Abstract
An impaired intestinal epithelial barrier is thought to be a major factor in the pathogenesis of human inflammatory bowel disease (IBD). IBD is frequently investigated by inducing a damaged barrier in murine models of colitis. This can be done by feeding mice with dextran sulfate sodium (DSS) polymers in their drinking water. Refinement measures should focus on alleviating unnecessary suffering during this probably painful condition. Appropriate parameters are needed to decide when to terminate the experiments. Our aim was to investigate whether a change in burrowing behaviour is a sensitive measure of animal welfare in murine models of colitis. Acute colitis was induced in C57BL/6 mice with 2.0% DSS over nine days. The burrowing test is based on the species-typical behaviour of mice to spontaneously displace items from tubes within their home cage. As a burrowing apparatus, a water bottle (250 mL, 150 mm length, 55 mm diameter) filled with 138–142 g of pellets of the animal’s diet was used. The presence of intestinal inflammation as a result of acute DSS-induced colitis was confirmed by a decrease in body weight, colon length and an increase of murine endoscopic index of colitis severity, histological score and spleen weight in the group receiving DSS as compared with the control group. An onset of intestinal inflammation correlated with a significant decrease in burrowing behaviour (P < 0.05). Altered adrenal gland histology indicated stress as a result of acute colitis. Our findings provide evidence that changes of spontaneous burrowing behaviour correlate with the onset of inflammation in acute DSS-induced colitis.
Laboratory mice are currently a widely-used animal species in biomedical research. Their current popularity may be due to the availability of a multitude of spontaneous or experimentally-induced mutants, allowing studies of in vivo functions of single genes. In recent years an increase in the use of genetically-modified mice in research has been observed. 1 Mice are also used to test hypotheses concerning the aetiology and pathogenesis of inflammatory bowel disease (IBD). IBD is a chronic, relapsing inflammation of unknown origin. IBD comprises two main disease conditions, ulcerative colitis (UC) and Crohn’s disease (CD). Disturbance of the epithelial barrier and epithelial transport processes has been proposed as a major factor in the pathogenesis of IBD. Epithelial apoptosis in mucosa from patients with UC 2 and CD 3 was found to be considerably upregulated followed by increased permeability of the epithelial barrier.4–6 Abnormal gut permeability is associated with the translocation of luminal antigens, intestinal bacteria and endotoxins.
The consequences of a disturbed barrier in human IBD are frequently investigated in mouse models of impaired epithelial cell function. Feeding mice for several days with dextran sulfate sodium (DSS) polymers in the drinking water induces acute colitis characterized by bloody diarrhoea, ulcerations and infiltrations of the intestinal mucosa with granulocytes. 7 It is generally believed that DSS is directly toxic to gut epithelial cells of the basal crypts and affects the integrity of the mucosal barrier. 8 DSS-induced colitis is readily reproducible in the common C57BL/6 strain, which shows intermediate to high susceptibility to DSS colitis but low mortality rates.
Appropriate parameters for monitoring the induction of colitis and impact on the wellbeing of mice are needed to decide when to terminate the experiments. On the one hand, a certain level of inflammation is necessary for observing the colitis-specific disease pattern, but on the other hand, animal wellbeing needs to be considered during the probably painful condition. While a certain level of inflammation is necessary for observing the colitis-specific disease pattern; for ethical reasons, distress from this probably painful condition should be minimized. Humane endpoints must be set to avoid unnecessary suffering in these mice. Therefore, appropriate parameters for monitoring the induction of colitis and the impact of intestinal inflammation on the wellbeing of mice are needed to decide when to terminate the experiments.9,10
To assess the grade of induced colitis in a DSS-treated animal, a retrospective histological examination of affected colon parts is usually performed, which requires sacrificing the animals. In the live animal, monitoring of the course of colitis and the progressing inflammation of the colon can be achieved via endoscopic examination in the end stage of the disease process or by observation of body weight loss. While endoscopy is associated with anaesthetic immobilization of the animal and may therefore be linked with physiological and emotional stress, the assessment of body weight loss alone may be a problematic parameter because of its retrospective character. 11 Changes in body weight can be associated with several impairments of the animal’s condition and therefore represent a relatively unspecific indicator, especially as animals suffer from diarrhoea during progressing colitis. For indication of impaired wellbeing or pain, body weight loss is therefore only relevant to a limited extent. 11 This situation requires additional parameters for the assessment of induced colitis, as well as animal wellbeing. Detection of pain and reduction of wellbeing in DSS-treated animals is essential for both scientific and ethical reasons. However, pain diagnosis in mice is challenging as mice often exhibit only subtle signs of pain, suffering or disability.11–14
A promising, simple approach to address this need is the assessment of changes in spontaneous burrowing behaviour. 15 The test is based on the species-typical behaviour of mice to spontaneously displace items from tubes within their home cage. Animals show some tendency to increase their burrowing with experience but these changes are not statistically significant. 16 Burrowing behaviour has been used as a tool to provoke and determine burrowing behaviour under experimental conditions in models of prion disease (scrapie),17–20 brain lesions 21 and complex regional pain syndrome 22 in C57BL/6 mice, lipopolysaccharide (LPS)-induced low-grade systemic inflammation 21 using C3H/HeJ and C3H/NeJ mice, and Alzheimer's disease using BL6/SJL mice. 23 In these models, a decrease in burrowing behaviour was interpreted as being correlated with chronic neurological or immunological disorders and was very efficient in detecting early signs of the diseases mentioned. Recently changes in burrowing behaviour were found to be sensitive indicators of post-operative pain after laparotomy in mice. 24 As burrowing behaviour is not an essential maintenance behaviour like food consumption or sleeping, 25 we suggest that it is sensitive to different, subtle kinds of impact on the animals’ general condition and wellbeing. Burrowing is a species-typical behaviour of mice. 26 Burrowing as a screening test for therapies may therefore offer great sensitivity, along with cheapness and simplicity. Changes in this species-typical behaviour might therefore be useful as an indicator of stress and pain under pathological conditions like induced colitis. We therefore investigated changes in species-typical burrowing behaviour during experimental colitis.
Materials and methods
The animal housing and experimental protocols were approved by the Cantonal Veterinary Department, Zurich, Switzerland, under licence no. 149/2009, and were in accordance with Swiss Animal Protection Law. Housing and experimental procedures also conformed to the European Convention for the Protection of Vertebrate Animals used for Experimental and other Scientific Purposes (Council of Europe no. 123, Strasbourg 1985) and to the Guide for the Care and Use of Laboratory Animals (Institute of Laboratory Animal Resources, National Research Council, National Academy of Sciences, 1996).
