Abstract
For oral glucose tolerance test (OGTT) in mice, multiple blood samples need to be taken within a few hours from conscious mice. Today, a number of essential parameters may be analysed on very small amounts of plasma, thus reducing the number of animals to be used. It is, however, crucial to obtain high-quality plasma or serum in order to avoid increased data variation and thereby increased group sizes. The aim of this study was to find the most valid and reproducible method for withdrawal of blood samples when performing OGTT. Four methods, i.e. amputation of the tail tip, lateral tail incision, puncture of the tail tip and periorbital puncture, were selected for testing at 21°C and 30°C after a pilot study. For each method, four blood samples were drawn from C57BL/6 mice at 30 min intervals. The presence of clots was registered, haemolysis was monitored spectrophotometrically at 430 nm, and it was noted whether it was possible to achieve 30–50 μL blood. Furthermore, a small amount of extra blood was sampled before and after the four samplings for testing of whether the sampling induced a blood glucose change over the 90 min test period. All methods resulted in acceptable amounts of plasma. Clots were observed in a sparse number of samples with no significant differences between the methods. Periorbital puncture did not lead to any haemolysed samples at all, and lateral tail incision resulted in only a few haemolysed samples, while puncture or amputation of the tail tip induced haemolysis in a significant number of samples. All methods, except for puncture of the tail tip, influenced blood glucose. Periorbital puncture resulted in a dramatic increase in blood glucose of up to 3.5 mmol/L indicating that it is stressful. Although lateral tail incision also had some impact on blood glucose, it seems to be the method of choice for OGTT, as it is likely to produce a clot-free non-haemolysed sample, while periorbital sampling, although producing a high quality of sample, induces such a dramatic change in blood glucose that it should not be applied for OGTT in mice.
For oral glucose tolerance test (OGTT), as well as in other studies in mice, multiple blood samples need to be taken within a few hours from conscious mice that typically have a total of approximately 2 mL of blood. To avoid impact on welfare as well as on serum parameters, it is considered good practice not to deplete the animal of more than 20% of its blood volume, i.e. approximately 300 μL within 24 h. 1 Previously, this might be problematic from an analytical point of view, and often entire mice were bled at different times, but today it is possible to analyse a number of essential parameters on very small amounts of plasma, e.g. 10 μL or even less, thus reducing the number of animals by using the same mouse during the entire test period. It is, however, crucial to obtain high-quality plasma or serum to avoid increased data variation and thereby increased group sizes.
A number of blood sampling methods have been recommended for multiple sampling in mice. Lateral tail bleeding is the most commonly applied method for OGTT. It can be made with a needle 2 or as a scalpel incision, and this method is recommended by the European Federation of Pharmaceutical Industries and Associations/European Centre for the Validation of Alternative Methods (EFPIA/ECVAM) group. 1 The tail incision can also be performed ventrally with a scalpel without pre-heating. 3 Although highly controversial, periorbital blood sampling is a method commonly used for OGTT, 4 e.g. for product control in companies working with insulin. For OGTT, anaesthesia is not possible as it interferes with insulin production, although a joint EFPIA/ECVAM working group in 2001 recommended that this method should only be used under anaesthesia. 1 This seems intuitively rational, but no references are given by the group, and very little scientific backup for this recommendation is actually available. Studies on periorbitally sampled mice are few. One paper describing catecholamine monitoring in mice concludes that the method is non-stressful. 5 In rats, the method seems more stressful than e.g. jugular sampling as monitored by cardiovascular telemetry, 6 but such stress is not reduced by ether or carbon dioxide anaesthesia. 6,7 In one study in rats, ether anaesthesia, which is a well-known stressor 8 and therefore not a fully covering example, increased stress hormones, while periorbital puncture itself did not. 9 In the absence of scientific support, legislative considerations of this method differ between European countries, and there is no general ban on omitting anaesthesia. One alternative is puncture of the lateral tarsal (saphenous) vein as recommended by the EFPIA/ECVAM group 1 for which the skin is shaved using a scalpel, 10 and the area is prepared with petroleum jelly, 11 ethanol 1 or silicone grease. 10 For multiple samplings, the clot is simply removed. 10 The tail is also commonly applied due to easy access and rich vascularization. Since the tail is used as a thermoregulatory organ, pre-heating at 30–50°C for 5 min may be useful. 12,13 The ‘farmers wife method’ or amputation of the tail tip, which is a very used method 14,15 recommended by a BVA/FRAME/RSPCA/UFAW joint working group, 16 may yield up to 1.1 mL, 17 and it may be used for multiple sampling by applying a vacuum. 18 When this method is used for multiple sampling, removing the clot is often sufficient, but in case re-cutting is necessary no more than 5 mm should be removed totally. 1
