Abstract
A large number of methods for obtaining blood from mice have been published. In our facility, saphenous venepuncture is considered the method of choice for bleeding mice and is believed to have a number of welfare advantages when performed correctly. The aim of this study was to compare levels of haemolysis and plasma glucose in blood samples obtained by saphenous venepuncture and tail vein incision. The results indicate that saphenous venepuncture is the preferred method.
Saphenous venepuncture is a convenient method for repeated blood sampling of mice without the need for an anaesthetic, 1 which in itself may be stressful. 2 The time needed for restraint can be reduced to a minimum, avoiding unnecessary stress that may adversely affect both the animal's welfare and the quality of the blood sample. The scab that forms at the puncture site may be gently rubbed off, enabling serial sampling from the original puncture site.
Haemolysis of blood samples is undesirable because it interferes with photospectrometric measurements, it releases enzymes from the erythrocytes that may decompose components in the plasma that are to be measured, and it results in clots that may make it impossible to work with small blood volumes.
Plasma glucose levels are one of a range of blood parameters used as indicators of stress in rodents, 3 since stress-mediated corticosterone production leads to gluconeogenesis and inhibition of insulin secretion. 4 Measurement of glucose levels without artefacts caused by stress is also an essential prerequisite for reliable results when conducting glucose tolerance tests.
In a recent paper, 5 a comparison was made of the plasma quality in samples taken from mice by four different methods: amputation of the tail tip, lateral tail incision, puncture of the tail tip and periorbital puncture. The authors concluded that incision of the tail vein was the method of choice. Puncture of the saphenous vein was not included in the study. Tail amputation and puncture resulted in a significant number of haemolysed samples, while periorbital puncture resulted in a considerable increase in plasma glucose levels. They concluded that lateral tail incision was the method of choice.
The aim of this study was to compare plasma glucose levels and the degree of haemolysis in multiple blood samples from mice that underwent saphenous venepuncture and lateral tail incision.
Materials and Methods
The study was performed at the Laboratory Animal Unit at the Norwegian School of Veterinary Science in Oslo, Norway. The unit is licensed by the Norwegian Animal Research Authority (www.fdu.no) and accredited by the Association for Assessment and Accreditation of Laboratory Animal Care (www.aaalac.org). The study was approved by the unit's animal ethics committee.
Animals, housing and husbandry
Male C57BL/6JBomTac mice (Taconic, Lille Skensved, Denmark) were used. For the main study, eight animals were allocated to each group (group A: saphenous vein and group B: tail vein). They weighed on average 33.9 g (range 30.3-35.8 g) at the time of the sampling. The animals were housed in open type III cages (Tecniplast, Buguggiate, Italy) in groups of four. The cages contained standard aspen bedding (Scanbur BK, Norway), cellulose nesting material and a Bio-Serv igloo (Bio-Serv, Frenchtown, NJ, USA) with an exercise wheel on top. The animals were given a standard maintenance diet (RM1 from SDS, Witham, UK) and tap water ad libitum. The animal room was on a 12:12 light-dark cycle, with a room temperature of 21 ± 2°C with 20 air changes per hour and 45 ± 5% relative humidity. The cages and bedding were changed twice a week and the water was changed daily by two experienced technicians. The mice were housed in the facility for nearly three months and then implanted subcutaneously with NanoTec® transponders (Lutronic, Rodange, Luxembourg) five days before the start of the study.
Calculation of animal numbers and blood volume
In the spirit of the 3Rs, 6 our study design used a Latin square design with eight animals per group, to minimize the number of animals without loss of power. Animals were allocated to groups randomly. Sampling was undertaken on days 1 and 15, alternating the group to which they belonged.
We wished to reduce the amount of blood to be taken to a minimum, on welfare grounds, since the animals were to be sampled twice. Based on the average weight of the mice (33.9 g) and available guidelines on blood sampling, the animals contained approximately 1.7 mL of blood, of which it is considered reasonable to remove 10% (170 μL) over a 2-3 week period. 7
Pilot study
A pilot study was conducted on one mouse in each group (group A: saphenous vein and group B: tail vein) to ensure that both the blood sampling methods and the blood analyses were optimal.
Blood sampling
On day 1 of the study, eight mice (group A) were sampled from the saphenous vein and eight mice (group B) were sampled from the tail vein. All the mice were sampled four times, at 30 min intervals. Samples of 30 μL were collected for measurement of haemolysis. Saphenous venepuncture (group A) was performed as described by Hem et al. 1 The animals were restrained for this purpose in a plastic tube (http://film.oslovet.veths.no/saphena). Tail puncture (group B) was performed as reported by Christensen et al. 3 Immediately before the first and the last blood sample, 5 μL of blood were collected for the analysis of plasma glucose.
