Abstract
The choice of laboratory cage bedding material is often based on both practical and husbandry issues, whereas behavioral outcomes rarely appear to be considered. It has been noted that a breeding success difference appears to be associated with the differential use of aspen chip and aspen shaving bedding in our facility; therefore, we sought to analyze breeding records maintained over a 20-month period. In fact, in all four mouse strains analyzed, shaving bedding was associated with a significant increase in average weanlings per litter relative to chip bedding. To determine whether these bedding types also resulted in differences in behaviors associated with wellbeing, we examined nest building, anxiety-like, depressive-like (or helpless-like), and social behavior in mice housed on chip versus shaving bedding. We found differences in the nests built, but no overall effect of bedding type on the other behaviors examined. Therefore, we argue that breeding success, perhaps especially in more challenging strains, is improved on shaving bedding and this is likely due to improved nest-building potential. For standard laboratory practices, however, these bedding types appear equivalent.
Laboratory rodent bedding choice varies from institute to institute and is often based on practical issues such as storage, reliability of source, and cost. Animal husbandry issues such as absorbency, potential for contamination or toxicity, resistance to microbial growth, and provision of warmth and insulation also influence bedding choice. 1 While optimization of animal behavior is an additional criterion for bedding choice, there have been relatively few controlled studies with behavioral endpoints. When our mouse colony personnel noticed a difference in breeding success between two bedding types used in the breeding facility, we were compelled to quantify this. In addition, we were interested in whether there were differences in behaviors sensitive to altered wellbeing between mice housed with the two substrates. Behavioral alterations constitute a parameter used in the assessment of animal welfare2–4 and can provide insight into physiological functions important for all animal uses in biomedical research.
Aspen bedding is a particularly popular choice because this hardwood has a low rate of contamination from tars and resins. Unlike some other wood bedding, aspen does not significantly affect liver enzymes and has thus far not been linked to any demonstrable physiological or behavioral confounding. For example, Beta Chip bedding (maple, birch, and beech) enhances mucosal immune responses, 5 and cedar and pine chip bedding both affect liver enzymes. 6 In addition, lower ammonia concentrations have been found when hardwood bedding has been used relative to pine, paper, pulp, and corncob. 7 Corncob bedding, although having excellent absorptive properties, 8 has been shown to have estrogenic properties and, in fact, has reduced social withdrawal behavior of female California mice raised on this bedding. 9
There have been a few published reports of bedding effects on breeding success in mice. For example, when equal numbers of rat pups were born on corncob, shredded aspen, and cedar shavings, the mortality rate was about 50% by three weeks of age on cedar shavings, 10 an effect attributed to a toxin either ingested or inhaled by the pups. Vermiculite also appeared to have a detrimental effect on breeding outcomes in mice. 11 In fact, in a comparison study, vermiculite had the most detrimental effect on breeding success, followed by pine shavings, with eucalyptus pulp having the best outcome of the three. 12 Pregnant CF1 mice chose to have their litters on flakewood and avoided cellulose bedding. 13 The chemical components of these substrates (for example volatile hydrocarbons in cedar and the deodorizing agent added to cellulose) were primarily blamed for these problems; however, the production and quality of nests using these bedding types were not described in these studies, nor was the size of the individual particles of bedding. However, a recent study found that supplying breeding cages with nesting material (8 g of either Enviro-dri or Nestlets) significantly improved breeding performance in C57BL/6 (B6), BALB/c, and CD-1 mice. 14
Tests of mouse bedding preference vary somewhat due to the specific bedding materials used in the studies. Both B6 and BALB/cBYJ mice preferred larger particles and ones that could be manipulated, 15 and female B6 mice preferred shaving to chip bedding. 16 In addition, given a choice of four different paper bedding materials, ICR mice preferred the one with the largest fibrous shape, and the authors noted that mice appeared to have the easiest time building their nests with this bedding. 17 Indeed, providing mice with a layer of natural corn husk material to allow burrowing was found to decrease bouts of aggression following the formation of new housing groups of aggressive mice. 18 In addition, ICR mice chose Agrebe (fibrous cloth nesting material from Globe Inc, Japan) over the less manipulable paper Shepherd Shack, wood cylinder, and polycarbonate Mouse-Igloo as a shelter material. 19 Overall, it appears that the nestability of the materials seems to be more important than the specific material used. 20
The Scripps Research Institute (TSRI) has been using #7090A Harlan Teklad Aspen Sani-Chip bedding (chip) for almost 30 years. Recently, when the breeding facility was outfitted with new caging including an automatic watering system, #12000 Newco Aspen Shaving bedding (shaving) was purchased with the idea that this bedding, made of larger pieces, would be less likely to make its way into the sipper tubes and therefore reduce cage flooding incidence. In the meantime, it was found that both types of bedding could be used successfully with the automatic watering system. However, the breeding colony staff noted that breeding success seemed to be greater when the shaving bedding was used.