The choice of sampling site and method is known to have deleterious impact on some haematological and clinico-chemical parameters, 19,20 e.g. because the stress induced by the various methods may change parameters such as glucose and insulin. Haemolysis, another frequently occurring problem, changes the ability of the serum to absorb light, and it releases erythrocytic proteases and peptidases decomposing the components that are to be analysed. To avoid haemolysis, it is important to avoid milking the tail, which is another argument for working with small serum volumes. Murine blood has a tendency to form clots which after centrifugation can be found in the plasma. To be able to work with small plasma volumes, it is crucial to avoid such clots. From 30 μL to 50 μL blood approximately 15–20 μL plasma may be obtained. Therefore, polymerase chain reaction (PCR) plates or specialized microtubes need to be applied instead of ordinary 96-well microtiter plates. Only in these plates, it is possible to work with volumes below 10 μL. Because 10 μL does not fill much even in these plates, it is impossible to avoid getting the clots into the pipette tips. As an increasing part of modern laboratory work has been automated, which is beneficial due to speed, reduction of stereotypical work and labour costs, and the elimination of human mistakes, visual or mechanical control to verify the absence of clots and haemolysis becomes difficult, and even if manual inspection is performed, visual estimation of haemolysis is rather subjective. 21,22 More objective methods, such as the reaction with tetramethylbenzidine 23 or direct spectrophotometry may be applied, although these methods would require sampling of additional plasma. From 30 μL to 50 μL blood, approximately 15–20 μL plasma may be obtained, and therefore, PCR plates with 384 wells or specialized microtubes need to be applied instead of ordinary 96-well microtiter plates. Only in these plates is it possible to work with volumes below 10 μL, and they increase the throughput in a busy laboratory. Commercial pipette clotting detection systems are only available for volumes above 10 μL and only on eight-channel pipettors. There are no 96- or 384-channel pipettors on the market with clot detection. Due to this fact, it is crucial to avoid plasma samples with clots when using automated equipment.
The aim of this study was to find the most valid and reproducible method for withdrawal of four blood samples from a conscious and unanaesthetized mouse with 30 min intervals between each sample. The samples should be suitable for performing OGTT, i.e. the sampling should be able to yield 30–50 μL blood in a non-haemolysed and clot-free sample, and the method itself should not interfere with serum glucose levels.
Materials and methods
Licence
The study was performed under a licence issued by the National Animal Experimentation Board under the Danish Ministry of Justice. Danish animal experimentation legislation is based upon the EU Directive 86/609 and the Council of Europe Convention ETS 123. The study was furthermore approved by the in-house ethical committee of Novo Nordisk.
Pilot study to select methods and to calculate group sizes
A total of 265 single blood samples were drawn from 75 C57BL/6.VBomTac-Lepob mice by lateral tail incision, amputation of the tail tip, puncture of the tail tip, periorbital puncture and puncture of the lateral tarsal vein at both 21°C and 30°C. These blood samples showed a mean absorbance (mean ± SD) of 0.437 ± 0.291 (lateral tail incision), 0.406 ± 0.171 (tail-tip amputation), 0.575 ± 0.240 (tail-tip puncture) and 0.161 ± 0.100 (periorbital puncture), whereas puncture of the lateral tarsal vein, with which the technicians had less experience, did not reveal enough blood. Based upon this, a group size of 20 mice per method was used for the main study, which would allow a difference of 0.250 to be shown (P < 0.05, π = 75–99%). The four methods listed in Table 1, i.e. amputation of the tail tip (Figure 1), lateral tail incision (Figure 2), puncture of the tail tip (Figure 3) and periorbital puncture (Figure 4), were selected for testing at two different temperatures, i.e. 21 ± 2°C and 30 ± 1°C, which were methods with which the technicians had several years of experience.

