Abstract
Blood is collected during animal experimentation to measure haematological and metabolic parameters. It cannot be assumed that circulating blood has the same composition irrespective of its location, and indeed, differences in the composition of blood sampled from the arterial and venous compartments have been reported. Here we investigated whether blood collected by cardiac puncture (CP) versus that collected following removal of the distal 1 mm of the tail tip (TT) differs with respect to glucose and lipid profiles in male C57BL/6J mice at 4, 7, 20 and 28 weeks of age. Blood was first collected from the TT of unanaesthetized mice, which were then immediately anaesthetized using ketamine/xylazine, and a second blood sample was collected by CP. The CP glucose concentration was significantly higher than TT glucose by a positive bias averaging +80% (P < 0.01), irrespective of the age of the mice. Conversely, the concentrations of the CP lipids, including total cholesterol, high-density lipoprotein cholesterol and triglyceride were lower than TT lipids by a negative bias averaging −25% (P < 0.05). These observations highlight the difficulty in measuring and comparing metabolic parameters such as glucose and lipid between one blood compartment and another. They illustrate the need to standardize sampling sites, especially when repeated blood sampling is required.
Assessment of blood parameters is often used to monitor animal health and to determine the success of dietary and pharmacological intervention in preclinical animal models. Cardiac puncture (CP) of anaesthetized animals is routinely used in rodent studies to collect large volumes of good quality central circulatory blood from the heart at the termination of an experiment. Smaller aliquots of peripheral blood are commonly obtained by removal of the distal 1 mm of the tail tip (TT) of conscious, unanaesthetized animals. Sampling blood from the TT is a less invasive procedure that allows repeated blood collection from the same animal without the confounding effects of anaesthesia. It is important to consider whether haematological and metabolic measurements might differ in relation to different blood sampling sites, and/or use of anaesthesia at the time of blood collection. More than one blood collection site may be used concurrently during a study, which would potentially confound the results. Further, metabolic parameters are sometimes compared across studies.
Rodents and humans share a similar cardiovascular circulatory system where the venae cavae carry deoxygenated blood from the tissues to the heart, which then pumps the blood via the pulmonary artery to the lungs. The oxygenated blood is pumped back to the heart via the pulmonary veins, and then to the rest of the body via the aorta. In the tail of the mouse, there are three veins and one artery, hence blood collection from the TT yields both venous and arterial blood. 1,2 Blood collected from the heart can yield venous or arterial blood or a mixture depending on the volume taken and site of puncture. Differences in the composition of the blood in an artery or a vein have been widely addressed in clinical medicine and clinical studies; 3,4 however, direct comparison remains less well known in animal studies and is often ignored. In humans, inconsistent differences in lipid and lipoprotein concentrations in arterial and venous blood have been reported. Miles et al. 5 reported that venous plasma triglyceride (TAG) concentrations were significantly lower than arterial values in eight healthy male subjects. In contrast, Kupke et al. 6 reported that the concentrations of lipids and lipoproteins measured in arterial blood serum were lower than in venous blood, and concluded that venous and arterial blood serum cannot be used interchangeably for these estimations.
There are little published animal study data on how levels of haematological and metabolic parameters differ in arterial and venous blood. Nevertheless, differences in the levels of glucose obtained from arterial versus venous blood have been documented in humans since the 1920s. 4,7–10 The levels of venous glucose generally reflect that of arterial glucose under fasting conditions; however, arterial and venous glucose levels can deviate in a similar but unpredictable manner after feeding or under a glucose load. The arterial value may be anywhere from 2% higher during fasting to as much as 70% higher in the postprandial state. 4,7–10 Other haematological parameters such as packed cell volume, haemoglobin, white blood cell, red blood cell and haematocrit have been reported to be higher in venous blood samples compared with blood samples obtained from the retro-orbital sinus or arterial heart of mice and rats. 11–15 In C57BL/6 mice the total leukocyte counts obtained by CP were reported to be significantly lower as compared with blood sourced from the saphenous vein, tail and foot. 16
Anaesthesia may affect the concentrations of a number of circulating metabolites. There are reports of higher levels of glucose in blood collected from rats anaesthetized with ether, pentobarbitone sodium or fentanyl plus droperidol compared with manually restrained unanaesthetized rats, 17 and similar findings have been observed in ketamine/xylazine anaesthetized rats. 18 Saha et al. 19 reported that anaesthesia with ketamine/xylazine caused acute hyperglycaemia associated with decreased insulin, adrenocorticotropic hormone and corticosterone in fed, but not fasted, rats. Hyperglycaemia and hypoinsulinaemia have also been documented during ketamine/xylazine anaesthesia in thoroughbred horses. 20 Hence, some commonly used anaesthetics can differentially affect a number of blood parameters in animal studies.