Animals
Twenty-three C57BL/6 mice weighing 20–22 g were obtained from our in-house breeding facility (Fuellinsdorf, Switzerland) at the age of nine weeks. Female mice were used as male mice frequently show aggressive behaviour upon DSS treatment. All the animals were housed for at least three weeks prior to testing in a specific pathogen free (SPF) facility. The animals were randomly assigned to cages and kept in groups of two (with the exception of one group of three) in type II long clear-transparent individually ventilated cages (IVCs, 365mm × 207mm × 140mm, Allentown, New Jersey, USA) with autoclaved dust-free bedding and tissue papers as nesting material. They were fed a pelleted and extruded mouse diet (R/M–H Extrudat, ssniff Spezialdiäten, Soest, Germany) ad libitum and had unrestricted access to sterilized drinking water. The light/dark cycle in the room was given through natural daylight (sunrise: 07:00 h, sunset: 18:00 h). The mice were weighed at 10:00 h every morning; burrowing tests were started 2 h prior to sunset. The temperature was set to 21 ± 1℃, with a relative humidity of 55 ± 5% and 75 complete changes of filtered air per hour (filter: Megalam MD H14, Camfil, Zug, Switzerland).
Induction and treatment of DSS colitis
Mice were randomly assigned to the cages and acute colitis was induced in 12 female C57BL/6 mice by feeding 2% DSS in drinking water over nine days as described previously.7,8 The colonic inflammation resulting from DSS depends on a number of unknown factors, e.g. the microbiome present. For the mice in our SPF facility we chose to induce acute colitis by feeding 2% DSS in drinking water over nine days. Reduced food consumption and an unshaped or spread stool consistency can frequently be observed upon DSS induction. Both contributed to body weight loss < 20% for the conditions present in our SPF facility. Body weight was determined by weighing the animals using a precision balance (PR 2003 Delta Range, Mettler-Toledo AG, Greifensee, Switzerland) especially designed for weighing moving animals. Stool consistency is part of the colonoscopy score described below. Eleven mice not fed with DSS were used as controls. All the animals were sacrificed on day 9 by cervical dislocation.
Assessment of colonoscopy score in mice
Murine endoscopic index of colitis severity (MEICS).
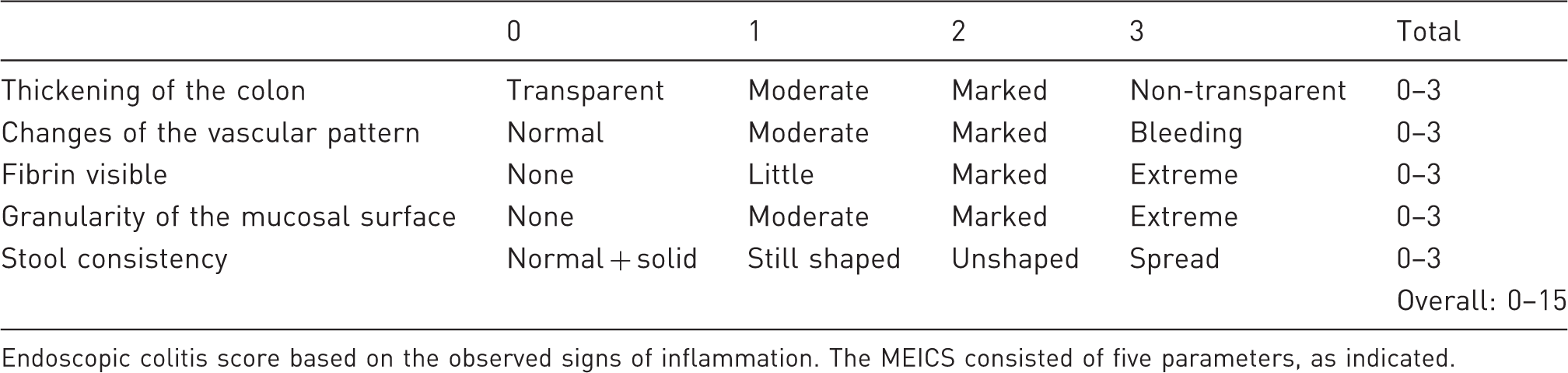
Endoscopic colitis score based on the observed signs of inflammation. The MEICS consisted of five parameters, as indicated.
Assessment of histological score in mice
The first distal centimetre of the colon was removed and used for histological analysis. The mice were scored individually by an independent investigator blinded to the type of treatment. The total histological score represented the sum of the epithelial and infiltration score, and thus ranged from 0 to 8 (maximum total score). Histology was scored as follows:
Epithelium: 0 = normal morphology; 1 = loss of goblet cells; 2 = loss of goblet cells in large areas; 3 = loss of crypts; 4 = loss of crypts in large areas. Infiltration: 0 = no infiltrate; 1 = infiltrate around crypt basis; 2 = infiltrate reaching to the L. muscularis mucosae; 3 = extensive infiltration reaching the L. muscularis mucosae and thickening of the mucosa with abundant oedema; 4 = infiltration of the L. submucosa.
Behavioural testing with burrowing apparatus
As burrowing apparatus, a plastic bottle (standard opaque water bottle, 250 mL, 150 mm length, 55 mm diameter) filled with 138–142 g of pellets of the animal’s diet was used. An empty bottle of the same dimensions was provided to serve as a shelter for the animals. 24 To test for effects of DSS-induced colitis on burrowing behaviour each animal was tested in the burrowing apparatus before (baseline) and during the DSS administration (experiment). The individual baseline values were used to compensate for inter-individual variation in burrowing behaviour. For acclimatization, the animals were housed in groups of two mice for three days in a standard IVC containing the experimental set-up as described above. Except for this prior experience with the burrowing apparatus, the animals had no experience with behavioural testing and were not selected for burrowing performance.
Burrowing tests for the measurements of baseline and experimental values of burrowing behaviour started 2 h before sunset and were carried out in the animal room. Pellets which had been removed or eaten by the mice during the preceding days were replaced in the apparatus before testing started. The burrowing apparatus was weighed 2 h after the start of the experiment to assess the weight of the removed pellets. The apparatus was again placed in the cage after the removed pellets were replaced and weighed 12 h later at the end of the dark phase. On day 9, the burrowed weight was additionally evaluated after 0.5 h. Burrowing tests were performed on days 4–9 of the DSS administration.