Blood sampling from a lateral tail incision in the mouse. The mouse was fixed in a v-trap and the lateral tail incision was performed on one side of the tail with a scalpel no. 21 (Lance Paragon Limited, Sheffield, UK). Approximately, 2 cm from the tail base a vertical incision with an adequate depth was made (a). The blood was drawn from the pool of blood on the surface of the tail. If not enough blood appeared in the incision, slight pressure was applied on the incision by running a finger on the opposite side of the tail (b). Subsequent samples were drawn by removing the clot or re-cutting when necessary
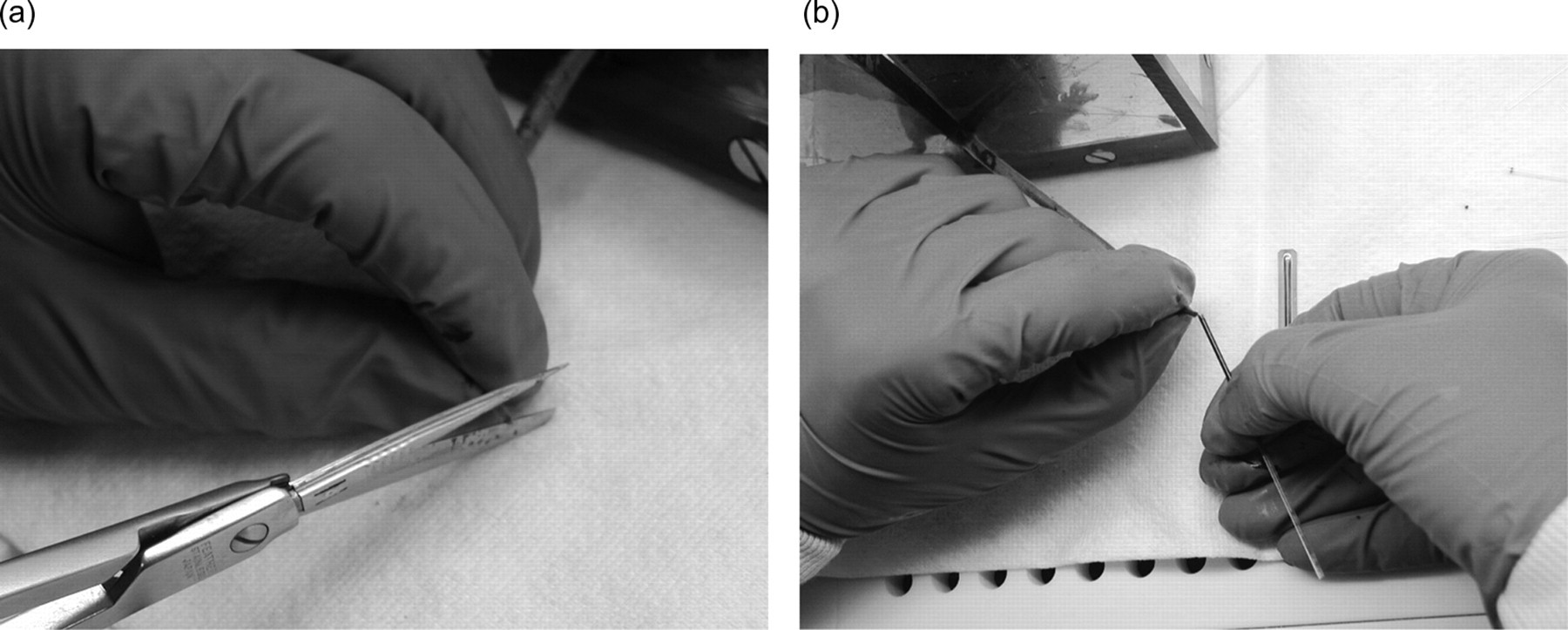
Blood sampling by the amputation of the tail tip in the mouse. The mouse was fixed in a v-trap and amputation of the tail tip was performed with a pair of scissors no. 145 with a sharp/blunt replaceable blade (Medizin AG, Cologne, Germany) and approximately 1 mm was removed (a). The blood was drawn from the pool of blood on the tip of the tail (b). If necessary, the blood was moved to the tip of the tail by gently ‘milking’ the tail. Subsequent samples were drawn by removing the clot or re-cutting when necessary

Blood sampling from a tail-tip puncture. The mouse was fixed in a v-trap and the puncture of the tail tip was performed with a lancet no. 366 (Karl Hecht KG, Sondheim, Germany) (a). The tip of the tail was punctured and the blood was drawn from the pool of blood on the tip of the tail. If necessary, the blood was moved to the tip of the tail by gently ‘milking’ the tail (b). Subsequent samples were drawn by removing the clot or re-puncturing when necessary