The goal of this experiment was to compare breeding success and animal wellbeing on chip versus shaving bedding. We selected tests that would measure subtle, but important, impacts on wellbeing: specifically, nesting behavior, anxiety-like, helpless-like or depressive-like, and social behaviors.
Materials and methods
Animals
Male and female mice were used in the present experiments. For the behavioral tests, group sizes were eight mice per bedding type, housing condition and sex. The behavioral tests were scored by trained technicians blinded to the group condition of the mice. All the mice were housed in individually ventilated caging (IVC) under sanitary conditions in light (12 h on, 12 h off) and temperature (21 ± 2℃) controlled vivarium rooms. TSRI maintains a centralized animal care and use program assured by the Public Health Service (PHS), registered with the United States Department of Agriculture (USDA), and accredited by the Association for Assessment and Accreditation of Laboratory Animal Care, International (AAALAC). Animals are maintained within facilities provided by the TSRI Department of Animal Resources (DAR). DAR is headed by a full-time TSRI laboratory animal veterinarian who is assisted by a staff of capable veterinary, animal care and technical support personnel. DAR personnel are responsible for the daily care and maintenance of animals housed at TSRI. Housing and care of animals is consistent with the PHS Policy, the Guide for the Care and Use of Laboratory Animals, the Animal Welfare Regulations, and other applicable state and local regulations. All procedures were approved by the TSRI Institutional Care and Use Committee and were in concordance with the National Research Council’s ‘Guide for the Care and Use of Laboratory Animals’. In addition, the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines of the National Centre for the Replacement, Refinement and Reduction of Animals in Research were followed in the design and presentation of this study.
Breeding assessment
While the personnel involved in colony management felt that breeding success was better on shaving bedding than on chip bedding, supportive data were needed. Records were compiled from the TSRI rodent breeding colony between January 2012 and August 2013 for the four strains that were maintained for TSRI investigator research and for the institutional sentinel program. These four strains were C57BL/6 J (B6; Jackson Laboratory, Bar Harbor, ME, USA), Hsd:Athymic Nude-Foxn1nu (Nude; Harlan Laboratories, Indianapolis, IN, USA), B6(Cg)-Tyrc-2J/J (B6-albino; Jackson Laboratory), and BALB/cByJ (BALB; Jackson Laboratory). The measure we focused on was the number of mice weaned per litter. Because we had pup numbers at birth as well as at weaning, we were also able to calculate the percent of litters with deaths occurring between birth and weaning. We considered examining numbers of litters per breeding pair, but we found that the variation in the time period each pair was bred would be a confounding factor. While this was not an a priori designed experiment, in examining these records we found that we had sufficient litter numbers per strain and per bedding type for adequate statistical power in comparing bedding types.
Behavioral analysis
B6 mice were chosen for the behavioral analyses because they make up more than half of the TSRI breeding colony and are the most commonly used mouse strain in research, partly because they are used in the generation of mutant lines. Eight-week-old male and female B6 mice obtained from the TSRI breeding colony were bred on #7084 Teklad pelleted paper bedding (Harlan Laboratories) to control the environment for all pups being born (i.e. all experimental mice underwent the same initial experience). Pups were weaned at 21–23 days and moved into cages with either #12000 Newco Virgin Aspen Hardwood Shavings (shaving; Newco, Rancho Cucamonga, CA, USA) or #7090A Teklad Aspen Sani-Chips (chip; Harlan Laboratories). Litters were separated by sex and then as equally as possible into the two bedding types, either singly- or pair-housed. Thus we were able to examine the effects of shaving versus chip bedding on behavior in males and females and in singly- versus pair-housed mice (eight groups in total). Other than the bedding difference, the mice were treated identically throughout the experiment. Cages were changed every two weeks and 1.0–1.3 g of fresh cotton nestlet material (Ancare, Bellmore, NY, USA) was included in all cages. The breeding, weaning, and experimental housing were staggered to keep the weaning ages consistent and to reach the goal of having eight mice per group, for a total of 64 mice.
Starting at 10 weeks of age, nests were examined, and the mice were tested in the light/dark transfer test, tail suspension test, and social interaction test, each separated by 4–7 days. These tests were chosen because they are standardized, validated assays of mouse anxiety-like, depressive-like or helpless-like, and social behavior, and thus are considered to be sensitive to mouse wellbeing.
