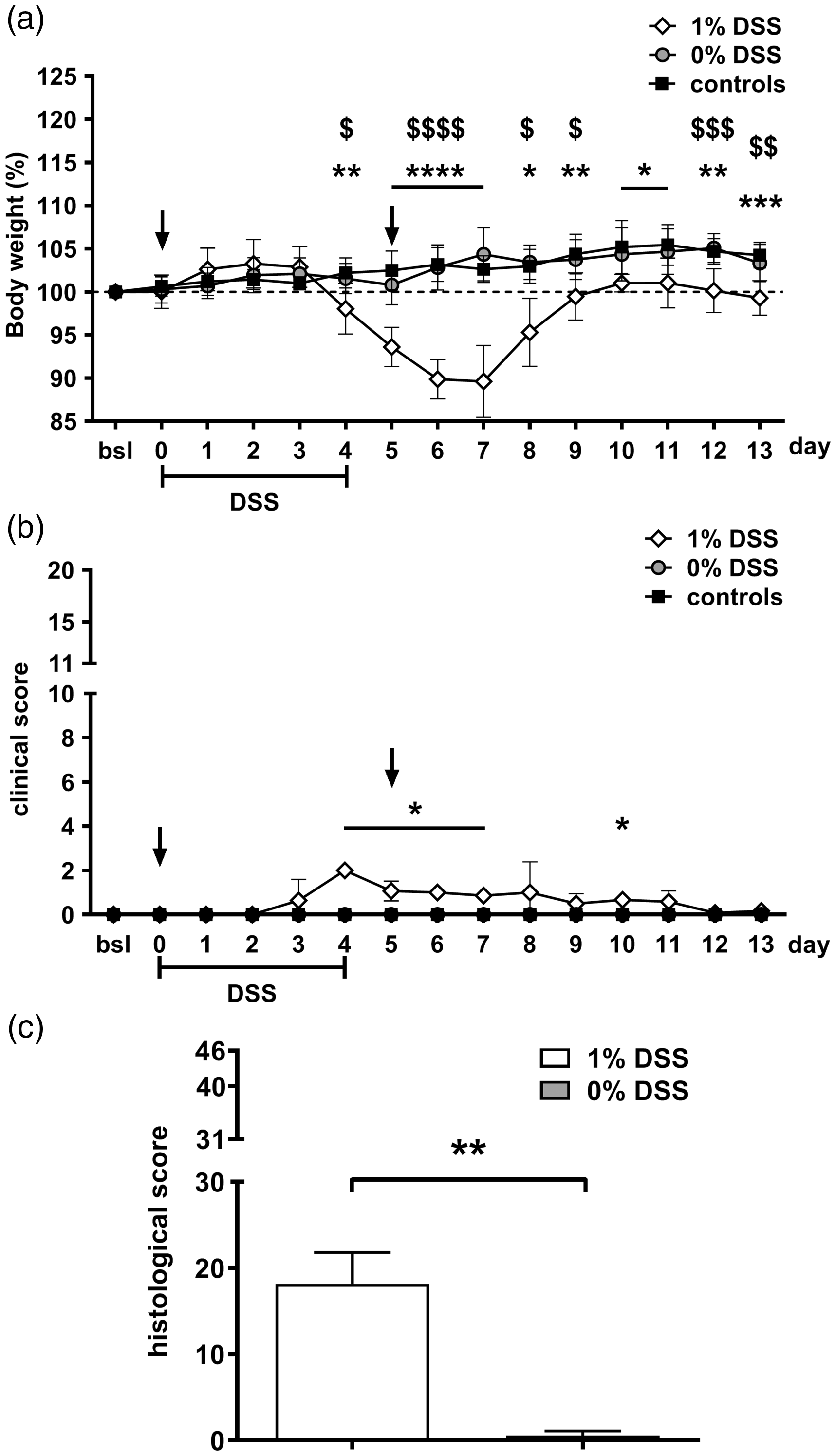
The clinical score displayed highest values in the 1% DSS group on day 4, which is attributable to most mice developing bloody faeces on the fifth day of DSS administration. After discontinuation of DSS treatment on day 5, the score decreased because faecal samples were soft but free of visible traces of blood (Figure 3(b)). The animals did not show any further clinical signs of disturbed wellbeing. Neither animals of the 0% DSS nor the control group exhibited any clinical signs of compromised welfare (Figure 3(b)).