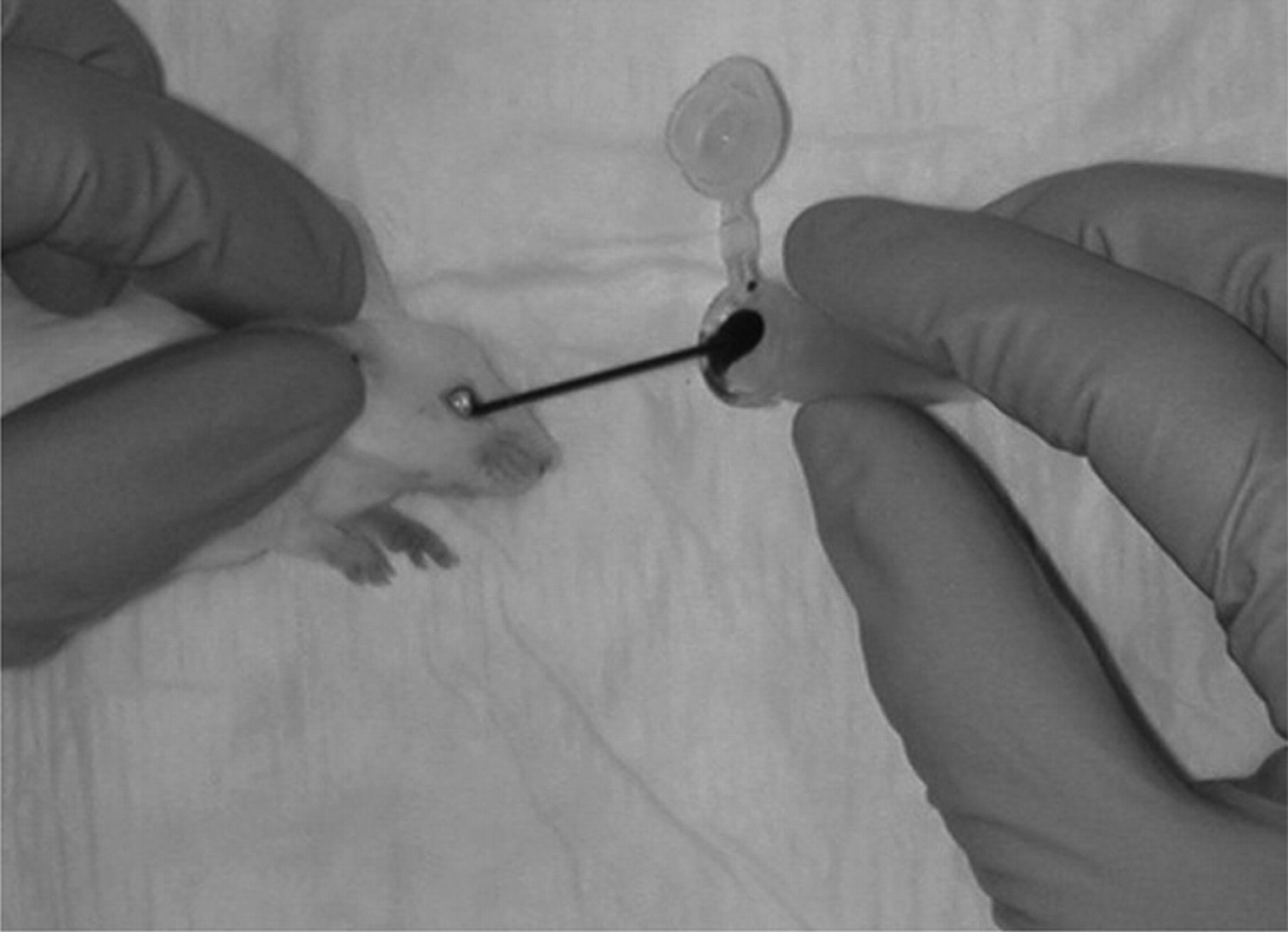
Blood sampling by periorbital puncture in the mouse. The mouse was grabbed by the scruff and fixed on one side towards the table. A 100 μL microhaematocrit tube was inserted in the medial cantus of the orbit and gently pressed and rotated until blood appeared in the tube. Subsequent samples were drawn from the opposite eye
Haemolysis, clotting and spontaneous change in blood glucose over 90 min after blood sampling from mice by four different methods at two different temperatures
Absorbance cut-off was 0.5 (OD 430) for 5 μL plasma volume in assay or 0.95 for 1 μL plasma volume in assay (dependent on assay technology)
*Change significantly different from 0 by P < 0.05
**Change significantly different from 0 by P < 0.01
***Change significantly different from 0 by P < 0.001
****Groups significantly different from one another with a P < 0.0001 in Fisher's exact test with Monte Carlo estimate
Groups having the same letter do not differ significantly from one another
Animals and housing
In a classical parallel design, 160 male C57BL/6JBomTac mice weighing 17.9–30.5 g were divided into 16 groups in a weight-balanced stratification and housed in a Tecniplast Makrolon type IV cage with a raised lid (Scanbur, Karlslunde, Denmark) with Tapvei aspen bedding, Enviro-Dri® paper-based