Nest construction
Nest building is a natural rodent behavior that relates to reproduction, temperature regulation, shelter, and social behaviors. In addition, burrowing and nest building have been shown to be sensitive to wellbeing in that these behaviors in mice are disrupted following surgery.21–23 In addition to the shaving or chip bedding, 1.0–1.3 g of cotton nestlet material (Ancare) was placed in each cage, and 24 h later the nests were assessed on a rating scale of 1–5 based on cotton nest construction. 24
Light/dark transfer test
The light/dark transfer procedure has been used to assess anxiety-like behavior in mice by capitalizing on the conflict between exploration of a novel environment and the avoidance of a brightly lit open field.25,26 The apparatus was a rectangular box made of Plexiglas divided by a partition into two environments. One compartment (14.5 × 27 × 26.5 cm) was dark (8–16 lux) and the other compartment (28.5 × 27 × 26.5 cm) was highly illuminated (400–600 lux) by a 60 W light source located above it. The compartments were connected by an opening (7.5 × 7.5 cm) located at floor level in the center of the partition. Mice were placed in the dark compartment to start the 5 min test. The time spent in the light compartment was used as a predictor of anxiety-like behavior, in that a greater amount of time in the light compartment was indicative of decreased anxiety-like behavior.
Tail suspension test
The tail suspension test is a well-characterized test for assessing depression-related and antidepressant-like behaviors.27,28 In this test, mice were suspended from a metal rod mounted 50 cm above the surface by fastening the tail to the rod with adhesive tape. The duration of the test was 6 min, and immobility was measured during the last 4 min as typically reported. 29 Immobility was defined as the absence of any limb or body movements, except those caused by respiration. Increased immobility is associated with increased depressive-like behavior.
Social interaction test
This test was originally developed to model in mice aspects of autism spectrum disorders in humans30,31 and has been used widely by behavioral neuroscientists. 32 Individuals on the autism spectrum show aberrant reciprocal social interaction, including low levels of social approach and unusual modes of interaction. The social interaction apparatus was a rectangular, three-chambered Plexiglas box, with each chamber measuring 20 cm × 40.5 cm × 22 cm (L × W × H). Dividing walls were clear with small semicircular openings (3.5 cm radius) allowing access into each chamber. The middle chamber was empty, and the two outer chambers contained small, round wire cages (Galaxy Cup: Spectrum Diversified Designs Inc, Streetsboro, OH, USA) during testing. The mice were habituated to the entire apparatus with the round wire cages removed for 5 min. To assess sociability, mice were returned to the middle chamber, this time with a stranger mouse (B6 of the same sex being tested, habituated to the wire cage) in one of the wire cages in an outer compartment, and with another identical wire cage in the opposite compartment. Time spent in the chamber with the stranger mouse and time spent in the chamber with the novel object were recorded for 5 min. For the social novelty preference test, mice were returned to the middle chamber, this time with the original mouse (familiar mouse) in its chamber and a new unfamiliar mouse (novel mouse) in the previously empty wire cage. Again, time spent in each chamber was recorded for 5 min. Young male B6 mice spent more time with the novel mouse in the sociability test and also more time with the new novel mouse in the social novelty test. 30
Statistical analysis
Kruskal–Wallis tests (SPSS software package; IBM Corp, Armonk, NY, USA) were used to assess the differences in births and weanlings between shaving and chip beddings for the four strains investigated. The nest building and light/dark transfer behavioral results were initially analyzed using three-way analysis of variance (ANOVA) with the between-subject factor bedding type, caging structure and sex. Tail suspension and social interaction data were initially analyzed using four-way repeated measure ANOVAs with the same between-subject factors as above in addition to the within-subject factor time (tail suspension) or compartment time (social interaction). In order to focus on the influence of bedding, ANOVAs were performed separately for males and females. Furthermore, if there were effects resulting from the caging, ANOVAs were performed separately for singly- and pair-housed mice, again, to allow focus on the bedding condition. Chi-square, F values and P levels were reported, with significance set at P < 0.05.
Results
Breeding assessment
Breeding characteristics associated with shaving and chip bedding in C57BL/6J (B6), Hsd:Athymic Nude-Foxn1nu (Nude), B6(Cg)-Tyrc-2J/J (B6-albino), and BALB/cByJ (BALB) mice.

Significantly greater than Chip condition using Kruskal–Wallis test (see text for details). Note that statistical differences were detected even when median and interquartile range (IQR) values were identical. This happens when the deviation scores around the median are altered by the factor in question, in this case the bedding type.
Behavioral analysis
Nest construction
Mice on shaving bedding did not always incorporate the cotton nestlet material into their nests; however, they made symmetrical nests with high sides, sometimes with a partial roof. Mice on chip bedding made nests using the nestlet material; therefore, these were scored using the scale of Deacon,
24
which was devised for assessing the use of this material. Male singly-housed mice had average nest scores of 4.0 ± 0.3, pair-housed males had scores of 3.0 ± 0.5, pair-housed females had scores of 4.0 ± 0.2, and singly-housed females had scores of 4.0 ± 0.0. Average scores of 3–4 were associated with the nestlet being mostly shredded to an identifiable, albeit flat, nest.