Hyperplasia and hypertrophy in adrenal gland
To assess cellular hyperplasia and hypertrophy in the adrenal cortex and medulla, paraffin-embedded sections were haematoxylin and eosin (H&E) stained as described above. Cell nuclei in the zona glomerulosa (ZG) and zona fasciculata (ZF) were counted in defined areas (5 regions each from 20 different mice) at 20-fold magnification. Numerical density (labelled cells per volume) was determined. An increase in nuclear density in the adrenal cortex was interpreted as evidence of a decreased cell size, whereas a decrease in nuclear density was interpreted as evidence of an increased cell size.28,29 In all cases, care was taken to avoid sampling regions of the medulla that contained large blood vessels, because this would have greatly affected cellular density measurements.
Statistical analysis
Statistical analysis was performed using the Mann–Whitney rank sum test. In weight curves, mean values ± SEM are displayed. Box plots express median, 25% quartiles, minimum and maximum. Differences were considered significant at a P value of <0.05 (*), highly significant at a P value of <0.01 (**) and very highly significant at a P value of <0.001 (***).
Results
DSS colitis negatively alters the colonic mucosa at a macroscopic level
To determine changes in burrowing behaviour during the onset of intestinal inflammation we used the DSS colitis mouse model. During induction of acute colitis, the mice received 2% DSS dissolved in drinking water (n = 12) or drinking water alone (n = 11) over nine days. Water consumption was not reduced in mice treated with DSS. Weight loss in acute colitis was determined over nine days (Figure 1). The percentage changes of body weight in the group receiving DSS were significantly different from the control group on day 7 (98.6 ± 3.1% versus 101.8 ± 2.5%, P < 0.05), day 8 (94.9 ± 7.2% versus 103.1 ± 3.8%, P < 0.01) and day 9 (91.4 ± 5.7% versus 104.6 ± 2.9%, P < 0.001).
Percentage body weight loss. Mice received either dextran sulfate sodium (DSS) (triangles, n = 12) or water (diamonds, n = 11). Induction of colitis was followed by a significant reduction of the weight. Bars represent mean ± SD. *P < 0.05, **P < 0.01 and ***P < 0.001.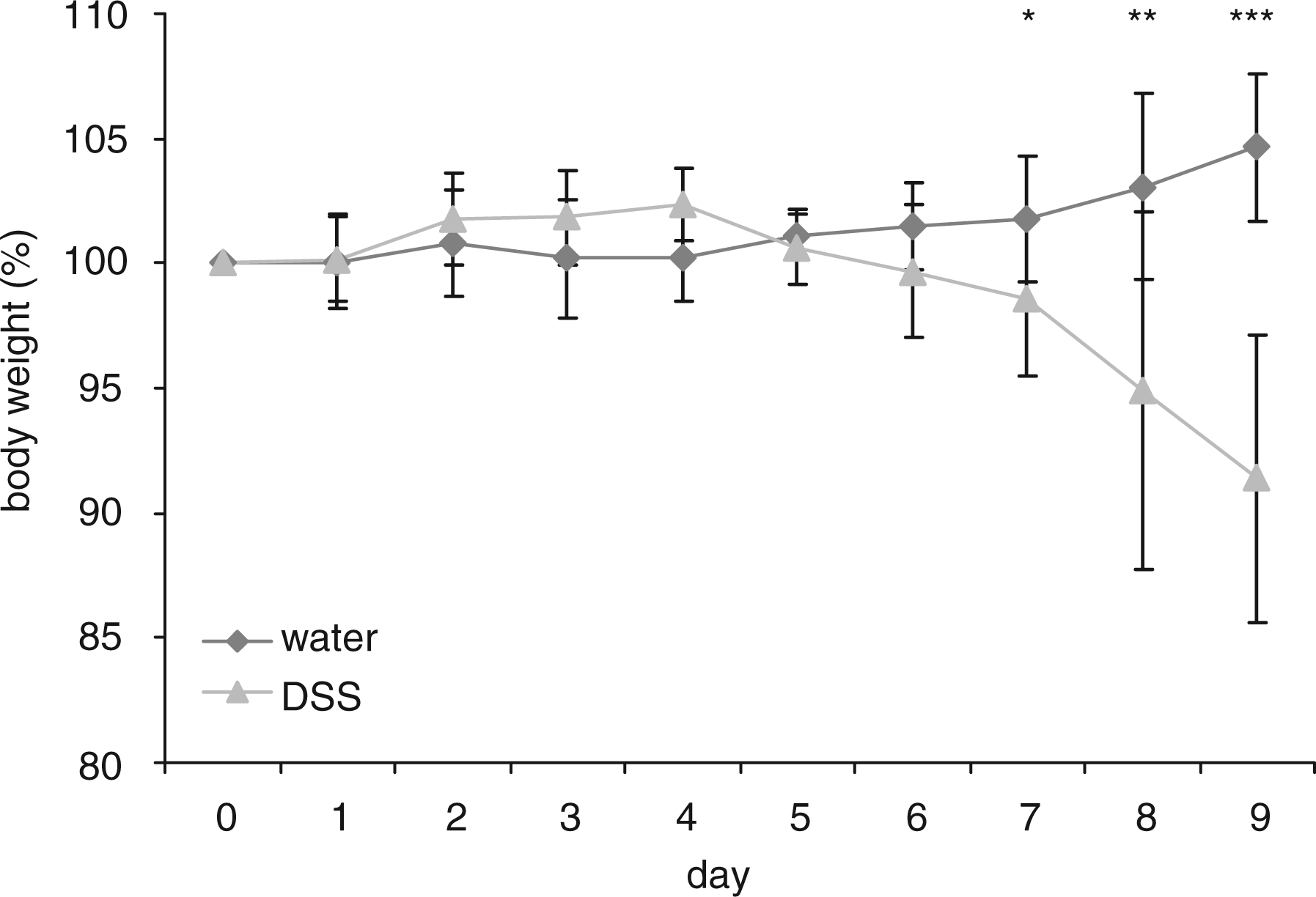
Macroscopic mucosal damage was assessed by endoscopy and colonoscopy score. During colonoscopy on day 9 mucosa from control mice displayed a smooth and transparent mucosa with a normal vascular pattern (MEICS 2.0 ± 0.4, n = 11, Figure 2a and b). A solid stool was visible. By contrast, mucosa from animals with DSS colitis appeared with an intransparent mucosa and altered vascular pattern (MEICS 12.0 ± 0.5, n = 12, P < 0.01; Figure 2c and d). The mucosal surface appeared more granular than with mice without DSS colitis, and the thickening of the colon was more prominent. Diseased regions often had a cobblestone-like appearance and casually mucosal bleeding. Unshaped stool was visible. The endoscopic colitis score indicated severe mucosal damage in the DSS colitis mice.