The aim of the present study therefore was to determine whether different blood sampling sites had a significant effect on measurements of circulating concentrations of glucose and lipids in male C57BL/6J mice. Blood samples were collected from the TT of unanaesthetized animals and then immediately from CP of ketamine/xylazine anaesthetized animals at 4, 7, 20 and 28 weeks of age.
Materials and methods
Animals and housing
Differences in glucose, total cholesterol, triglyceride (TAG) and high-density lipoprotein cholesterol (HDL-C) concentrations between cardiac puncture (CP) and tail tip (TT) blood
Values are expressed as mean difference (range) between CP and TT bleeds
NS: not significant as compared with the TT group of mice
*P < 0.01, # P < 0.05
Collection of blood samples
TT blood: Blood samples were collected from the TT of conscious, unanaesthetized mice at 4, 7, 20 and 28 weeks of age. Mice were fasted for 4 h on the morning of blood collection. Animals were placed in a restrainer and the tail section was covered with a clean gauze swab leaving the last 5 mm clear. The last 1 mm of the tip of the tail was then removed using sterilized surgical scissors. The tail was placed in a serum separator tube (Becton Dickinson Cat. No. 365956; Becton, Dickinson and Co, Franklin Lakes, NJ, USA), and gently milked to ensure an adequate blood flow. Approximately 200 µL of blood was collected on each occasion.
Cardiac puncture: Following collection of the TT sample, the mice were immediately anaesthetized by intraperitoneal injection with ketamine (100 mg/kg body weight) and xylazine (5 mg/kg body weight). They then underwent laparotomy and a 22 G needle attached to a 1 mL syringe was used to puncture through the diaphragm into the heart and 800 µL of blood was collected, representing most of the animal's blood volume, and expected to be a mix of venous and arterial blood. The blood was placed in a serum separator tube (Becton Dickinson Cat. No. 365956).
Analyses
The blood samples were centrifuged at 3000 rpm for 10 min, and the sera were collected and transferred to fresh microtubes, and stored at −80°C until analysed. Blood biochemistry was analysed using a Vitalab Flexor clinical chemistry analyser (Vitalab Flexor E, AC Dieren, The Netherlands). The serum levels of glucose were measured by the hexokinase method; 21 total cholesterol (TC) was measured by an enzymatic method using cholesterol esterase and cholesterol oxidase/peroxidase; TAG was measured by the lipase/glycerol kinase method; and high-density lipoprotein cholesterol (HDL-C) was measured by an enzymatic method using Roche assay kits (Roche Diagnostics GmbH, Mannheim, Germany).
Statistical methods
All data are expressed as mean ± standard error of the mean (SEM) unless otherwise stated. Cross comparisons between the blood collection sites were calculated using a paired t-test. Pearson's correlation coefficients were calculated to determine associations between the blood sampling sites and the various blood parameters. Linear regression analyses were used to determine the partial R 2 (Microsoft Office Excel 2007, Professional Edition, Microsoft Corporation). P < 0.05 was considered statistically significant.