24
Male pair-housed mice had lower nest building scores than male singly-housed mice (P < 0.05), but in general the chip-housed mice used the nestlet material to build their nests. The nests built of shavings appeared to be sturdier. Unlike the cotton nests in the chip cages, the shaving nests were not destroyed by sudden movements of the mice. We also found that the mice in the shaving bedding used the larger shaving pieces to build their nests and kept their nest areas clean and dry. An example of each nest type is shown in Figure 1.
Nest examples in cages with aspen shaving bedding (top) and aspen chip bedding (bottom).
Light/dark transfer test
There were no significant differences between bedding types regarding either the time spent in the light compartment or the number of transitions between the dark and light compartments (Figure 2). There were also no significant effects from housing condition (single versus pair), sex, or any combination of these factors. These results suggest that anxiety-like behavior as estimated using this test is not differentially impacted by chip versus shaving bedding, nor does bedding type interact with housing condition or sex in these measures.
Light/dark transfer test results. Time spent in the light compartment in males (a) and females (b) and number of dark-to-light compartment transitions in males (c) and females (d).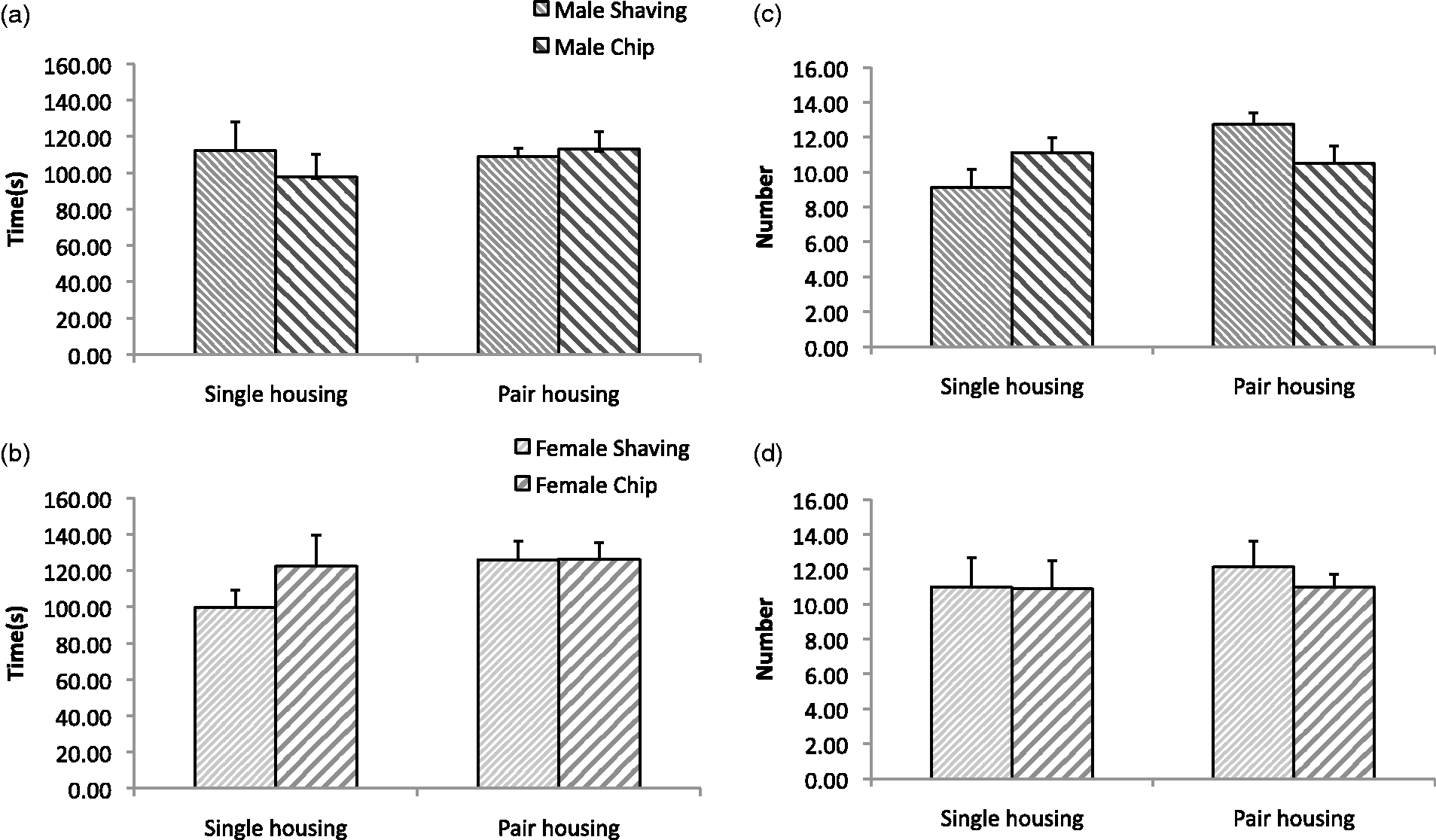
Tail suspension test
There was no effect of bedding type on immobility times in this test, nor were there any significant interactions between bedding and housing or sex (Figure 3). There was, however, an overall effect from housing conditions (F(1,52) = 6.3, P < 0.05), and further investigation showed that this effect came from male pair-housed mice having increased immobility times than male singly-housed mice (F(1,27) = 9.0, P < 0.01). These results suggest that depressive-like (helpless-like) behavior as assessed using this test is not differentially affected by chip versus shaving bedding.