Histological analysis
Histological analysis for evaluation of inflammatory changes in the colon of mice treated with 1% DSS at the end of the experiment still reached a score of 18 out of 46 points maximal score, demonstrating ongoing inflammation without full recovery. Pathological changes were dominated by a moderate infiltration of inflammatory cells into lamina propria, submucosa and lamina muscularis, also leading to peritonitis in three out of six mice. Additionally, alterations of crypt architecture and minimal oedema were found in DSS treated mice, while all respective non-treated mice depicted a physiological condition of the intestinal wall (Figure 3(c)).
Application of the cluster model
Representative transfer of day 7 data to the above-mentioned cluster model 5 revealed distinct differences between all three groups. The cluster model had been developed based on body weight and VWR training data from another DSS colitis experiment and the utilization of a k-means algorithm, resulting in the definition of two borders which allocate data points to one of three severity levels (levels 0, 1 and 2). These levels were suggested to be within the range of no to moderate impact of experimental procedures on the overall wellbeing of mice.
Clustering showed most animals of the 1% DSS group allocated to severity level 2, while 0% DSS treated animals predominantly clustered into level 1 and control animals mainly fall into level 0 (Figure 4(a) to (c)). The percentage distribution into severity level 2 was 86% in the 1% DSS group, 28% in 0% DSS group and 14% in the control group (Figure 4(d)).
All data points of the experiment based on voluntary wheel running (VWR) and body weight data (a): allocation of individual mice of the 1% dextran sodium sulphate (DSS) group, the 0% DSS group and the control group on day 7 ((b); n = 7 each) to severity levels 0, 1 and 2. (c) Percentage distribution in each group.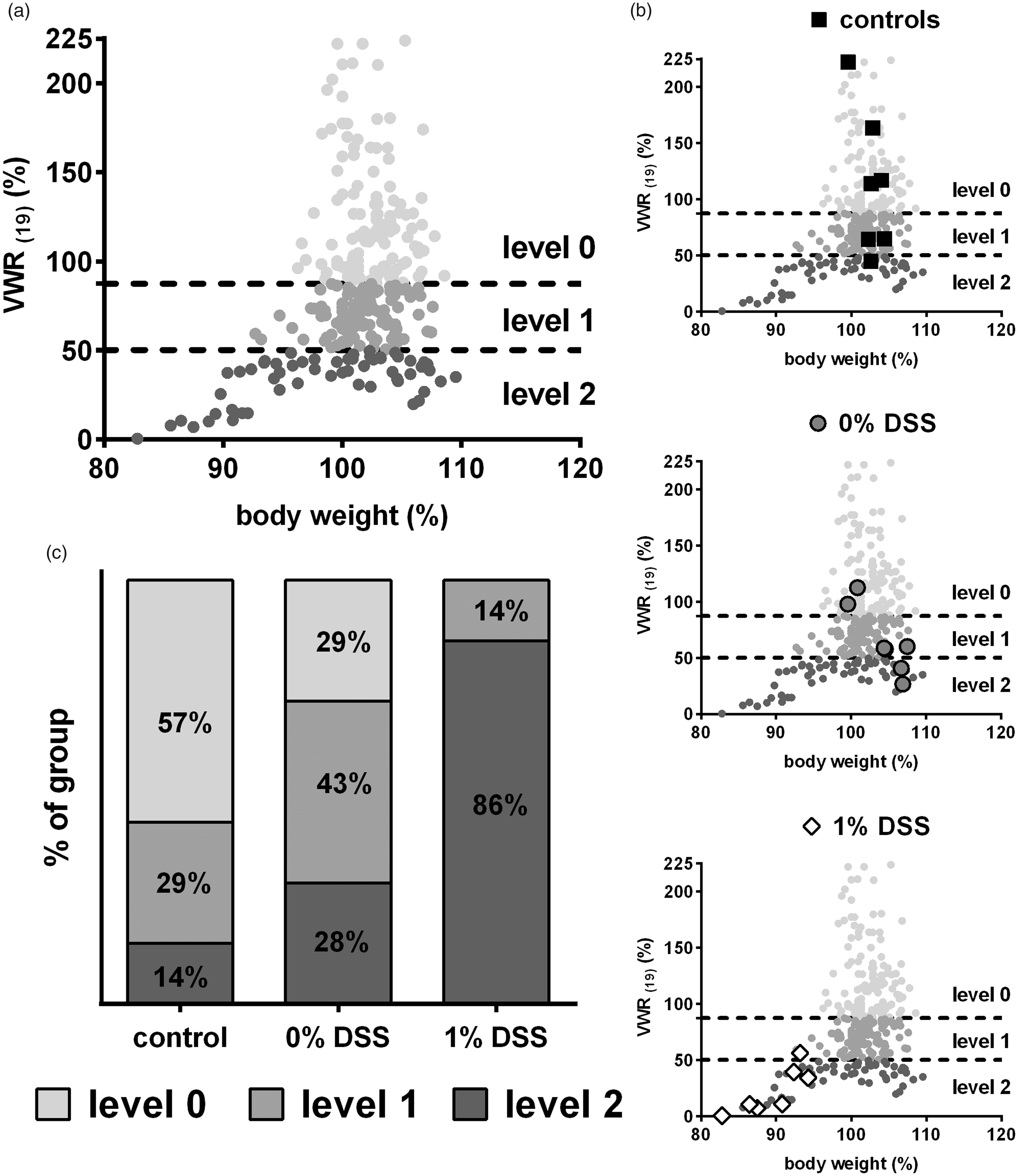
Discussion