Endoscopic images of dextran sulfate sodium (DSS) colitis and murine endoscopic index of colitis severity (MEICS) (day 9). Control mice (a and b). Mice upon DSS treatment (c and d). Control mice showed no signs of inflammation and solid stool compared with a thickened colon and granular mucosal surface upon DSS induction. Images are representative of five mice. MEICS (e). Statistical analysis was performed using the Mann–Whitney rank sum test. Box plots express median, 25% quartiles, minimum and maximum. **P < 0.01.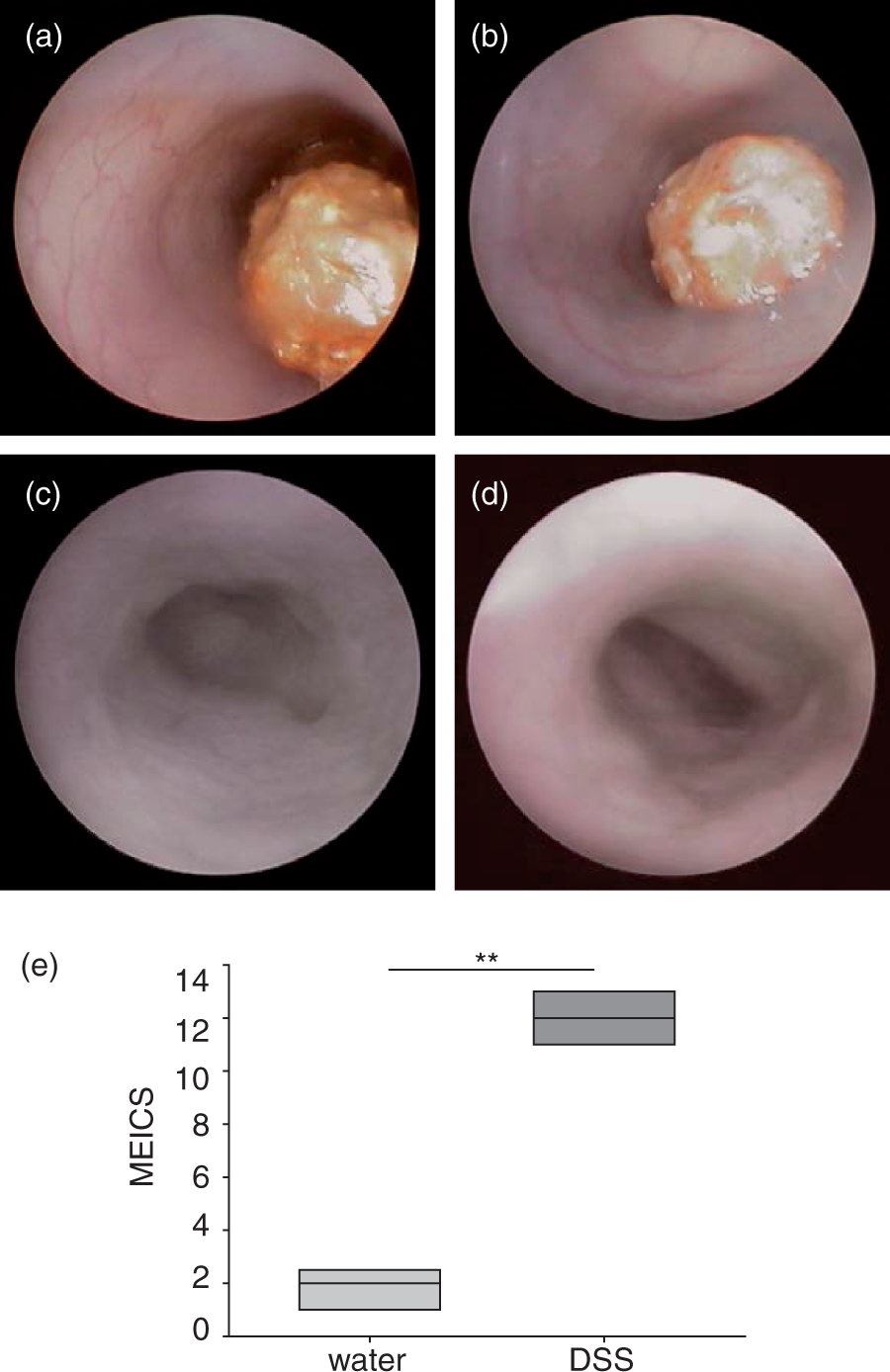
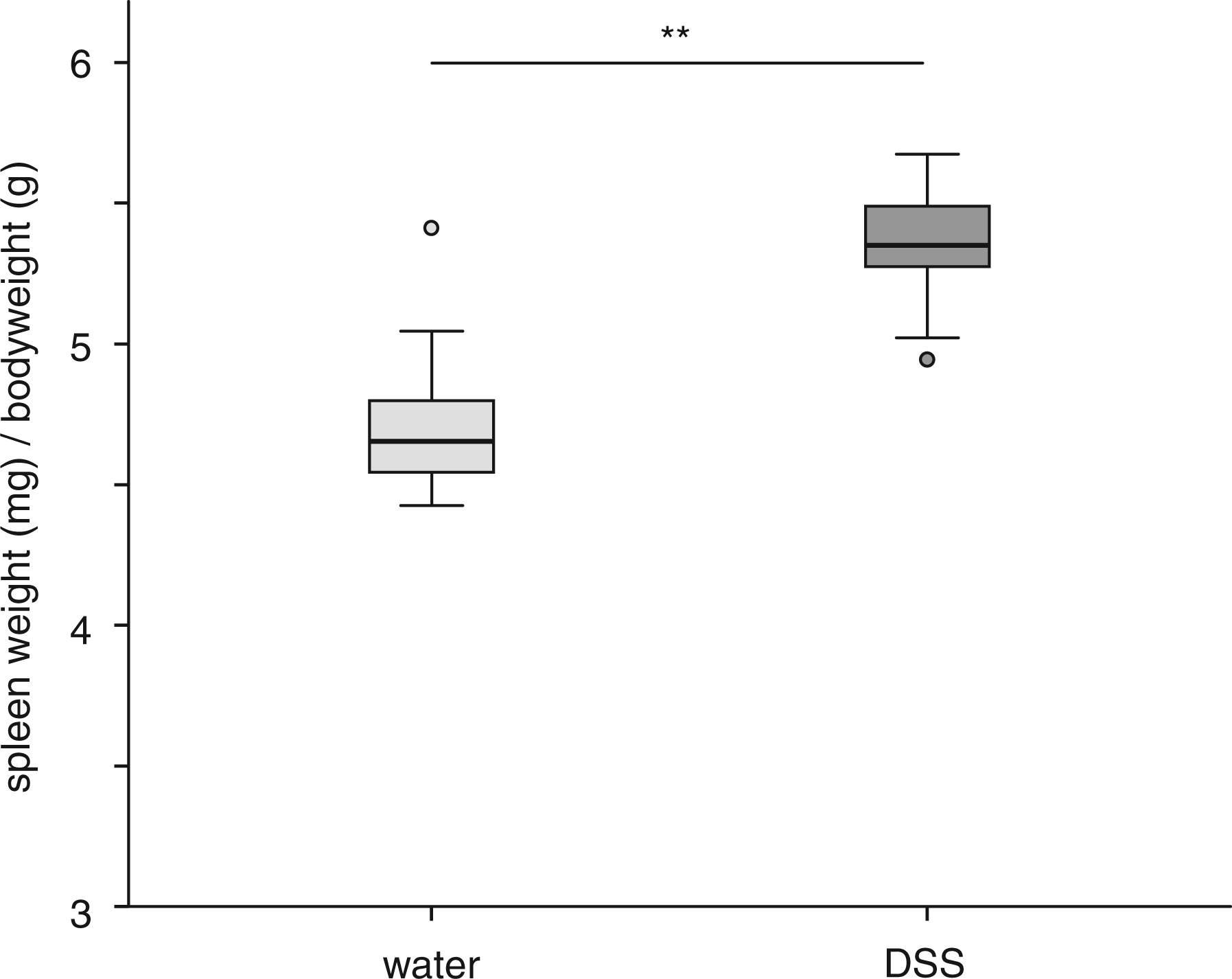
Induction of colitis was also followed by a significant reduction of the colon length as compared with the control animals (6.4 ± 0.6 cm versus 9.4 ± 0.5 cm, P < 0.001; Figure 3). Additionally, the spleen weight, normalized to the body weight, was significantly increased during acute colitis as compared with the control animals (5.4 ± 0.6 mg/g versus 4.7 ± 0.4 mg/g, respectively, P < 0.001; Figure 4). Both these parameters demonstrated the induction of a severe inflammation in response to DSS treatment.