Tail suspension test results. Seconds immobile in minutes 3–6 in singly-housed males (a), singly-housed females (b), pair-housed males (c), and pair-housed females (d).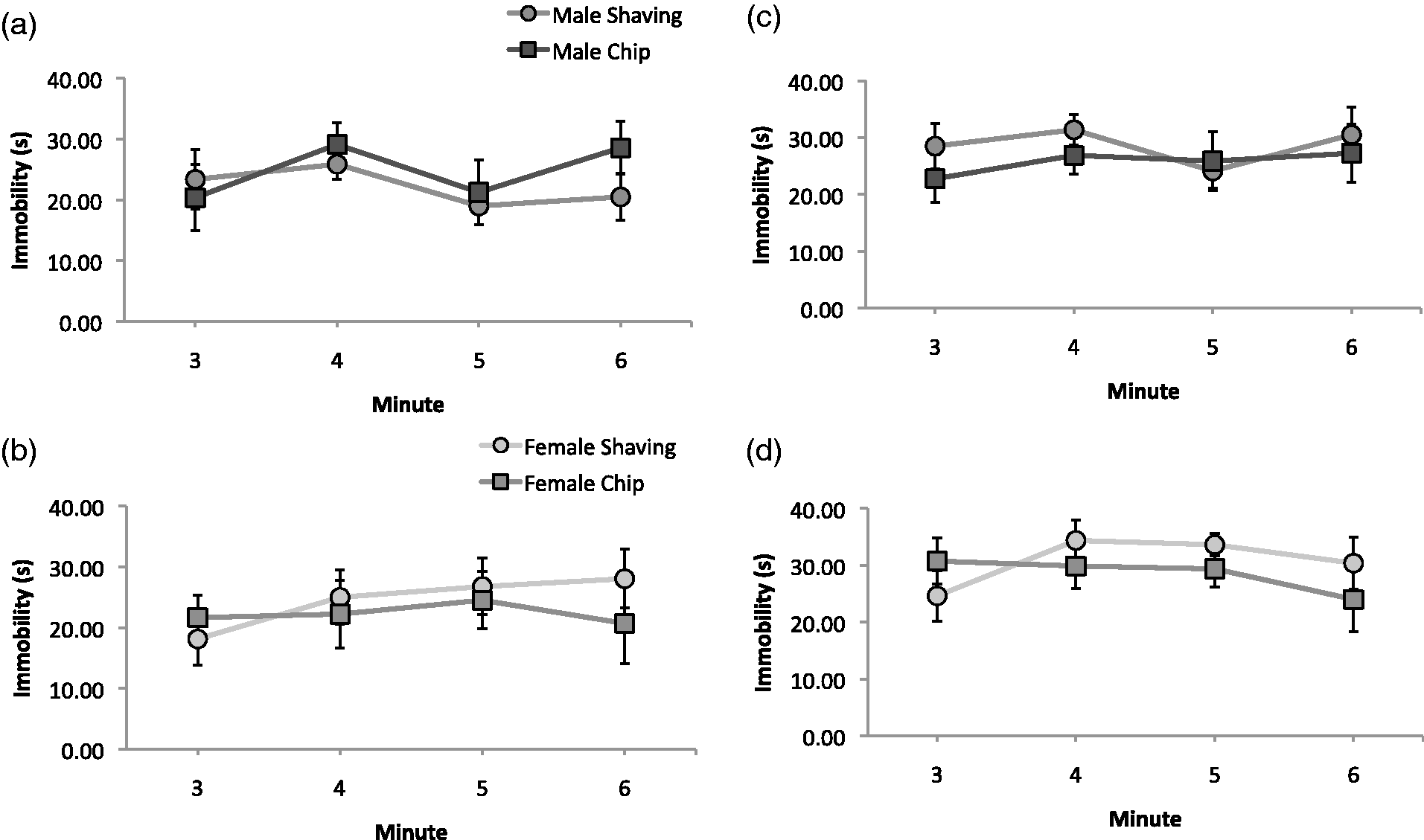
Social interaction test
Two measures of social interaction were gleaned from this test: sociability (Figures 4a and 4b: comparison of time spent with a novel object versus a novel mouse); and social novelty (Figures 4c and 4d: comparison of time spent with a familiar mouse versus a novel mouse). Overall, in the sociability test, there was a significant difference between time spent in the two distal compartments (novel mouse > novel object; F(1,56) = 31.6, P < 0.0001). There were no effects from bedding, nor any interaction involving bedding. There was however a significant effect of housing on sociability (F(1,56) = 6.0, P < 0.05). Looking at sociability in each group separately, although almost significant in several groups, only the male pair-housed shaving bedding (F(1,7) = 9.9, P < 0.05), female pair-housed shaving bedding (F(1,7) = 14.9, P < 0.01), and female singly-housed chip bedding (F(1,7) = 9.6, P < 0.05) groups spent significantly more time with the novel mouse than with the novel object. In the social novelty test, there was only a moderately significant difference between time spent in the two distal compartments (novel mouse > familiar mouse; F(1,56) = 4.2, P > 0.05) and no effects from bedding. Social novelty was only significant in male pair-housed shaving bedding (F(1,7) = 8.9, P < 0.05) and female singly-housed chip bedding (F(1,7) = 10.8, P < 0.05) groups. Overall, there were no consistent trends toward increased sociability or social novelty seeking related to bedding types.
Social interaction test results. Sociability (time spent in chamber with novel mouse versus novel object) in males (a) and females (b) and social novelty (time spent in chamber with new novel mouse versus familiar mouse) in males (c) and females (d). *P < 0.05 difference between time spent with novel mouse and novel object or familiar mouse and new novel mouse.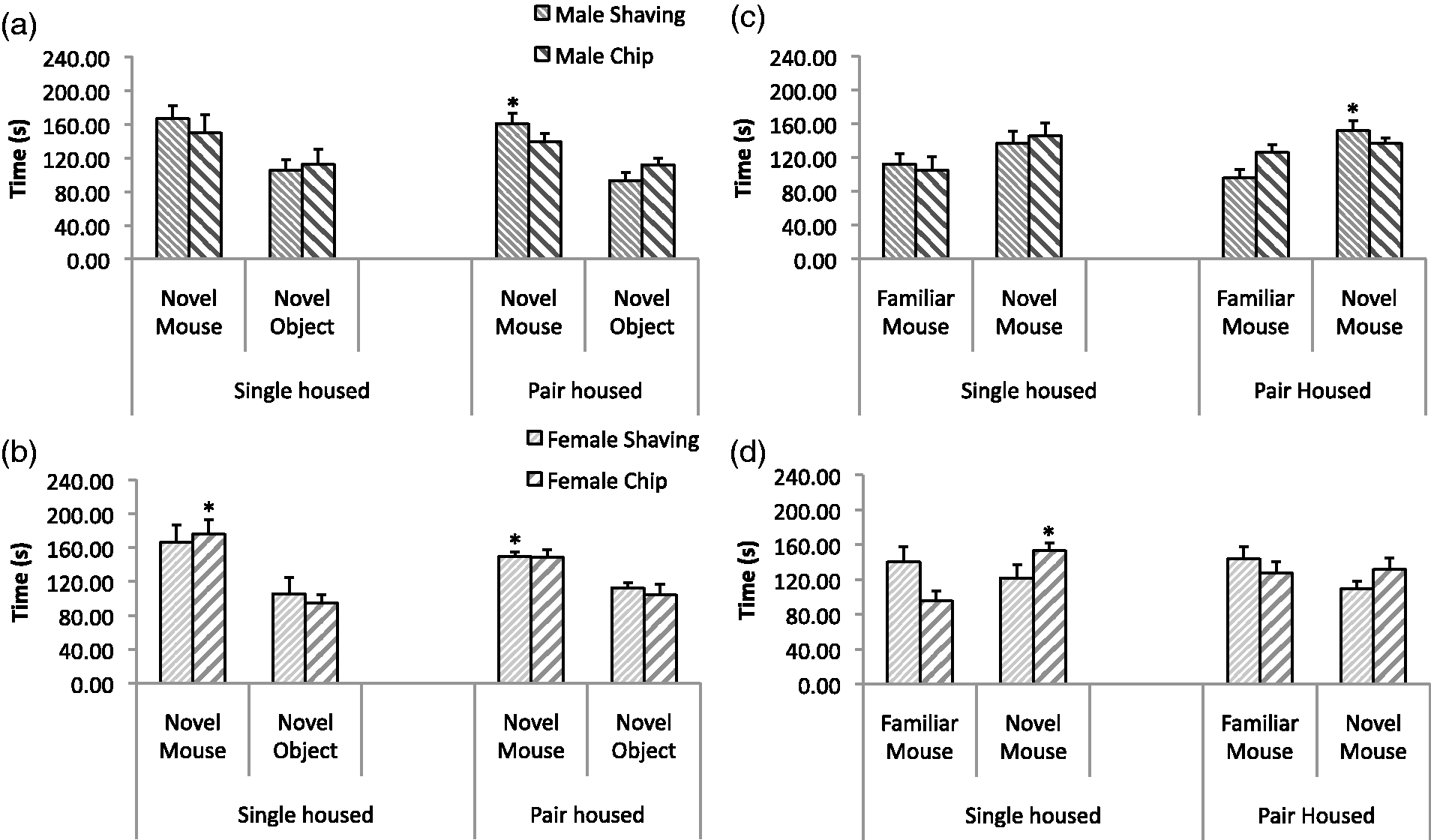
Discussion
The observation of improved breeding outcome on shaving versus chip bedding was supported by an analysis of records compiled over a 20-month period. Specifically, the number of mice weaned per litter was significantly greater with shaving bedding than with chip bedding across all four standard strains bred at TSRI. Interestingly, while the number of mice born per litter also differed between bedding types, these differences were not consistent across the strains and were of smaller magnitude. This indicates that the primary breeding difference was in post-birth survival, suggesting