Results
Measurement of blood glucose concentrations
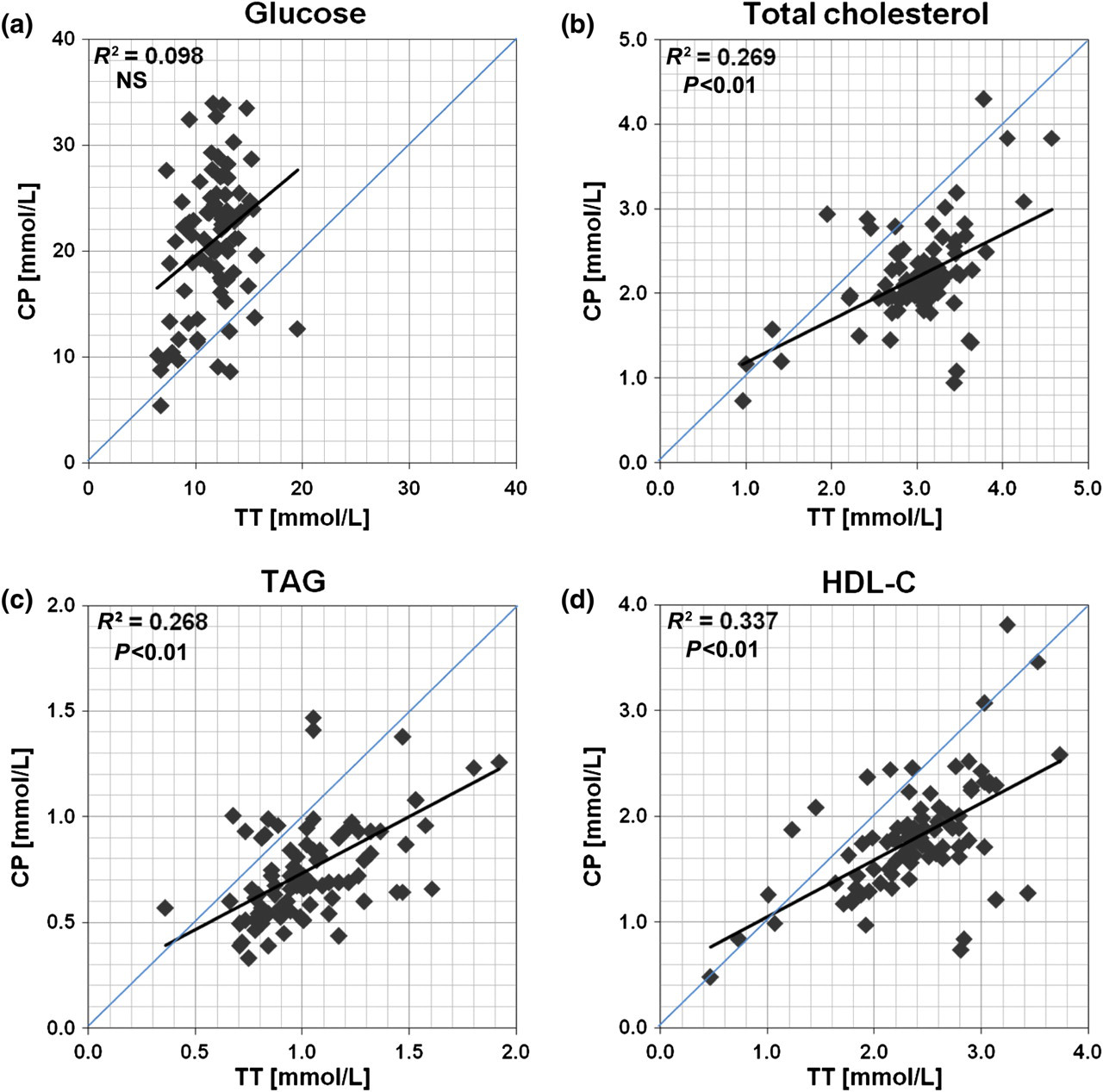
The mean glucose concentration was significantly different between blood obtained from the TT and by CP, irrespective of the age of the animals (Figure 1a; Table 1). CP glucose was significantly higher than TT glucose in 4-week-old (23.56 ± 1.34 versus 13.00 ± 0.29 mmol/L, P < 0.01), 7-week-old (20.55 ± 0.70 versus 13.13 ± 0.38 mmol/L, P < 0.01), 20-week-old (16.57 ± 1.24 versus 10.16 ± 0.60 mmol/L, P < 0.01) and 28-week-old (20.88 ± 1.63 versus 9.89 ± 0.54 mmol/L, P < 0.01) mice. CP glucose concentrations for all age groups combined exhibited a wide range of values (range 8.63–33.98 mmol/L) compared with TT glucose (range 8.15–15.69 mmol/L), and there was no correlation between the two sampling sites (glucose R
2: 0.098, P > 0.05) (Figure 2a). Further, more than 95% of the paired determinations had a higher glucose concentration in the blood obtained by CP than from the TT. The mean bias of the CP glucose concentrations versus TT glucose concentrations was approximately +80% (mean, SD) across all concentrations.