Colon lengths. (a) Mice received either dextran sulfate sodium (DSS) (n = 12) or water (n = 11). Induction of colitis was followed by a significant reduction of the colon length. Statistical analysis was performed using the Mann–Whitney rank sum test. Box plots express median, 25% quartiles, minimum and maximum. **P < 0.001. (b) Colon of a mouse which received water (left) or DSS (right). Images are representative of 12 and 11 mice, respectively. Spleen weights. Mice received either dextran sulfate sodium (DSS) (n = 12) or water (n = 11). Induction of colitis was followed by a significant increase of the spleen weight. Statistical analysis was performed using the Mann–Whitney rank sum test. Box plots express median, 25% quartiles, minimum and maximum. **P < 0.01.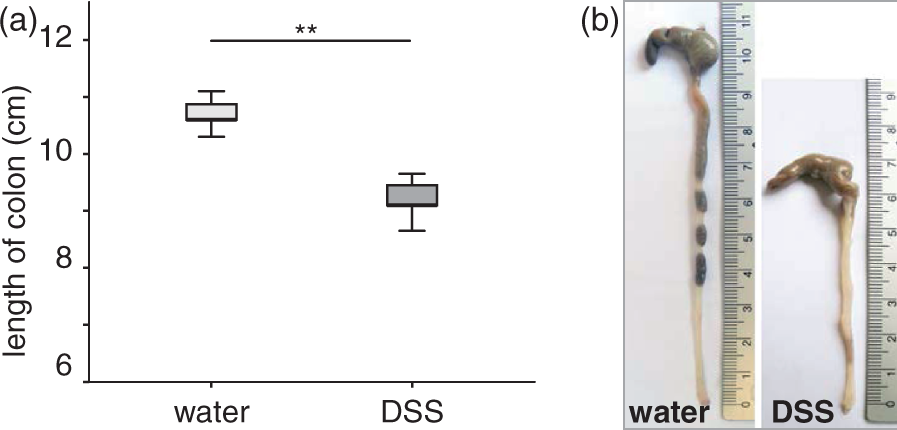
DSS colitis negatively alters the colonic mucosa at a microscopic level
The histological score for the mice receiving DSS was significantly increased compared with that of the water-treated animals (7.5 ± 0.5 versus 0.4 ± 0.4, respectively, P < 0.001; Figure 5a). Colonic sections taken from the mice with DSS colitis showed a loss of crypts, extensive epithelial damage, and both infiltration and thickening of the mucosa over large areas (Figure 5d and e) compared with that of the water controls (Figure 5b and c). These data show that in mice with DSS colitis the colonic mucosa is also negatively altered at a microscopic level.