that the beddings differentially impact maternal care and/or nest quality. The B6, B6-albino, and BALB mice are Jackson Labs strains and are described as ‘good breeders’ (regularly producing 3–7 pups per litter), and the Nude mice are from Harlan Laboratories, where litter size is about 7. Our breeding efficiency is within these standards. Interestingly, the increase in number of weanlings on shaving bedding than on chip bedding was greater in the three ‘good’ breeding strains than in the ‘very good’ Nude breeders.
Mice on chip bedding built nests using only the cotton nestlet material, whereas mice on shaving bedding used primarily the shavings, with some incorporation of the cotton material. The shaving nests were taller than the cotton nests in the chip cages, and the sides were almost always above the level of the inhabitants. These nests also appeared to be of studier construction because movements of the mice did not disrupt the nests when staff removed cage tops. Finally, the shaving nest material appeared to be kept cleaner and drier by the mice, and even at the two-week changing time nests were noted in these cages. Cotton nests in the chip bedding, on the other hand, grew disorganized and messy by about 3–4 days post cage changing and seemed much more susceptible to disarray from movement in the cage. While this nest difference in combination with the overall bedding difference did not impact the sensitive behaviors we examined, it might at least partly account for the difference in breeding efficiency observed with these two bedding types. For example, a deeper, sturdier nest would more likely enable pup temperature regulation, safety, and cleanliness than a shallow, less stable, and dirtier nest.
Overall, bedding type had no effect on tests of anxiety-like, depressive-like or helpless-like, or social behaviors in young adult B6 mice, suggesting that there were no differences in overall wellbeing associated with these two bedding types. Behavioral changes in response to stress have been assessed using procedures such as open field and cage emergence tests, which examine activity levels, exploratory drive, and anxiety-like behavior in mice.33,34 The light/dark transfer test used in this study measures activity levels, exploratory drive, and how mice deal with the conflict between exploratory drive (exploring novel surroundings) and being in fear-provoking situations (brightly lit, open novel surroundings). 35 The tail suspension test puts mice into an uncomfortable situation (hanging by their tail) and assesses immobility as a measure of the emergence of hopelessness 36 and may model liability to general distress. 37 We have used these tests in phenotypic batteries routinely in TSRI’s Mouse Behavioral Assessment Core and have found that mice are sensitive to anxiolytic and antidepressant administration. For example, the anxiolytic benzodiazepine chlordiazepoxide decreased anxiety-like behavior in the light/dark transfer test, 38 and the antidepressant citalopram decreased immobility in the tail suspension test. 39