Levels of (a) glucose, (b) total cholesterol, (c) triglyceride (TAG) and (d) high-density lipoprotein cholesterol (HDL-C) in serum samples of male C57BL/6J mice from blood collected from the tail tip (TT) and by cardiac puncture (CP) at 4 (n = 40), 7 (n = 10), 20 (n = 20) and 28 (n = 20) weeks of age. n = number of animal samples; values are expressed as mean ± SEM. *P < 0.01 and #
P < 0.05 as compared with the TT group of mice Deviations of the levels of (a) glucose, (b) total cholesterol, (c) triglyceride (TAG) and (d) high-density lipoprotein cholesterol (HDL-C) in serum samples from blood collected from the tail tip (TT) and by cardiac puncture (CP) (n = 90); NS: not significant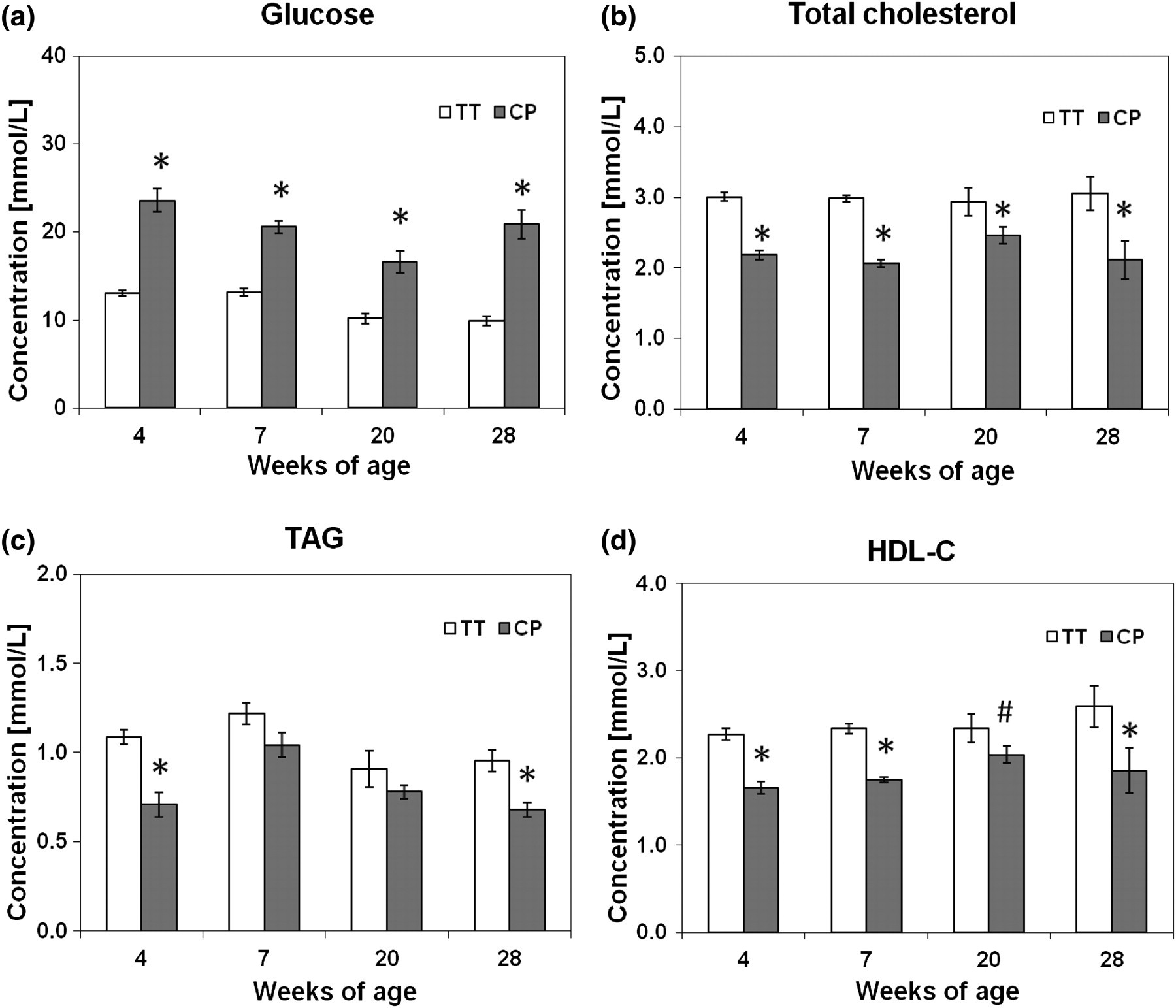
Measurement of blood lipid concentrations
In contrast to the glucose results, the levels of TC, HDL-C and TAG in arterial blood collected by CP from anaesthetized mice at 4, 7, 20 and 28 weeks of age were consistently lower than venous blood obtained from the TT (Figures 1b, c and d). These effects were significant irrespective of the age of the mice, with the exception of TAG at 7 and 20 weeks where there was no significant difference between the two sampling sites (Table 1).
There was a positive correlation between levels of TC, HDL-C and TAG measured from TT blood and those obtained by CP under anaesthesia (TC r 2: 0.27, P < 0.01; TAG r 2: 0.27, P < 0.01; HDL-C r 2: 0.34, P < 0.01; combined data from 4-, 7-, 20- and 28-week-old mice) (Figures 2b, c and d). Approximately 90% of the paired determinations of TC, TAG and HDL-C were lower in CP blood than in TT blood, yielding a mean negative bias of approximately −25% (mean, SD) across all concentrations.
Discussion
In the present study, we showed firstly that the commonly measured metabolic parameters of glucose, TC, HDL-C and TAG differed significantly between the two sampling sites irrespective of the age of the mice, with higher circulating glucose and lower circulating lipid concentrations in CP blood. Secondly, that when all age groups were combined there were significant positive correlations between TT and CP blood as lipid concentrations increased, but conversely no significant correlation between concentrations of glucose when collected from the TT and by CP. When working with rodents, blood is commonly collected from both the TT and/or heart. However, the choice of method and source of blood collection may vary, and hence differentially affect a number of blood parameters. 