Monitoring of VWR behaviour has been successfully used to assess individual severity levels in single housed mice. 5 The rationale of this study was to investigate whether this approach is also applicable in group-housed mice. Therefore, the impairment of wellbeing caused by DSS colitis was evaluated by VWR behaviour, clinical scoring and histological analysis of the colon. DSS treated mice showed a significant reduction in VWR and body weight and a significant elevation of the histological score, but very few signs of disturbed wellbeing when considering clinical scoring. Mice merely submitted to facial vein phlebotomy and faecal sampling also showed decreased VWR compared with control mice, while other parameters remained unaffected. Reduction of VWR has already been proven to indicate disturbed wellbeing, for example, in a model of migraine in rats, 16 post-surgical pain after partial hepatectomy 17 and in models of DSS colitis and restraint stress. 5 However, based on VWR and body weight data, the discrimination of individual levels of experimental severity was possible by integrating the data into a cluster model as described recently. 5 Applying this model, DSS-treated mice were allocated to the, within this study, highest severity level on day 7. However, a considerable number of mice that were solely subjected to sampling procedures did allocate to severity levels 1 and 2, also indicating a level of disturbed wellbeing. Therefore, the current study underlines and supports the applicability of a mathematical tool for severity assessment.
Wheel running itself is an intensely studied, but not yet fully understood, phenomenon, which has been attributed to various sources of intrinsic and extrinsic motivation, including exploration behaviour, escape, play and body weight maintenance. 1 Furthermore, there are concerns about VWR being a pathological, stereotypic behaviour because of its lack of purposeful function, 18 even though it is a behaviour also exerted by wild mice. 19 In this study, a stable use of running wheels was observed despite the vast opportunity for social interaction and the large area provided, suggesting a non-stereotypic behaviour due to other intrinsic or extrinsic physiological factors. In addition, none of the animals showed an over-excessive use of running wheels.