Mice devote a significant portion of their natural behavioral repertoire to social behaviors such as play (particularly in juveniles), patrolling, dominance/submission behaviors, sniffing, fighting, and reproduction-related behaviors. 40 The social interaction test used in the present experiment has two components. In the sociability test, time spent with and in the vicinity of a novel mouse is compared with time spent exploring or near a novel object. In the social novelty test, time spent with a new novel mouse is compared with time spent with the now familiar mouse. B6 mice have been shown to spend more time with the novel mouse in both tests, 30 suggesting that they have a preference for social interaction and for novelty in social situations. Behavior in this test is sensitive to disruption by drugs that affect the glutamatergic, dopaminergic, and serotonergic systems. 41 These systems have been implicated in mood, motivation, and general wellbeing and are affected by inflammation 42 and stress.43,44
A behavioral readout associated with mouse stress would suggest the presence of distress. However, the acceptance of behavioral changes as indicators of distress requires their correlation with stress-induced biological changes. 45 One of the conditions studied in great depth at our institute is drug and alcohol withdrawal. Withdrawal from chronic drug and alcohol exposure is associated with a negative affect in humans and laboratory rodents46,47and stress system sensitization, 48 as well as dysregulated pain processing. 49 Evidence of this comes from extensive physiological, motivational, and behavioral testing spanning over 20 years. Therefore it is widely accepted that withdrawal produces a state of discomfort, distress, and stress. Increases in anxiety-like behavior during alcohol withdrawal have been evaluated using the light/dark transfer test (example 50 ). Similarly, amphetamine withdrawal has been shown to increase immobility in the tail suspension test. 51 By contrast, while alcohol withdrawal decreases exploration in the social interaction test, it does not disrupt sociability in a small group of young mice, suggesting that sociability can be dissociated from anxiety-like behavior. 41 Therefore, while the tests chosen presently appear to be sensitive to wellbeing, they measure different aspects of this state.