11–15,18,22,23 It is therefore important to consider any possible disparity in measured blood constituents caused by the technique of collection, to avoid untoward experimental bias.
The glucose concentrations in blood samples obtained by CP were determined to be higher than those obtained from the TT. The current understanding is that under conditions of basal, steady-state glucose metabolism, the glucose concentration in the artery is consistently higher than that in the vein. Henry 24 predicted on a physiological basis that arterial glucose concentrations are approximately 0.20 mmol/L higher than venous values in fasting individuals. In the present study, the combined mean difference for all animals across all age groups was 8.85 mmol/L (80% bias individual sample), hence we speculate that either such extreme differences between the two sampling site are either not entirely physiological, or this represents a physiological difference between humans and mice.
Higher blood glucose levels were reported from anaesthetized rodents compared with unanaesthetized manually restrained counterparts. 17 Recent studies by Rodrigues et al. 18 and Saha et al. 19 also showed similar findings where anaesthesia with ketamine/xylazine caused significant hyperglycaemic effects in non-fasted Sprague–Dawley rats after 20 min that continued to increase for 3 h post-anaesthesia via an α2-adrenoceptor pathway. Interestingly, ketamine/xylazine-induced anaesthesia did not cause hyperglycaemia in fasted rats, though there was a small and insignificant increase in blood glucose levels. In the present study, the mice were fasted for 4 h on the morning of blood collection and the time between ketamine/xylazine-induced anaesthesia and cardiac bleeding was less than 2 min, hence it is unlikely that the anaesthesia induced hyperglycaemia, though it cannot be ruled out as partly explaining why the TT and CP glucose levels are so different.