Histological parameters and H&E staining of terminal colon. (a) Mice received either dextran sulfate sodium (DSS) (n = 12) or water (n = 11). Induction of colitis was followed by a significant increase of the histological score. Statistical analysis was performed using the Mann–Whitney rank sum test. Box plots express median, 25% quartiles, minimum and maximum. **P < 0.005. (b–e) H&E staining. Control mice (b and c) were treated without DSS. Taken from mice with DSS-induced colitis (d and e). Control mice showed no signs of inflammation and an intact epithelial barrier compared with a thickened mucosa and loss of crypts upon DSS. Images are representative of 12 mice.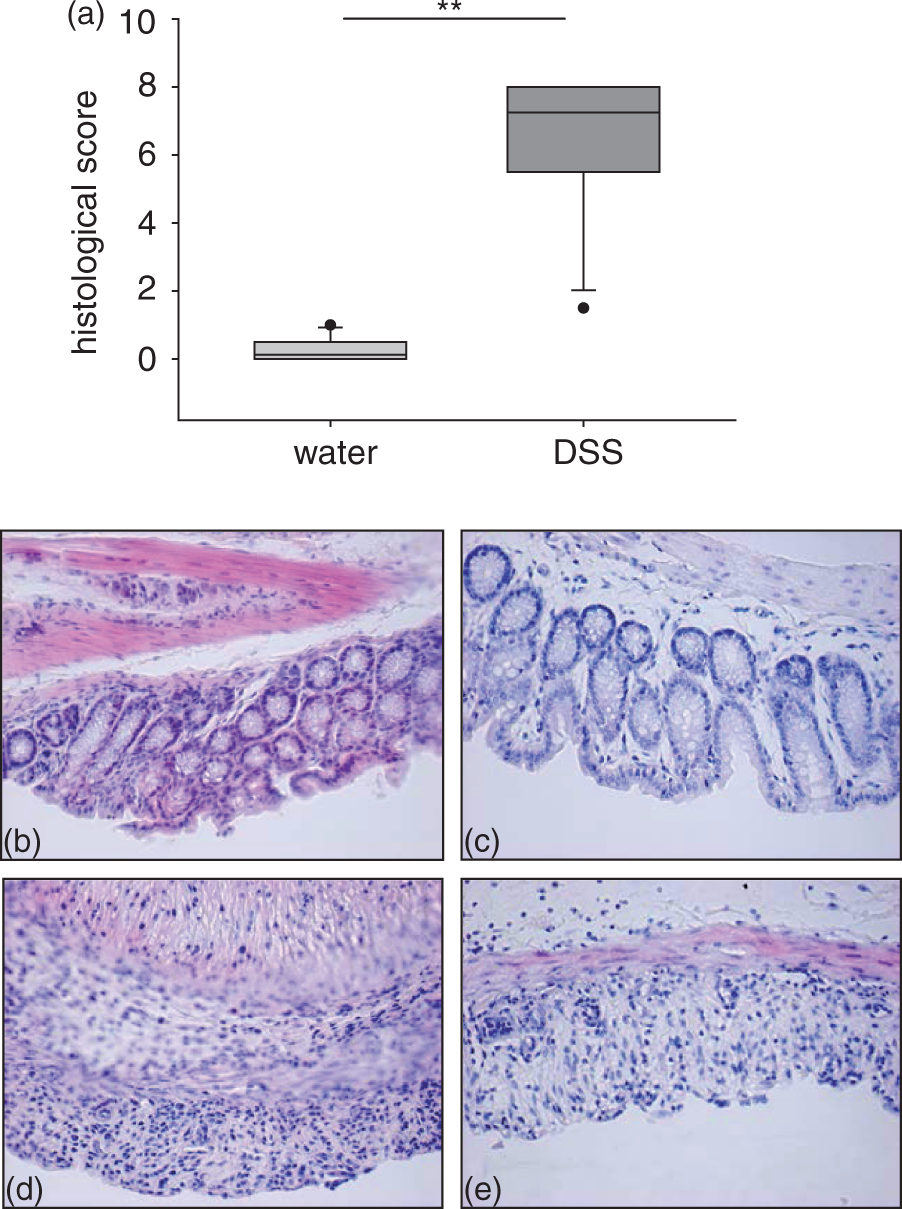
Onset of intestinal inflammation correlates with decrease in burrowing behaviour
The burrowing test (modified from descriptions by Deacon et al. 30 ), was evaluated in regard to its feasibility as a means to assess general condition and presumable pain in animals suffering from experimentally-induced colitis. The test proved to be easy to perform in an experimental setting within the animals’ home cage and burrowing performance responded sensitively to the DSS treatment. We observed that healthy mice started burrowing (i.e. displaced substantial numbers of food pellets from a tube) in general within several minutes after this pellet-filled tube was provided to them, while mice treated with DSS started burrowing later or not at all. The measured weight of removed pellet material in the default time frame of 2 and 12 h (referred to by Deacon et al. 30 ) was significantly reduced during the treatment (P < 0.05 each).
Figure 6 presents these changes in percentage weight of removed pellets by two mice after 2 h and 12 h of testing during the experimental period. During baseline measurements after 2 h and 12 h each pair of mice burrowed a mean weight of 70.7 ± 10.8 g and 83.5 ± 2.7 g, respectively. While the control animals showed only a slight decrease in burrowing performance during the following days of observation, the DSS-treated mice removed fewer pellets during the progression of colitis. Compared with the control animals, the mice treated with DSS removed significantly fewer pellets after 2 h on day 8 (49.2 ± 28.8 g versus 79.6 ± 6.8 g per two mice, P < 0.05) and day 9 (37.7 ± 35.1 g versus 75.4 ± 8.7 g per two mice, P < 0.05, Figure 6a). After 12 h, the weight of pellets removed by the DSS-treated mice was significantly lower than that of the control group on day 8 (64.2 ± 11.9 g versus 80.2 ± 2.2 g per two mice, P < 0.01, Figure 6b). On day 9 a very similar trend was observed. Additional measurements after 0.5 h on days 8 and 9 revealed a significant decrease in pellets removed by the DSS-treated animals as compared with the water controls (13.7 ± 24.7 g versus 65.8 ± 26.4 g per two mice, P < 0.05, and 16.5 ± 25.6 versus 62.2 ± 20.2 g per two mice, respectively, P < 0.05).