Our experimental design involved the examination of behavior in mice born and weaned on a third bedding type, with mice being assigned to the two study beddings at weaning. This was done so that litters could be evenly divided between the two experimental conditions. It is important to distribute littermates across different treatment groups even when testing genetically identical inbred mice because prenatal environment, parental care, and cage mate social interactions can contribute to behavioral phenotypes.52–54 However, in controlling for this potential experimental confounding, we may have revealed another: while we controlled for pre-weaning environmental conditions, it was possible that there might have been pre-weaning or even prenatal effects on adult behaviors from different bedding types.
Other studies examining the effects of bedding types on animal wellbeing with respect to behavioral parameters have typically focused on home cage behaviors such as grooming, feeding, drinking, nesting, sleeping, climbing, and aggression. For example, while cage density and sanitation frequency have no effect, mice maintained on a combination of aspen and cellulose display overgrooming behavior and have higher mortality rates relative to mice maintained on either of these bedding types alone. 55 However, these effects cannot be related to in-cage ammonia levels, lung pathology, or fecal corticosterone levels and therefore are deemed to be of little biological significance. As mentioned above, certain bedding materials have been shown to affect physiological parameters and these could certainly have an impact on behavior. For example, corncob bedding alters social withdrawal behavior of female California mice, likely due to its estrogenic properties. 9
Interestingly, there were few differences between males and females in the tests examined. While sex is clearly an important factor mediating behavior, interactions between sex and genotype and sex and housing conditions (housing density and isolation) are often more profound than the main effects of sex. 56 Indeed, we found a significant immobility × sex × housing interaction in the tail suspension test, with singly-housed males showing decreased immobility relative to pair-housed males. While main effects of sex in B6 mice in the tail suspension test have been reported (i.e. Ref 37), the housing of five mice per cage in this case may have been a contributing factor. Sex differences in anxiety-like behavior in B6 mice have been shown in some tests (for example, elevated plus maze open arm time), but not in others (for example, open field and elevated plus maze open arm entries), and principal components analyses of measures in these tests have shown differential loading of activity versus anxiety in males and females. 57 Finally, while sex differences in direct social interaction in B6 mice have been found, 57 this is primarily due to differences in sniffing, grooming, and digging. In our case, we focused on time spent in the chamber with the mouse or object and not time spent in direct sniffing, and it is therefore likely that sex differences were diluted. We chose to use the most robust measures in each of the tests to limit the focus to bedding effects.
In conclusion, while mouse wellbeing appears to be equal under both bedding conditions, breeding efficiency is significantly improved on shaving bedding. The shaving bedding used in this study was approximately 50% less expensive than the chip bedding used. Therefore, we argue that, for standard laboratory practices, both choices are fine. Additionally both bedding types have been, and are being, used successfully in IVC systems. However, in the case of breeding facilities, and perhaps especially with strains that are poor-to-good breeders, such as three of the strains we assessed, shaving bedding is not only a less expensive option, but can also increase breeding efficiency. Ultimately, the most important consideration is to ensure that breeding mice have ample nesting material and that it is sufficiently supplemented in cases in which chip-style bedding is used.
Footnotes
Acknowledgements
The authors thank Laura Beasley, Diana Saucedo, Corinne Trillo, Ana Barrios, Barb Engle, Mercedes Gutierrez, Julie Culver, Charlee Henschel, Hazel Garcia and Benigno Flores for excellent animal care and record maintenance; and Sandra Hively for critically reading this manuscript. The Mouse Behavioral Assessment Core and Department of Animal Resources of TSRI supported this work.
Funding
FundRef Funding Sources TSRI Department of Animal Resources, TSRI Mouse Behavioral Assessment Core.
Declaration of conflicting interests
None declared.