When external stimuli such as stress, even from routine laboratory procedures including animal handling and blood collection, are applied to animals, significant physiological changes correlated with stress have been reported in a number of studies, in particular stress-mediated increases in serum or plasma concentrations of corticosterone, glucose, growth hormone or prolactin, heart rate and blood pressure. 25,26 Periorbital puncture resulted in a dramatic increase in blood glucose in conscious mice indicating that this is a stressful procedure. 27 Tail puncture resulted in an increase in blood glucose in conscious mice, compared with saphenous venepuncture indicating that this is a stressful procedure. 28 Further Strawitz et al. 29 showed that fasted rats subjected to the standard shock procedure consisting of tail bleeding can mobilize glucose into the blood stream and cause severe hyperglycaemia independent of their initial glucose reserve. It is also possible that our mice may have experienced some form of haemorrhagic shock following the initial TT procedure which triggered the mobilization of glucose into the blood stream and caused an increase in blood glucose concentrations prior to the second CP procedure. Hypoperfusion is a known factor in the underestimation of point-of-care glucose testing, especially in critical care individuals. 30 Alternatively, milking the tail to collect a sufficient volume of blood could have led to dilution of blood by interstitial fluid as a contributory factor for the lower glucose concentrations.
The widely used C57BL/6J mouse strain is susceptible to obesity and atherosclerosis, which is exacerbated when the mice are fed a high-fat diet. 31–34 It is increasingly used as a diet-induced fatty mouse model of the metabolic syndrome that mimics high fat/energy-fed humans. 34 The present study shows that the levels of TC, HDL-C and TAG in blood obtained by CP from anaesthetized mice at 4, 7, 20 and 28 weeks of age were consistently lower than levels in TT blood. In contrast to glucose, this difference cannot be accounted for by simple dilution of venous blood with interstitial fluid in the TT sample due to milking the tail since the levels of lipids derived from TT blood were significantly higher than those obtained by CP. The increasing age of the mice did not seem to influence this pattern with the exception of circulating TAG at 7 and 20 weeks where it was just insignificant between the two sampling sites. The overall inter-individual variations (combined data) of CP blood lipid levels were narrower, and around 90% of the paired determinations measured were lower than those for TT blood yielding an average negative bias of approximately −25% across all concentrations irrespective of the age of the animals. We also found that circulating concentrations of TC, HDL-C and TAG collected from the TT were positively associated with the levels obtained by CP; however, the correlations were weak and TT values could not be used to predict CP concentrations of any lipid or lipoprotein component. In the fasting state used here glucose will be synthesized in the liver to be secreted into the circulation via the hepatic vein, whereas lipids will be released from both the liver and peripheral adipose tissues into the blood. Glucose extraction in the periphery may mean lower glucose levels in the tail and limbs than centrally. Whatever the exact reason for the consistent differences seen, the current observation suggests a probable true biological difference in lipid concentrations existing between these two sampling sites, and consequently we suggest that TT and CP blood should not be used interchangeably when assessing the levels of serum glucose and/or lipids within one experimental model.
In conclusion, the present study demonstrated that the blood sampling site can profoundly influence a number of commonly tested blood parameters such as glucose and lipid profiles including TC, HDL-C and TAG. Our findings emphasize the need to standardize the sampling site and collection method, especially in longitudinal research studies where multiple and repetitive routine blood samples are required.
Footnotes
ACKNOWLEDGEMENTS
The authors gratefully acknowledge Yih-Chih Chan, Carol Zhu and Katy Wiessing for their technical assistance. We thank staff members of the Biomedical Research Unit (BRU), University of Otago, Wellington, and at the Vernon Jansen Unit (VJU), University of Auckland for their assistance with daily animal husbandry. We thank staff of the Bioactivity Investigation Group and Trinity Bioactives Ltd, Wellington for technical assistance. This study was supported by funding from the Foundation for Research Science and Technology, and the Fonterra Co-operative Group via the consortium Lactopharma, New Zealand.