On day 15, blood sampling was repeated in exactly the same way, except that the mice in group A were allocated to group B, and vice versa. After this second session, the animals were humanely killed by cervical dislocation.
Blood analysis
All the 64 blood samples for analysis of haemolysis (30 μL each) were collected in 75 μL Assistent® micro-haematocrit tubes (Jan F Andersen, Jevnaker, Norway). After sampling, the tube was sealed in one end with Assistent® wax and kept at room temperature until the blood clotted. They were then centrifuged at 8000 rpm for 5 min in a Servomed® High Speed Centrifuge model SH120-2 (Jan F Andersen). All samples were then processed immediately without addition of anticoagulants. Twenty microlitres of serum from each of the blood samples were placed in a microcuvette and analysed for haemolysis in a HemoCue® Plasma/low Hb (HemoCue, Oslo, Norway). The 5 μL samples for analysis of plasma glucose were placed in a microcuvette and analysed in a HemoCue® Glucose 201+.
Statistical analysis
The data were recorded in Microsoft Office Excel 2003 and then transferred to JMP® 7.0 Statistical Discovery Software from SAS for statistical analysis. After descriptive analyses of data using standard summary measures and graphical presentation, two least-squares models were established, with haemolysis and glucose level as outcomes. First, the effect of treatment (ordinal, tail vein versus saphenous vein), day (ordinal, 15 versus 1) and sample number (ordinal, 1, 2, 3, 4) on haemolysis was tested. Then a similar model was established for glucose, but with a reduced number of samples. The individual animal was included in the models as a random effect, as no parameter estimates of each animal were of interest. The model gave a standard analysis of variance table and the corresponding regression coefficients. Model fit was assessed using graphical techniques, plotting actual versus predicted values, and residuals were assessed using the normal quantile plot. Graphs were made in Stata (SE/10 for Windows; StataCorp, College Station, TX, USA).
Results
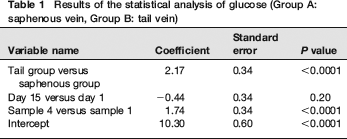
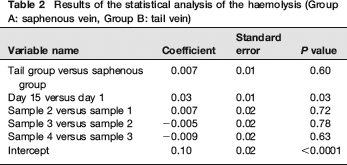
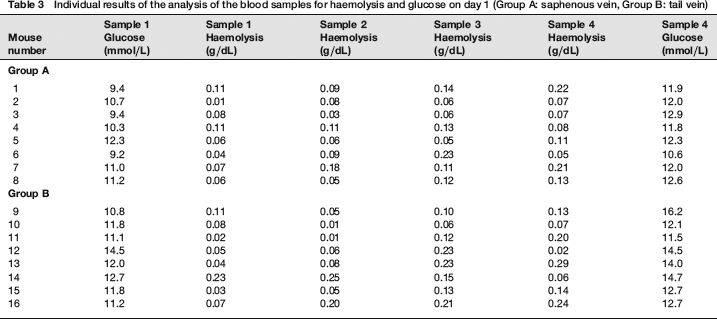
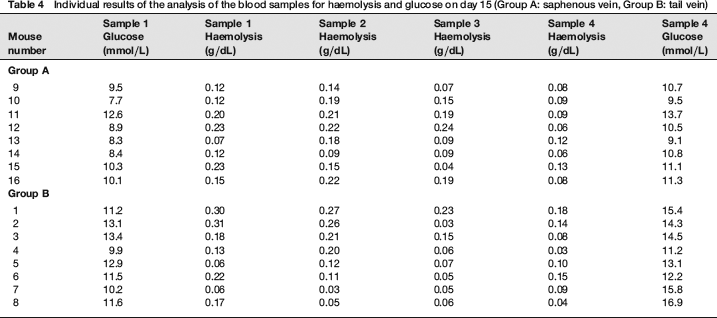
The results of the statistical analysis are shown in Tables 1 and 2 and Figures 1 and 2. The individual results of the analyses of the blood samples are shown in Tables 3 (day 1) and 4 (day 15). The levels of haemolysis were not significantly different between the two groups, but there was a significantly (P < 0.0001) lower level of glucose in the blood samples from the mice when they were sampled from the saphenous vein compared with the tail vein.