Generally, in light of mice being gregarious animals, it is recommended to avoid individual housing whenever possible.8,9 Indeed, many studies have compared individual versus group housing of rodents under laboratory conditions. Späni et al. 20 observed a higher heart rate in singly-housed male mice compared with group-housed individuals, pointing to potential discomfort. However, regarding behavioural tests of group- versus individually-housed mice, results are diverse. Female BALB/c mice housed without conspecifics have been reported to show higher levels of parameters related to anxiety in contrast to C57BL/6 females. 11 In addition, no effects of individual housing on behaviour were observed in the modified hole board test and single housing did not lead to increased stress markers.10,11,21 Furthermore, in a study investigating behaviour and recovery after minor laparotomy, no negative effects related to single housing were observed 10 . However, the fact that some studies did not report any negative effects of single housing does not convincingly advocate for a recommendation of single housing. In a preference study 12 even subordinate male mice preferred companionship when presented with the choice between an empty cage and a cage inhabited by another male mouse. However, aggressive behaviour in male mice might require separation to prevent severe injury or even death. As this can apply even to female mice of certain strains, genetic influences on social behaviour should also be taken into consideration.
Companionship itself has a marked influence on behaviour in mice with regard to home cage activity 22 and social interaction has a profound effect on certain parameters. For example, in mice subjected to restraint stress, the presence of a conspecific diminishes the negative impact of the treatment on working memory. 23 Furthermore, significantly elevated stress hormone levels were detected in non-tested members of a group, in which only some of the mice were submitted to behavioural testing. 11 Empathic behaviour has also been described in a three-chambered social approach test. 24 Therefore, the condition of an individual might alter the behaviour of other group members and vice versa.
However, for VWR, single housing was required due to technical limitations to assess individual behaviour within a group. Moreover, the effects of social interaction or the deprivation thereof on VWR have not yet been characterized in depth. Interestingly, in this study, group-housed mice showed about 50% less VWR compared with single-housed mice analysed in a previous study. 5 This marked reduction in VWR related to social housing is also in accordance with results from Sherwin, 22 who found a marked decrease in motivation of mice to access a running wheel when housed in groups. In addition, Dewan et al. 25 observed a reluctance to run in wheels which had previously been used by other individuals. Also, behavioural testing of single-housed male B6 and DBA mice showed varying results, but a distinguishable effect in terms of hyperactivity related to isolation. 26 Thus, group-housing may avoid confounding VWR data by hyperactivity or stereotypic behaviour. Therefore, a system that allows assessment of VWR on an individual level despite group-housing is highly desirable.
In summary, the present study proves VWR to be a suitable indicator of disturbed wellbeing, also in group-housed mice. The implementation of RFID technology into an automated wheel running system enables individual severity assessment without impairment of natural social behaviour of the animals.
Supplemental Material
LAN879455 Supplemetal Material - Supplemental material for Wheel running behaviour in group-housed female mice indicates disturbed wellbeing due to DSS colitis
Supplemental material, LAN879455 Supplemetal Material for Wheel running behaviour in group-housed female mice indicates disturbed wellbeing due to DSS colitis by Nora Weegh, Jonas Füner, Oliver Janke, York Winter, Christian Jung, Birgitta Struve, Laura Wassermann, Lars Lewejohann, André Bleich and Christine Häger in Laboratory Animals
Footnotes
Acknowledgment
AB and CH share senior authorship.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Federal Ministry of Economics and Energy, Zentrales Innovationsprogramm Mittelstand (ZIM) (grant/award number: KF3465361TS4), Deutsche Forschungsgemeinschaft (grant/award number: FOR 2591, ‘BL953/10-1’).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