Burrowing test. Percentage weight of pellets removed by two mice out of test apparatus. Mice received dextran sulfate sodium (DSS) (triangles, n = 6) or water (diamonds, n = 5). Burrowing was tested for 2 h (a) and 12 h (b). Induction of colitis was followed by a significant reduction of the weight of the removed pellets. Mann–Whitney rank sum test was performed. Bars represent mean ± SEM, *P < 0.05.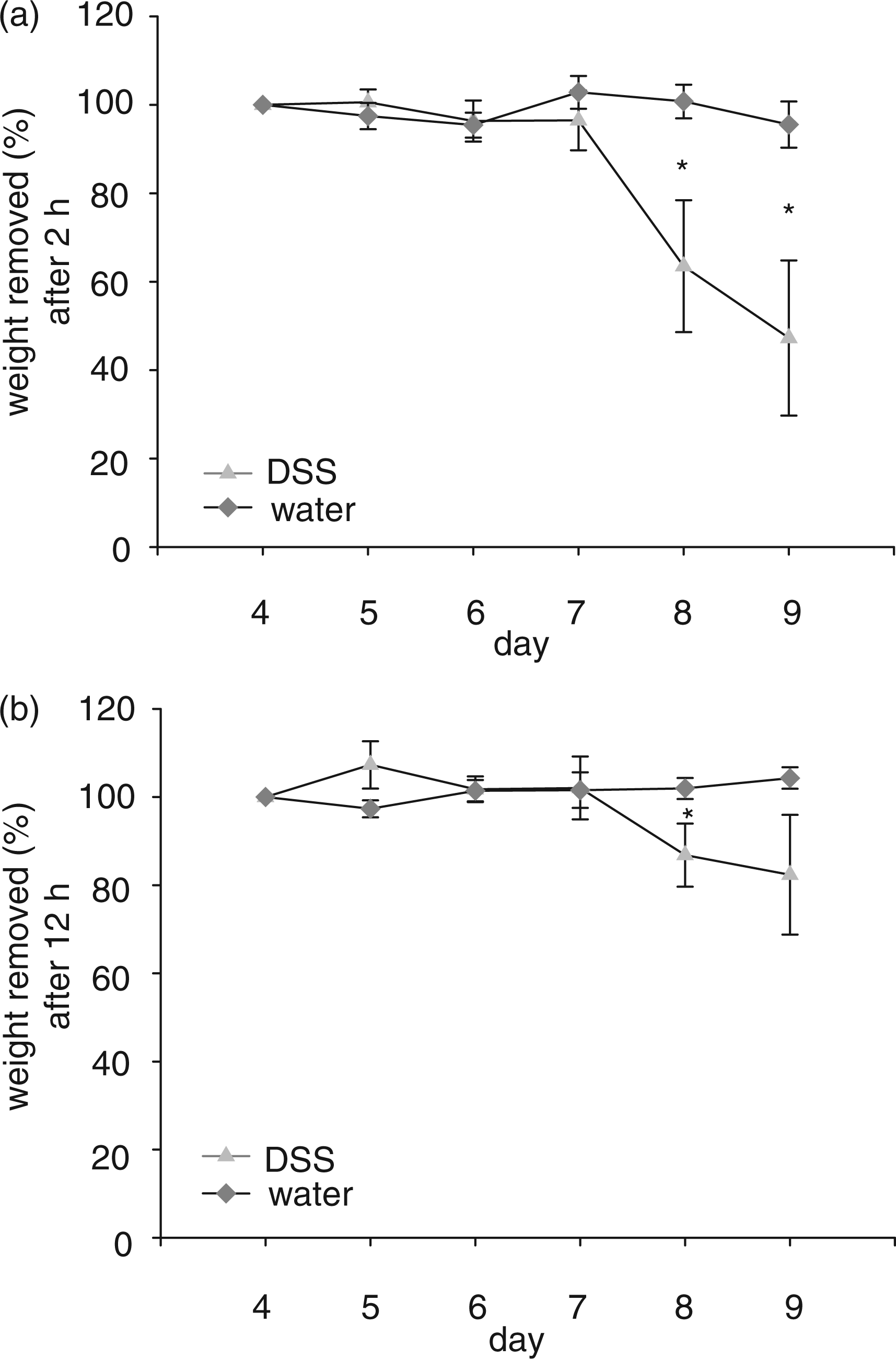
An altered adrenal gland suggests onset of stress upon DSS-induced colitis
Chronic stress induces changes in adrenal growth and function. Therefore we determined whether treatment with DSS is associated with cellular hypertrophy in the medulla and/or decreased cell size in the cortex of adrenal glands. Induction of colitis was followed by a decrease of the ZF/ZG ratio (mean ± SD: 68 ± 15 versus 61 ± 12, Figure 7a). Nuclear density in the ZG was increased in the control mice (Figure 7b) compared with the mice that received DSS (Figure 7c). In the adrenal medulla DSS decreased nuclear density was found, suggesting increased cell size in this region.
Hyperplasia and hypertrophy in adrenal gland. (a) Adrenal sections collected from mice which received either water (n = 11) or dextran sulfate sodium (DSS) (n = 9). Induction of colitis was followed by a decrease of the zona fasciculata (ZF)/zona glomerulosa (ZG) ratio. (b) H&E staining of an adrenal gland section from a mouse which received water (representative of 11 mice). (c) DSS colitis induced cellular hypertrophy in the inner ZF and medulla, compared with a decreased cell size in the ZG (representative of nine mice). Nuclei were counted in five areas in both the ZG (green boxes – see online version for colour reference) and the ZF (black boxes) and summed up for result.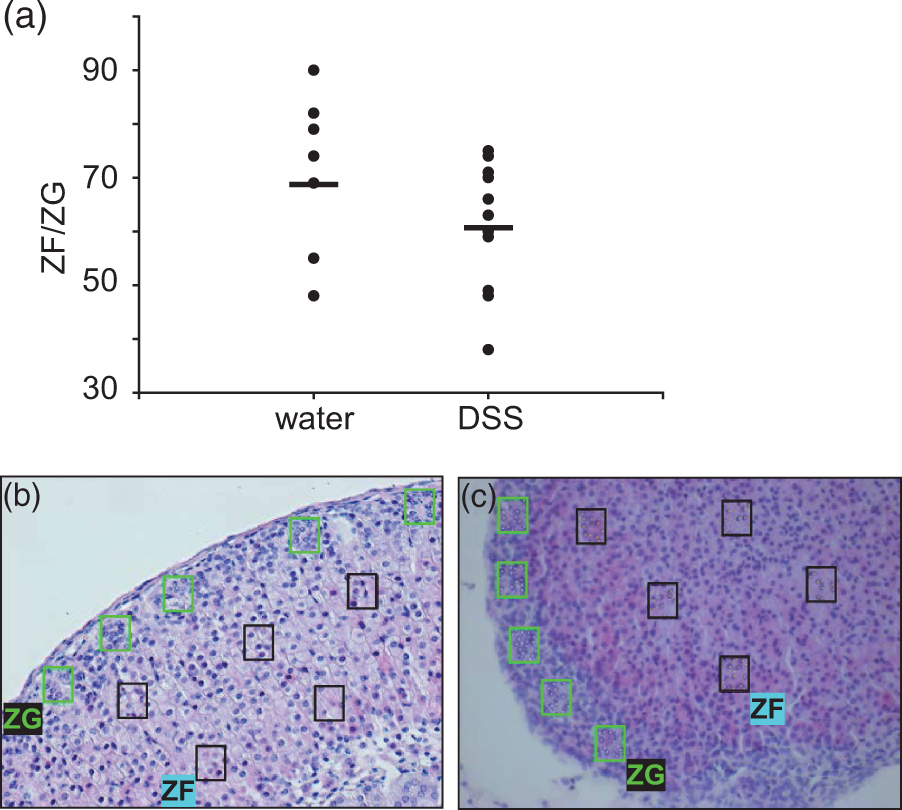
Discussion
In this study we have suggested a simple but sensitive behavioural test system for assessing the impact of colitis on animal wellbeing. We used acute DSS-induced colitis to induce intestinal inflammation. Parameters collected during the induction of acute DSS-induced colitis documented the onset of inflammation in mice. A significant decrease in body weight and a significant increase of MEICS in the group receiving DSS compared with the control group indicated mucosal damage in the DSS colitis mice. Parameters collected retrospectively confirmed the presence of inflammation. A significant decrease in colon length as well as a significant increase in both histological score and spleen weight for animals receiving DSS compared with the control group indicated the induction of inflammation in response to DSS treatment. As expected, DSS colitis also negatively altered the colonic mucosa at both macroscopic and microscopic levels. The murine model of acute DSS-induced colitis is comparable with acute colitis in human patients. Animals completely recover from acute colitis after discontinuing DSS without chronic manifestation of inflammation. This includes weight gain, decrease of MEICS, normalization of colon length and decrease of histological score and spleen weight. Acute DSS-induced colitis does not affect the survival rate.