nesting material, a Tapvei aspen wood biting stick (Brogaarden, Gentofte, Denmark) and a hide (Novo Nordisk, Måløv, Denmark) in a ScantainerNOVO cage system (Scanbur). In each group, the mice were earmarked with numbers from 1 to 10. The mice were fed Altromin 1320 diet (Brogaarden) plus a small amount of corn and wheat mixed into the bedding material. Tap water was provided automatically ad libitum. The light was on from 07:00 h to 19:00 h, the temperature was 20 ± 2°C and the air humidity was 45 ± 5%.
Prior to sampling, all mice were weighed and the weight of the mice from each method was tested for normal distribution by Shapiro-Wilk's test, and the means of the groups were compared by analysis of variance (ANOVA), while the means of mice sampled at different temperature were compared by student's t-test.
Blood sampling and testing
The complete blood sampling process is described in Figure 5. Each blood sampling method was performed on a group of 20 unanaesthetized mice. Each method was tested at room temperatures of both 21°C and 30°C, i.e. 80 mice were used for testing at each temperature. Each group was divided into two subgroups of 10, and the subgroups were sampled on two different days at intervals of a few days. After the session, the mice were euthanized. Two skilled technicians were each handling one subgroup. Subgroups were compared by the statistical methods described below for each parameter to reveal any difference due to these factors.