Results of the statistical analysis of glucose (Group A: saphenous vein, Group B: tail vein)
Results of the statistical analysis of the haemolysis (Group A: saphenous vein, Group B: tail vein)
Individual results of the analysis of the blood samples for haemolysis and glucose on day 1 (Group A: saphenous vein, Group B: tail vein)
Individual results of the analysis of the blood samples for haemolysis and glucose on day 15 (Group A: saphenous vein, Group B: tail vein)
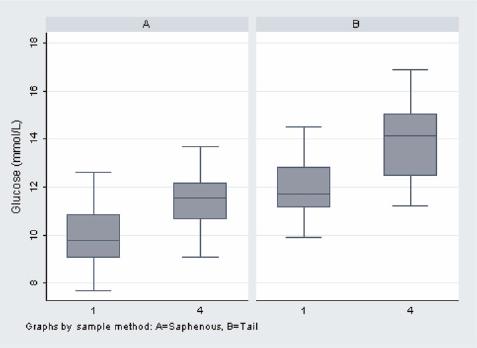
Level of glucose (mmol/L) for both groups on days 1 and 15 (Group A: saphenous vein, Group B: tail vein)
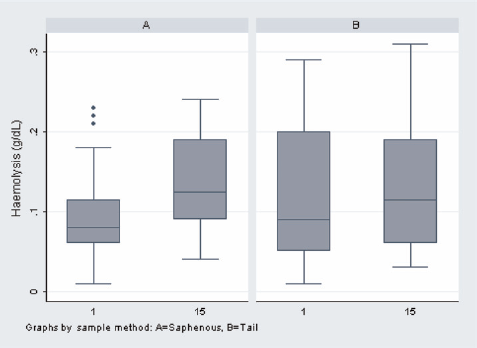
Amounts of haemolysis (g/dL) in both groups on days 1 and 15 (Group A: saphenous vein, Group B: tail vein)
Discussion
Mice sampled from the tail vein had significantly higher glucose levels and showed larger increases from the first sample to the fourth sample. Although glucose levels alone are not definitive indicators of stress, this indicates that, in this controlled study, the mice sampled from the tail vein were more stressed than the mice sampled by saphenous venepuncture. There were no significant differences in the degree of haemolysis between methods, indicating that the physical quality of blood taken with the two methods is comparable.
None of the methods requires anaesthesia, but both require some form of physical restraint. The saphenous vein is clearly visible (unlike periorbital puncture), enabling full control over perforation and any haemorrhage after the procedure. Multiple samples can be taken by removing the scab that forms over the puncture site, whereas in the case of the tail vein, a new incision has to be made each time. 8 If the saphenous vein is not located by the operator, the needle will probably penetrate muscle, as opposed to the tail method where more sensitive structures such as bone may be pierced. Even if the vein is hit first time, an experiment requiring four blood samples per day results in four holes in the tail. For welfare reasons, the number of blood samples taken by tail incision is therefore likely to be less than with saphenous venepuncture. Since the tail is frequently used to lift the animals, in addition to its primary role in balance and thermoregulation, damage to this organ is clearly undesirable.
Tail vein incision requires the animals to be warmed in order to achieve vasodilation prior to sampling. This may be stressful, it is time-consuming and it may cause dehydration due to excessive salivation, as well as increasing the metabolic rate that may affect blood parameters (NC3Rs, http://www.nc3rs.org.uk/bloodsamplingmicrosite/default.asp?id=313). Indeed, any method that relies on peripheral vasodilation, either by increasing the environmental temperature or by using chemical agents, may be potentially harmful by causing excessive bleeding. This may result in the animal losing more blood than the recommended amount, after the sample has been collected.
Saphenous venepuncture has the advantage of ease of restraint. 1 If the restraining tube is covered with opaque material, mice will often enter it of their own accord. The restraining methods used for puncture of the tail vein are, in contrast, more unnatural and may increase stress. During saphenous venepuncture, it is extremely simple to apply pressure above the site (for presampling haemostasis) or over the puncture wound (if bleeding persists after sampling). This is considerably more difficult if the tail vein is used. In the present study, the mice sampled from the tail vein were more vocal both during restraint and bleeding than the mice sampled from the saphenous vein. This was true for both groups of mice and on both sampling days. It is difficult to avoid the subjective conclusion that the method of tail sampling is more stressful.
We have demonstrated in the present study that saphenous venepuncture has clear advantages compared with tail vein incision, and we believe the method also has welfare benefits, in line with the increasing focus on this field. 9