In parallel we investigated the changes in burrowing behaviour under these conditions. The onset of intestinal inflammation indicated by a weight loss in DSS-treated mice compared with the controls on days 7, 8 and 9 correlated with a reduction of burrowing performance on days 8 and 9. DSS-treated mice removed significantly fewer pellets during the progression of colitis compared with their respective control animals. Behavioural testing with the burrowing apparatus provided reliable results when an experimental period of 0.5 or 2 h was applied. In our experimental set-up two mice were kept in a cage during the burrowing test. Acute DSS-induced colitis, just as any other induced disease, does not develop in each mouse with the same onset or severity of inflammation. To identify outliers animals need to be housed separately for the duration of the burrowing test. Further, we determined whether treatment with DSS is associated with cellular hypertrophy and decreased cell size in the adrenal glands. An altered adrenal gland is an indication of first signs of chronic stress upon acute DSS-induced colitis at a cellular level. This suggests that altered burrowing behaviour could also be a sensitive readout parameter for nascent chronic stress.
A correlation between inflammation and burrowing as well as pain and burrowing has been shown in a number of recent studies. The immune system communicates with the brain in times of both good health and illness. Inflammation can be associated with behavioural changes, collectively referred to as sickness behaviour, characterized by symptoms such as cognitive dysfunction, anxiety, depression and lethargy. Injection of double-stranded RNA into C57BL/6 mice mimics an acute phase of viral infection. This treatment induced severe sickness behaviour in animals as revealed by a burrowing test performed 6 h post injection. 31 Low-grade systemic inflammation was induced in mice using LPS to mimic aspects of bacterial infection. Sub-pyrogenic inflammation resulted in changes in burrowing. 21 Further, in a mouse model of chronic neurodegeneration additional transient systemic inflammation by injection of LPS led to an increase of inflammatory markers like IL-1β, TNF and IFN-β. This inflammation caused exaggerated impairments in burrowing and locomotor activity. 32 Selective and non-selective inhibitors of cyclooxygenase (COX)-1 like piroxicam, indomethacin and ibuprofen reversed the effect of LPS-induced systemic inflammation on behaviour. This suggests that inflammation-driven changes in burrowing depend on COX-1. 33
As the construction of burrows provides protection from natural threats (e.g. predation and detrimental environmental conditions) in the wild, burrowing is a highly motivated behaviour persisting as a spontaneous behaviour in the laboratory mouse.25,34,35
Knowledge of disease progression during acute DSS-induced colitis is of immediate importance for establishing humane endpoints and for monitoring wellbeing. In studies based on this model useful indicators of the onset of inflammation, disease progress and pain are rare. Directly accessible parameters like appearance, common behaviour, overall activity, stool consistency, visible blood on stool and prolapse formation must be determined upon DSS-induced colitis to comply with basic ethical needs. But a mouse without overt pathological findings is neither necessarily free of pain nor reliably non-inflamed. Colonoscopy should not be performed on a daily basis because of the risk of mucosal perforation. Cytokine levels could be determined from peripheral blood but blood should also not be taken daily. The inflammatory parameter calprotectin present in neutrophils in mice can be determined from stool samples. But as inflammation is mainly initiated by epithelial lesions in the mentioned model, calprotectin is often not a reliable parameter in acute DSS-induced colitis. Therefore body weight loss is the only physical indicator for the onset of inflammation that can be measured repeatedly. However, body weight may also be dependent on specific treatments, e.g. during nutrition or pharmacological trials. Further this could play a major role if long-term models of colitis were used. Chronic manifestation of inflammation can be initiated using the murine model of chronic DSS-induced colitis. Here the administration of DSS for several cycles (e.g. 7 days DSS, 14 days water) results in chronic colitis after a time period of two to three months. Despite body weight being a robust physical parameter it is of only limited use for determining impaired wellbeing or pain. 11 Behaviour, like burrowing performance, in contrast to most physiological and clinical parameters, can be observed easily in a non-invasive manner and has been described as a promising approach to assess animal wellbeing.15,36–39 Here we provide evidence that testing burrowing performance could be a sensitive behavioural test system for assessing the impact of colitis on animal wellbeing. Our data indicate that, next to body weight loss and MEICS, burrowing behaviour might be a useful parameter that can be easily documented during the induction of DSS colitis to confirm the onset of inflammation in mice.
Footnotes
Acknowledgements
This study was supported by grants from the Swiss National Science Foundation (SNF 31003A_127247) to M Hausmann and (SNF 310030 120312) to G Rogler, by the Federal Veterinary Office (Bern, Switzerland) and the UBS Foundation to M Arras. We also acknowledge the support from the Zurich Center for Integrative Human Physiology (ZIHP) to M Hausmann and G Rogler and the support from the Swiss Inflammatory Bowel Disease Cohort Study (SIBDC) to G Rogler. The authors would like to thank Robin Schneider and the staff of the central biological laboratory for support in housing the mice.
The study concept came from K Leucht, P Jirkof, N Cesarovic, M Arras and M Hausmann. Acquisition of data was performed by K Leucht, P Jirkof and N Cesarovic. Critical revision of the manuscript was undertaken by K Leucht, P Jirkof, N Cesarovic, G Rogler, M Arras and M Hausmann. Technical support was provided by M Caj and F Nicholls.
Declaration of conflicting interests
G Rogler discloses grant support from Abbot, Ardeypharm, Essex, FALK, Flamentera, Novartis, Tillots, UCB and Zeller. All other authors have no conflict of interest to disclose.