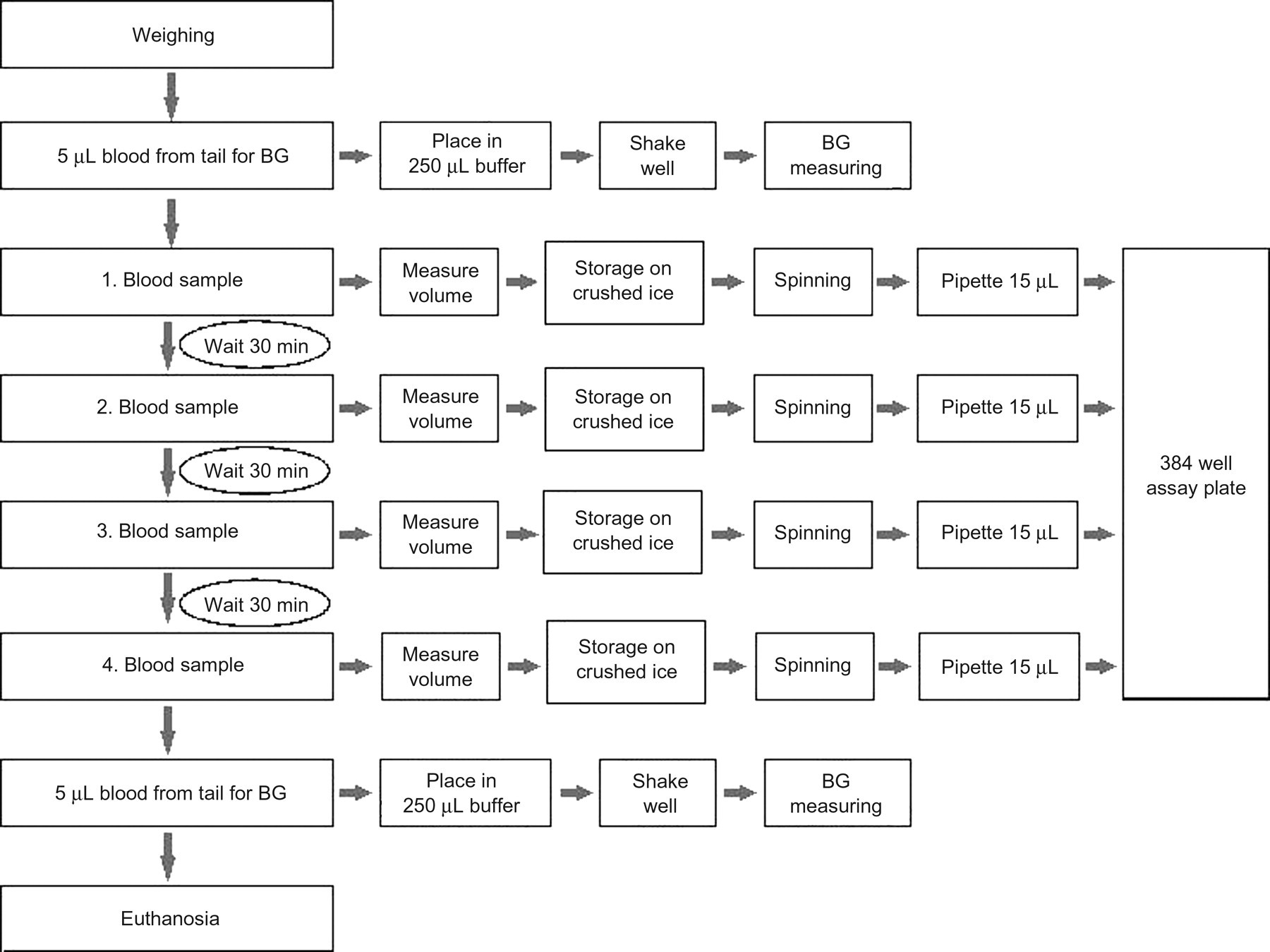
Blood sampling schedule for comparing the effectiveness of four different blood sampling techniques for mice
Four blood samples were drawn at 30 min intervals. All samples were collected in 100 μL microhaematocrit tubes with 8 mmol/m ethylenediaminetetraacetic acid (EDTA) as anticoagulant (Modulohm A/S, Herlev, Denmark) aiming at an optimal volume of 50 μL with a minimum of 30 μL accepted. After sampling, the tube was closed at one end with VITREX Sigillum Wax (Modulohm A/S) and stored on crushed ice for no longer than 30 min before being centrifuged at 8000 rpm at 4°C for 10 min. A total of 15 μL of the plasma was transferred with a pipette to a well in a 384-well assay plate no. 781182 (Greiner Bio-One GmbH, Frickenhausen, Germany) placed on dry ice. After sampling was completed, the plate was frozen at –18°C, and the mouse was euthanized by CO2/O2. Mice with clearly visible trauma from fighting were excluded from the study.
Before the first and after the fourth blood sample, 5 μL additional blood was drawn by puncture of the tail tip with a lancet no. 366 (Karl Hecht KG, Sondheim, Germany). These samples were collected in a 5 μL end-to-end micropipette (Modulohm A/S) and placed in an Eppendorf tube with 250 μL Ebio buffer (Prahl Biolab A/S, Græsted, Denmark). The tube was thoroughly shaken, and the blood glucose was measured within half an hour in an EBIO® plus unit (Eppendorf AG, Hamburg, Germany). The difference between the blood glucose values before and after the withdrawal of the four blood samples was calculated as T90–T0 and the differences were tested for normal distribution with Shapiro-Wilk within each method. By comparing the differences with a t-test against 0, it was tested if any of the methods induced a significant change in the glucose level. The means of the differences from each method were compared with each other using ‘PROC MIXED’ ANOVA. The residuals from the ANOVA were plotted as normal distribution and tested by Anderson-Darling. The distribution between the predicted values and the residuals was plotted.
For each method, the number of samples not yielding the accepted minimum volume of 30 μL and the number of samples not yielding the optimal volume of 50 μL were counted and compared using Fisher's exact test.
After re-thawing, samples were mixed with 20 μL of water and vibrated for 3 min. The absorbance was measured using a Spectraflour Plus microplate reader (Tecan, Salzburg, Austria) with a wavelength at 430 nm and a reference at 590 nm. Values from wells with insufficient amounts of plasma or wells in which clots appeared before or after the addition of water were not accepted. The values within each method were tested for normal distribution with Anderson-Darling. The means of the absorbance values from each method were compared with each other using ‘PROC MIXED’ ANOVA with autoregressive covariance structure, since the absorbance values from the same mouse were not independent. The residuals from the ANOVA were plotted as normal distribution and tested by Anderson-Darling. The distribution between the predicted values and the residuals was plotted. Two cut-off values for characterizing a sample as ‘haemolysed’ have previously been established at Novo Nordisk A/S as 0.95 when using 1 μL plasma in assays, and 0.50 when using 5 μL plasma in assays. Fisher's exact test with Monte Carlo estimate was then used to test whether some of the methods more frequently led to haemolysed samples.
In a stereo magnifier, the samples were inspected visually before the addition of water under 10× magnification to see if any clots had developed in the plasma, and the numbers of clotted samples for each method were compared by Fisher's exact test.
All data were analysed using SAS V.8.02 (SAS Institute Inc, Cary, NC, USA).
Results
Three mice being exposed to tail-tip amputation at 30°C were excluded during the study due to visible fighting wounds, leaving only 17 mice in this group. There were no incidents in the mice due to sampling. Mean weights of the groups were normally distributed and ranged from 23.7 g (periorbital puncture at 21°C) to 25.8 g (tail-tip puncture at 30°C). There was a significant difference between the weight of the groups (P < 0.01) due to a difference between mice sampled at 21°C (24.1 ± 2.25) and mice sampled at 30°C (25.7 ± 2.24), which again was due to the practical problem that mice at 21°C had to be sampled first, while the remaining groups had to be sampled a few days later when the laboratory had been heated to 30°C. There were no significant differences between subgroups sampled at the same temperature.
Out of 80 samples for each method (only 79 for puncture of tail tip at 21°C and 68 for amputation of the tail tip at 21°C, as some tubes broke during centrifugation), all yielded an amount of at least 50 μL blood, except for one lateral tail incision at 21°C, two tail-tip punctures at 21°C and one tail-tip puncture at 30°C, and therefore there were no significant differences between the groups in this respect (P = 0.303).
Absorbance values for the plasma samples are shown in Table 1. Periorbital puncture was the only method that did not cause haemolysis in any of the samples, and for lateral tail incision, the number of haemolysed samples was also rather minimal. However, amputation or puncture of the tail tip produced a significant number of haemolysed samples, i.e. from 14% to 61% of the samples at 30°C.
The prevalence of clotted samples before adding water to the plasma was found to be between 0% and 2.9% (Table 1). The lateral tail incision was the only method not causing the blood to clot in any of the samples, but there were no significant differences between the methods.
All data on the change in blood glucose from initiating the OGTT (T0) to finalizing it (T90) (Table 1) were normally distributed. Amputation of the tail tip in itself led to a decrease in blood glucose, which was significant at 30°C (P < 0.05), whereas periorbital puncture at both temperatures significantly increased the blood glucose by more than 3 mmol/L (P < 0.001).
Discussion
Three key elements would be crucial for blood sampling for OGTT and similar studies: high quality of the sample, i.e. the absence of haemolysis and clots, reproducibility, i.e. possibility of achieving multiple samples of high quality in which the sampling technique itself does not influence the parameters to be monitored, and last but not least animal welfare, i.e. that the animal experiences little discomfort as a cause of the procedure and leaves the study without compromised health or wellbeing.
In the pilot study, we were unsuccessful in receiving sufficient yields by lateral tarsal vein punctures. As this is in contrast to previous recommendations, 1,10,11 it may be considered that the outcome of the method might be improved by training and might also be easier in non-obese mice. However, for the four selected methods, i.e. amputation of the tail tip, lateral tail incision, puncture of the tail tip and periorbital puncture, we experienced no problems at any time with achieving a sample of 30–50 μL blood, so in this sense, the methods are equal. Clotting of the samples was not a major issue and as such not relevant to deselecting, although it is noticeable that lateral tail incision was the only method in which there was no clot formation at all. Haemolysis disqualifies puncture and may also disqualify amputation of the tail tip. Lateral tail incision caused two samples to haemolyse at both temperatures, which may be too little to have major impact on the data evaluation, whereas periorbital puncture was the only method resulting in non-haemolysed plasma in all samples, which was also clear from the fact that these samples showed the lowest mean absorbance. So, from a sample quality point of view, it is clear that periorbital puncture is superior to the other methods. Lateral tail incision is also an acceptable method, whereas amputation and especially puncture of the tail tip are not recommendable methods. It may be an idea to include absorbance reading in an automatic assay protocol. It should, of course, be considered that the exact way in which the method is performed may differ between laboratories, and therefore also some of these methods may be refined to produce better plasma. For example, a scalpel may be used for tail-tip amputation, which could be less traumatic compared with scissors, which may not recently have been sharpened and therefore may cause more haemolysis. It is of interest to note that heating the room to 30°C did not increase the quality of plasma, and there was no problem getting acceptable yields at 21°C, consequently, this should not be considered necessary.
It is of essential interest how amputation or punctuation of the tail tip, lateral tail incision and not at least periorbital puncture influence glucose monitoring. While changes in blood glucose of 1 mmol/L may be acceptable, an increase in blood glucose of 3.5 mmol/L, as observed in plasma after periorbital sampling due to the technique itself, renders this method totally out of the question for OGTT although commonly applied. 4 In rats, periorbital puncture has been shown to be stressful, 6 and the dramatic increase it causes in blood glucose in the present study indicates that this is also the case in mice, as increased blood glucose is a well-known stress response in rodents. 24,25 Obtaining blood by periorbital bleeding has previously been shown to induce decreased glucose and increased insulin compared with blood obtained by tail bleeding in mice, 26 so these findings are in contrast to our findings. However, it is known that stressors such as chronic restraint 27 and forced swimming 28 decrease insulin secretion and increase serum glucose in rodents. The other methods are probably stress-inducing as well, but probably to a smaller degree than periorbital sampling as far as can be judged from the blood glucose data. Obviously, smaller or larger deviations in the performance of the techniques, e.g. due to more or less routine, will have importance for the stress induced to the animals.
Although periorbital puncture 4 and also amputation of the tail vein 29 are applied by some groups, lateral tail vein incision is the most commonly applied method for OGTT. It can be concluded that this is for good reasons, while periorbital puncture should be disregarded in the future due to its invalidating impact on blood glucose and because it seems to stress the mice.
Footnotes
ACKNOWLEDGEMENTS
The authors gratefully acknowledge Hanne Jensen-Holm, Mikkel Schrøder Jørgensen, Ulla Ebbesen and Henriette Løwe Press at Novo Nordisk A/S for their excellent technical support during the blood sampling, the animal caretakers at Novo Nordisk A/S for their skilful animal care and Helle Vinberg Schrøder for her linguistic corrections.
